Human Angiotensin I-Converting Enzyme Produced by Different Cells: Classification of the SERS Spectra with Linear Discriminant Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. ACE Isolation from Different Sources
2.2. ACE Characterization
2.3. SERS Substrate Fabrication and Characterization
2.4. SERS Measurements
2.5. Preprocessing Spectra for LDA
2.6. ACE Classification with LDA
3. Results and Discussion
3.1. SERS Substrate Characterization
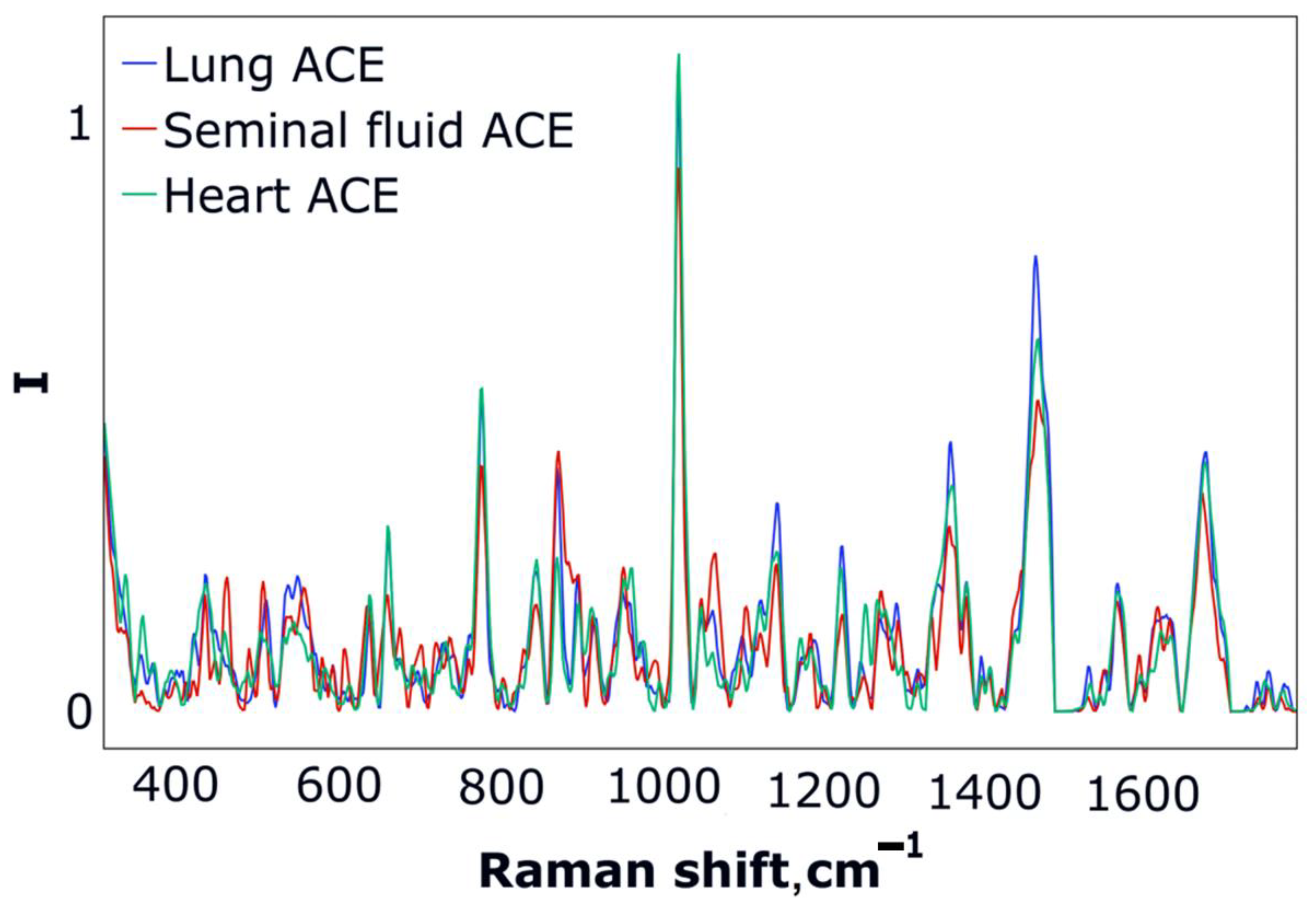
3.2. ACE SERS Measurement
3.3. ACE Classification with LDA
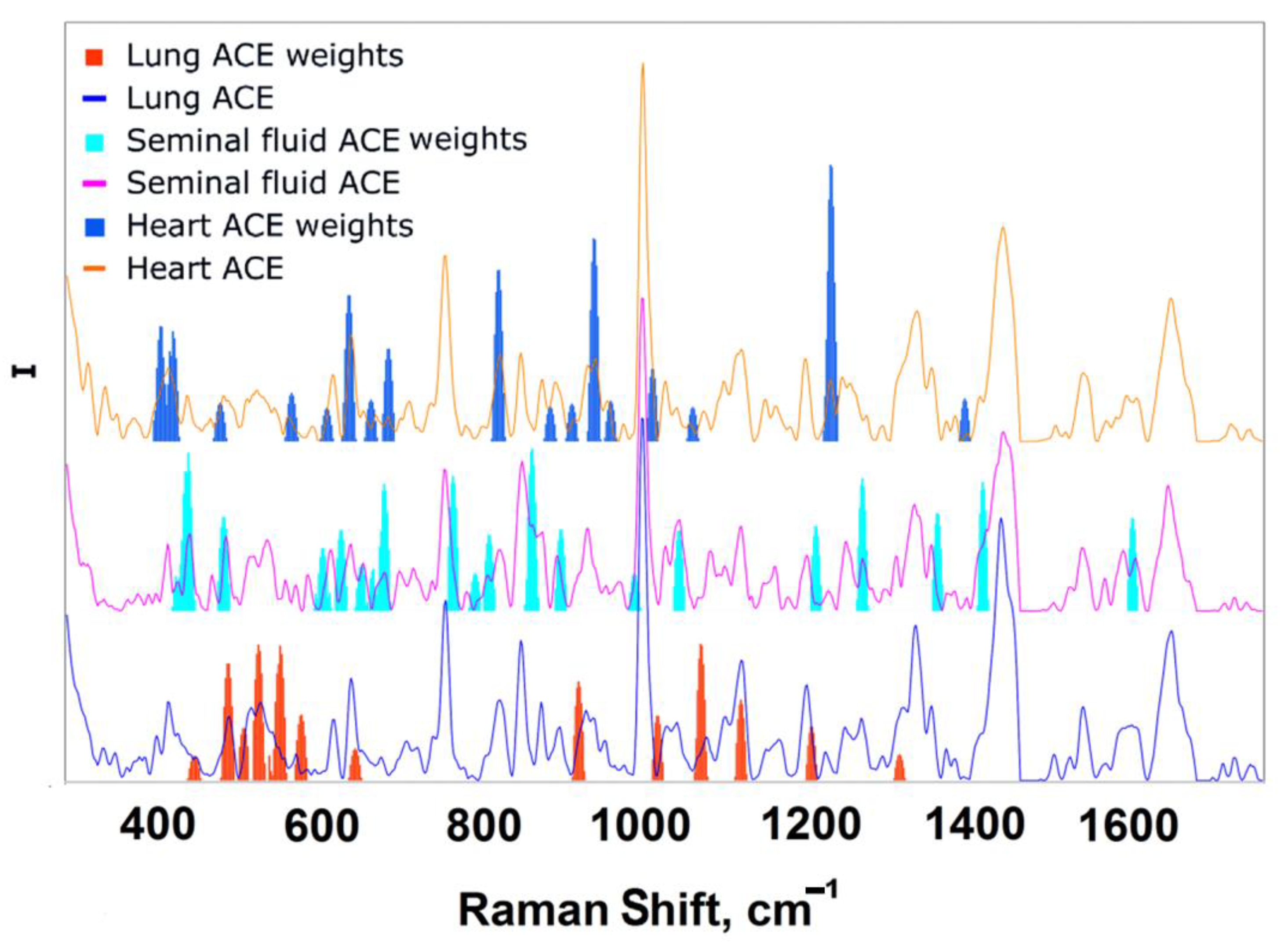
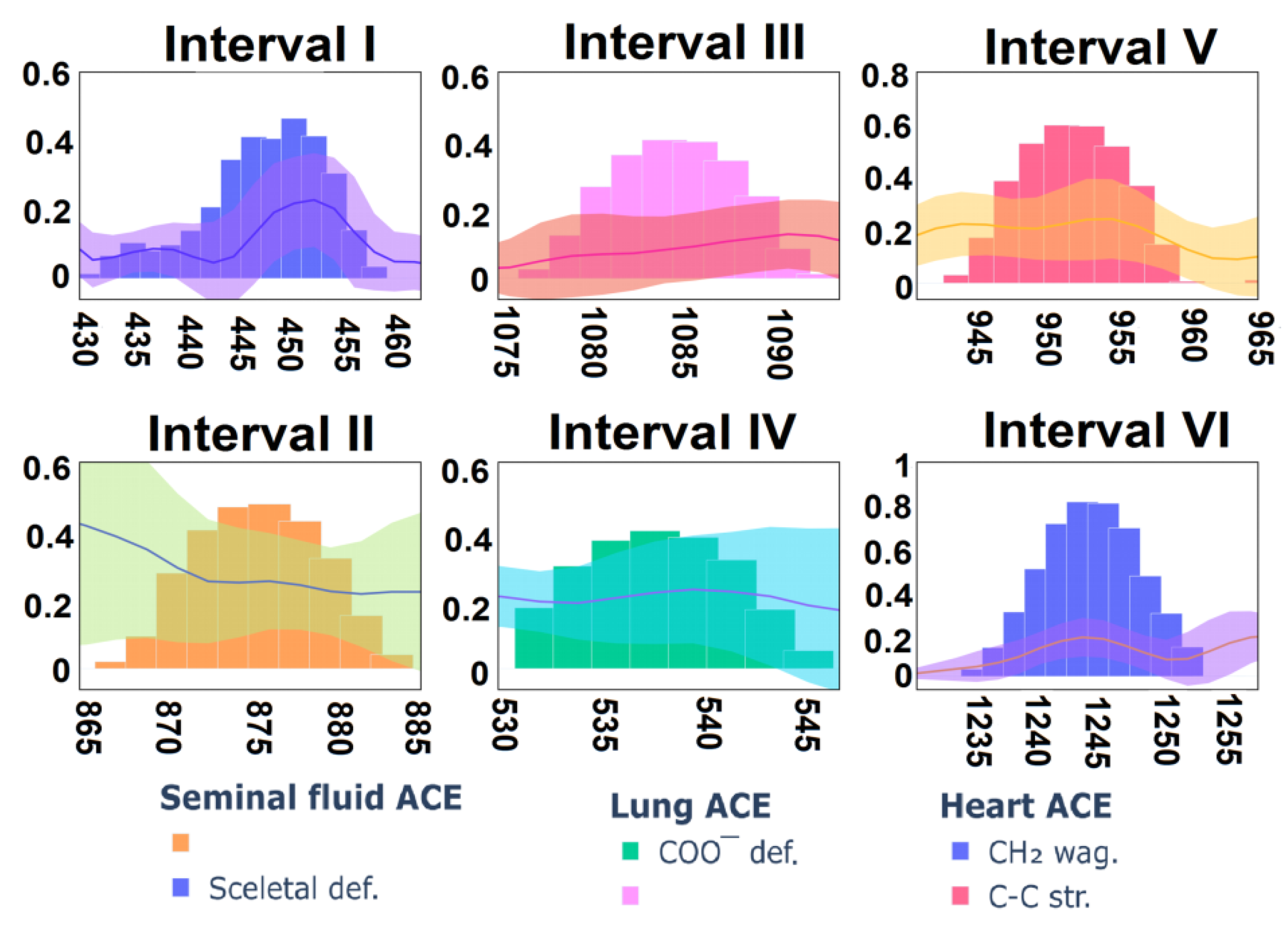
3.4. Analysis
- Contribution value (importance) to the separation exceeds 95% quantile in absolute value;
- Contributions at the boundaries of the studied range were not taken into account in the analysis.
