Chemical Ecology of the Colorado Potato Beetle, Leptinotarsa decemlineata (Say) (Coleoptera: Chrysomelidae), and Potential for Alternative Control Methods
Abstract
:1. Introduction
| Category | ControlApproaches | References |
|---|---|---|
| Cultural modifications | Crop rotation | [14,18] |
| Delayed or early planting | [15] | |
| Trap crops | [15,19] | |
| Physical barriers | Straw mulch ground cover | [20] |
| Traps with plastic trenches | [21] | |
| Mechanical control | Use of propane flamers | [22] |
| Vacuum collection devices | [23] | |
| Biological control | Predators or parasitoids | [24,25] |
| Nematodes | [26,27] | |
| Fungi | [28] | |
| Genetical modifications | Transgenic plants with Bacillus thuringiensis | [29,30] |
| Molecular biology | RNA interference | [31] |
2. Chemicals Involved in Host Plant Selection
2.1. Role of Glycoalkaloids in Acceptance, Repellency and Defense
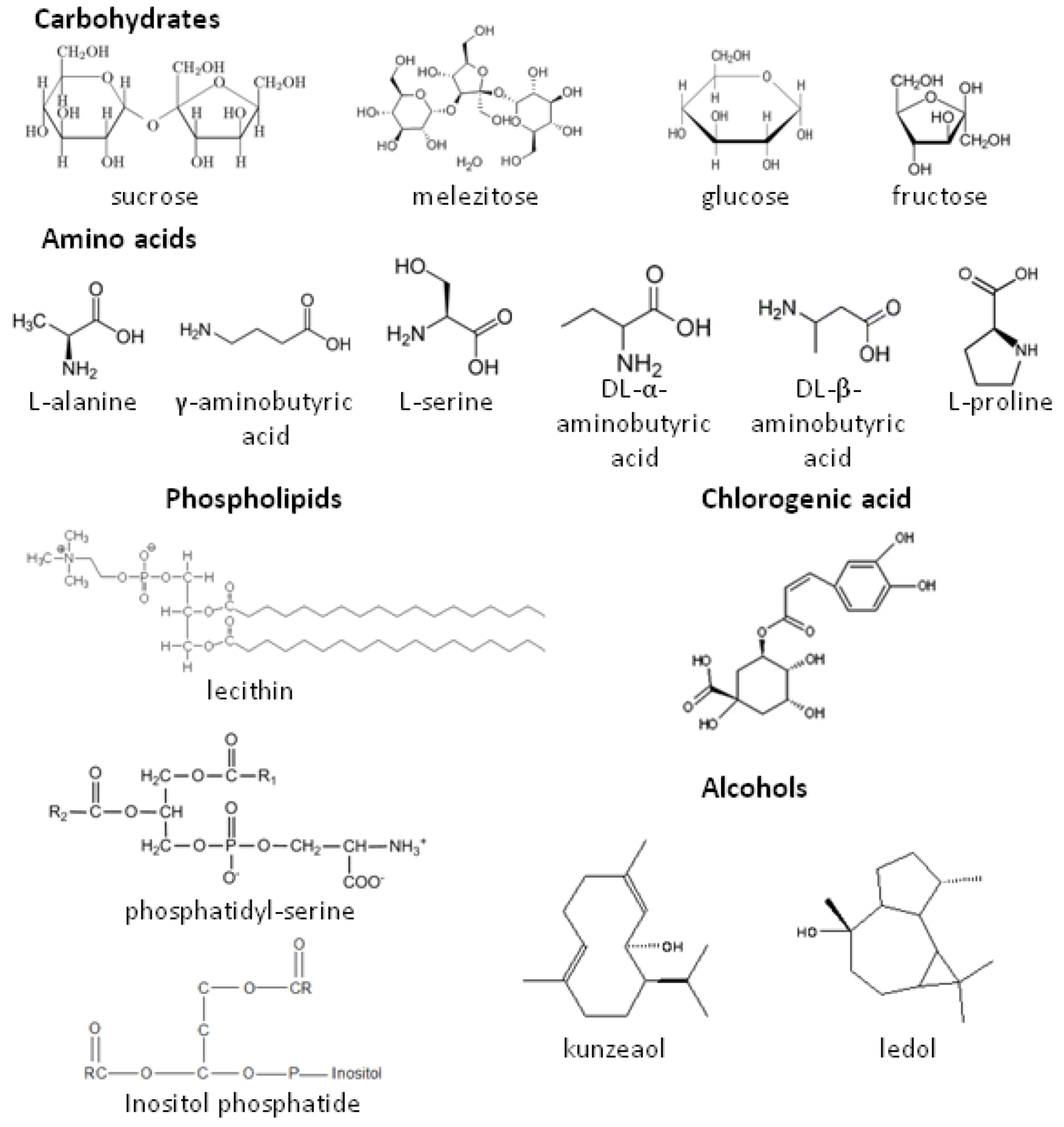
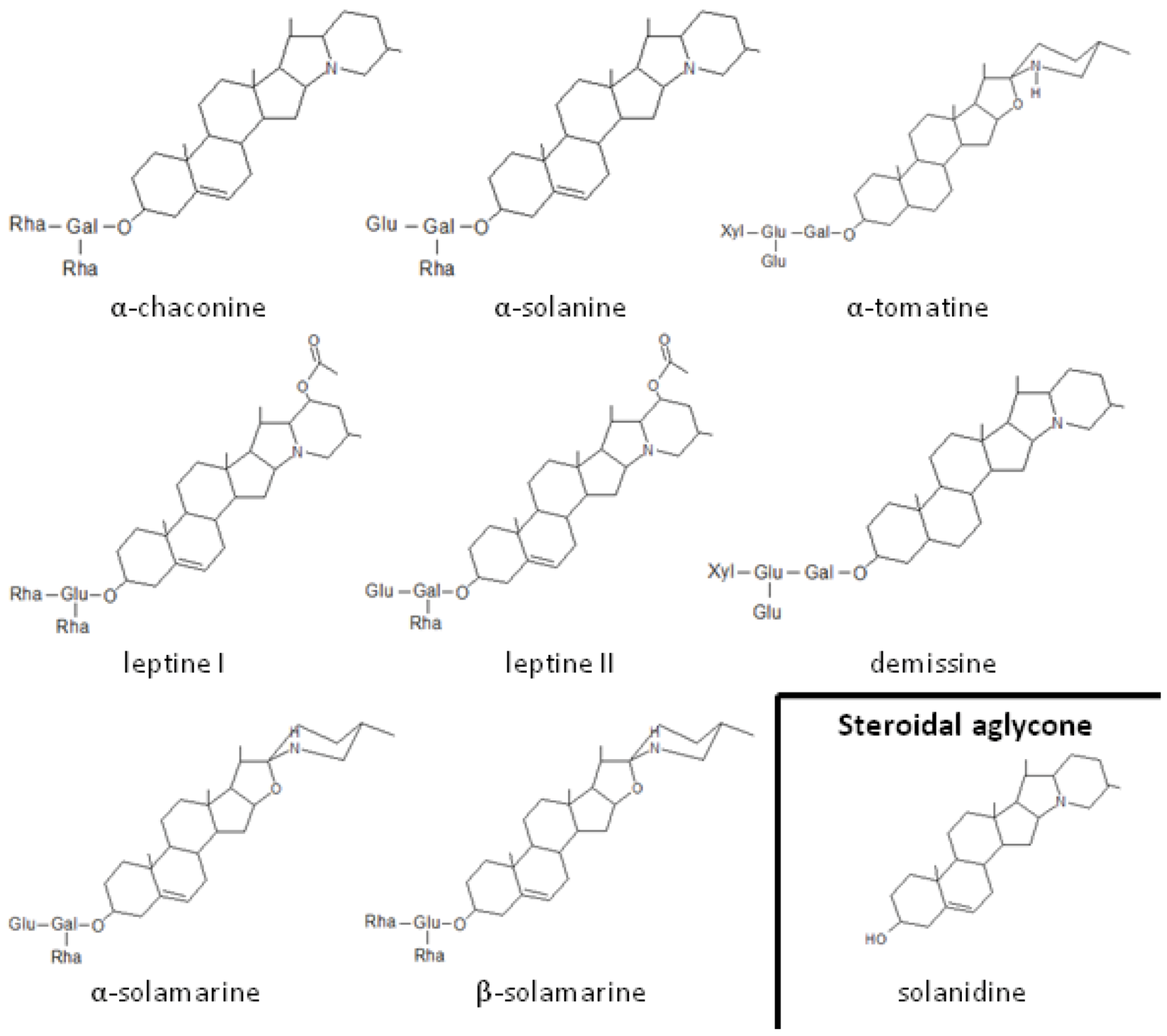
2.2. Role of Plant Volatiles in CPB Orientation

3. Intraspecific Communication in CPB
3.1. Sexual Recognition and Sex Pheromone
3.2. Aggregation Pheromone and Aggregation Behavior

4. Interspecific Interactions
4.1. Defensive Chemicals
4.2. Volatile Perception by Predators
4.3. Interactions between Different Pest Species
5. Semiochemical-Based IPM Strategies
5.1. Disorientation of CPB Adults with Masking Odors
| Name | Schemas | Brief description |
|---|---|---|
| Masking odors |  | CPB cannot detect its host plant because GLVs of potatoes were mixing with others odors from cultures intercropping |
| Trap crops |  | CPB are attracted in a specific part of the field with semiochemicals and only this part is treated to eliminate the beetles |
| Antifeedants |  | Chemical molecules were sprayed on potatoes to deter CPB feeding and damages are lesser on potato foliage |
| Genetic manipulations |  | Potatoes are genetically modified to detect CPB presence and to express more their defense mechanisms to prevent CPB colonization |
5.2. Use of Attractants and Aggregation Pheromone
5.3. Antifeedants
5.4. Genetic Manipulations
6. Conclusions
Acknowledgements
References and Notes
- Casagrande, R.A. The Colorado potato beetle: 125 years of mismanagement. Bull. Entomol. Soc. Am. 1987, 33, 142–150. [Google Scholar]
- Riley, C.V. Seven Annual Report on the Noxious, Beneficial, and Other Insect of the State of Missouri; Regan & Carter: Jefferson City, MS, USA, 1875. [Google Scholar]
- du Chatenet, G. Coléoptères Phytophages d'Europe : Chrysomelidae; N.A.P Editions: Verrières Le Buisson, France, 2002; Vol. 2, p. 258. [Google Scholar]
