Optimization of HIV-1 Envelope DNA Vaccine Candidates within Three Different Animal Models, Guinea Pigs, Rabbits and Cynomolgus Macaques
Abstract
:1. Introduction
2. Experimental Section
2.1. DNA Vaccine Plasmids
2.2. Recombinant Glycoprotein (gp)140
2.3. Animal Immunizations
2.4. Anti-Env Antibody ELISA
2.5. Neutralization Assays and env Selection
2.6. Statistical Analysis
3. Results
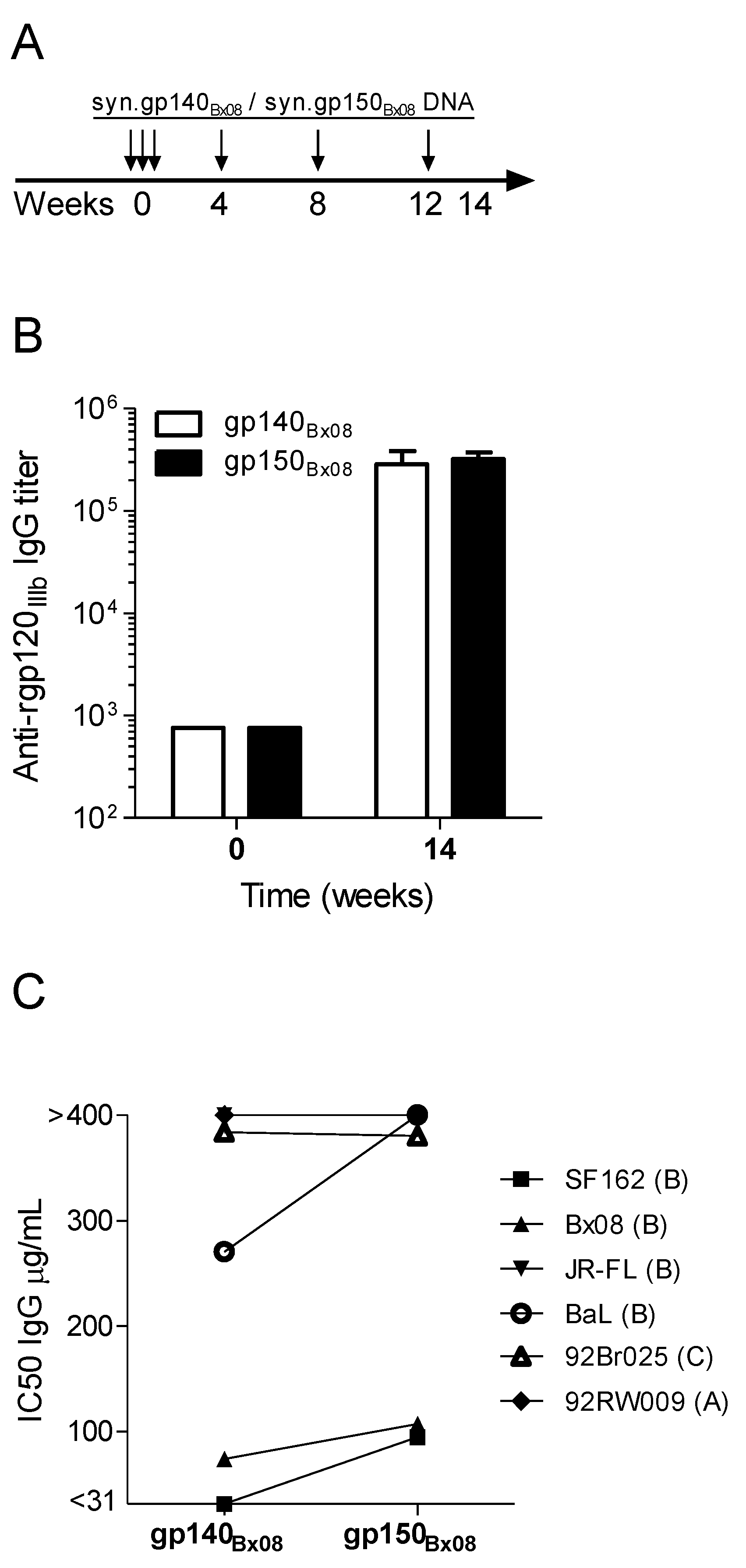
3.1. Immunogenicity and Heterologous Neutralization Elicited by DNA Vaccine Encoding Secreted gp140Bx08 versus Membrane-Bound gp150Bx08 Gene Product
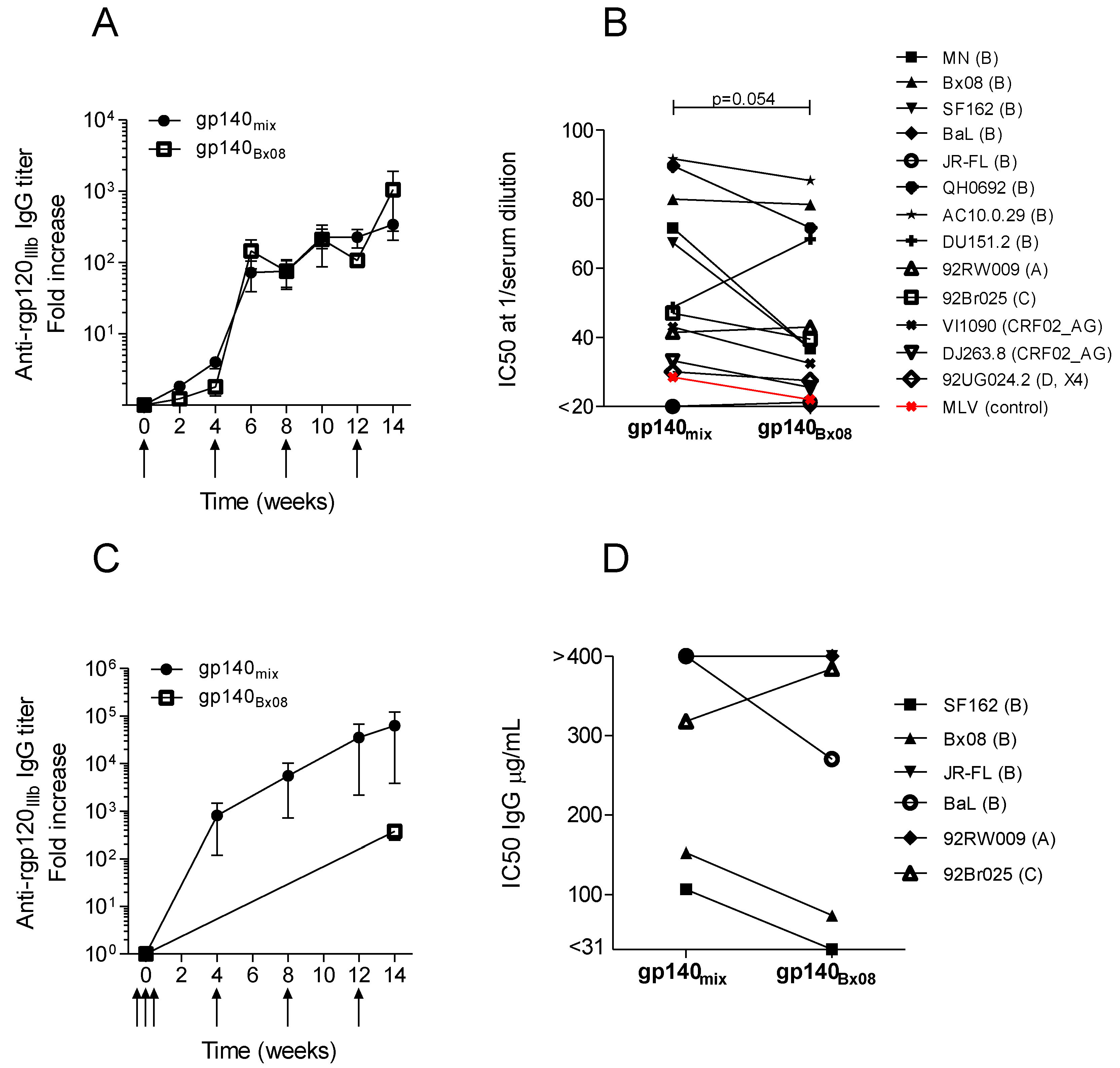
3.2. Neutralization Induced by Trivalent versus Monovalent env DNA Vaccines
| Serum ID | HIV-1 panel (subtype) | ||||
|---|---|---|---|---|---|
| Bx08 § (B) | SF162(B) | BaL(B) | JR-FL(B) | DK1 # (A1D) | |
| ctl31 | >625 | 6 | 103 | 14 | <5 |
| ctl21 | 273 | 625 | 158 | 72 | 38 |
| ctl33 | 178 | 59 | 19 | 20 | <5 |
| ctl27 | 63 | 254 | 106 | 113 | 19 |
| ctl47 | 50 | 31 | >5 | 66 | <5 |
| ctl25 | 47 | 11 | >625 | 19 | 8 |
| ctl38 | 42 | 281 | 438 | 55 | <5 |
| ctl30 | 42 | 16 | 43 | 39 | 123 |
| ctl37 | 32 | 5 | 306 | 22 | 8 |
| ctl18 | 29 | 17 | 57 | 11 | 10 |
| ctl17 | 27 | 11 | 135 | 13 | 15 |
| ctl12 | 23 | 8 | 9 | 6 | 51 |
| ctl43 | 18 | 37 | >5 | 149 | 8 |
| ctl44 | 16 | 6 | >5 | 64 | 13 |
| ctl40 | 15 | 11 | >5 | 64 | <5 |
| ctl11 | 12 | 13 | 102 | 11 | 24 |
| ctl15 | 12 | 13 | 10 | 10 | 11 |
| ctl14 | 8 | >5 | 36 | 299 | 32 |
| ctl23 | 8 | 15 | >625 | 11 | <5 |
| ctl19 | >5 | >5 | >5 | >5 | >5 |
| ctl29 | <5 | 12 | 166 | 60 | 13 |
| ctl24 | <5 | >5 | >5 | >5 | >5 |
| ctl39 | >5 | 15 | >5 | <5 | <5 |
| ctl20 | 5 | >5 | >5 | <5 | <5 |
| ctl46 | >5 | >5 | >5 | <5 | <5 |
| ctl26 | >5 | >5 | >5 | <5 | <5 |
| ctl36 | >5 | <5 | 12 | >5 | <5 |
| ctl22 | <5 | >5 | >5 | <5 | >5 |
| ctl34 | >5 | <5 | >5 | <5 | <5 |
| ctl45 | >5 | <5 | >5 | <5 | <5 |
| ctl28 | >5 | <5 | >5 | <5 | <5 |
| ctl32 | >5 | <5 | <5 | <5 | <5 |
| ctl35 | <5 | >5 | 211 | <5 | <5 |
| ctl13 | <5 | >5 | >5 | <5 | <5 |
| ctl16 | <5 | >5 | >5 | <5 | <5 |
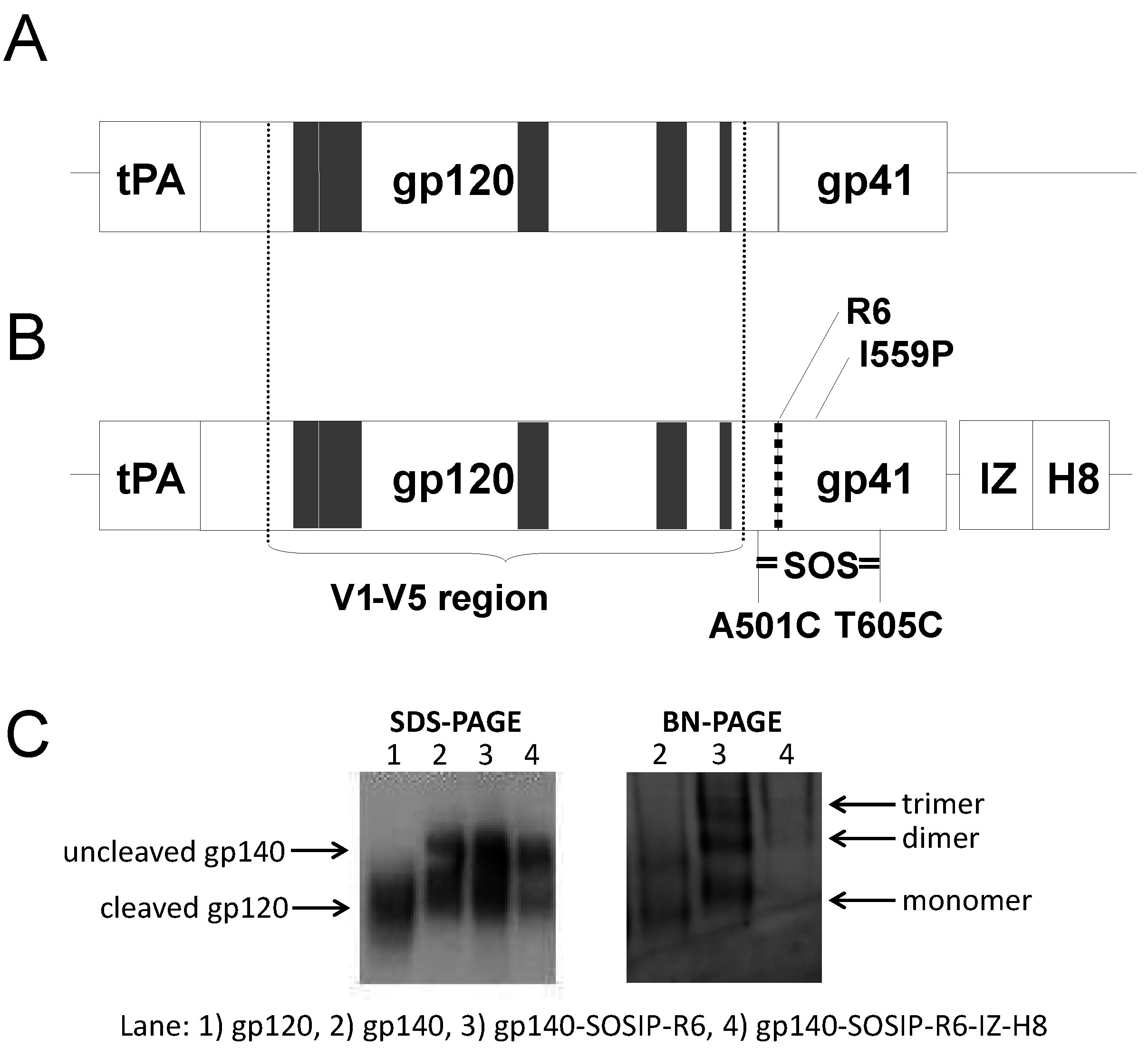
3.3. Trimeric env Modifications of DNA Vaccines Hold Different Immunogenicity in Guinea Pigs and Rabbits
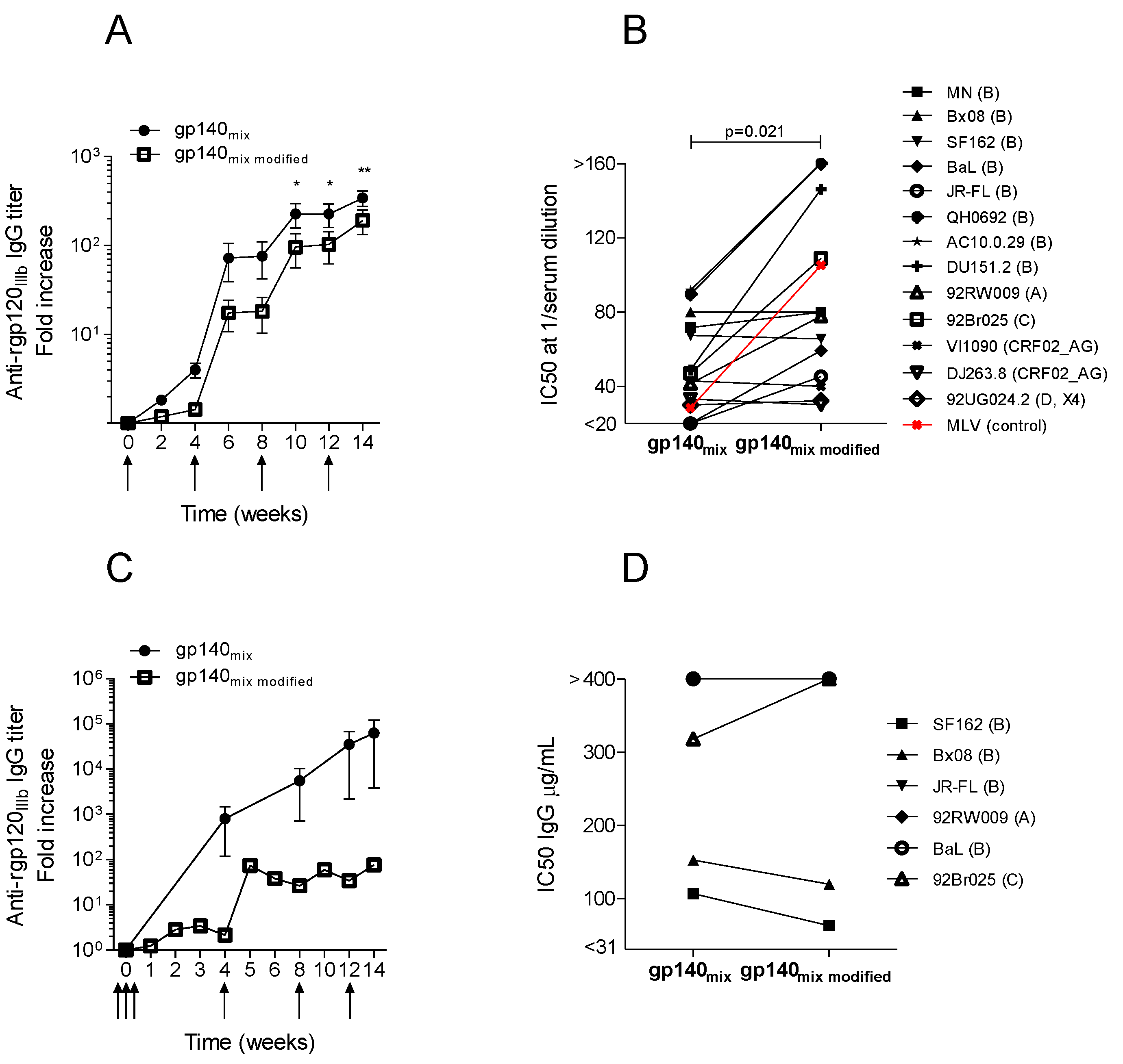
3.4. Immunization with the Same Optimized DNA Vaccine Induces Different Neutralizing Responses in Different Animal Models
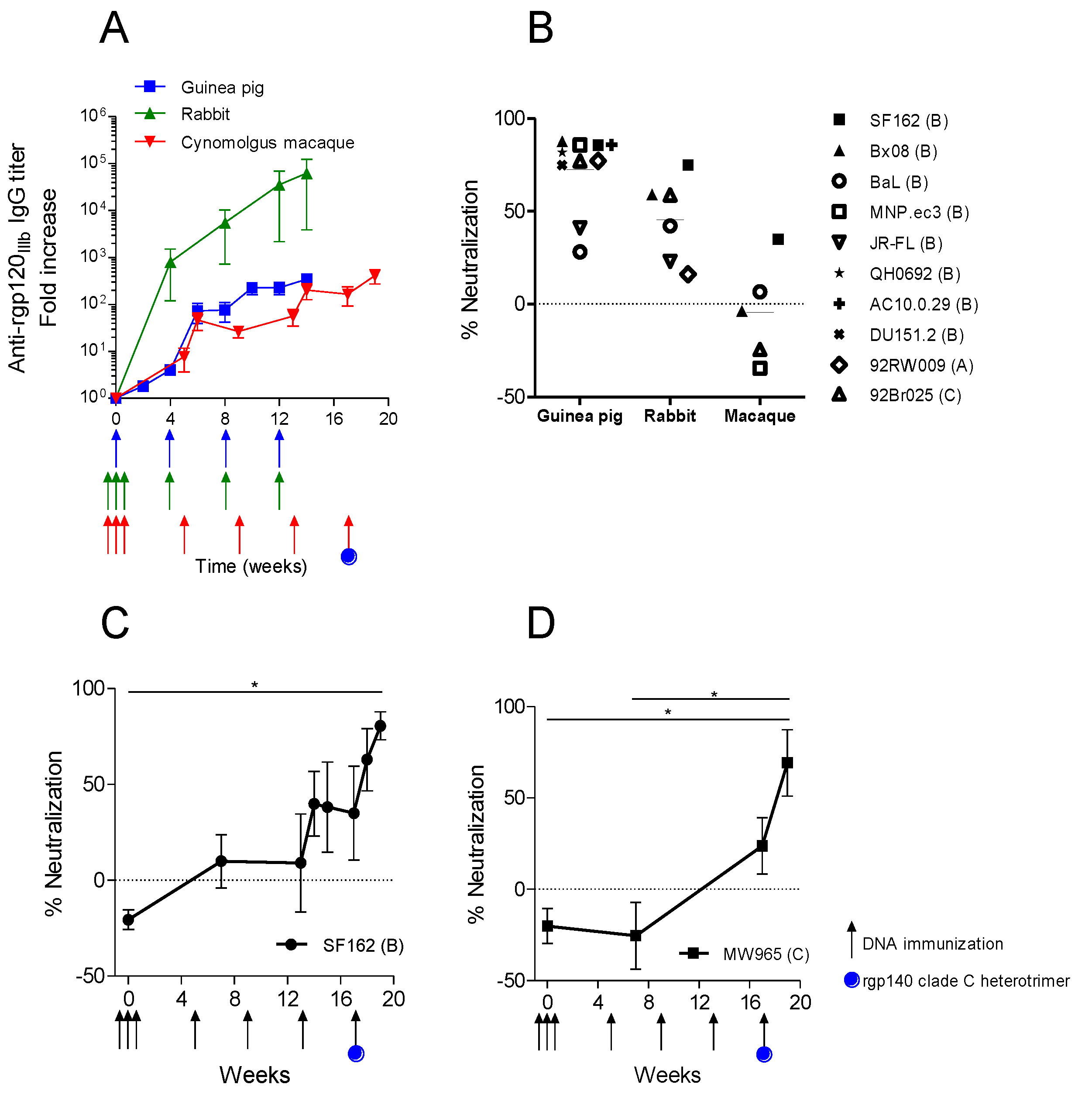
4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Conflict of Interest
References
- Van Gils, M.J.; Sanders, R.W. Broadly neutralizing antibodies against HIV-1: Templates for a vaccine. Virology 2013, 435, 46–56. [Google Scholar] [CrossRef]