- The values of the bar plot were sorted in descending order;
- The first band was taken and the forecast was based on it;
- The mean value of the target metric with its standard deviation on random train and test sampling from X was recorded;
- The process continued until convergence.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Krafft, C.; Sergo, V. Biomedical applications of Raman and infrared spectroscopy to diagnose tissues. Spectroscopy 2006, 20, 195–218. [Google Scholar] [CrossRef]
- Krafft, C.; Popp, J. The many facets of Raman spectroscopy for biomedical analysis. Anal. Bioanal. Chem. 2015, 407, 699–717. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, M.; Hendra, P.J.; McQuillan, A.J. Raman spectra of pyridine adsorbed at a silver electrode. Chem. Phys. Lett. 1974, 26, 163–166. [Google Scholar] [CrossRef]
- Boginskaya, I.; Sedova, M.; Baburin, A.; Afanas’ev, K.; Zverev, A.; Echeistov, V.; Ryzhkov, V.; Rodionov, I.; Tonanaiskii, B.; Ryzhikov, I.; et al. SERS-active substrates nanoengineering based on e-beam evaporated self-assembled silver films. Appl. Sci. 2019, 9, 3988. [Google Scholar] [CrossRef]
- Drachev, V.P.; Nashine, V.C.; Thoreson, M.D.; Ben-Amotz, D.; Jo Davisson, V.; Shalaev, V.M. Adaptive silver films for detection of antibody-antigen binding. Langmuir 2005, 21, 8368–8373. [Google Scholar] [CrossRef]
- Guo, L.; Cao, H.; Cao, L.; Yang, Y.; Wang, M. SERS study of wheat leaves substrates with two different structures. Opt. Commun. 2022, 510, 127921. [Google Scholar] [CrossRef]
- Drachev, V.P.; Thoreson, M.D.; Khaliullin, E.N.; Davisson, V.J.; Shalaev, V.M. Surface-enhanced Raman difference between human insulin and insulin lispro detected with adaptive nanostructures. J. Phys. Chem. B 2004, 108, 18046–18052. [Google Scholar] [CrossRef]
- Nechaeva, N.L.; Boginskaya, I.A.; Ivanov, A.V.; Sarychev, A.K.; Eremenko, A.V.; Ryzhikov, I.A.; Lagarkov, A.N.; Kurochkin, I.N. Multiscale flaked silver SERS-substrate for glycated human albumin biosensing. Anal. Chim. Acta 2020, 1100, 250–257. [Google Scholar] [CrossRef]
- Nechaeva, N.; Prokopkina, T.; Makhaeva, G.; Rudakova, E.; Boltneva, N.; Dishovsky, C.; Eremenko, A.; Kurochkin, I. Quantitative butyrylcholinesterase activity detection by surface-enhanced Raman spectroscopy. Sens. Actuators B Chem. 2018, 259, 75–82. [Google Scholar] [CrossRef]
- Ma, H.; Tang, X.; Liu, Y.; Han, X.X.; He, C.; Lu, H.; Zhao, B. Surface-enhanced Raman scattering for direct protein function investigation: Controlled immobilization and orientation. Anal. Chem. 2019, 91, 8767–8771. [Google Scholar] [CrossRef]
- Das, G.; Gentile, F.; Coluccio, M.L.; Perri, A.M.; Nicastri, A.; Mecarini, F.; Cojoc, G.; Candeloro, P.; Liberale, C.; De Angelis, F.; et al. Principal component analysis based methodology to distinguish protein SERS spectra. J. Mol. Struct. 2011, 993, 500–505. [Google Scholar] [CrossRef]
- Ding, S.-Y.; You, E.-M.; Tian, Z.-Q.; Moskovits, M. Electromagnetic theories of surface-enhanced Raman spectroscopy. Chem. Soc. Rev. 2017, 46, 4042–4076. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Guo, L.; Sun, Z.; Jiao, T.; Wang, M. Surface-enhanced fluorescence and application study based on Ag-wheat leaves. Chin. Phys. B 2022, 31, 037803. [Google Scholar] [CrossRef]
- Hao, Z.; Li, N.; Cao, H.; Guo, L.; Cao, H.; Cao, L.; Liu, H.L.; Jiao, T.; Wang, M. Modified Ag nanoparticles on the regular array structure to improve the optical properties. J. Lumin. 2022, 243, 118684. [Google Scholar] [CrossRef]
- Kong, X.; Yu, Q.; Zhang, X.; Du, X.; Gong, H.; Jiang, H. Synthesis and application of surface enhanced Raman scattering (SERS) tags of Ag@SiO2 core/shell nanoparticles in protein detection. J. Mater. Chem. 2012, 22, 7767–7774. [Google Scholar] [CrossRef]
- Feliu, N.; Hassan, M.; Garcia Rico, E.; Cui, D.; Parak, W.; Alvarez-Puebla, R. SERS quantification and characterization of proteins and other biomolecules. Langmuir 2017, 33, 9711–9730. [Google Scholar] [CrossRef]
