Increased Remnant Lipoproteins in Apo E Deficient Mice Induce Coronary Atherosclerosis following Transverse Aortic Constriction and Aggravate the Development of Pressure Overload-Induced Cardiac Hypertrophy and Heart Failure
Abstract
1. Introduction
2. Materials and Methods
2.1. In Vivo Experiments
2.2. Blood Sampling
2.3. Separation of Lipoproteins by Gel Filtration
2.4. Quantification of Lipid Peroxidation Products in Plasma
2.5. In Vivo Hemodynamic Measurements
2.6. Histological Analysis
2.7. Statistical Analysis
3. Results
3.1. Markedly Increased Mortality in C57BL/6N Apo E−/− Mice Compared to C57BL/6N Mice following Transverse Aortic Constriction
3.2. Pressure Overload Induced by TAC Induces Coronary Atherosclerosis in C57BL/6N Apo E−/− Mice
3.3. Pressure Overload Induced by TAC Results in Markedly Increased Cardiac Hypertrophy and More Pronounced Heart Failure in C57BL/6N Apo E−/− Mice in Comparison to C57BL/6N Mice
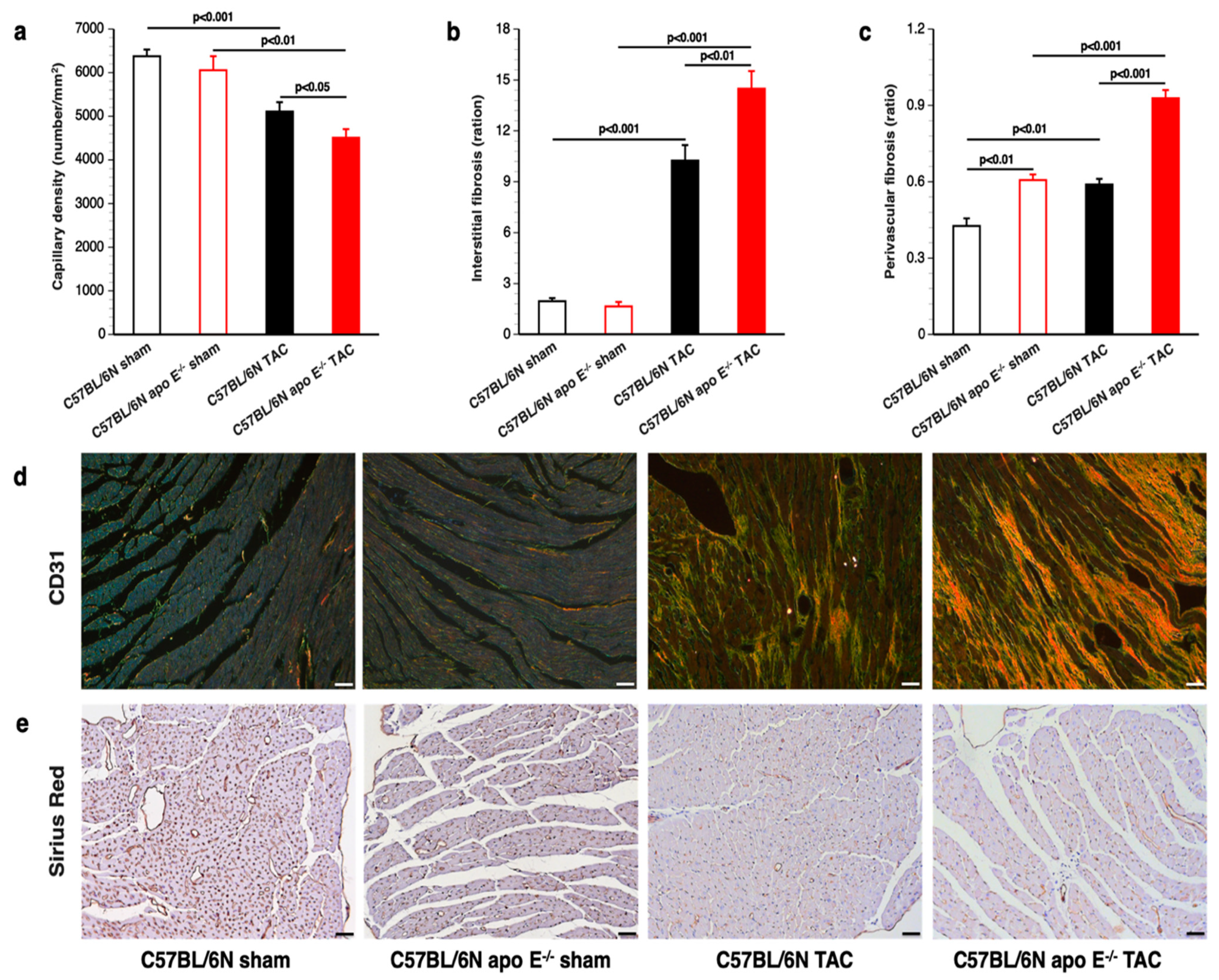
3.4. Increased Myocardial Fibrosis and Capillary Rarefaction following TAC in Apo E Deficient Mice
3.5. Deterioration of Cardiac Function Induced by TAC Is Significantly More Pronounced in Apo E Deficient Mice Compared to Wild-Type Mice
3.6. Apo E Deficiency Induces Pronounced Oxidative Stress in Both Sham Mice and TAC Mice
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Williams, K.J.; Tabas, I. The response-to-retention hypothesis of early atherogenesis. Arter. Thromb. Vasc. Biol. 1995, 15, 551–561. [Google Scholar] [CrossRef] [PubMed]
- Fogelstrand, P.; Boren, J. Retention of atherogenic lipoproteins in the artery wall and its role in atherogenesis. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Boren, J.; Olin, K.; Lee, I.; Chait, A.; Wight, T.N.; Innerarity, T.L. Identification of the principal proteoglycan-binding site in LDL. A single-point mutation in apo-B100 severely affects proteoglycan interaction without affecting LDL receptor binding. J. Clin. Investig. 1998, 101, 2658–2664. [Google Scholar] [CrossRef]
- Flood, C.; Gustafsson, M.; Richardson, P.E.; Harvey, S.C.; Segrest, J.P.; Boren, J. Identification of the proteoglycan binding site in apolipoprotein B48. J. Biol. Chem. 2002, 277, 32228–32233. [Google Scholar] [CrossRef] [PubMed]
- Wight, T.N. A role for proteoglycans in vascular disease. Matrix Biol. 2018, 71–72, 396–420. [Google Scholar] [CrossRef]
- Bornfeldt, K.E.; Linton, M.F.; Fisher, E.A.; Guyton, J.R. JCL roundtable: Lipids and inflammation in atherosclerosis. J. Clin. Lipidol. 2021, 15, 3–17. [Google Scholar] [CrossRef]
- McGill, H.C., Jr.; McMahan, C.A. Determinants of atherosclerosis in the young. Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group. Am. J. Cardiol. 1998, 82, 30T–36T. [Google Scholar] [CrossRef]
- McGill, H.C., Jr.; McMahan, C.A.; Gidding, S.S. Preventing heart disease in the 21st century: Implications of the Pathobiological Determinants of Atherosclerosis in Youth (PDAY) study. Circulation 2008, 117, 1216–1227. [Google Scholar] [CrossRef]