- Weber, D.C. Colorado beetle: Pest on the move. Pesticide Outlook 2003, 14, 256–259. [Google Scholar]
- Hare, J.D. Impact of defoliation by the Colorado potato beetle on potato yields. J. Econ. Entomol. 1980, 73, 369–373. [Google Scholar]
- Hare, J.D. Ecology and management of the Colorado potato beetle. Annu. Rev. Entomol. 1990, 35, 81–100. [Google Scholar] [CrossRef]
- Harcourt, D.G. Population dynamics of Leptinotarsa decemlineata (Say) in eastern Ontario. III. Major population processes. Can. Entomol. 1971, 103, 1049–1061. [Google Scholar] [CrossRef]
- Ferro, D.N.; Logan, J.A.; Voss, R.H.; Elkinton, J.S. Colorado potato beetle (Coleoptera: Chrysomelidae) temperature-dependent growth and feeding rates. Environ. Entomol. 1985, 14, 343–348. [Google Scholar]
- Walgenbach, J.F.; Wyman, J.A. Colorado potato beetle (Coleoptera: Chrysomelidae) development in relation to temperature in Wisconsin. Ann. Entomol. Soc. Am. 1984, 77, 604–609. [Google Scholar]
- Alyokhin, A.; Baker, M.; Mota-Sanchez, D.; Dively, G.; Grafius, E. Colorado potato beetle resistance to insecticides. Am. J. Potato Res. 2008, 85, 395–413. [Google Scholar] [CrossRef]
- Alyokhin, A. Colorado potato beetle management on potatoes: Current challenges and future prospects. Fruit Vegetable Cereal Sci. Biotechnol. 2009, 3, 10–19. [Google Scholar]
- Bishop, B.A.; Grafius, E.J. Insecticide resistance in the Colorado potato beetle. In Chrysomelidae Biology; Jolivet, P., Hsiao, T.H., Eds.; SBP Academic Publishing: Amsterdam, The Netherlands, 1996; Volume 1, pp. 355–377. [Google Scholar]
- Clark, J.M.; Argentine, J.A. Biochemical mechanisms of insecticide resistance in the Colorado potato beetle. In Advances in Potato Pest Biology and Management; Zehnder, G.W., Powelson, M.L., Jansson, R.K., Raman, K.V., Eds.; APS Press: St. Paul, Minnesota, USA, 1994; pp. 294–308. [Google Scholar]
- Weisz, R.; Smilowitz, Z.; Christ, B. Distance, rotation, and border crops affect Colorado potato beetle (Coleoptera, Chrysomelidae) colonization and population-density and early blight (Alternaria-solani) severity in rotated potato fields. J. Econ. Entomol. 1994, 87, 723–729. [Google Scholar]
- Weber, D.C.; Ferro, D.N.; Buonaccorsi, J.; Hazzard, R.V. Disrupting spring colonization of Colorado potato beetle to nonrotated potato fields. Entomol. Exp. Appl. 1994, 73, 39–50. [Google Scholar] [CrossRef]
- Coombs, J.J.; Douches, D.S.; Li, W.B.; Grafius, E.J.; Pett, W.L. Combining engineered (Bt-cry3A) and natural resistance mechanisms in potato for control of Colorado potato beetle. J. Am. Soc. Hortic. Sci. 2002, 127, 62–68. [Google Scholar]