- Boginskaya, I.; Nechaeva, N.; Tikhomirova, V.; Kryukova, O.; Evdokimov, V.; Bulaeva, N.; Golukhova, E.; Ryzhikov, I.; Kost, O.; Afanasev, K.; et al. Human angiotensin I-converting enzyme study by surface-enhanced Raman spectroscopy. J. Raman Spectrosc. 2021, 52, 1529–1539. [Google Scholar] [CrossRef]
- Varki, A. Biological roles of glycans. Glycobiology 2017, 27, 3–49. [Google Scholar] [CrossRef]
- Fuster, M.M.; Esko, J.D. The sweet and sour of cancer: Glycans as novel therapeutic targets. Nat. Rev. Cancer 2005, 5, 526–542. [Google Scholar] [CrossRef]
- Wang, Y.N.; Lee, H.H.; Hsu, J.L.; Yu, D.; Hung, M.C. The impact of PD-L1 N-linked glycosylation on cancer therapy and clinical diagnosis. J. Biomed. Sci. 2020, 27, 77. [Google Scholar] [CrossRef]
- West, M.B.; Segu, Z.M.; Feasley, C.L.; Kang, P.; Klouckova, I.; Li, C.; Novotny, M.V.; West, C.M.; Mechref, Y.; Hanigan, M.H. Analysis of site-specific glycosylation of renal and hepatic γ-glutamyl transpeptidase from normal human tissue. J. Biol. Chem. 2010, 285, 29511–29524. [Google Scholar] [CrossRef] [PubMed]
- Ikegami, K.; Liao, X.; Hoshino, Y.; Ono, H.; Ota, W.; Ito, Y.; Yamada, M.; Murata, Y.; Refetoff, S.; Yoshimura, T. Tissue-specific post-translational modification allows functional targeting of thyrotropin. Cell Rep. 2014, 9, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Boyce, A.K.J.; Epp, A.L.; Nagarajan, A.; Swayne, L.A. Transcriptional and post-translational regulation of pannexins. Biochim. Biophys. Acta Biomembr. 2018, 1860, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Brogren, H.; Sihlbom, C.; Wallmark, K.; Lönn, M.; Deinum, J.; Karlsson, L.; Jern, S. Heterogeneous glycosylation patterns of human PAI-1 may reveal its cellular origin. Thromb. Res. 2008, 122, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, K.E.; Ong, F.S.; Blackwell, W.-L.B.; Shah, K.H.; Giani, J.F.; Gonzalez-Villalobos, R.A.; Shen, X.Z.; Fuchs, S.; Touyz, R.M. A modern understanding of the traditional and nontraditional biological functions of angiotensin-converting enzyme. Pharmacol. Rev. 2013, 65, 1–46. [Google Scholar] [CrossRef] [PubMed]
- Khurana, V.; Goswami, B. Angiotensin converting enzyme (ACE). Clin. Chim. Acta 2022, 524, 113–122. [Google Scholar] [CrossRef]
- Beneteau-Burnat, B.; Baudin, B. Angiotensin-converting enzyme: Clinical applications and laboratory investigations on serum and other biological fluids. Crit. Rev. Clin. Lab. Sci. 1991, 28, 337–356. [Google Scholar] [CrossRef]
- Danilov, S.M.; Balyasnikova, I.V.; Danilova, A.S.; Naperova, I.A.; Arablinskaya, N.E.; Borisov, S.E.; Metzger, R.; Franke, F.E.; Schwartz, D.E.; Gachok, I.V.; et al. Conformational fingerprinting of the angiotensin I-converting enzyme (ACE). 1. Application in sarcoidosis. J. Proteome Res. 2010, 9, 5782–5793. [Google Scholar] [CrossRef]
- Petrov, M.N.; Shilo, V.Y.; Tarasov, A.V.; Schwartz, D.E.; Garcia, J.G.N.; Kost, O.A.; Danilov, S.M. Conformational changes of blood ACE in chronic uremia. PLoS ONE 2012, 7, 32–36. [Google Scholar] [CrossRef][Green Version]
- Danilov, S.M.; Tikhomirova, V.E.; Metzger, R.; Naperova, I.A.; Bukina, T.M.; Goker-Alpan, O.; Tayebi, N.; Gayfullin, N.M.; Schwartz, D.E.; Samokhodskaya, L.M.; et al. ACE phenotyping in Gaucher disease. Mol. Genet. Metab. 2018, 123, 501–510. [Google Scholar] [CrossRef]
- Orth, T.; Voronov, S.; Binevski, P.; Saenger, W.; Kost, O. Glycosylation of bovine pulmonary angiotensin-converting enzyme modulates its catalytic properties. FEBS Lett. 1998, 431, 255–258. [Google Scholar] [CrossRef]
- Kryukova, O.V.; Tikhomirova, V.E.; Golukhova, E.Z.; Evdokimov, V.V.; Kalantarov, G.F.; Trakht, I.N.; Schwartz, D.E.; Dull, R.O.; Gusakov, A.V.; Uporov, I.V.; et al. Tissue specificity of human angiotensin I-converting enzyme. PLoS ONE 2015, 10, e0143455. [Google Scholar] [CrossRef][Green Version]