- VanderLaan, P.A.; Reardon, C.A.; Getz, G.S. Site specificity of atherosclerosis: Site-selective responses to atherosclerotic modulators. Arter. Thromb. Vasc. Biol. 2004, 24, 12–22. [Google Scholar] [CrossRef]
- Feussner, G.; Wagner, A.; Kohl, B.; Ziegler, R. Clinical features of type III hyperlipoproteinemia: Analysis of 64 patients. Clin. Investig. 1993, 71, 362–366. [Google Scholar] [CrossRef]
- Paquette, M.; Bernard, S.; Pare, G.; Baass, A. Dysbetalipoproteinemia: Differentiating Multifactorial Remnant Cholesterol Disease From Genetic ApoE Deficiency. J. Clin. Endocrinol. Metab. 2022, 107, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Boot, C.S.; Luvai, A.; Neely, R.D.G. The clinical and laboratory investigation of dysbetalipoproteinemia. Crit. Rev. Clin. Lab. Sci. 2020, 57, 458–469. [Google Scholar] [CrossRef] [PubMed]
- Chacon-Camacho, O.F.; Pozo-Molina, G.; Mendez-Catala, C.F.; Reyes-Reali, J.; Mendez-Cruz, R.; Zenteno, J.C. Familial Hypercholesterolemia: Update and Review. Endocr. Metab. Immune Disord. Drug Targets 2022, 22, 198–211. [Google Scholar] [CrossRef] [PubMed]
- Van Craeyveld, E.; Jacobs, F.; Feng, Y.; Thomassen, L.C.; Martens, J.A.; Lievens, J.; Snoeys, J.; De Geest, B. The relative atherogenicity of VLDL and LDL is dependent on the topographic site. J. Lipid Res. 2010, 51, 1478–1485. [Google Scholar] [CrossRef] [PubMed]
- Van Craeyveld, E.; Gordts, S.C.; Jacobs, F.; De Geest, B. Correlation of atherosclerosis between different topographic sites is highly dependent on the type of hyperlipidemia. Heart Vessel. 2012, 27, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Oppi, S.; Luscher, T.F.; Stein, S. Mouse Models for Atherosclerosis Research-Which Is My Line? Front. Cardiovasc. Med. 2019, 6, 46. [Google Scholar] [CrossRef] [PubMed]
- Braun, A.; Trigatti, B.L.; Post, M.J.; Sato, K.; Simons, M.; Edelberg, J.M.; Rosenberg, R.D.; Schrenzel, M.; Krieger, M. Loss of SR-BI expression leads to the early onset of occlusive atherosclerotic coronary artery disease, spontaneous myocardial infarctions, severe cardiac dysfunction, and premature death in apolipoprotein E-deficient mice. Circ. Res. 2002, 90, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Robicsek, F.; Thubrikar, M.J. The freedom from atherosclerosis of intramyocardial coronary arteries: Reduction of mural stress--a key factor. Eur. J. Cardiothorac. Surg. 1994, 8, 228–235. [Google Scholar] [CrossRef]
- Nakanishi, R.; Baskaran, L.; Gransar, H.; Budoff, M.J.; Achenbach, S.; Al-Mallah, M.; Cademartiri, F.; Callister, T.Q.; Chang, H.J.; Chinnaiyan, K.; et al. Relationship of Hypertension to Coronary Atherosclerosis and Cardiac Events in Patients With Coronary Computed Tomographic Angiography. Hypertension 2017, 70, 293–299. [Google Scholar] [CrossRef]