- Grafius, E.J.; Douches, D.S. The present and future role of insect resistant, genetically modified potato cultivars in potato IPM. In Integration of Insect-Resistant GM Crops within IPM Programs; Romeis, J., Shelton, A.M., Kennedy, G.G., Eds.; Springer: New York, NY, USA, 2008; pp. 195–222. [Google Scholar]
- Lashomb, J.H.; Ng, Y.S. Colonization by Colorado potato beetles, Leptinotarsa decemlineata (Say) (Coleoptera: Chrysomelidae), in rotated and nonrotated potato fields. Environ. Entomol. 1984, 13, 1352–1356. [Google Scholar]
- Hoy, C.W.; Vaughn, T.T.; East, D.A. Increasing the effectiveness of spring trap crops for Leptinotarsa decemlineata. Entomol. Exp. Appl. 2000, 96, 193–204. [Google Scholar]
- Zehnder, G.W.; Hough-Goldstein, J. Colorado potato beetle (Coleoptera: Chrysomelidae) population development and effects on yield of potatoes with and without straw mulch. J. Econ. Entomol. 1989, 83, 1982–1987. [Google Scholar]
- Misener, G.C.; Boiteau, G.; McMillan, L.P. A plastic-lining trenching device for the control of Colorado potato beetle - beetle excluder. Am. Potato J. 1993, 70, 903–908. [Google Scholar] [CrossRef]
- Pelletier, Y.; McLeod, C.D.; Bernard, G. Description of sublethals injuries caused to the Colorado potato beetle (Coleoptera, Chrysomelidae) by propane flamer treatement. J. Econ. Entomol. 1995, 88, 1203–1205. [Google Scholar]
- Boiteau, G.; Misener, G.C.; Singh, R.P.; Bernard, G. Evaluation of vacuum collector for insect pest-control in potato. Am. Potato J. 1992, 69, 157–166. [Google Scholar] [CrossRef]
- Hough-Goldstein, J.A.; Heimpel, G.E.; Bechmann, H.E.; Mason, C.E. Arthropod natural enemies of the Colorado potato beetle. Crop. Prot. 1993, 12, 324–334. [Google Scholar] [CrossRef]
- Ferro, D.N. Biological control of the Colorado potato beetle. In Advances in Potato Pest Biology and Management; Zehnder, G.W., Jansson, R.K., Powelson, M.L., Raman, K.V., Eds.; APS Press: St. Paul, MI, USA, 1994; pp. 357–375. [Google Scholar]
- Berry, R.E.; Liu, J.; Reed, G. Comparison of endemic and exotic entomopathogenic nematode species for control of Colorado potato beetle (Coleoptera:Chrysomelidae). J. Econ. Entomol. 1997, 90, 1528–1533. [Google Scholar]
- Ebrahimi, L.; Niknam, G.; Lewis, E.E. Lethal and sublethal effects of Iranian isolates of Steinernema feltiae and Heterorhabditis bacteriophora on the Colorado potato beetle, Leptinotarsa decemlineata. Biocontrol 2011, 56, 781–788. [Google Scholar] [CrossRef]