- Tikhomirova, V.E.; Kost, O.A.; Kryukova, O.V.; Golukhova, E.Z.; Bulaeva, N.I.; Zholbaeva, A.Z.; Bokeria, L.A.; Garcia, J.G.N.; Danilov, S.M. ACE phenotyping in human heart. PLoS ONE 2017, 12, e0181976. [Google Scholar] [CrossRef] [PubMed]
- Kost, O.A.; Tikhomirova, V.E.; Kryukova, O.V.; Gusakov, A.V.; Bulaeva, N.I.; Evdokimov, V.V.; Golukhova, E.Z.; Danilov, S.M. Conformational “fingerprint” of the angiotensin-converting enzyme. Russ. J. Bioorganic Chem. 2018, 44, 48–60. [Google Scholar] [CrossRef]
- Soubrier, F.; Alhenc-gelas, F.O.I.S.; Hubert, C.; Allegrini, J.; Johnt, M.; Tregeart, G.; Corvol, P. Two putative active centers in human angiotensin I-converting enzyme revealed by molecular cloning. Proc. Natl. Acad. Sci. USA 1988, 85, 9386–9390. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.C.; Sturrock, E.D.; Wu, Z.; Biemann, K.; Ehlers, M.R.; Riordan, J.F. Identification of N-linked glycosylation sites in human testis angiotensin-converting enzyme and expression of an active deglycosylated form. J. Biol. Chem. 1997, 272, 3511–3519. [Google Scholar] [CrossRef] [PubMed]
- Ripka, J.E.; Ryan, J.W.; Valido, F.A.; Chung, A.Y.K.; Peterson, C.M.; Urry, R.L. N-glycosylation of forms of angiotensin converting enzyme from four mammalian species. Biochem. Biophys. Res. Commun. 1993, 196, 503–508. [Google Scholar] [CrossRef]
- Liu, T.; Qian, W.-J.; Gritsenko, M.A.; Camp, D.G.; Monroe, M.E.; Moore, R.J.; Smith, R.D. Human plasma N-glycoproteome analysis by immunoaffinity subtraction, hydrazide chemistry, and mass spectrometry. J. Proteome Res. 2005, 4, 2070–2080. [Google Scholar] [CrossRef]
- Danilov, S.M.; Tikhomirova, V.E.; Kryukova, O.V.; Balatsky, A.V.; Bulaeva, N.I.; Golukhova, E.Z.; Bokeria, L.A.; Samokhodskaya, L.M.; Kost, O.A. Conformational fingerprint of blood and tissue ACEs: Personalized approach. PLoS ONE 2018, 13, e0209861. [Google Scholar] [CrossRef]
- Lussier, F.; Thibault, V.; Charron, B.; Wallace, G.Q.; Masson, J.F. Deep learning and artificial intelligence methods for Raman and surface-enhanced Raman scattering. TrAC Trends Anal. Chem. 2020, 124, 115796. [Google Scholar] [CrossRef]
- Cheng, W.C.; Chen, L.H.; Jiang, C.R.; Deng, Y.M.; Wang, D.W.; Lin, C.H.; Jou, R.; Wang, J.K.; Wang, Y.L. Sensible functional linear discriminant analysis effectively discriminates enhanced Raman spectra of mycobacterium species. Anal. Chem. 2021, 93, 2785–2792. [Google Scholar] [CrossRef] [PubMed]
- Vance, T.; Reljin, N.; Lazarevic, A.; Pokrajac, D.; Kecman, V.; Melikechi, N.; Marcano, A.; Markushin, Y.; McDaniel, S. Classification of LIBS protein spectra using support vector machines and adaptive local hyperplanes. In Proceedings of the 2010 International Joint Conference on Neural Networks (IJCNN), Barcelona, Spain, 18–23 July 2010; pp. 1–7. [Google Scholar] [CrossRef]
- Barucci, A.; D’Andrea, C.; Farnesi, E.; Banchelli, M.; Amicucci, C.; De Angelis, M.; Hwang, B.; Matteini, P. Label-free SERS detection of proteins based on machine learning classification of chemo-structural determinants. Analyst 2021, 146, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Rinnan, Å.; van den Berg, F.; Engelsen, S.B. Review of the most common pre-processing techniques for near-infrared spectra. TrAC Trends Anal. Chem. 2009, 28, 1201–1222. [Google Scholar] [CrossRef]
- Conlin, A.K.; Martin, E.B.; Morris, A.J. Data augmentation: An alternative approach to the analysis of spectroscopic data. Chemom. Intell. Lab. Syst. 1998, 44, 161–173. [Google Scholar] [CrossRef]
- Fan, Z.; Kong, F.; Zhou, Y.; Chen, Y.; Dai, Y. Review article intelligence algorithms for protein classification by mass spectrometry. BioMed Res. Int. 2018, 2018, 2862458. [Google Scholar] [CrossRef] [PubMed]