- Wren, L.; Kent, S.J. HIV vaccine efficacy trial: Glimmers of hope and the potential role of antibody-dependent cellular cytotoxicity. Hum. Vaccines 2011, 7, 466–473. [Google Scholar] [CrossRef]
- Burton, D.R.; Ahmed, R.; Barouch, D.H.; Butera, S.T.; Crotty, S.; Godzik, A.; Kaufmann, D.E.; McElrath, M.J.; Nussenzweig, M.C.; Pulendran, B.; et al. A blueprint for HIV vaccine discovery. Cell Host Microbe 2012, 12, 396–407. [Google Scholar] [CrossRef]
- Binley, J.M.; Sanders, R.W.; Clas, B.; Schuelke, N.; Master, A.; Guo, Y.; Kajumo, F.; Anselma, D.J.; Maddon, P.J.; Olson, W.C.; et al. A recombinant human immunodeficiency virus type 1 envelope glycoprotein complex stabilized by an intermolecular disulfide bond between the gp120 and gp41 subunits is an antigenic mimic of the trimeric virion-associated structure. J. Virol. 2000, 74, 627–643. [Google Scholar] [CrossRef]
- Sanders, R.W.; Vesanen, M.; Schuelke, N.; Master, A.; Schiffner, L.; Kalyanaraman, R.; Paluch, M.; Berkhout, B.; Maddon, P.J.; Olson, W.C.; et al. Stabilization of the soluble, cleaved, trimeric form of the envelope glycoprotein complex of human immunodeficiency virus type 1. J. Virol. 2002, 76, 8875–8889. [Google Scholar] [CrossRef]
- Yang, X.; Farzan, M.; Wyatt, R.; Sodroski, J. Characterization of stable, soluble trimers containing complete ectodomains of human immunodeficiency virus type 1 envelope glycoproteins. J. Virol. 2000, 74, 5716–5725. [Google Scholar]
- Yang, X.; Florin, L.; Farzan, M.; Kolchinsky, P.; Kwong, P.D.; Sodroski, J.; Wyatt, R. Modifications that stabilize human immunodeficiency virus envelope glycoprotein trimers in solution. J. Virol. 2000, 74, 4746–4754. [Google Scholar]
- Binley, J.M.; Sanders, R.W.; Master, A.; Cayanan, C.S.; Wiley, C.L.; Schiffner, L.; Travis, B.; Kuhmann, S.; Burton, D.R.; Hu, S.L.; et al. Enhancing the proteolytic maturation of human immunodeficiency virus type 1 envelope glycoproteins. J. Virol. 2002, 76, 2606–2616. [Google Scholar] [CrossRef]
- Beddows, S.; Franti, M.; Dey, A.K.; Kirschner, M.; Iyer, S.P.; Fisch, D.C.; Ketas, T.; Yuste, E.; Desrosiers, R.C.; Klasse, P.J.; et al. A comparative immunogenicity study in rabbits of disulfide-stabilized, proteolytically cleaved, soluble trimeric human immunodeficiency virus type 1 gp140, trimeric cleavage-defective gp140 and monomeric gp120. Virology 2007, 360, 329–340. [Google Scholar] [CrossRef]