- Fuchs, F.D.; Whelton, P.K. High Blood Pressure and Cardiovascular Disease. Hypertension 2020, 75, 285–292. [Google Scholar] [CrossRef]
- Muthuramu, I.; Mishra, M.; Aboumsallem, J.P.; Postnov, A.; Gheysens, O.; De Geest, B. Cholesterol lowering attenuates pressure overload-induced heart failure in mice with mild hypercholesterolemia. Aging 2019, 11, 6872–6891. [Google Scholar] [CrossRef] [PubMed]
- Muthuramu, I.; Amin, R.; Postnov, A.; Mishra, M.; Aboumsallem, J.P.; Dresselaers, T.; Himmelreich, U.; Van Veldhoven, P.P.; Gheysens, O.; Jacobs, F.; et al. Cholesterol-Lowering Gene Therapy Counteracts the Development of Non-ischemic Cardiomyopathy in Mice. Mol. Ther. 2017, 25, 2513–2525. [Google Scholar] [CrossRef] [PubMed]
- Muthuramu, I.; Amin, R.; Aboumsallem, J.P.; Mishra, M.; Robinson, E.L.; De Geest, B. Hepatocyte-Specific SR-BI Gene Transfer Corrects Cardiac Dysfunction in Scarb1-Deficient Mice and Improves Pressure Overload-Induced Cardiomyopathy. Arter. Thromb. Vasc. Biol. 2018, 38, 2028–2040. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Benn, M.; Schnohr, P.; Tybjaerg-Hansen, A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA 2007, 298, 299–308. [Google Scholar] [CrossRef]
- Varbo, A.; Nordestgaard, B.G. Remnant Cholesterol and Triglyceride-Rich Lipoproteins in Atherosclerosis Progression and Cardiovascular Disease. Arter. Thromb. Vasc. Biol. 2016, 36, 2133–2135. [Google Scholar] [CrossRef] [PubMed]
- Muthuramu, I.; Amin, R.; Postnov, A.; Mishra, M.; Jacobs, F.; Gheysens, O.; Van Veldhoven, P.P.; De Geest, B. Coconut Oil Aggravates Pressure Overload-Induced Cardiomyopathy without Inducing Obesity, Systemic Insulin Resistance, or Cardiac Steatosis. Int. J. Mol. Sci. 2017, 18, 1565. [Google Scholar] [CrossRef] [PubMed]
- Aboumsallem, J.P.; Mishra, M.; Amin, R.; Muthuramu, I.; Kempen, H.; De Geest, B. Successful treatment of established heart failure in mice with recombinant HDL (Milano). Br. J. Pharmacol. 2018, 175, 4167–4182. [Google Scholar] [CrossRef]
- Van Linthout, S.; Collen, D.; De Geest, B. Effect of promoters and enhancers on expression, transgene DNA persistence, and hepatotoxicity after adenoviral gene transfer of human apolipoprotein A-I. Hum. Gene Ther. 2002, 13, 829–840. [Google Scholar] [CrossRef]
- Amin, R.; Muthuramu, I.; Aboumsallem, J.P.; Mishra, M.; Jacobs, F.; De Geest, B. Selective HDL-Raising Human Apo A-I Gene Therapy Counteracts Cardiac Hypertrophy, Reduces Myocardial Fibrosis, and Improves Cardiac Function in Mice with Chronic Pressure Overload. Int. J. Mol. Sci. 2017, 18, 2012. [Google Scholar] [CrossRef]
- Junqueira, L.C.; Bignolas, G.; Brentani, R.R. Picrosirius staining plus polarization microscopy, a specific method for collagen detection in tissue sections. Histochem. J. 1979, 11, 447–455. [Google Scholar] [CrossRef]
- Van Craeyveld, E.; Jacobs, F.; Gordts, S.C.; De Geest, B. Low-density lipoprotein receptor gene transfer in hypercholesterolemic mice improves cardiac function after myocardial infarction. Gene Ther. 2012, 19, 860–871. [Google Scholar] [CrossRef] [PubMed]