- Gyftokostas, N.; Stefas, D.; Kokkinos, V.; Bouras, C.; Couris, S. Laser-induced breakdown spectroscopy coupled with machine learning as a tool for olive oil authenticity and geographic discrimination. Sci. Rep. 2021, 11, 5360. [Google Scholar] [CrossRef] [PubMed]
- Kost, O.A.; Bovin, N.V.; Chemodanova, E.E.; Nasonov, V.V.; Orth, T.A. New feature of angiotensin-converting enzyme: Carbohydrate-recognizing domain. J. Mol. Recognit. 2000, 13, 360–369. [Google Scholar] [CrossRef]
- Hooper, N.M.; Turner, A.J. Isolation of two differentially glycosylated forms of peptidyl-dipeptidase A (angiotensin converting enzyme) from pig brain: A re-evaluation of their role in neuropeptide metabolism. Biochem. J. 1987, 241, 625–633. [Google Scholar] [CrossRef]
- Piquilloud, Y.; Reinharz, A.; Roth, M. Studies on the angiotensin converting enzyme with different substrates. Biochim. Biophys. Acta 1970, 206, 136–142. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Arzumanyan, G.M.; Gur’ev, A.S.; Kravtsunova, D.E.; Mamatkulov, K.Z.; Marchenko, A.S.; Vereshchagin, K.A.; Volkov, A.Y. Micro Raman spectroscopy for NETosis detection. J. Raman Spectrosc. 2020, 5, 1960. [Google Scholar] [CrossRef]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar] [CrossRef]
- Durmanov, N.N.; Guliev, R.R.; Eremenko, A.V.; Boginskaya, I.A.; Ryzhikov, I.A.; Trifonova, E.A.; Putlyaev, E.V.; Mukhin, A.N.; Kalnov, S.L.; Balandina, M.V.; et al. Non-labeled selective virus detection with novel SERS-active porous silver nanofilms fabricated by electron beam physical vapor deposition. Sens. Actuators B Chem. 2018, 257, 37–47. [Google Scholar] [CrossRef]
- Rakić, A.D.; Djurišić, A.B.; Elazar, J.M.; Majewski, M.L. Optical properties of metallic films for vertical-cavity optoelectronic devices. Appl. Opt. 1998, 37, 5271–5283. [Google Scholar] [CrossRef]
- Madzharova, F.; Heiner, Z.; Kneipp, J. Surface enhanced hyper-Raman scattering of the amino acids tryptophan, histidine, phenylalanine, and tyrosine. J. Phys. Chem. C 2017, 121, 1235–1242. [Google Scholar] [CrossRef]
- Kumar, S.; Rai, A.K.; Singh, V.B.; Rai, S.B. Vibrational spectrum of glycine molecule. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2005, 61, 2741–2746. [Google Scholar] [CrossRef]
- Zhu, G.; Zhu, X.; Fan, Q.; Wan, X. Raman spectra of amino acids and their aqueous solutions. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 78, 1187–1195. [Google Scholar] [CrossRef]
- Aliaga, A.E.; Garrido, C.; Leyton, P.; Diaz, F.G.; Gomez-Jeria, J.S.; Aguayo, T.; Clavijo, E.; Campos-Vallette, M.M.; Sanchez-Cortes, S. SERS and theoretical studies of arginine. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 76, 458–463. [Google Scholar] [CrossRef]
- Stewart, S.; Fredericks, P.M. Surface-enhanced Raman spectroscopy of amino acids adsorbed on an electrochemically prepared silver surface. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 1999, 55, 1641–1660. [Google Scholar] [CrossRef]
- Aliaga, A.E.; Osorio-Roman, I.; Garrido, C.; Leyton, P.; Cárcamo, J.; Clavijo, E.; Gómez-Jeria, J.S.; Díaz, F.G.; Campos-Vallette, M.M. Surface enhanced Raman scattering study of L-lysine. Vib. Spectrosc. 2009, 50, 131–135. [Google Scholar] [CrossRef]
- Podstawka, E.; Ozaki, Y.; Proniewicz, L.M. Part II: Surface-enhanced Raman spectroscopy investigation of methionine containing heterodipeptides adsorbed on colloidal silver. Appl. Spectrosc. 2004, 58, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Podstawka, E.; Ozaki, Y.; Proniewicz, L.M. Part I: Surface-enhanced Raman spectroscopy investigation of amino acids and their homodipeptides adsorbed on colloidal silver. Appl. Spectrosc. 2004, 58, 570–580. [Google Scholar] [CrossRef] [PubMed]
- Xing, L.; Lin, K.; Zhou, X.; Liu, S.; Luo, Y. Multistate mechanism of lysozyme denaturation through synchronous analysis of Raman spectra. J. Phys. Chem. B 2016, 120, 10660–10667. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumar Rai, A.; Rai, S.B.; Rai, D.K.; Singh, A.N.; Singh, V. Infrared, Raman and electronic spectra of alanine: A comparison with ab intio calculation. J. Mol. Struct. 2006, 791, 23–29. [Google Scholar] [CrossRef]