- Beddows, S.; Schulke, N.; Kirschner, M.; Barnes, K.; Franti, M.; Michael, E.; Ketas, T.; Sanders, R.W.; Maddon, P.J.; Olson, W.C.; et al. Evaluating the immunogenicity of a disulfide-stabilized, cleaved, trimeric form of the envelope glycoprotein complex of human immunodeficiency virus type 1. J. Virol. 2005, 79, 8812–8827. [Google Scholar]
- Kang, Y.K.; Andjelic, S.; Binley, J.M.; Crooks, E.T.; Franti, M.; Iyer, S.P.; Donovan, G.P.; Dey, A.K.; Zhu, P.; Roux, K.H.; et al. Structural and immunogenicity studies of a cleaved, stabilized envelope trimer derived from subtype a HIV-1. Vaccine 2009, 27, 5120–5132. [Google Scholar] [CrossRef]
- Kutzler, M.A.; Weiner, D.B. DNA vaccines: Ready for prime time? Nat. Rev. Genet. 2008, 9, 776–788. [Google Scholar] [CrossRef]
- Liu, M.A. DNA vaccines: An historical perspective and view to the future. Immunol. Rev. 2011, 239, 62–84. [Google Scholar] [CrossRef]
- Hutnick, N.A.; Myles, D.J.; Bian, C.B.; Muthumani, K.; Weiner, D.B. Selected approaches for increasing HIV DNA vaccine immunogenicity in vivo. Curr. Opin. Virol. 2011, 1, 233–240. [Google Scholar] [CrossRef]
- Gudmundsdotter, L.; Wahren, B.; Haller, B.K.; Boberg, A.; Edback, U.; Bernasconi, D.; Butto, S.; Gaines, H.; Imami, N.; Gotch, F.; et al. Amplified antigen-specific immune responses in HIV-1 infected individuals in a double blind DNA immunization and therapy interruption trial. Vaccine 2011, 29, 5558–5566. [Google Scholar] [CrossRef]
- Rosenberg, E.S.; Graham, B.S.; Chan, E.S.; Bosch, R.J.; Stocker, V.; Maenza, J.; Markowitz, M.; Little, S.; Sax, P.E.; Collier, A.C.; et al. Safety and immunogenicity of therapeutic DNA vaccination in individuals treated with antiretroviral therapy during acute/early HIV-1 infection. PLoS One 2010, 5, e10555. [Google Scholar] [CrossRef]
- Wilson, C.C.; Newman, M.J.; Livingston, B.D.; MaWhinney, S.; Forster, J.E.; Scott, J.; Schooley, R.T.; Benson, C.A. Clinical phase 1 testing of the safety and immunogenicity of an epitope-based DNA vaccine in human immunodeficiency virus type 1-infected subjects receiving highly active antiretroviral therapy. Clin. Vaccine Immunol. 2008, 15, 986–994. [Google Scholar] [CrossRef]
- Corbet, S.; Vinner, L.; Hougaard, D.M.; Bryder, K.; Nielsen, H.V.; Nielsen, C.; Fomsgaard, A. Construction, biological activity, and immunogenicity of synthetic envelope DNA vaccines based on a primary, ccr5-tropic, early HIV type I isolate (bx08) with human codons. AIDS Res. Hum. Retroviruses 2000, 16, 1997–2008. [Google Scholar] [CrossRef]
- Vinner, L.; Nielsen, H.V.; Bryder, K.; Corbet, S.; Nielsen, C.; Fomsgaard, A. Gene gun DNA vaccination with rev-independent synthetic HIV-I gp160 envelope gene using mammalian codons. Vaccine 1999, 17, 2166–2175. [Google Scholar] [CrossRef]
