Bio-Guided Targeting for Preservative and Anti-Ageing Cosmetic Ingredient Development
Abstract
:1. Introduction
2. Experimental Section
2.1. Reagents
2.2. Kalanchoe pinnata Extract
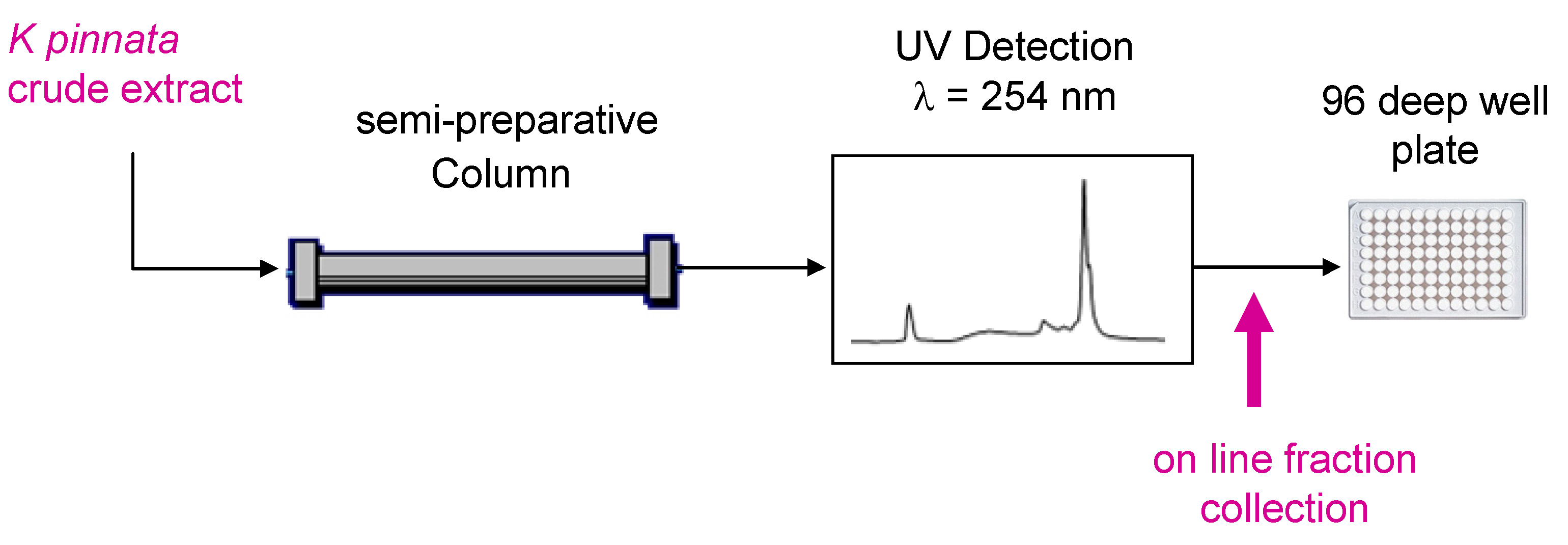
2.3. Semi-Preparative HPLC Fractionation
2.4. Mass Spectrometry
2.5. Antibacterial Assay
2.6. Cell Culture
2.7. Skin Cell Viability Test
2.8. Antioxidant Assay on Normal Human Epidermis Keratinocytes
3. Results and Discussion
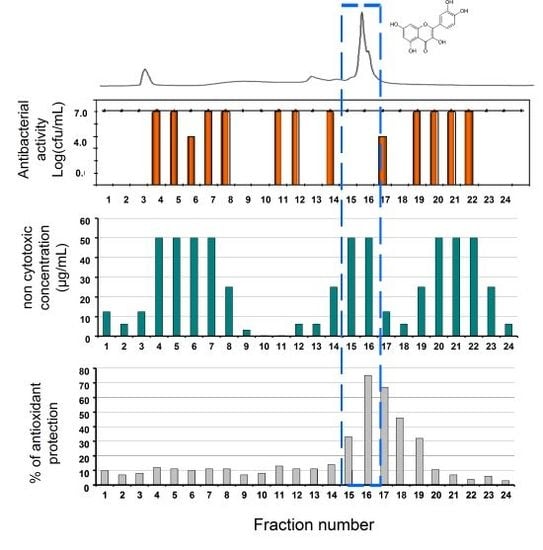
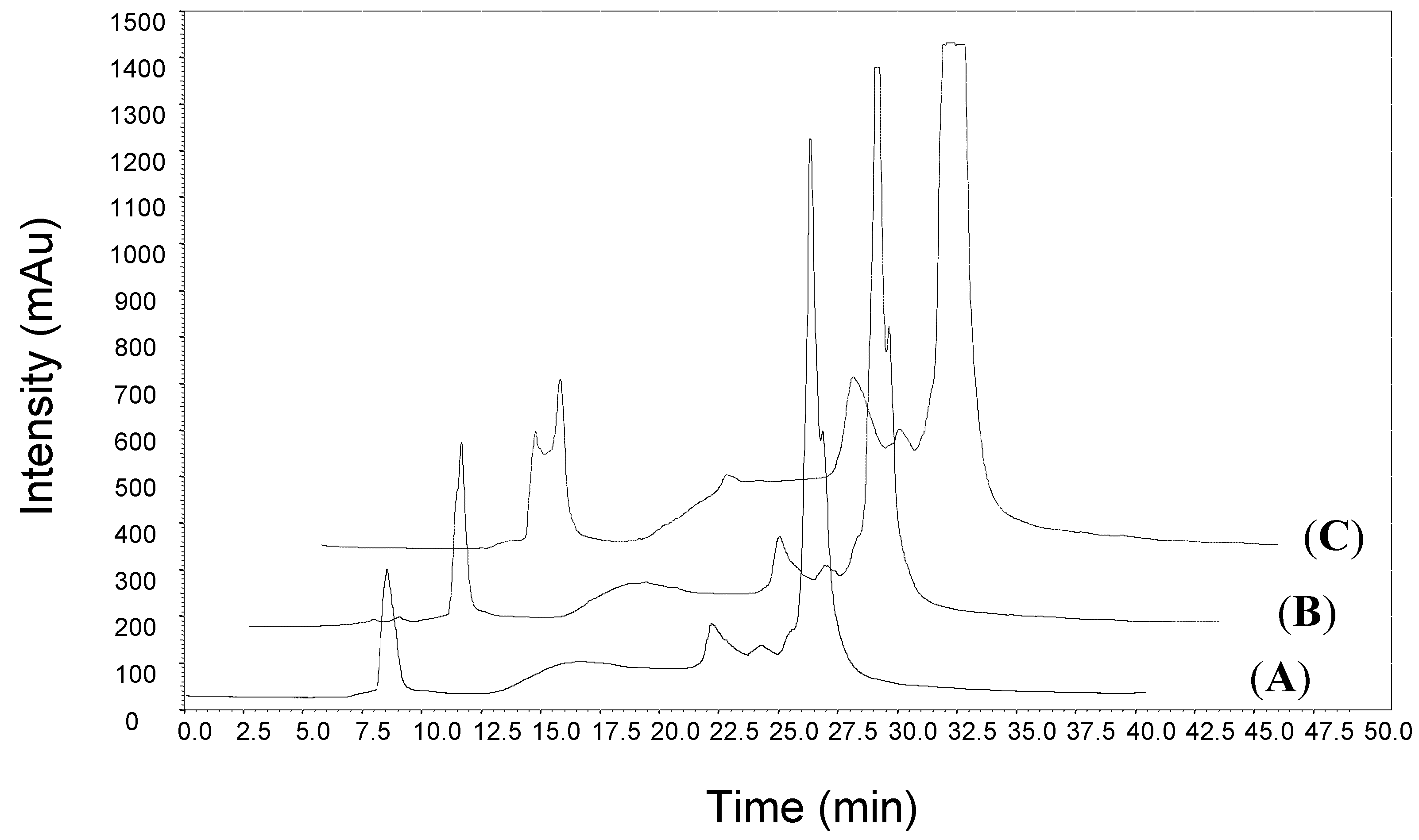
3.1. Semi-Preparative HPLC Fractionation
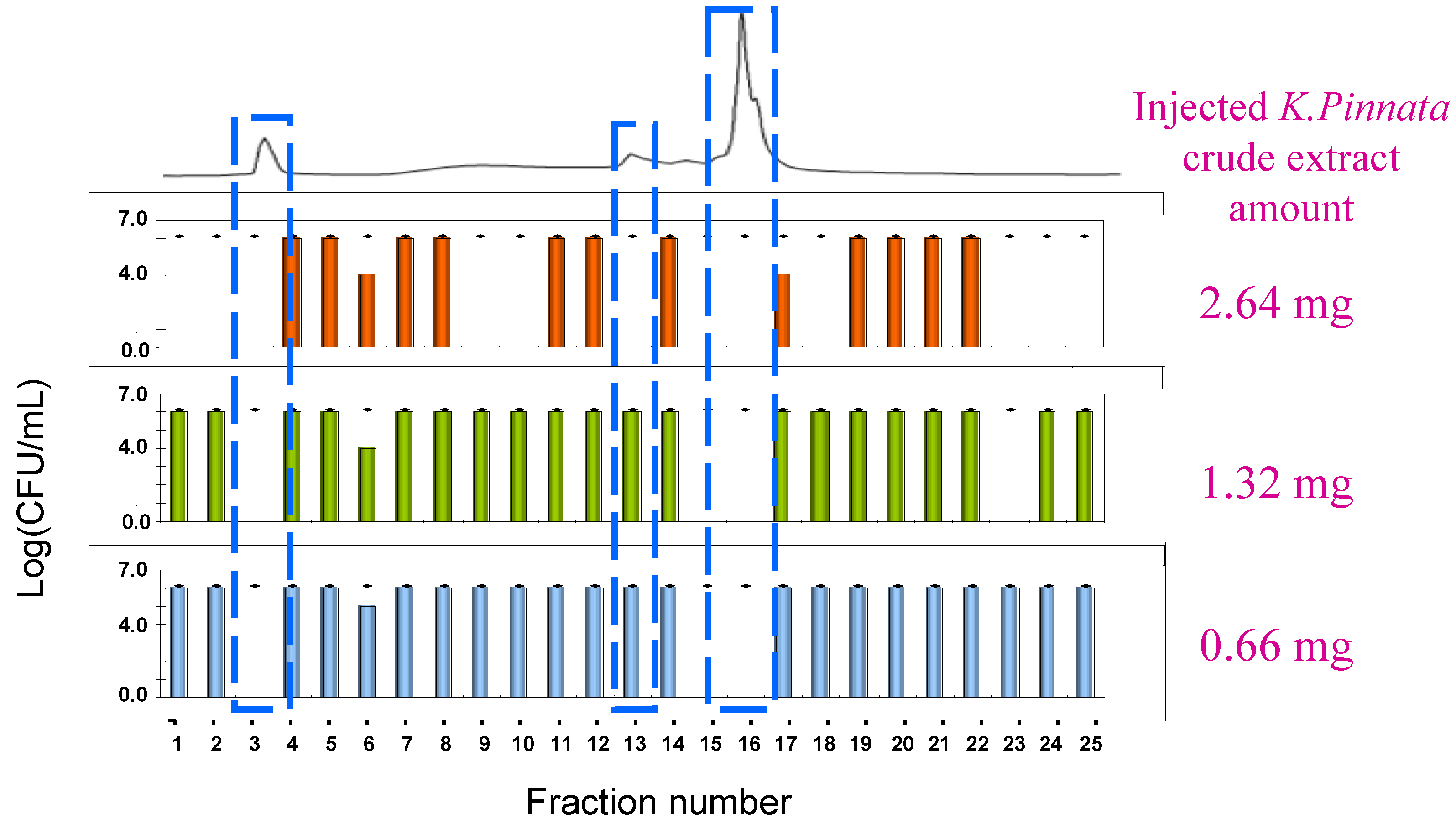
3.2. Antibacterial Assay
| Samples | Ec | Sa | Pa | Ca | Ab |
|---|---|---|---|---|---|
| CE 66 µg mL−1 | − | ++ | − | ++ | − |
| CE 132 µg mL−1 | + | + | − | ++ | − |
| CE 264 µg mL−1 | + | + | +/− | + | + |
| Fraction 1 | + | + | − | +/− | − |
| Fraction 2 | + | + | − | +/− | − |
| Fraction 3 | ++ | + | +/− | +/− | − |
| Fraction 4 | − | + | +/− | +/− | − |
| Fraction 5 | − | + | − | +/− | − |
| Fraction 6 | +/− | + | − | +/− | − |
| Fraction 7 | − | + | − | ++ | − |
| Fraction 8 | − | + | + | ++ | − |
| Fraction 9 | + | + | + | ++ | − |
| Fraction 10 | + | + | − | +/− | − |