- Stewart, S.; Fredericks, P.M. Surface-enhanced Raman spectroscopy of peptides and proteins adsorbed on an electrochemically prepared silver surface. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 1999, 55, 1615–1640. [Google Scholar] [CrossRef]
- Podstawka, E.; Ozaki, Y.; Proniewicz, L.M. Part III: Surface-enhanced Raman scattering of amino acids and their homodipeptide monolayers deposited onto colloidal gold surface. Appl. Spectrosc. 2005, 59, 1516–1526. [Google Scholar] [CrossRef]
- Nabiev, I.R.; Savchenko, V.A.; Efremov, E.S. Surface-enhanced Raman spectra of aromatic amino acids and proteins adsorbed by silver hydrosols. J. Raman Spectrosc. 1983, 14, 375–379. [Google Scholar] [CrossRef]
- Paiva, F.M.; Batista, J.C.; Rêgo, F.S.C.; Lima, J.A.; Freire, P.T.C.; Melo, F.E.A.; Mendes Filho, J.; de Menezes, A.S.; Nogueira, C.E.S. Infrared and Raman spectroscopy and DFT calculations of DL amino acids: Valine and lysine hydrochloride. J. Mol. Struct. 2017, 1127, 419–426. [Google Scholar] [CrossRef]
- Jarmelo, S.; Carey, P.R.; Fausto, R. The Raman spectra of serine and 3,3-dideutero-serine in aqueous solution. Vib. Spectrosc. 2007, 43, 104–110. [Google Scholar] [CrossRef]
- Han, X.X.; Zhao, B.; Ozaki, Y. Surface-enhanced Raman scattering for protein detection. Anal. Bioanal. Chem. 2009, 394, 1719–1727. [Google Scholar] [CrossRef] [PubMed]
- Pilot, R.; Signorini, R.; Durante, C.; Orian, L.; Bhamidipati, M.; Fabris, L. A review on surface-enhanced Raman scattering. Biosensors 2019, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Ramchandran, R.; Sen, G.C.; Misono, K.; Sen, I. Regulated cleavage-secretion of the membrane-bound angiotensin-converting enzyme. J. Biol. Chem. 1994, 269, 2125–2130. [Google Scholar] [CrossRef]
- Beldent, V.; Michaud, A.; Bonnefoy, C.; Chauvet, M.T.; Corvol, P. Cell surface localization of proteolysis of human endothelial angiotensin I-converting enzyme: Effect of the amino-terminal domain in the solubilization process. J. Biol. Chem. 1995, 270, 28962–28969. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, M.R.W.; Chen, Y.N.P.; Riordan, J.F. The unique N-terminal sequence of testis angiotensin-converting enzyme is heavily O-glycosylated and unessential for activity or stability. Biochem. Biophys. Res. Commun. 1992, 183, 199–205. [Google Scholar] [CrossRef]
- Naperova, I.A.; Balyasnikova, I.V.; Schwartz, D.E.; Watermeyer, J.; Sturrock, E.D.; Kost, O.A.; Danilov, S.M. Mapping of conformational mAb epitopes to the C domain of human angiotensin l-converting enzyme. J. Proteome Res. 2008, 7, 3396–3411. [Google Scholar] [CrossRef]
- Naim, H.Y. Human small intestinal angiotensin-converting enzyme: Intracellular transport, secretion and glycosylation. Biochem. J. 1993, 296, 607–615. [Google Scholar] [CrossRef]
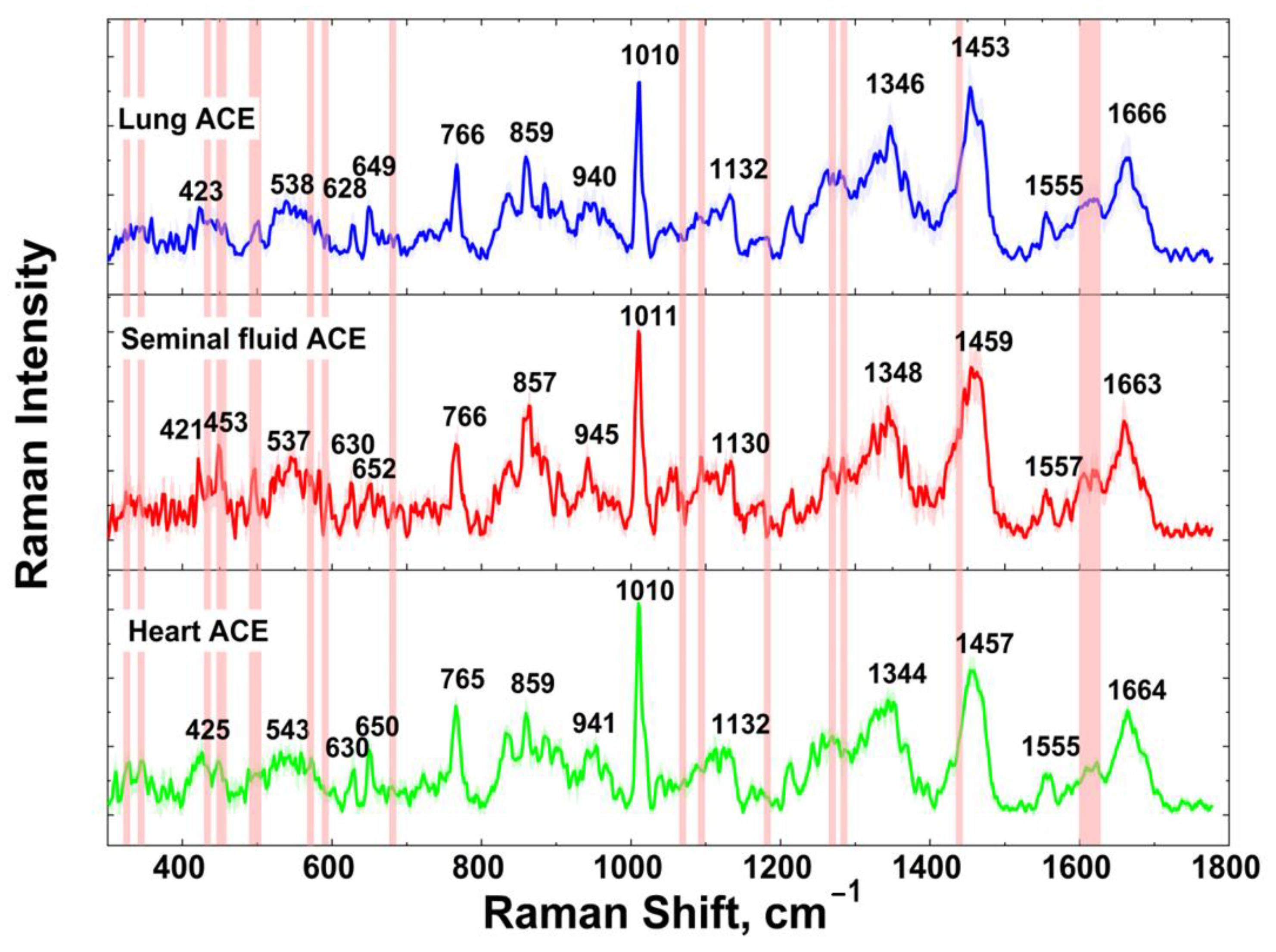


| Seminal Fluid | Lung | Heart | Band Assignment | Amino Acid or Dipeptide | Reported Band | Ref |
|---|---|---|---|---|---|---|
| 324 | - | 328 | - | - | - | - |
| 344 | - | 346 | - | - | - | - |
| 421 | 423 | 425 | Skeletal def. | - | - | - |
| 435 | - | - | Skeletal def. | - | - | - |
| 445 | - | 448 | - | - | - | - |
| 453 | - | - | Benzene ring def. | Trp | 454 | [57] |
| 492 | - | - | COO− bend. + CH2 | Gly | 497, 496 | [58,59] |
| - | 502 | - | - | - | - | - |
| 537 | 538 | 543 | COO− def. | Arg | 535 | [60] |
| 572 | - | - | COO− rock. | Thr | 568 | [61] |
| 591 | - | - | NH def. | Trp | 595 | [62] |
| 630 | 628 | 630 | C-S str. | Met | 632 | [63] |
| 652 | 649 | 650 | Imidazole ring breathing | His | 657 | [59] |
| 683 | - | - | C-S str. | Met-Leu | 685 | [64] |
| 766 | 766 | 765 | CH2 rock. | Met | 765 | [59] |