- Wraight, S.P.; Ramos, M.E. Application parameters affecting field efficacy of Beauveria bassiana foliar treatments against Colorado potato beetle Leptinotarsa decemlineata. Biol. Control 2002, 23, 164–178. [Google Scholar] [CrossRef]
- Perlak, F.J.; Stone, T.B.; Muskopf, Y.M.; Petersen, L.J.; Parker, G.B.; McPherson, S.A.; Wyman, J.; Love, S.; Reed, G.; Biever, D.; Fischhoff, D.A. Genetically improved potatoes - protection from damage by Colorado potato beetles. Plant Mol. Biol. 1993, 22, 313–321. [Google Scholar] [CrossRef]
- Coombs, J.J.; Douches, D.S.; Li, W.B.; Grafius, E.J.; Pett, W.L. Field evaluation of natural, engineered, and combined resistance mechanisms in potato for control of colorado potato beetle. J. Am. Soc. Hortic. Sci. 2003, 128, 219–224. [Google Scholar]
- Zhu, F.; Xu, J.J.; Palli, R.; Ferguson, J.; Palli, S.R. Ingested RNA interference for managing the populations of the Colorado potato beetle, Leptinotarsa decemlineata. Pest Manag. Sci. 2011, 67, 175–182. [Google Scholar] [CrossRef]
- Hsiao, T.H.; Fraenkel, G. Selection and specificity of the Colorado potato beetle for solanaceous and nonsolanaceous plants. Ann. Entomol. Soc. Am. 1968, 61, 493–503. [Google Scholar]
- Harrison, G.D. Host-plant discrimination and evolution of feeding preference in the Colorado potato beetle Leptinotarsa-decemlineata. Physiol. Entomol. 1987, 12, 407–415. [Google Scholar] [CrossRef]
- Hsiao, T.H.; Fraenkel, G. The influence of nutrient chemicals on the feeding behavior of the Colorado potato beetle, Leptinotarsa decemlineata (Coleoptera:Chrysomelidae). Ann. Entomol. Soc. Am. 1968, 61, 44–54. [Google Scholar]
- Hsiao, T.H. Chemical basis of host selection and plant resistance in oligophagous insects. Entomol. Exp. Appl. 1969, 12, 777–788. [Google Scholar] [CrossRef]
- Szafranek, B.; Chrapkowska, K.; Waligora, D.; Palavinskas, R.; Banach, A.; Szafranek, J. Leaf surface sesquiterpene alcohols of the potato (Solanum tuberosum) and their influence on Colorado beetle (Leptinotarsa decemlineata Say) feeding. J. Agric. Food Chem. 2006, 54, 7729–7734. [Google Scholar] [CrossRef]
- Chen, Z.B.; Miller, A.R. Steroidal alkaloids in solanaceous vegetable crops. Horticultural Reviews 2001, 25, 171–196. [Google Scholar]
- Herb, S.F.; Fitzpatrick, T.J.; Osman, S.F. Separation of potato glycoalkaloids by gas chromatography. J. Agric. Food Chem. 1975, 23, 520–523. [Google Scholar] [CrossRef]
- Flanders, K.L.; Hawkes, J.G.; Radcliffe, E.B.; Lauer, F.I. Insect resistance in potatoes - sources, evolutionary relantionships, morphological and chemical defenses, and ecogeographical associations. Euphytica 1992, 61, 83–111. [Google Scholar] [CrossRef]