| Fraction 11 | − | ++ | − | +/− | − |
| Fraction 12 | − | ++ | − | +/− | − |
| Fraction 13 | + | ++ | − | +/− | − |
| Fraction 14 | − | ++ | − | ++ | − |
| Fraction 15 | ++ | − | − | ++ | − |
| Fraction 16 | ++ | + | + | ++ | − |
| Fraction 17 | +/− | + | + | +/− | − |
| Fraction 18 | + | + | + | +/− | − |
| Fraction 19 | − | + | − | +/− | − |
| Fraction 20 | − | + | − | ++ | − |
| Fraction 21 | − | + | − | ++ | − |
| Fraction 22 | − | + | − | ++ | − |
| Fraction 23 | + | ++ | − | +/− | − |
| Fraction 24 | + | + | − | +/− | − |
| Fraction 25 | + | + | + | ++ | − |
3.3. Viability Test
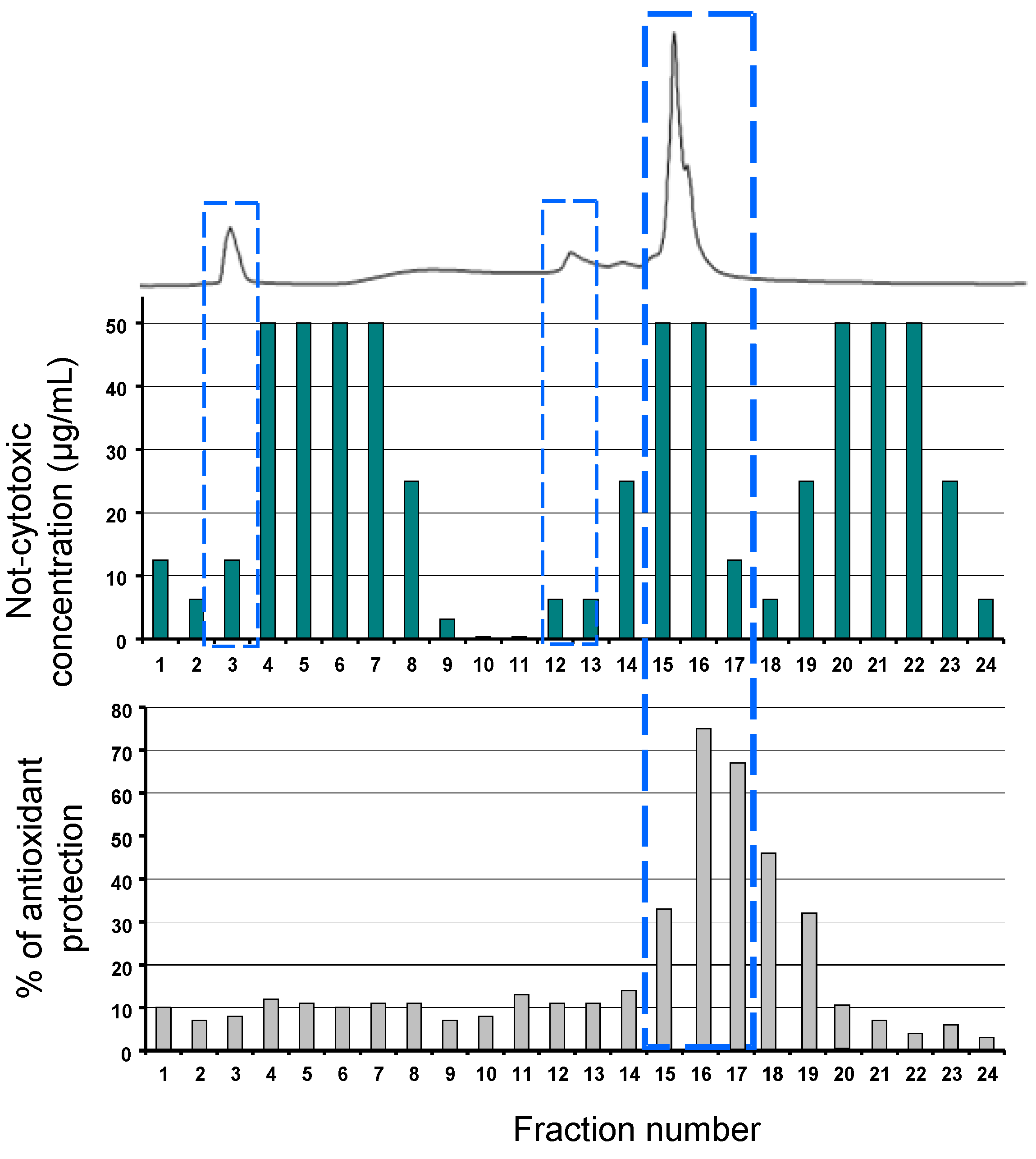
3.4. Antioxidant Assay on Normal Human Epidermis Keratinocytes
3.5. Mass Spectrometry
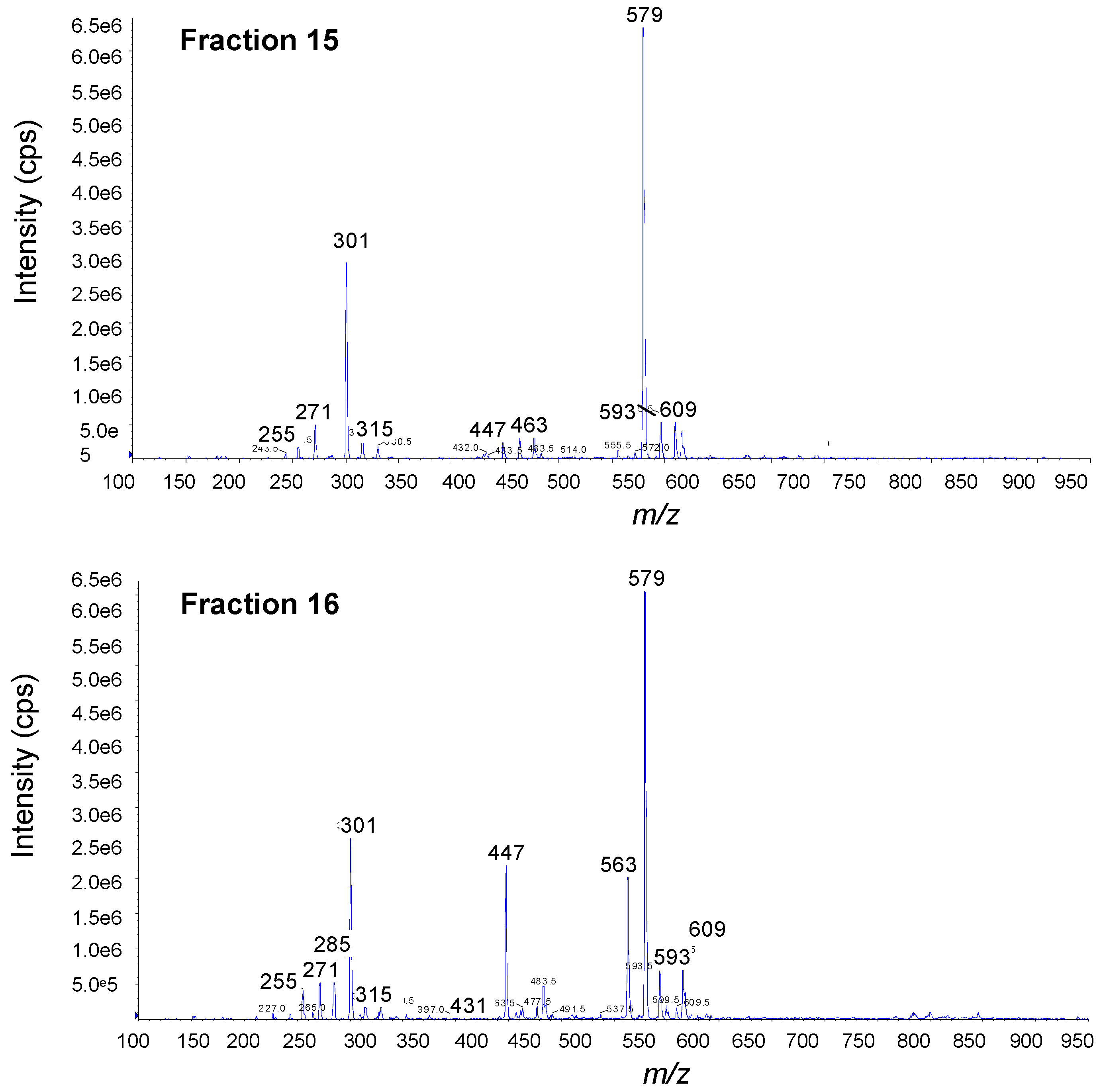
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Potterat, O.; Hamburger, M. Concepts and technologies for tracking bioactive compounds in natural product extracts: Generation of libraries, and hyphenation of analytical processes with bioassays. Nat. Prod. Rep. 2013, 30, 546–564. [Google Scholar] [CrossRef]
- Kamboj, A.; Saluja, A.K. Byophyllum pinnatum (Lam.) Kruz;. Phytochemical and pharmacological profile: A review. Pharmacon. Rev. 2009, 3, 364–374. [Google Scholar]
