Microarray-Based Comparative Genomic and Transcriptome Analysis of Borrelia burgdorferi
Abstract
:1. Introduction
B. burgdorferi Microarray Methodology
2. Comparative Genomic Studies
3. Global Transcriptome Studies
3.1. Response to Temperature
3.2. Transcriptome of B. burgdorferi in the Host-Adapted State
3.3. Transcriptome of B. burgdorferi in the Tick Vector
4. Transcriptional Regulation
4.1. Rrp2-RpoN-RpoS Regulatory Cascade
4.2. Borrelia Oxidative Stress Regulator (BosR)
4.3. Hk1-Rrp1 Regulatory Circuit
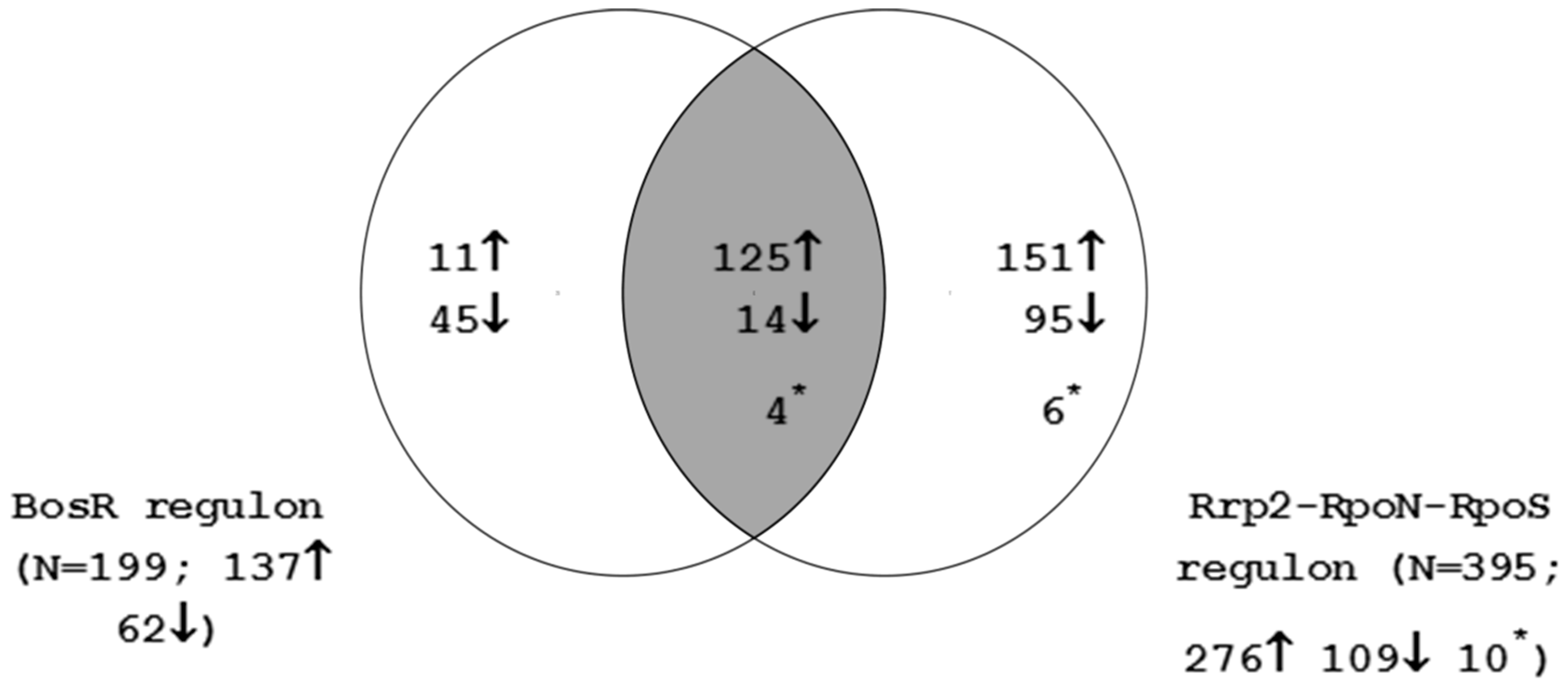
4.4. Interaction between the Rrp2-RpoN-RpoS-BosR and Hk1-Rrp1 Regulatory Circuits
5. Conclusions and Prospects for the Future
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Steere, A.C.; Grodzicki, R.L.; Kornblatt, A.N.; Craft, J.E.; Barbour, A.G.; Burgdorfer, W.; Schmid, G.P.; Johnson, E.; Malawista, S.E. The spirochetal etiology of Lyme disease. N. Engl. J. Med. 1983, 308, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Benach, J.L.; Bosler, E.M.; Hanrahan, J.P.; Coleman, J.L.; Habicht, G.S.; Bast, T.F.; Cameron, D.J.; Ziegler, J.L.; Barbour, A.G.; Burgdorfer, W.; et al. Spirochetes isolated from the blood of two patients with Lyme disease. N. Engl. J. Med. 1983, 308, 740–742. [Google Scholar] [CrossRef] [PubMed]
- Mead, P.S. Epidemiology of Lyme disease. Infect. Dis. Clin. N. Am. 2015, 29, 187–210. [Google Scholar] [CrossRef] [PubMed]
- Radolf, J.D.; Caimano, M.J.; Stevenson, B.; Hu, L.T. Of ticks, mice and men: Understanding the dual-host lifestyle of Lyme disease spirochaetes. Nat. Rev. Microbiol. 2012, 10, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Samuels, D.S. Gene regulation in Borrelia burgdorferi. Annu. Rev. Microbiol. 2011, 65, 479–499. [Google Scholar] [CrossRef] [PubMed]
- Fraser, C.M.; Casjens, S.; Huang, W.M.; Sutton, G.G.; Clayton, R.; Lathigra, R.; White, O.; Ketchum, K.A.; Dodson, R.; Hickey, E.K.; et al. Genomic sequence of a Lyme disease spirochaete, Borrelia burgdorferi. Nature 1997, 390, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Casjens, S.; Palmer, N.; van Vugt, R.; Huang, W.M.; Stevenson, B.; Rosa, P.; Lathigra, R.; Sutton, G.; Peterson, J.; Dodson, R.J.; et al. A bacterial genome in flux: The twelve linear and nine circular extrachromosomal DNAs in an infectious isolate of the Lyme disease spirochete Borrelia burgdorferi. Mol. Microbiol. 2000, 35, 490–516. [Google Scholar] [CrossRef] [PubMed]
- Revel, A.T.; Talaat, A.M.; Norgard, M.V. DNA microarray analysis of differential gene expression in Borrelia burgdorferi, the Lyme disease spirochete. Proc. Natl. Acad. Sci. USA 2002, 99, 1562–1567. [Google Scholar] [CrossRef] [PubMed]
- Ojaimi, C.; Brooks, C.; Akins, D.; Casjens, S.; Rosa, P.; Elias, A.; Barbour, A.; Jasinskas, A.; Benach, J.; Katonah, L.; et al. Borrelia burgdorferi gene expression profiling with membrane-based arrays. Methods Enzymol. 2002, 358, 165–177. [Google Scholar] [PubMed]