- Otten, G.; Schaefer, M.; Doe, B.; Liu, H.; Srivastava, I.; zur Megede, J.; O’Hagan, D.; Donnelly, J.; Widera, G.; Rabussay, D.; et al. Enhancement of DNA vaccine potency in rhesus macaques by electroporation. Vaccine 2004, 22, 2489–2493. [Google Scholar] [CrossRef]
- Widera, G.; Austin, M.; Rabussay, D.; Goldbeck, C.; Barnett, S.W.; Chen, M.; Leung, L.; Otten, G.R.; Thudium, K.; Selby, M.J.; et al. Increased DNA vaccine delivery and immunogenicity by electroporation in vivo. J. Immunol. 2000, 164, 4635–4640. [Google Scholar]
- Hallengard, D.; Brave, A.; Isaguliants, M.; Blomberg, P.; Enger, J.; Stout, R.; King, A.; Wahren, B. A combination of intradermal jet-injection and electroporation overcomes in vivo dose restriction of DNA vaccines. Genet. Vaccines Ther. 2012, 10, 5. [Google Scholar]
- Bragstad, K.; Vinner, L.; Hansen, M.S.; Nielsen, J.; Fomsgaard, A. A polyvalent influenza a DNA vaccine induces heterologous immunity and protects pigs against pandemic a(h1n1)pdm09 virus infection. Vaccine 2013, 31, 2281–2288. [Google Scholar] [CrossRef]
- Wu, T.T.; Johnson, G.; Kabat, E.A. Length distribution of cdrh3 in antibodies. Proteins 1993, 16, 1–7. [Google Scholar] [CrossRef]
- Johnson, G.; Wu, T.T. Preferred cdrh3 lengths for antibodies with defined specificities. Int. Immunol. 1998, 10, 1801–1805. [Google Scholar] [CrossRef]
- Ofek, G.; Tang, M.; Sambor, A.; Katinger, H.; Mascola, J.R.; Wyatt, R.; Kwong, P.D. Structure and mechanistic analysis of the anti-human immunodeficiency virus type 1 antibody 2f5 in complex with its gp41 epitope. J. Virol. 2004, 78, 10724–10737. [Google Scholar]
- Cardoso, R.M.; Zwick, M.B.; Stanfield, R.L.; Kunert, R.; Binley, J.M.; Katinger, H.; Burton, D.R.; Wilson, I.A. Broadly neutralizing anti-HIV antibody 4e10 recognizes a helical conformation of a highly conserved fusion-associated motif in gp41. Immunity 2005, 22, 163–173. [Google Scholar] [CrossRef]
- Saphire, E.O.; Parren, P.W.; Pantophlet, R.; Zwick, M.B.; Morris, G.M.; Rudd, P.M.; Dwek, R.A.; Stanfield, R.L.; Burton, D.R.; Wilson, I.A. Crystal structure of a neutralizing human igg against HIV-1: A template for vaccine design. Science 2001, 293, 1155–1159. [Google Scholar] [CrossRef]
- Pancera, M.; McLellan, J.S.; Wu, X.; Zhu, J.; Changela, A.; Schmidt, S.D.; Yang, Y.; Zhou, T.; Phogat, S.; Mascola, J.R.; et al. Crystal structure of pg16 and chimeric dissection with somatically related pg9: Structure-function analysis of two quaternary-specific antibodies that effectively neutralize HIV-1. J. Virol. 2010, 84, 8098–8110. [Google Scholar]