- Tingey, W.M. Glycoalkaloids as pest resistance factors. Am. Potato J. 1984, 61, 157–167. [Google Scholar] [CrossRef]
- Sinden, S.L.; Sanford, L.L.; Cantelo, W.W.; Deahl, K.L. Leptine glycoalkaloids and resistance to the Colorado potato beetle (Coleoptera: Chrysomelidae) in Solanum chacoense. Environ. Entomol. 1986, 15, 1057–1062. [Google Scholar]
- Sinden, S.L.; Sanford, L.L.; Cantelo, W.W.; Deahl, K.L. Bioassays of segregating plants - a strategy for studying chemical defenses. J. Chem. Ecol. 1988, 14, 1941–1950. [Google Scholar] [CrossRef]
- Yencho, G.C.; Kowalski, S.P.; Kennedy, G.G.; Sanford, L.L. Segregation of leptine glycoalkaloids and resistance to Colorado potato beetle (Leptinotarsa decemlineata (Say)) in F2 Solanum tuberosum (4x) x S-chacoense (4x) potato progenies. Am. J. Potato Res. 2000, 77, 167–178. [Google Scholar] [CrossRef]
- Hollister, B.; Dickens, J.C.; Perez, F.; Deahl, K.L. Differential neurosensory responses of adult Colorado potato beetle, Leptinotarsa decemlineata, to glycoalkaloids. J. Chem. Ecol. 2001, 27, 1105–1118. [Google Scholar] [CrossRef]
- Mitchell, B.K.; Harrison, G.D. Characterization of galeal chemosensilla in the adult Colorado beetle, Leptinotarsa decemlineata. Physiol. Entomol. 1984, 9, 49–56. [Google Scholar] [CrossRef]
- Mitchell, B.K. Interactions of alkaloids with galeal chemosensory cells of Colorado potato beetle. J. Chem. Ecol. 1987, 13, 2009–2022. [Google Scholar] [CrossRef]
- Hare, J.D. Growth of Leptinotarsa-decemlineata larvae in response to simultaneous variation in protein and glycoalkaloid concentration. J. Chem. Ecol. 1987, 13, 39–46. [Google Scholar] [CrossRef]
- Kowalski, S.P.; Domek, J.M.; Deahl, K.L.; Sanford, L.L. Performance of Colorado potato beetle larvae, Leptinotarsa decemlineata (Say), reared on synthetic diets supplemented with Solanum glycoalkaloids. Am. J. Potato Res. 1999, 76, 305–312. [Google Scholar] [CrossRef]
- Lyytinen, A.; Lindstrom, L.; Mappes, J.; Julkunen-Tiitto, R.; Fasulati, S.R.; Tiilikkala, K. Variability in host plant chemistry: behavioural responses and life-history parameters of the Colorado potato beetle (Leptinotarsa decemlineata). Chemoecology 2007, 17, 51–56. [Google Scholar] [CrossRef]
- Armer, C.A. Colorado potato beetle toxins revisited: Evidence the beetle does not sequester host plant glycoalkaloids. J. Chem. Ecol. 2004, 30, 883–888. [Google Scholar] [CrossRef]
- Kruzmane, D.; Jankevica, L.; Ievinsh, G. Effect of regurgitant from Leptinotarsa decemlineata on wound responses in Solanum tuberosum and Phaseolus vulgaris. Physiol. Plant. 2002, 115, 577–584. [Google Scholar] [CrossRef]