- Ojaimi, C.; Brooks, C.; Casjens, S.; Rosa, P.; Elias, A.; Barbour, A.; Jasinskas, A.; Benach, J.; Katona, L.; Radolf, J.; et al. Profiling of temperature-induced changes in Borrelia burgdorferi gene expression by using whole genome arrays. Infect. Immun. 2003, 71, 1689–1705. [Google Scholar] [CrossRef] [PubMed]
- Terekhova, D.; Iyer, R.; Wormser, G.P.; Schwartz, I. Comparative genome hybridization reveals substantial variation among clinical isolates of Borrelia burgdorferi sensu stricto with different pathogenic properties. J. Bacteriol. 2006, 188, 6124–6134. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Caimano, M.J.; Iyer, R.; Eggers, C.H.; Gonzalez, C.; Morton, E.A.; Gilbert, M.A.; Schwartz, I.; Radolf, J.D. Analysis of the RpoS regulon in Borrelia burgdorferi in response to mammalian host signals provides insight into RpoS function during the enzootic cycle. Mol. Microbiol. 2007, 65, 1193–1217. [Google Scholar] [CrossRef] [PubMed]
- Liang, F.T.; Nelson, F.K.; Fikrig, E. DNA microarray assessment of putative Borrelia burgdorferi lipoprotein genes. Infect. Immun. 2002, 70, 3300–3303. [Google Scholar] [CrossRef] [PubMed]
- Livengood, J.A.; Schmit, V.L.; Gilmore, R.D., Jr. Global transcriptome analysis of Borrelia burgdorferi during association with human neuroglial cells. Infect. Immun. 2008, 76, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.L.; Karna, S.L.; Seshu, J. Borrelia host adaptation regulator (BadR) regulates rpoS to modulate host adaptation and virulence factors in Borrelia burgdorferi. Mol. Microbiol. 2013, 88, 105–124. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Barbour, A.G. Cross-species hybridization of a Borrelia burgdorferi DNA array reveals infection- and culture-associated genes of the unsequenced genome of the relapsing fever agent Borrelia hermsii. Mol. Microbiol. 2004, 51, 729–748. [Google Scholar] [CrossRef] [PubMed]
- Ojaimi, C.; Mulay, V.; Liveris, D.; Iyer, R.; Schwartz, I. Comparative transcriptional profiling of Borrelia burgdorferi clinical isolates differing in capacities for hematogenous dissemination. Infect. Immun. 2005, 73, 6791–6802. [Google Scholar] [CrossRef] [PubMed]
- Brooks, C.S.; Hefty, P.S.; Jolliff, S.E.; Akins, D.R. Global analysis of Borrelia burgdorferi genes regulated by mammalian host-specific signals. Infect. Immun. 2003, 71, 3371–3383. [Google Scholar] [CrossRef] [PubMed]
- Tokarz, R.; Anderton, J.M.; Katona, L.I.; Benach, J.L. Combined effects of blood and temperature shift on Borrelia burgdorferi gene expression as determined by whole genome DNA array. Infect. Immun. 2004, 72, 5419–5432. [Google Scholar] [CrossRef] [PubMed]
- Anderton, J.M.; Tokarz, R.; Thill, C.D.; Kuhlow, C.J.; Brooks, C.S.; Akins, D.R.; Katona, L.I.; Benach, J.L. Whole-genome DNA array analysis of the response of Borrelia burgdorferi to a bactericidal monoclonal antibody. Infect. Immun. 2004, 72, 2035–2044. [Google Scholar] [CrossRef] [PubMed]