- Gordts, S.C.; Muthuramu, I.; Nefyodova, E.; Jacobs, F.; Van Craeyveld, E.; De Geest, B. Beneficial effects of selective HDL-raising gene transfer on survival, cardiac remodelling and cardiac function after myocardial infarction in mice. Gene Ther. 2013, 20, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Maeda, N.; Johnson, L.; Kim, S.; Hagaman, J.; Friedman, M.; Reddick, R. Anatomical differences and atherosclerosis in apolipoprotein E-deficient mice with 129/SvEv and C57BL/6 genetic backgrounds. Atherosclerosis 2007, 195, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, Y.; Plump, A.S.; Raines, E.W.; Breslow, J.L.; Ross, R. ApoE-deficient mice develop lesions of all phases of atherosclerosis throughout the arterial tree. Arter. Thromb. 1994, 14, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Van Craeyveld, E.; Gordts, S.C.; Nefyodova, E.; Jacobs, F.; De Geest, B. Regression and stabilization of advanced murine atherosclerotic lesions: A comparison of LDL lowering and HDL raising gene transfer strategies. J. Mol. Med. 2011, 89, 555–567. [Google Scholar] [CrossRef]
- Van Craeyveld, E.; Gordts, S.C.; Singh, N.; Jacobs, F.; De Geest, B. A critical reassessment of murine and rabbit models of atherosclerosis: Focus on lesion progression and remodelling. Acta Cardiol. 2012, 67, 11–21. [Google Scholar] [CrossRef]
- Fernandez, B.; Duran, A.C.; Fernandez, M.C.; Fernandez-Gallego, T.; Icardo, J.M.; Sans-Coma, V. The coronary arteries of the C57BL/6 mouse strains: Implications for comparison with mutant models. J. Anat. 2008, 212, 12–18. [Google Scholar] [CrossRef]
- Kumar, D.; Hacker, T.A.; Buck, J.; Whitesell, L.F.; Kaji, E.H.; Douglas, P.S.; Kamp, T.J. Distinct mouse coronary anatomy and myocardial infarction consequent to ligation. Coron Artery Dis. 2005, 16, 41–44. [Google Scholar] [CrossRef]
- Klosinska, D.; Ciszek, B.; Majchrzak, B.; Badurek, I.; Ratajska, A. Diversity of coronary arterial tree in laboratory mice. Folia Morphol. 2020, 79, 255–264. [Google Scholar] [CrossRef]
- Scher, A.M. Absence of atherosclerosis in human intramyocardial coronary arteries: A neglected phenomenon. Atherosclerosis 2000, 149, 1–3. [Google Scholar] [CrossRef]
- Nakaura, T.; Nagayoshi, Y.; Awai, K.; Utsunomiya, D.; Kawano, H.; Ogawa, H.; Yamashita, Y. Myocardial bridging is associated with coronary atherosclerosis in the segment proximal to the site of bridging. J. Cardiol. 2014, 63, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Botta, D.M., Jr.; Elefteriades, J.A. Why are the intramyocardial portions of the coronary arteries spared from arteriosclerosis? Clinical implications. Int. J. Angiol. 2009, 18, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Nishimiya, K.; Matsumoto, Y.; Wang, H.; Piao, Z.; Ohyama, K.; Uzuka, H.; Hao, K.; Tsuburaya, R.; Takahashi, J.; Ito, K.; et al. Absence of adventitial vasa vasorum formation at the coronary segment with myocardial bridge—An optical coherence tomography study. Int. J. Cardiol. 2018, 250, 275–277. [Google Scholar] [CrossRef] [PubMed]