| 833 | 835 | 833 | Ring breathing mode and out-of-plane | Tyr | 837 | [65] |
| 857 | 859 | 859 | Ring def. Fermi resonance | 857 | [65] | |
| 885 | 885 | 886 | C-N str. + Cβ-Cδ str. | Ala | 885 | [66] |
| 905 | 906 | 902 | Asp | 902 | [59] | |
| 945 | 940 | 941 | C-C str. | His | 948 | [57] |
| 954 | 950 | 953 | C-C str. | Gly(Gly-Glu) | 956 | [67] |
| 963 | 963 | 966 | C-C str. | Pro-Leu | 961 | [67] |
| 1011 | 1010 | 1010 | Indole asym. ring breathing | Trp(Trp-Leu) | 1011 | [67] |
| 1041 | 1043 | 1037 | C-N str. | Pro(Pro-Leu) | 1044 | [67] |
| 1053 | 1052 | 1051 | Cα-N str., C-N str. | Met-Leu, Ala-Ala | 1056, 1050 | [63,67] |
| 1069 | - | - | C-N str. | Glu(Gly-Glu) | 1066 | [67] |
| 1095 | - | - | Cα-C-N str. asym., NH2 twist. | Pro-Pro, Met-Leu | 1092, 1095 | [63,68] |
| 1130 | 1132 | 1132 | N-H wag. | Lys | 1142 | [61] |
| 1169 | 1174 | 1173 | N-H wag. | His | 1160 | [61] |
| - | 1183 | 1183 | - | Glu-Gly, Leu-Gly | 1194, 1174 | [67] |
| 1212 | 1215 | 1213 | Ring def. | Phe | 1214 | [59] |
| 1241 | 1240 | 1243 | CH2 wag. | Leu(Leu-Gly) | 1241 | [67] |
| 1260 | 1262 | 1256 | Amide III | - | 1264 | [61] |
| - | 1270 | 1270 | CH2 wag. | Leu(Leu-Glu), Ser-Gly | 1276, 1266 | [67] |
| 1283 | 1279 | - | CH2 wag. | Trp(Trp-leu) | 1283 | [67] |
| 1307 | 1307 | 1307 | CH2 wag. | Glu(Gly-Glu) | 1307 | [67] |
| 1324 | 1326 | 1327 | C-NH2 str. | Met-Leu | 1323 | [67] |
| 1339 | 1333 | 1335 | C-H bend. | Asp | 1336 | [69] |
| 1348 | 1346 | 1344 | - | Glu | 1346 | [59] |
| 1366 | 1366 | 1367 | Indole vibration | Trp(Trp-Leu) | 1363 | [67] |
| 1385 | 1385 | 1386 | - | - | - | - |
| 1392 | 1394 | 1396 | COO− sym. str. | Leu-Leu | 1396 | [67] |
| 1441 | - | - | CH2 sciss. | Gly(Leu-Gly) | 1440 | [67] |
| 1459 | 1453 | 1457 | CH2 sciss. | Gly(Leu-Gly) | 1454 | [61] |
| 1473 | 1467 | 1468 | Cγ, Cδ bend. | Arg | 1477 | [59] |
| 1557 | 1555 | 1555 | Cβ-Cγ def. | Lys | 1556 | [70] |
| 1605 | 1600 | - | Ring C-C str. | Phe | 1602 | [61] |
| - | 1613 | 1611 | Sym.ring C-C str. | Gly(Tyr-Gly) | 1613 | [67] |
| 1618 | - | 1623 | Indole NH | Trp(Trp-Leu) | 1621 | [61] |
| 1663 | 1666 | 1664 | Amide I | - | 1664 | [61] |
| Precision | Sensitivity | F score | Quantity | |
|---|---|---|---|---|
| Lung ACE | 1.00 | 1.00 | 1.00 | 33 |
| Seminal fluid ACE | 1.00 | 1.00 | 1.00 | 31 |
| Heart ACE | 1.00 | 1.00 | 1.00 | 35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boginskaya, I.; Safiullin, R.; Tikhomirova, V.; Kryukova, O.; Nechaeva, N.; Bulaeva, N.; Golukhova, E.; Ryzhikov, I.; Kost, O.; Afanasev, K.; et al. Human Angiotensin I-Converting Enzyme Produced by Different Cells: Classification of the SERS Spectra with Linear Discriminant Analysis. Biomedicines 2022, 10, 1389. https://doi.org/10.3390/biomedicines10061389
Boginskaya I, Safiullin R, Tikhomirova V, Kryukova O, Nechaeva N, Bulaeva N, Golukhova E, Ryzhikov I, Kost O, Afanasev K, et al. Human Angiotensin I-Converting Enzyme Produced by Different Cells: Classification of the SERS Spectra with Linear Discriminant Analysis. Biomedicines. 2022; 10(6):1389. https://doi.org/10.3390/biomedicines10061389
Chicago/Turabian StyleBoginskaya, Irina, Robert Safiullin, Victoria Tikhomirova, Olga Kryukova, Natalia Nechaeva, Naida Bulaeva, Elena Golukhova, Ilya Ryzhikov, Olga Kost, Konstantin Afanasev, and et al. 2022. "Human Angiotensin I-Converting Enzyme Produced by Different Cells: Classification of the SERS Spectra with Linear Discriminant Analysis" Biomedicines 10, no. 6: 1389. https://doi.org/10.3390/biomedicines10061389
APA StyleBoginskaya, I., Safiullin, R., Tikhomirova, V., Kryukova, O., Nechaeva, N., Bulaeva, N., Golukhova, E., Ryzhikov, I., Kost, O., Afanasev, K., & Kurochkin, I. (2022). Human Angiotensin I-Converting Enzyme Produced by Different Cells: Classification of the SERS Spectra with Linear Discriminant Analysis. Biomedicines, 10(6), 1389. https://doi.org/10.3390/biomedicines10061389