- Ojewole, J.A.O. Antinociceptive, anti-inflammatory and antidiabetic effects of Bryophyllum pinnatum (Crassulaceae) leaf aqueous extract. J. Ethnopharmacol. 2005, 99, 13–19. [Google Scholar] [CrossRef]
- Akinpelu, D.A. Antimicrobial activity of Bryophyllum pinnatum leaves. Fitoterapia 2000, 71, 193–194. [Google Scholar] [CrossRef]
- Afzal, M.; Gupta, G.; Kazmi, I.; Rahman, M.; Afzal, O.; Alam, J.; Hakeem, K.R.; Pravez, M.; Gupta, R.; Anwar, F. Anti-inflammatory and analgesic potential of a novel steroidal derivative from Bryophyllum pinnatum. Fitoterapia 2012, 83, 853–858. [Google Scholar] [CrossRef]
- Yadav, N.P.; Dixit, V.K. Hepatoprotective activity of leaves of Kalanchoe pinnata Pers. J. Ethnopharmacol. 2003, 86, 197–202. [Google Scholar] [CrossRef]
- Almeida, A.P.; da Silva, S.A.; Souza, M.L.; Lima, L.M.; Rossi-Bergmann, B.; Goncalves de Moraes, V.L.; Costa, S.S. Isolation and chemical analysis of a fatty acid fraction of Kalanchoe pinnata with a potent lymphocyte suppressive activity. Planta Med. 2000, 66, 134–137. [Google Scholar] [CrossRef]