- Lawrence, S.D.; Novak, N.G.; Blackburn, M.B. Inhibition of proteinase inhibitor transcripts by Leptinotarsa decemlineata regurgitant in Solanum lycopersicum. J. Chem. Ecol. 2007, 33, 1041–1048. [Google Scholar] [CrossRef]
- Lawrence, S.D.; Novak, N.G.; Ju, C.J.T.; Cooke, J.E.K. Potato, Solanum tuberosum, defense against colorado potato beetle, Leptinotarsa decemlineata (Say): Microarray gene expression profiling of potato by colorado potato beetle regurgitant treatment of wounded leaves. J. Chem. Ecol. 2008, 34, 1013–1025. [Google Scholar] [CrossRef]
- Chung, S.H.; Felton, G.W. Specificity of induced resistance in tomato against specialist lepidopteran and coleopteran species. J. Chem. Ecol. 2011, 37, 378–386. [Google Scholar] [CrossRef]
- McIndoo, N.E. An insect olfactometer. J. Econ. Entomol. 1926, 19, 545–571. [Google Scholar]
- Chin, C.T. Studies on the physiological relation between the larvae of Leptinotarsa decemlineata Say and some solanaceous plants. Eur. J. Plant Pathol. 1950, 56, 1–88. [Google Scholar]
- De Wilde, J.; Hille Ris Lambers-Suverkropp, K.; Van Tol, A. Responses to air flow and airborne plant odour in the Colorado beetle. Neth. J. Plant Pathol. 1969, 75, 53–57. [Google Scholar] [CrossRef]
- Visser, J.H.; Nielsen, J.K. Specificity in the olfactory orientation of the Colorado beetle, Leptinotarsa decemlineata. Entomol. Exp. Appl. 1977, 21, 14–22. [Google Scholar] [CrossRef]
- Visser, J.H. The design of a low-speed wind tunnel as an instrument for the study of olfactory orientation in the Colorado beetle (Leptinotarsa decemlineata). Entomol. Exp. Appl. 1976, 20, 275–288. [Google Scholar] [CrossRef]
- Thiery, D.; Visser, J.H. Satiation effects on olfactory orientations patterns of Colorado potato beetle females. Comptes Rendus Acad. Sci. Ser. III-Sci. Vie-Life Sci. 1995, 318, 105–111. [Google Scholar]
- Visser, J.H.; Straten, S.V.; Maarse, H. Isolation and identification of volatiles in the foliage of potato, Solanum tuberosum, a host plant of the Colorado beetle, Leptinotarsa decemlineata. J. Chem. Ecol. 1979, 5, 13–25. [Google Scholar] [CrossRef]
- Visser, J.H. Electroantennogram responses of the Colorado beetle, Leptinotarsa decemlineata, to plant volatiles. Entomol. Exp. Appl. 1979, 25, 86–97. [Google Scholar] [CrossRef]
- Dickens, J.C. Sexual maturation and temporal variation of neural responses in adult Colorado potato beetles to volatiles emitted by potato plants. J. Chem. Ecol. 2000, 26, 1265–1279. [Google Scholar] [CrossRef]