- Hyde, J.A.; Seshu, J.; Skare, J.T. Transcriptional profiling of Borrelia burgdorferi containing a unique bosR allele identifies a putative oxidative stress regulon. Microbiology 2006, 152, 2599–2609. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Z.; Kumar, M.; Kariu, T.; Haq, S.; Goldberg, M.; Pal, U.; Norgard, M.V. BosR (BB0647) governs virulence expression in Borrelia burgdorferi. Mol. Microbiol. 2009, 74, 1331–1343. [Google Scholar] [CrossRef] [PubMed]
- Boardman, B.K.; He, M.; Ouyang, Z.; Xu, H.; Pang, X.; Yang, X.F. Essential role of the response regulator Rrp2 in the infectious cycle of Borrelia burgdorferi. Infect. Immun. 2008, 76, 3844–3853. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Z.; Blevins, J.S.; Norgard, M.V. Transcriptional interplay among the regulators Rrp2, RpoN and RpoS in Borrelia burgdorferi. Microbiology 2008, 154, 2641–2658. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.A.; Grimm, D.; Henion, A.K.; Elias, A.F.; Stewart, P.E.; Rosa, P.A.; Gherardini, F.C. Borrelia burgdorferi σ54 is required for mammalian infection and vector transmission but not for tick colonization. Proc. Natl. Acad. Sci. USA 2005, 102, 5162–5167. [Google Scholar] [CrossRef] [PubMed]
- Rogers, E.A.; Terekhova, D.; Zhang, H.M.; Hovis, K.M.; Schwartz, I.; Marconi, R.T. Rrp1, a cyclic-di-GMP-producing response regulator, is an important regulator of Borrelia burgdorferi core cellular functions. Mol. Microbiol. 2009, 71, 1551–1573. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Ouyang, Z.; Troxell, B.; Xu, H.; Moh, A.; Fu, X.-Y.; Piesman, J.; Norgard, M.V.; Gomelsky, M.; Yang, X.F. Cyclic di-GMP is essential for the survival of Borrelia burgdorferi in ticks. PLoS Pathog. 2011, 7, e1002133. [Google Scholar] [CrossRef] [PubMed]
- Bugrysheva, J.V.; Pappas, C.J.; Terekhova, D.A.; Iyer, R.; Godfrey, H.P.; Schwartz, I.; Cabello, F.C. Characterization of the RelBbu regulon in Borrelia burgdorferi reveals modulation of glycerol metabolism by (p)ppGpp. PLoS ONE 2015, 10, e0118063. [Google Scholar] [CrossRef] [PubMed]
- Salman-Dilgimen, A.; Hardy, P.O.; Radolf, J.D.; Caimano, M.J.; Chaconas, G. HrpA, an RNA helicase involved in RNA processing, is required for mouse infectivity and tick transmission of the Lyme disease spirochete. PLoS Pathog. 2013, 9, e1003841. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, S.; Caimano, M.J.; Liang, F.T.; Santiago, F.; Laskowski, M.; Philipp, M.T.; Pachner, A.R.; Radolf, J.D.; Fikrig, E. Borrelia burgdorferi transcriptome in the central nervous system of non-human primates. Proc. Natl. Acad. Sci. USA 2003, 100, 15953–15958. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, S.; Santiago, F.; Koski, R.A.; Brei, B.; Anderson, J.F.; Fish, D.; Fikrig, E. Examination of the Borrelia burgdorferi transcriptome in Ixodes scapularis during feeding. J. Bacteriol. 2002, 184, 3122–3125. [Google Scholar] [CrossRef] [PubMed]