- Pal, S.; Chaudhuri, A.K.N. Studies on the anti-ulcer activity of a Bryophyllum pinnatum leaf extract in experimental animals. J. Ethnopharmacol. 1991, 33, 97–102. [Google Scholar] [CrossRef]
- Pal, S.; Nag Chaudhuri, A.K. Anti-inflammatory action of Bryophullum pinnatum leaf extract. Fitoterapia 1990, 61, 527–533. [Google Scholar]
- Pal, S.; Nag Chaudhuri, A.K. Further studies on the anti-inflammatory profile of the methanolic fraction of the fresh leaf extract of Bryophyllum pinnatum. Fitoterapia 1992, 63, 451–459. [Google Scholar]
- Biswas, S.K.; Chowdhury, A.; Raihan, S.Z.; Muhit, M.A.; Akbar, M.A.; Mowla, R. Phytochemical investigation with assessment of cytotoxicity and antibacterial activities of chloroform extract of the leaves of Kalanchoe pinnata. Am. J. Plant Physiol. 2012, 7, 41–46. [Google Scholar]
- Abdellaoui, S.; Destandau, E.; Toribio, A.; Elfakir, C.; Lafosse, M.; Renimel, I.; André, P.; Cancellieri, P.; Landemarre, L. Bioactive molecules in Kalanchoe pinnata leaves: Extraction, purification, and identification. Anal. Bioanal. Chem. 2010, 398, 1329–1338. [Google Scholar] [CrossRef]
- Yamagishim, T.; Haruna, M.; Yan, X.Z.; Chang, J.J.; Lee, K.H. Antitumor agent; 110. Bryophyllin B, a novel potent cytotoxic bufadienolide from Bryophyllium Pinnatum. J. Nat. Prod. 1989, 52, 1071–1079. [Google Scholar] [CrossRef]