- Pejchal, R.; Walker, L.M.; Stanfield, R.L.; Phogat, S.K.; Koff, W.C.; Poignard, P.; Burton, D.R.; Wilson, I.A. Structure and function of broadly reactive antibody pg16 reveal an h3 subdomain that mediates potent neutralization of HIV-1. Proc. Natl. Acad. Sci. USA 2010, 107, 11483–11488. [Google Scholar] [CrossRef]
- Dewan, M.Z.; Terashima, K.; Ahmed, S.; Ohba, K.; Taruishi, M.; Yamamoto, N. Mouse serum factor(s) down-modulate the cd4 and cxcr4 molecules on human T cells conferring resistance to HIV infection in nog mice. Med. Microbiol. Immunol. 2005, 194, 175–180. [Google Scholar] [CrossRef]
- Moog, C.; Fleury, H.J.; Pellegrin, I.; Kirn, A.; Aubertin, A.M. Autologous and heterologous neutralizing antibody responses following initial seroconversion in human immunodeficiency virus type 1—Infected individuals. J. Virol. 1997, 71, 3734–3741. [Google Scholar]
- Vinner, L.; Therrien, D.; Wee, E.; Laursen, I.; Hanke, T.; Corbet, S.L.; Fomsgaard, A. Immune response in rhesus macaques after mixed modality immunisations with DNA, recombinant adenovirus and recombinant gp120 from human immunodeficiency virus type 1. Acta Pathol. Microbiol. Immunol. Scand. 2006, 114, 690–699. [Google Scholar]
- Vinner, L.; Wee, E.G.; Patel, S.; Corbet, S.; Gao, G.P.; Nielsen, C.; Wilson, J.M.; Ertl, H.C.; Hanke, T.; Fomsgaard, A. Immunogenicity in Mamu-A*01 rhesus macaques of a CCR5-tropic human immunodeficiency virus type 1 envelope from the primary isolate (Bx08) after synthetic DNA prime and recombinant adenovirus 5 boost. J. Gen. Virol. 2003, 84, 203–213. [Google Scholar] [CrossRef]
- Haas, J.; Park, E.C.; Seed, B. Codon usage limitation in the expression of HIV-1 envelope glycoprotein. Curr. Biol. 1996, 6, 315–324. [Google Scholar] [CrossRef]
- Sanders, R.W.; Schiffner, L.; Master, A.; Kajumo, F.; Guo, Y.; Dragic, T.; Moore, J.P.; Binley, J.M. Variable-loop-deleted variants of the human immunodeficiency virus type 1 envelope glycoprotein can be stabilized by an intermolecular disulfide bond between the gp120 and gp41 subunits. J. Virol. 2000, 74, 5091–5100. [Google Scholar]
- Harbury, P.B.; Zhang, T.; Kim, P.S.; Alber, T. A switch between two-, three-, and four-stranded coiled coils in gcn4 leucine zipper mutants. Science 1993, 262, 1401–1407. [Google Scholar]
- Harbury, P.B.; Kim, P.S.; Alber, T. Crystal structure of an isoleucine-zipper trimer. Nature 1994, 371, 80–83. [Google Scholar] [CrossRef]
- Melchers, M.; Bontjer, I.; Tong, T.; Chung, N.P.; Klasse, P.J.; Eggink, D.; Montefiori, D.C.; Gentile, M.; Cerutti, A.; Olson, W.C.; et al. Targeting HIV-1 envelope glycoprotein trimers to B cells by using april improves antibody responses. J. Virol. 2012, 86, 2488–2500. [Google Scholar]