- Muller, O.; Pyxaras, S.A.; Trana, C.; Mangiacapra, F.; Barbato, E.; Wijns, W.; Taylor, C.A.; De Bruyne, B. Pressure-diameter relationship in human coronary arteries. Circ. Cardiovasc. Interv. 2012, 5, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Henrichs, K.J.; Unger, T.; Berecek, K.H.; Ganten, D. Is arterial media hypertrophy in spontaneously hypertensive rats a consequence of or a cause for hypertension? Clin. Sci. 1980, 59, 331s–333s. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, S.H.; Reddick, R.L.; Piedrahita, J.A.; Maeda, N. Spontaneous hypercholesterolemia and arterial lesions in mice lacking apolipoprotein E. Science 1992, 258, 468–471. [Google Scholar] [CrossRef]
- Plump, A.S.; Smith, J.D.; Hayek, T.; Aalto-Setala, K.; Walsh, A.; Verstuyft, J.G.; Rubin, E.M.; Breslow, J.L. Severe hypercholesterolemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell 1992, 71, 343–353. [Google Scholar] [CrossRef]
- De Geest, B.; Zhao, Z.; Collen, D.; Holvoet, P. Effects of adenovirus-mediated human apo A-I gene transfer on neointima formation after endothelial denudation in apo E-deficient mice. Circulation 1997, 96, 4349–4356. [Google Scholar] [CrossRef]
- Ishibashi, S.; Brown, M.S.; Goldstein, J.L.; Gerard, R.D.; Hammer, R.E.; Herz, J. Hypercholesterolemia in low density lipoprotein receptor knockout mice and its reversal by adenovirus-mediated gene delivery. J. Clin. Investig. 1993, 92, 883–893. [Google Scholar] [CrossRef]
- Jacobs, F.; Van Craeyveld, E.; Feng, Y.; Snoeys, J.; De Geest, B. Adenoviral low density lipoprotein receptor attenuates progression of atherosclerosis and decreases tissue cholesterol levels in a murine model of familial hypercholesterolemia. Atherosclerosis 2008, 201, 289–297. [Google Scholar] [CrossRef]
- Ishibashi, S.; Herz, J.; Maeda, N.; Goldstein, J.L.; Brown, M.S. The two-receptor model of lipoprotein clearance: Tests of the hypothesis in “knockout” mice lacking the low density lipoprotein receptor, apolipoprotein E, or both proteins. Proc. Natl. Acad. Sci. USA 1994, 91, 4431–4435. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P.H.; Khokhar, A.A.; Massaro, J.M.; Lirette, S.T.; Griswold, M.E.; Martin, S.S.; Blaha, M.J.; Kulkarni, K.R.; Correa, A.; D’Agostino, R.B., Sr.; et al. Remnant Lipoprotein Cholesterol and Incident Coronary Heart Disease: The Jackson Heart and Framingham Offspring Cohort Studies. J. Am. Heart Assoc. 2016, 5, e002765. [Google Scholar] [CrossRef] [PubMed]
- Getz, G.S.; Reardon, C.A. Apoprotein E as a lipid transport and signaling protein in the blood, liver, and artery wall. J. Lipid Res. 2009, 50, S156–S161. [Google Scholar] [CrossRef] [PubMed]