- Da Silva, S.A.G.; Costa, S.S.; Mendonça, S.C.F.; Silva, E.M.; Moraes, V.L.G.; Rossi-Bergmann, B. Therapeutic effect of oral Kalanchoe pinnata leaf extract in murine leishmaniasis. Acta Trop. 1995, 60, 201–210. [Google Scholar] [CrossRef]
- Muzitano, M.F.L.; Tinoco, W.; Guette, C.; Kaiser, C.R.; Rossi-Bergmann, B.; Costa, S.S. The antileishmanial activity assessment of unusual flavonoids from Kalanchoe pinnata. Phytochemistry 2006, 67, 2071–2077. [Google Scholar] [CrossRef]
- Muzitano, M.F.; Falcão, C.A.; Cruz, E.A.; Bergonzi, M.C.; Bilia, A.R.; Vincieri, F.F.; Rossi-Bergmann, B.; Costa, S.S. Oral metabolism and efficacy of Kalanchoe pinnata flavonoids in a murine model of cutaneous leishmaniasis. Planta Med. 2009, 75, 307–311. [Google Scholar] [CrossRef]
- Nascimento, L.B.S.; Leal-Costa, M.V.; Coutinho, M.A.S.; Moreira, N.D.S.; Lage, C.L.S.; Barbi, N.D.S.; Costa, S.S.; Tavares, E.S. Increased antioxidant activity and changes in phenolic profile of Kalanchoe pinnata (lamarck) Persoon (crassulaceae) specimens grown under supplemental blue light. Photochem. Photobiol. 2013, 89, 391–399. [Google Scholar] [CrossRef]