- Melchers, M.; Matthews, K.; de Vries, R.P.; Eggink, D.; van Montfort, T.; Bontjer, I.; van de Sandt, C.; David, K.; Berkhout, B.; Moore, J.P.; et al. A stabilized HIV-1 envelope glycoprotein trimer fused to CD40 ligand targets and activates dendritic cells. Retrovirology 2011, 8, 48. [Google Scholar] [CrossRef]
- Gomez Roman, V.R.; Vinner, L.; Grevstad, B.; Hansen, J.J.; Wegmann, F.; Spetz, A.L.; Fomsgaard, A. Development of standard operating procedures to obtain longitudinal vaginal specimens from nulliparous rabbits as part of HIV vaccine mucosal immunogenicity studies. J. Immunol. Method. 2010, 363, 29–41. [Google Scholar] [CrossRef]
- Heyndrickx, L.; Heath, A.; Sheik-Khalil, E.; Alcami, J.; Bongertz, V.; Jansson, M.; Malnati, M.; Montefiori, D.; Moog, C.; Morris, L.; et al. International network for comparison of HIV neutralization assays: The neutnet report II. PLoS One 2012, 7, e36438. [Google Scholar] [CrossRef]
- Montefiori, D.C. Measuring HIV neutralization in a luciferase reporter gene assay. Meth. Mol. Biol. 2009, 485, 395–405. [Google Scholar] [CrossRef]
- Fenyo, E.M.; Heath, A.; Dispinseri, S.; Holmes, H.; Lusso, P.; Zolla-Pazner, S.; Donners, H.; Heyndrickx, L.; Alcami, J.; Bongertz, V.; et al. International network for comparison of HIV neutralization assays: The neutnet report. PLoS One 2009, 4, e4505. [Google Scholar] [CrossRef]
- Nielsen, C.M.; Bygbjerg, I.C.; Vestergaard, B.F. Detection of HIV antigens in eluates from whole blood collected on filterpaper. Lancet 1987, 1, 566–567. [Google Scholar]
- Corbet, S.; Nielsen, H.V.; Vinner, L.; Lauemoller, S.; Therrien, D.; Tang, S.; Kronborg, G.; Mathiesen, L.; Chaplin, P.; Brunak, S.; et al. Optimization and immune recognition of multiple novel conserved hla-a2, human immunodeficiency virus type 1-specific ctl epitopes. J. Gen. Virol. 2003, 84, 2409–2421. [Google Scholar] [CrossRef]
- Fomsgaard, A.; Vinner, L.; Therrien, D.; Jorgensen, L.B.; Nielsen, C.; Mathiesen, L.; Pedersen, C.; Corbet, S. Full-length characterization of a1/d intersubtype recombinant genomes from a therapy-induced HIV type 1 controller during acute infection and his noncontrolling partner. AIDS Res. Hum. Retroviruses 2008, 24, 463–472. [Google Scholar] [CrossRef]
- Wang, S.; Pal, R.; Mascola, J.R.; Chou, T.H.; Mboudjeka, I.; Shen, S.; Liu, Q.; Whitney, S.; Keen, T.; Nair, B.C.; et al. Polyvalent HIV-1 env vaccine formulations delivered by the DNA priming plus protein boosting approach are effective in generating neutralizing antibodies against primary human immunodeficiency virus type 1 isolates from subtypes A, B, C, D and E. Virology 2006, 350, 34–47. [Google Scholar] [CrossRef]