- Pal, U.; Dai, J.; Li, X.; Neelakanta, G.; Luo, P.; Kumar, M.; Wang, P.; Yang, X.; Anderson, J.F.; Fikrig, E. A differential role for BB0365 in the persistence of Borrelia burgdorferi in mice and ticks. J. Infect. Dis. 2008, 197, 148–155. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Iyer, R.; Caimano, M.J.; Luthra, A.; Axline, D., Jr.; Corona, A.; Iacobas, D.A.; Radolf, J.D.; Schwartz, I. Stage-specific global alterations in the transcriptomes of Lyme disease spirochetes during tick feeding and following mammalian host adaptation. Mol. Microbiol. 2015, 95, 509–538. [Google Scholar] [CrossRef] [PubMed]
- Schutzer, S.E.; Fraser-Liggett, C.M.; Casjens, S.R.; Qiu, W.G.; Dunn, J.J.; Mongodin, E.F.; Luft, B.J. Whole-genome sequences of thirteen isolates of Borrelia burgdorferi. J. Bacteriol. 2011, 193, 1018–1020. [Google Scholar] [CrossRef] [PubMed]
- Casjens, S.R.; Mongodin, E.F.; Qiu, W.G.; Luft, B.J.; Schutzer, S.E.; Gilcrease, E.B.; Huang, W.M.; Vujadinovic, M.; Aron, J.K.; Vargas, L.C.; et al. Genome stability of Lyme disease spirochetes: Comparative genomics of Borrelia burgdorferi plasmids. PLoS ONE 2012, 7, e33280. [Google Scholar] [CrossRef] [PubMed]
- Mongodin, E.F.; Casjens, S.R.; Bruno, J.F.; Xu, Y.; Drabek, E.F.; Riley, D.R.; Cantarel, B.L.; Pagan, P.E.; Hernandez, Y.A.; Vargas, L.C.; et al. Inter- and intra-specific pan-genomes of Borrelia burgdorferi sensu lato: Genome stability and adaptive radiation. BMC Genom. 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Akins, D.R.; Bourell, K.W.; Caimano, M.J.; Norgard, M.V.; Radolf, J.D. A new animal model for studying Lyme disease spirochetes in a mammalian host-adapted state. J. Clin. Investig. 1998, 101, 2240–2250. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Popova, T.G.; Hagman, K.E.; Wikel, S.K.; Schoeler, G.B.; Caimano, M.J.; Radolf, J.D.; Norgard, M.V. Identification, characterization, and expression of three new members of the Borrelia burgdorferi mlp (2.9) lipoprotein gene family. Infect. Immun. 1999, 67, 6008–6018. [Google Scholar] [PubMed]
- Caimano, M.J.; Eggers, C.H.; Gonzalez, C.A.; Radolf, J.D. Alternate sigma factor RpoS is required for the in vivo-specific repression of Borrelia burgdorferi plasmid lp54-borne ospA and lp6.6 genes. J. Bacteriol. 2005, 187, 7845–7852. [Google Scholar] [CrossRef] [PubMed]
- Mulay, V.B.; Caimano, M.J.; Iyer, R.; Dunham-Ems, S.; Liveris, D.; Petzke, M.M.; Schwartz, I.; Radolf, J.D. Borrelia burgdorferi bba74 is expressed exclusively during tick feeding and is regulated by both arthropod- and mammalian host-specific signals. J. Bacteriol. 2009, 191, 2783–2794. [Google Scholar] [CrossRef] [PubMed]
- Pappas, C.J.; Iyer, R.; Petze, M.M.; Caimano, M.J.; Radolf, J.D.; Schwartz, I. Borrelia burgdorferi requires glycerol for maximum fitness during the tick phase of the enzootic cycle. PLoS Pathog. 2011, 7, e1002102. [Google Scholar] [CrossRef] [PubMed]