- Tatsimo, S.J.N.; Tamokou, J.D.D.; Havyarimana, L.; Csupor, D.; Forgo, P.; Hohmann, J.; Kuiate, J.R.; Tane, P. Antimicrobial and antioxidant activity of kaempferol rhamnoside derivatives from Bryophyllum pinnatum. BMC Res. Notes 2012, 5, 158. [Google Scholar] [CrossRef]
- Gaind, K.N.; Gupta, R.L. Phenolic compounds from the leaves of Kalanchoe Pinnata. Planta Med. 1973, 23, 149–153. [Google Scholar] [CrossRef]
- Orth, D.S.; Eck, K.S. Use of triphenyltetrazolium chloride in preservative efficacy testing. Int. J. Cosmet. Sci. 2005, 27, 353. [Google Scholar] [CrossRef]
- Roehm, N.W.; Rodgers, G.H.; Hatfield, S.M.; Glasebrook, A.L. An improved colorimetric assay for cell proliferation and viability utilizing the tetrazolium salt XTT. J. Immunol. Methods 1991, 142, 257–265. [Google Scholar] [CrossRef]
- Lebel, C.P.; Ischriropoulos, H.; Bondy, S.C. Evaluation of the probe 2',7'-dichlorofluorescence an indicator of reactive oxygen species formation and oxidative stress. Chem. Res. Toxicol. 1992, 5, 227–231. [Google Scholar] [CrossRef]
- Jenkins, G. Molecular mechanisms of skin ageing. Mech. Ageing Dev. 2002, 123, 801–810. [Google Scholar] [CrossRef]