- Catanzaro, A.T.; Roederer, M.; Koup, R.A.; Bailer, R.T.; Enama, M.E.; Nason, M.C.; Martin, J.E.; Rucker, S.; Andrews, C.A.; Gomez, P.L.; et al. Phase 1 clinical evaluation of a six-plasmid multiclade HIV-1 DNA candidate vaccine. Vaccine 2007, 25, 4085–4092. [Google Scholar] [CrossRef]
- Rollman, E.; Brave, A.; Boberg, A.; Gudmundsdotter, L.; Engstrom, G.; Isaguliants, M.; Ljungberg, K.; Lundgren, B.; Blomberg, P.; Hinkula, J.; et al. The rationale behind a vaccine based on multiple HIV antigens. Microbes Infect. 2005, 7, 1414–1423. [Google Scholar]
- Eller, M.A.; Eller, L.A.; Opollo, M.S.; Ouma, B.J.; Oballah, P.O.; Galley, L.; Karnasuta, C.; Kim, S.R.; Robb, M.L.; Michael, N.L.; et al. Induction of HIV-specific functional immune responses by a multiclade HIV-1 DNA vaccine candidate in healthy ugandans. Vaccine 2007, 25, 7737–7742. [Google Scholar] [CrossRef]
- Jelley-Gibbs, D.M.; Swain, S.L. T cell memory generation in the face of persistent antigen presentation. Curr. Immunol. Rev. 2007, 3, 240–250. [Google Scholar] [CrossRef]
- Farrell, H.M., Jr.; Jimenez-Flores, R.; Bleck, G.T.; Brown, E.M.; Butler, J.E.; Creamer, L.K.; Hicks, C.L.; Hollar, C.M.; Ng-Kwai-Hang, K.F.; Swaisgood, H.E. Nomenclature of the proteins of cows’ milk—Sixth revision. J. Dairy Sci. 2004, 87, 1641–1674. [Google Scholar] [CrossRef]
- Jeffs, S.A.; Goriup, S.; Kebble, B.; Crane, D.; Bolgiano, B.; Sattentau, Q.; Jones, S.; Holmes, H. Expression and characterisation of recombinant oligomeric envelope glycoproteins derived from primary isolates of HIV-1. Vaccine 2004, 22, 1032–1046. [Google Scholar] [CrossRef]
- Muster, T.; Steindl, F.; Purtscher, M.; Trkola, A.; Klima, A.; Himmler, G.; Ruker, F.; Katinger, H. A conserved neutralizing epitope on gp41 of human immunodeficiency virus type 1. J. Virol. 1993, 67, 6642–6647. [Google Scholar]
- Stiegler, G.; Kunert, R.; Purtscher, M.; Wolbank, S.; Voglauer, R.; Steindl, F.; Katinger, H. A potent cross-clade neutralizing human monoclonal antibody against a novel epitope on gp41 of human immunodeficiency virus type 1. AIDS Res. Hum. Retroviruses 2001, 17, 1757–1765. [Google Scholar] [CrossRef]
- Zwick, M.B.; Jensen, R.; Church, S.; Wang, M.; Stiegler, G.; Kunert, R.; Katinger, H.; Burton, D.R. Anti-human immunodeficiency virus type 1 (HIV-1) antibodies 2f5 and 4e10 require surprisingly few crucial residues in the membrane-proximal external region of glycoprotein gp41 to neutralize HIV-1. J. Virol. 2005, 79, 1252–1261. [Google Scholar]