- Caimano, M.J.; Dunham-Ems, S.; Allard, A.M.; Cassera, M.B.; Kenedy, M.; Radolf, J.D. Cyclic di-GMP modulates gene expression in Lyme disease spirochetes at the tick-mammal interface to promote spirochete survival during the blood meal and tick-to-mammal transmission. Infect. Immun. 2015, 83, 3043–3060. [Google Scholar] [CrossRef] [PubMed]
- Drecktrah, D.; Lybecker, M.; Popitsch, N.; Rescheneder, P.; Hall, L.S.; Samuels, D.S. The Borrelia burgdorferi RelA/SpoT homolog and stringent response regulate survival in the tick vector and global gene expression during starvation. PLoS Pathog. 2015, 11, e1005160. [Google Scholar] [CrossRef] [PubMed]
- Hubner, A.; Yang, X.; Nolen, D.M.; Popova, T.G.; Cabello, F.C.; Norgard, M.V. Expression of Borrelia burgdorferi OspC and DbpA is controlled by a RpoN-RpoS regulatory pathway. Proc. Natl. Acad. Sci. USA 2001, 98, 12724–12729. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.H.; Blevins, J.S.; Bachlani, G.N.; Yang, X.F.; Norgard, M.V. Evidence that RpoS (σs) in Borrelia burgdorferi is controlled directly by RpoN (σ54/ σn). J. Bacteriol. 2007, 189, 2139–2144. [Google Scholar] [CrossRef] [PubMed]
- Boylan, J.A.; Posey, J.E.; Gherardini, F.C. Borrelia oxidative stress response regulator, BosR, a distinctive Zn-dependent transcriptional activator. Proc. Natl. Acad. Sci. USA 2003, 100, 11684–11689. [Google Scholar] [CrossRef] [PubMed]
- Katona, L.I.; Tokarz, R.; Kuhlow, C.J.; Benach, J.; Benach, J.L. The fur homologue in Borrelia burgdorferi. J. Bacteriol. 2004, 186, 6443–6456. [Google Scholar] [CrossRef] [PubMed]
- Hyde, J.A.; Shaw, D.K.; Smith III, R.; Trzeciakowski, J.P.; Skare, J.T. The BosR regulatory protein of Borrelia burgdorferi interfaces with the RpoS regulatory pathway and modulates both the oxidative stress response and pathogenic properties of the Lyme disease spirochete. Mol. Microbiol. 2009, 74, 1344–1355. [Google Scholar] [CrossRef] [PubMed]
- Samuels, D.S.; Radolf, J.D. Who is the BosR around here anyway? Mol. Microbiol. 2009, 74, 1295–1299. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Z.; Deka, R.K.; Norgard, M.V. BosR (BB0647) controls the RpoN-RpoS regulatory pathway and virulence expression in Borrelia burgdorferi by a novel DNA-binding mechanism. PLoS Pathog. 2011, 7, e1001272. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Dadhwal, P.; Cheng, Z.; Zianni, M.R.; Rikihisa, Y.; Liang, F.T.; Li, X. Borrelia burgdorferi oxidative stress regulator BosR directly represses lipoproteins primarily expressed in the tick during mammalian infection. Mol. Microbiol. 2013, 89, 1140–1153. [Google Scholar] [CrossRef] [PubMed]

| Experimental Condition | Strain | Microarray Type | Reference |
|---|---|---|---|
| Comparative genomics | B31 | Glass slide | Liang et al., Infect. Immun., 2002 [13] |
| Comparative genomics | B31 | membrane | Zhong & Barbour, Mol. Microbiol., 2004 [16] |