- Chung, H.Y.; Cesari, M.; Anton, S.; Marzetti, E.; Giovannini, S.; Seo, A.Y.; Carter, C.; Yu, B.; Leeuwenburgh, C. Molecular inflammation: Underpinnings of aging and age-related diseases. Ageing Res. Rev. 2009, 8, 18–30. [Google Scholar] [CrossRef]
- Cuyckens, F.; Claeys, M. Mass spectrometry in the structural analysis of flavonoids. J. Mass Spectrom. 2004, 39, 1–15. [Google Scholar] [CrossRef]
- De Vos, S.; Brach, M.; Budnik, A.; Grewe, M.; Herrmann, F.; Krutmann, J. Post-transcriptional regulation of interleukin-6 gene expression in human keratinocytes by ultraviolet B radiation. J. Investig. Dermatol. 1994, 103, 92–96. [Google Scholar]
- Aragane, Y.; Yamada, H.; Schwarz, A.; Poppelmann, B.; Luger, T.A.; Tezuka, T. Transforming growth factor-alpha induces interleukin-6 in the human keratinocyte cell line HaCaT mainly by transcriptional activation. J. Investig. Dermatol. 1996, 106, 1192–1197. [Google Scholar]
- Suh, D.H.; Kwon, T.E.; Youn, J.I. Changes of comedonal cytokines and sebum secretion after UV irradiation in acne patients. Eur. J. Dermatol. 2002, 12, 139–144. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Destandau, E.; Krolikiewicz-Renimel, I.; El Abdellaoui, S.; Cancellieri, P.; Fougère, L.; Toribio, A.; Landemarre, L.; André, P.; Elfakir, C. Bio-Guided Targeting for Preservative and Anti-Ageing Cosmetic Ingredient Development. Cosmetics 2014, 1, 14-28. https://doi.org/10.3390/cosmetics1010014
Destandau E, Krolikiewicz-Renimel I, El Abdellaoui S, Cancellieri P, Fougère L, Toribio A, Landemarre L, André P, Elfakir C. Bio-Guided Targeting for Preservative and Anti-Ageing Cosmetic Ingredient Development. Cosmetics. 2014; 1(1):14-28. https://doi.org/10.3390/cosmetics1010014
Chicago/Turabian StyleDestandau, Emilie, Isabelle Krolikiewicz-Renimel, Saida El Abdellaoui, Perrine Cancellieri, Laetitia Fougère, Alix Toribio, Ludovic Landemarre, Patrice André, and Claire Elfakir. 2014. "Bio-Guided Targeting for Preservative and Anti-Ageing Cosmetic Ingredient Development" Cosmetics 1, no. 1: 14-28. https://doi.org/10.3390/cosmetics1010014
APA StyleDestandau, E., Krolikiewicz-Renimel, I., El Abdellaoui, S., Cancellieri, P., Fougère, L., Toribio, A., Landemarre, L., André, P., & Elfakir, C. (2014). Bio-Guided Targeting for Preservative and Anti-Ageing Cosmetic Ingredient Development. Cosmetics, 1(1), 14-28. https://doi.org/10.3390/cosmetics1010014