- Wortmann, M.; Arshad, M.; Peters, A.S.; Hakimi, M.; Bockler, D.; Dihlmann, S. The C57Bl/6J mouse strain is more susceptible to angiotensin II-induced aortic aneurysm formation than C57Bl/6N. Atherosclerosis 2021, 318, 8–13. [Google Scholar] [CrossRef]
- Williams, J.L.; Paudyal, A.; Awad, S.; Nicholson, J.; Grzesik, D.; Botta, J.; Meimaridou, E.; Maharaj, A.V.; Stewart, M.; Tinker, A.; et al. Mylk3 null C57BL/6N mice develop cardiomyopathy, whereas Nnt null C57BL/6J mice do not. Life Sci. Alliance 2020, 3, e201900593. [Google Scholar] [CrossRef]
- Ronchi, J.A.; Figueira, T.R.; Ravagnani, F.G.; Oliveira, H.C.; Vercesi, A.E.; Castilho, R.F. A spontaneous mutation in the nicotinamide nucleotide transhydrogenase gene of C57BL/6J mice results in mitochondrial redox abnormalities. Free Radic. Biol. Med. 2013, 63, 446–456. [Google Scholar] [CrossRef]
- Mekada, K.; Abe, K.; Murakami, A.; Nakamura, S.; Nakata, H.; Moriwaki, K.; Obata, Y.; Yoshiki, A. Genetic differences among C57BL/6 substrains. Exp. Anim. 2009, 58, 141–149. [Google Scholar] [CrossRef]
- Vozenilek, A.E.; Vetkoetter, M.; Green, J.M.; Shen, X.; Traylor, J.G.; Klein, R.L.; Orr, A.W.; Woolard, M.D.; Krzywanski, D.M. Absence of Nicotinamide Nucleotide Transhydrogenase in C57BL/6J Mice Exacerbates Experimental Atherosclerosis. J. Vasc. Res. 2018, 55, 98–110. [Google Scholar] [CrossRef]
- Kang, B.Y.; Wang, W.; Palade, P.; Sharma, S.G.; Mehta, J.L. Cardiac hypertrophy during hypercholesterolemia and its amelioration with rosuvastatin and amlodipine. J. Cardiovasc Pharmacol. 2009, 54, 327–334. [Google Scholar] [CrossRef]
- Csont, T.; Bereczki, E.; Bencsik, P.; Fodor, G.; Gorbe, A.; Zvara, A.; Csonka, C.; Puskas, L.G.; Santha, M.; Ferdinandy, P. Hypercholesterolemia increases myocardial oxidative and nitrosative stress thereby leading to cardiac dysfunction in apoB-100 transgenic mice. Cardiovasc. Res. 2007, 76, 100–109. [Google Scholar] [CrossRef]
- Gordts, S.C.; Van Craeyveld, E.; Muthuramu, I.; Singh, N.; Jacobs, F.; De Geest, B. Lipid Lowering and HDL Raising Gene Transfer Increase Endothelial Progenitor Cells, Enhance Myocardial Vascularity, and Improve Diastolic Function. PLoS ONE 2012, 7, e46849. [Google Scholar] [CrossRef]
- Maczewski, M.; Maczewska, J. Hypercholesterolemia exacerbates ventricular remodeling in the rat model of myocardial infarction. J. Card. Fail. 2006, 12, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Aboumsallem, J.P.; Muthuramu, I.; Mishra, M.; De Geest, B. Cholesterol-Lowering Gene Therapy Prevents Heart Failure with Preserved Ejection Fraction in Obese Type 2 Diabetic Mice. Int. J. Mol. Sci. 2019, 20, 2222. [Google Scholar] [CrossRef] [PubMed]
- Csonka, C.; Sarkozy, M.; Pipicz, M.; Dux, L.; Csont, T. Modulation of Hypercholesterolemia-Induced Oxidative/Nitrative Stress in the Heart. Oxidative Med. Cell. Longev. 2016, 2016, 3863726. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.S.; Li, T.D.; Zeng, Z.H. Mechanisms underlying direct actions of hyperlipidemia on myocardium: An updated review. Lipids Health Dis. 2020, 19, 23. [Google Scholar] [CrossRef] [PubMed]