- Cho, M.W.; Kim, Y.B.; Lee, M.K.; Gupta, K.C.; Ross, W.; Plishka, R.; Buckler-White, A.; Igarashi, T.; Theodore, T.; Byrum, R.; et al. Polyvalent envelope glycoprotein vaccine elicits a broader neutralizing antibody response but is unable to provide sterilizing protection against heterologous simian/human immunodeficiency virus infection in pigtailed macaques. J. Virol. 2001, 75, 2224–2234. [Google Scholar] [CrossRef]
- Seaman, M.S.; Leblanc, D.F.; Grandpre, L.E.; Bartman, M.T.; Montefiori, D.C.; Letvin, N.L.; Mascola, J.R. Standardized assessment of nab responses elicited in rhesus monkeys immunized with single- or multi-clade HIV-1 envelope immunogens. Virology 2007, 367, 175–186. [Google Scholar] [CrossRef]
- Moog, C.; Spenlehauer, C.; Fleury, H.; Heshmati, F.; Saragosti, S.; Letourneur, F.; Kirn, A.; Aubertin, A.M. Neutralization of primary human immunodeficiency virus type 1 isolates: A study of parameters implicated in neutralization in vitro. AIDS Res. Hum. Retroviruses 1997, 13, 19–27. [Google Scholar] [CrossRef]
- Kulkarni, V.; Jalah, R.; Ganneru, B.; Bergamaschi, C.; Alicea, C.; von Gegerfelt, A.; Patel, V.; Zhang, G.M.; Chowdhury, B.; Broderick, K.E.; et al. Comparison of immune responses generated by optimized DNA vaccination against SIV antigens in mice and macaques. Vaccine 2011, 29, 6742–6754. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Borggren, M.; Vinner, L.; Andresen, B.S.; Grevstad, B.; Repits, J.; Melchers, M.; Elvang, T.L.; Sanders, R.W.; Martinon, F.; Dereuddre-Bosquet, N.; et al. Optimization of HIV-1 Envelope DNA Vaccine Candidates within Three Different Animal Models, Guinea Pigs, Rabbits and Cynomolgus Macaques. Vaccines 2013, 1, 305-327. https://doi.org/10.3390/vaccines1030305
Borggren M, Vinner L, Andresen BS, Grevstad B, Repits J, Melchers M, Elvang TL, Sanders RW, Martinon F, Dereuddre-Bosquet N, et al. Optimization of HIV-1 Envelope DNA Vaccine Candidates within Three Different Animal Models, Guinea Pigs, Rabbits and Cynomolgus Macaques. Vaccines. 2013; 1(3):305-327. https://doi.org/10.3390/vaccines1030305
Chicago/Turabian StyleBorggren, Marie, Lasse Vinner, Betina Skovgaard Andresen, Berit Grevstad, Johanna Repits, Mark Melchers, Tara Laura Elvang, Rogier W Sanders, Frédéric Martinon, Nathalie Dereuddre-Bosquet, and et al. 2013. "Optimization of HIV-1 Envelope DNA Vaccine Candidates within Three Different Animal Models, Guinea Pigs, Rabbits and Cynomolgus Macaques" Vaccines 1, no. 3: 305-327. https://doi.org/10.3390/vaccines1030305