| Comparative genomics | B31 | 70 m oligo glass slide | Terekhova et al., J. Bacteriol., 2006 [11] |
| Temperature response | B31 | Membrane | Ojaimi et al., Infect. Immun., 2003 [10] |
| Strain transcriptome comparison | B31 | Membrane | Ojaimi et al., Infect. Immun., 2005 [17] |
| In vitro and host-adapted (DMC) | B31 | Glass slide | Revel et al., PNAS, 2002 [8] |
| In vitro and host-adapted (DMC) | B31 | Membrane | Brooks et al., Infect. Immun., 2003 [18] |
| Blood co-incubation | B31 | Membrane | Tokarz et al., Infect. Immun., 2004 [19] |
| Monoclonal OspB antibody co-cultivation | B31 | Membrane | Anderton et al., Infect. Immun., 2004 [20] |
| Neuroglial cell co-incubation | B31 | Affymetrix slide | Livengood et al., Infect. Immun., 2008 [14] |
| RpoS regulon | 297 | 70 m oligo glass slide | Caimano et al., Mol. Microbiol., 2007 [12] |
| BosR regulon | B31 | Membrane array | Hyde et al., Microbiology, 2006 [21] |
| BosR regulon | B31 | 70 m oligo glass slide | Ouyang et al., Mol. Microbiol., 2009 [22] |
| Rrp2 regulon | B31 | 70 m oligo glass slide | Boardman et al., Infect. Immun., 2008 [23] |
| Rrp2/RpoN/RpoS regulon | 297 | 70 m oligo glass slide | Ouyang et al., Microbiology, 2008 [24] |
| RpoN/RpoS regulon | B31 | 70 m oligo glass slide | Fisher et al., PNAS, 2005 [25] |
| Rrp1 regulon | B31 | 70 m oligo glass slide | Rogers et al., Mol. Microbiol., 2009 [26] |
| Rrp1 regulon | B31 | 70 m oligo glass slide | He et al., PLoS Pathog., 2011 [27] |
| RelBbu regulon | 297 | 70 m oligo glass slide | Bugrysheva et al., PLoS ONE, 2015 [28] |
| BadR regulon | B31 | Nimblegen | Miller et al., Mol. Microbiol., 2013 [15] |
| HrpA regulon | B31 | Nimblegen | Salman-Dilgimen et al., PLoS Pathog., 2013 [29] |
| Non-human primate tissues | N40, JD1 | Glass slide | Narasimhan et al., PNAS, 2003 [30] |
| Fed Ticks | N40 | Glass slide | Narasimhan et al., J. Bacteriol., 2002 [31] |
| Mouse tissues | B31 | 70 m oligo glass slide | Pal et al., J. Infect. Dis., 2008 [32] |
| Tick feeding stages and host-adapted (DMC) | B31 | 70 m oligo glass slide | Iyer et al., Mol. Microbiol., 2015 [33] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iyer, R.; Schwartz, I. Microarray-Based Comparative Genomic and Transcriptome Analysis of Borrelia burgdorferi. Microarrays 2016, 5, 9. https://doi.org/10.3390/microarrays5020009
Iyer R, Schwartz I. Microarray-Based Comparative Genomic and Transcriptome Analysis of Borrelia burgdorferi. Microarrays. 2016; 5(2):9. https://doi.org/10.3390/microarrays5020009
Chicago/Turabian StyleIyer, Radha, and Ira Schwartz. 2016. "Microarray-Based Comparative Genomic and Transcriptome Analysis of Borrelia burgdorferi" Microarrays 5, no. 2: 9. https://doi.org/10.3390/microarrays5020009
APA StyleIyer, R., & Schwartz, I. (2016). Microarray-Based Comparative Genomic and Transcriptome Analysis of Borrelia burgdorferi. Microarrays, 5(2), 9. https://doi.org/10.3390/microarrays5020009