- De Geest, B.; Mishra, M. Role of Oxidative Stress in Heart Failure: Insights from Gene Transfer Studies. Biomedicines 2021, 9, 1645. [Google Scholar] [CrossRef]
- Glazer, H.P.; Osipov, R.M.; Clements, R.T.; Sellke, F.W.; Bianchi, C. Hypercholesterolemia is associated with hyperactive cardiac mTORC1 and mTORC2 signaling. Cell Cycle 2009, 8, 1738–1746. [Google Scholar] [CrossRef]
- Sciarretta, S.; Volpe, M.; Sadoshima, J. Mammalian target of rapamycin signaling in cardiac physiology and disease. Circ. Res. 2014, 114, 549–564. [Google Scholar] [CrossRef]
- Tenger, C.; Zhou, X. Apolipoprotein E modulates immune activation by acting on the antigen-presenting cell. Immunology 2003, 109, 392–397. [Google Scholar] [CrossRef]
- Ali, K.; Middleton, M.; Pure, E.; Rader, D.J. Apolipoprotein E suppresses the type I inflammatory response in vivo. Circ. Res. 2005, 97, 922–927. [Google Scholar] [CrossRef]
- Kawamura, A.; Baitsch, D.; Telgmann, R.; Feuerborn, R.; Weissen-Plenz, G.; Hagedorn, C.; Saku, K.; Brand-Herrmann, S.M.; von Eckardstein, A.; Assmann, G.; et al. Apolipoprotein E interrupts interleukin-1beta signaling in vascular smooth muscle cells. Arter. Thromb. Vasc. Biol. 2007, 27, 1610–1617. [Google Scholar] [CrossRef] [PubMed]
- Reina-Couto, M.; Pereira-Terra, P.; Quelhas-Santos, J.; Silva-Pereira, C.; Albino-Teixeira, A.; Sousa, T. Inflammation in Human Heart Failure: Major Mediators and Therapeutic Targets. Front. Physiol. 2021, 12, 746494. [Google Scholar] [CrossRef] [PubMed]
- Munzel, T.; Gori, T.; Keaney, J.F., Jr.; Maack, C.; Daiber, A. Pathophysiological role of oxidative stress in systolic and diastolic heart failure and its therapeutic implications. Eur. Heart J. 2015, 36, 2555–2564. [Google Scholar] [CrossRef] [PubMed]
- Goonasekara, C.L.; Balse, E.; Hatem, S.; Steele, D.F.; Fedida, D. Cholesterol and cardiac arrhythmias. Expert Rev. Cardiovasc. Ther. 2010, 8, 965–979. [Google Scholar] [CrossRef] [PubMed]
- Balycheva, M.; Faggian, G.; Glukhov, A.V.; Gorelik, J. Microdomain-specific localization of functional ion channels in cardiomyocytes: An emerging concept of local regulation and remodelling. Biophys. Rev. 2015, 7, 43–62. [Google Scholar] [CrossRef]





| C57BL/6N(n = 5) | C57BL/6N Apo E−/−(n = 10) | |
|---|---|---|
| Total cholesterol | 68.3 ± 2.4 | 296 ± 4 **** |
| Non-HDL cholesterol | 9.39 ± 1.50 | 274 ± 4 **** |
| HDL cholesterol | 58.9 ± 1.2 | 22.5 ± 1.3 **** |
| C57BL/6N Sham (n = 9) | C57BL/6N Apo E−/− Sham (n = 12) | C57BL/6N TAC (n = 16) | C57BL/6N Apo E−/−TAC (n = 24) | |
|---|---|---|---|---|
| EEL area (µm2) | 30,000 ± 1800 | 29,300 ± 2700 | 65,600 ± 4900 §§§ | 67,800 ± 8100 !!! |
| IEL area (µm2) | 17,800 ± 400 | 18,500 ± 1700 | 44,400 ± 3700 §§§ | 43,200 ± 5400 !! |
| Media area (µm2) | 12,200 ± 1600 | 10,800 ± 1100 | 21,200 ± 2100 §§ | 24,700 ± 2800 !!! |
| Intima area (µm2) | 0.00 ± 0.01 | 0.00 ± 0.01 | 0.00 ± 0.01 | 11,300 ±3500 !!!*** |
| Luminal area (µm2) | 17,800 ± 400 | 18,500 ± 1700 | 44,400 ± 3700 §§§ | 31,900 ± 3200 !!* |
| Stenosis (%) | 0.00 ± 0.01 | 0.00 ± 0.01 | 0.00 ± 0.01 | 18.9 ± 4.1 !!!*** |
| C57BL/6N Sham (n = 19) | C57BL/6N Apo E−/− Sham (n = 8) | |
|---|---|---|
| LEFT VENTRICLE | ||
| Peak systolic pressure (mm Hg) | 101 ± 2 | 96.1 ± 1.3 |
| End-diastolic pressure (mm Hg) | 0.677 ± 0.475 | 1.96 ± 0.46 |
| dP/dtmax (mm Hg/ms) | 12.1 ± 0.4 | 10.5 ± 0.7 |
| dP/dtmin (mm Hg/ms) | −9.91 ± 0.35 | −8.61 ± 0.42° |
| Tau (ms) | 4.42 ± 0.13 | 4.94 ± 0.15° |
| Heart rate (bpm) | 609 ± 13 | 599 ± 12 |
| AORTA | ||
| Mean pressure (mm Hg) | 81.6 ± 1.7 | 79.5 ± 2.0 |
| Systolic pressure (mm Hg) | 99.4 ± 1.7 | 95.6 ± 1.8 |
| Diastolic pressure (mm Hg) | 64.4 ± 2.6 | 64.1 ± 4.7 |
| C57BL/6N TAC (n = 18) | C57BL/6N Apo E−/− TAC (n = 21) | |
|---|---|---|
| LEFT VENTRICLE | ||
| Peak systolic pressure (mm Hg) | 173 ± 9 | 183 ± 8 |
| End-diastolic pressure (mm Hg) | 2.29 ± 0.90 | 6.32 ± 1.04 * |
| dP/dtmax (mm Hg/ms) | 11.9 ± 0.8 | 11.9 ± 0.7 |
| dP/dtmin (mm Hg/ms) | −11.5 ± 0.6 | −11.3 ± 0.7 |
| Tau (ms) | 5.10 ± 0.23 | 6.00 ± 0.33 * |
| Heart rate (bpm) | 631 ± 11 | 601 ± 12 |
| AORTA | ||
| Mean pressure (mm Hg) | 104 ± 6 | 109 ± 4 |
| Systolic pressure (mm Hg) | 172 ± 11 | 183 ± 8 |
| Diastolic pressure (mm Hg) | 61.8 ± 5.5 | 65.1 ± 5.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muthuramu, I.; Mishra, M.; De Geest, B. Increased Remnant Lipoproteins in Apo E Deficient Mice Induce Coronary Atherosclerosis following Transverse Aortic Constriction and Aggravate the Development of Pressure Overload-Induced Cardiac Hypertrophy and Heart Failure. Biomedicines 2022, 10, 1592. https://doi.org/10.3390/biomedicines10071592
Muthuramu I, Mishra M, De Geest B. Increased Remnant Lipoproteins in Apo E Deficient Mice Induce Coronary Atherosclerosis following Transverse Aortic Constriction and Aggravate the Development of Pressure Overload-Induced Cardiac Hypertrophy and Heart Failure. Biomedicines. 2022; 10(7):1592. https://doi.org/10.3390/biomedicines10071592
Chicago/Turabian StyleMuthuramu, Ilayaraja, Mudit Mishra, and Bart De Geest. 2022. "Increased Remnant Lipoproteins in Apo E Deficient Mice Induce Coronary Atherosclerosis following Transverse Aortic Constriction and Aggravate the Development of Pressure Overload-Induced Cardiac Hypertrophy and Heart Failure" Biomedicines 10, no. 7: 1592. https://doi.org/10.3390/biomedicines10071592
APA StyleMuthuramu, I., Mishra, M., & De Geest, B. (2022). Increased Remnant Lipoproteins in Apo E Deficient Mice Induce Coronary Atherosclerosis following Transverse Aortic Constriction and Aggravate the Development of Pressure Overload-Induced Cardiac Hypertrophy and Heart Failure. Biomedicines, 10(7), 1592. https://doi.org/10.3390/biomedicines10071592