- Mitchell, B.K.; McCashin, B.G. Tasting green leaf volatiles by larvae and adults of Colorado potato beetle, Leptinotarsa-decemlineata. J. Chem. Ecol. 1994, 20, 753–769. [Google Scholar] [CrossRef]
- Visser, J.H.; Ave, D.A. General green leaf volatiles in the olfactory orientation of the Colorado beetle, Leptinotarsa decemlineata. Entomol. Exp. Appl. 1978, 24, 738–749. [Google Scholar] [CrossRef]
- Hitchner, E.M.; Kuhar, T.P.; Dickens, J.C.; Youngman, R.R.; Schultz, P.B.; Pfeiffer, D.G. Host plant choice experiments of Colorado potato beetle (Coleoptera : Chrysomelidae) in Virginia. J. Econ. Entomol. 2008, 101, 859–865. [Google Scholar] [CrossRef]
- Jermy, T.; Szentesi, A.; Horvath, J. Host plant finding in phytophagous insects - the case of the Colorado potato beetle. Entomol. Exp. Appl. 1988, 49, 83–98. [Google Scholar] [CrossRef]
- Thiery, D.; Visser, J.H. Masking of host plant odour in the olfactory orientation of the Colorado potato beetle. Entomol. Exp. Appl. 1986, 41, 165–172. [Google Scholar] [CrossRef]
- Thiery, D.; Visser, J.H. Misleading the Colorado potato beetle with an odor blend. J. Chem. Ecol. 1987, 13, 1139–1146. [Google Scholar] [CrossRef]
- Bolter, C.J.; Dicke, M.; vanLoon, J.J.A.; Visser, J.H.; Posthumus, M.A. Attraction of Colorado potato beetle to herbivore-damaged plants during herbivory and after its termination. J. Chem. Ecol. 1997, 23, 1003–1023. [Google Scholar] [CrossRef]
- Landolt, P.J.; Tumlinson, J.H.; Alborn, D.H. Attraction of Colorado potato beetle (Coleoptera : Chrysomelidae) to damaged and chemically induced potato plants. Environ. Entomol. 1999, 28, 973–978. [Google Scholar]
- Schutz, S.; Weissbecker, B.; Klein, A.; Hummel, H.E. Host plant selection of the Colorado potato beetle as influenced by damage induced volatiles of the potato plant. Naturwissenschaften 1997, 84, 212–217. [Google Scholar] [CrossRef]
- Alborn, H.T.; Turlings, T.C.J.; Jones, T.H.; Stenhagen, G.; Loughrin, J.H.; Tumlinson, J.H. An elicitor of plant volatiles from beet armyworm oral secretion. Science 1997, 276, 945–949. [Google Scholar] [CrossRef]
- Farmer, E.E.; Ryan, C.A. Interplant communication - airborne methyl jasmonate induces synthesis of proteinase-inhibitors in plant-leaves. Proc. Natl. Acad. Sci. USA 1990, 87, 7713–7716. [Google Scholar] [CrossRef]
- Dickens, J.C. Behavioural responses of larvae of Colorado potato beetle, Leptinotarsa decemlineata (Coleoptera: Chrysomelidae), to host plant volatile blends attractive to adults. Agric. For. Entomol. 2002, 4, 309–314. [Google Scholar] [CrossRef]
- Hammock, J.A.; Vinyard, B.; Dickens, J.C. Response to host plant odors and aggregation pheromone by larvae of the Colorado potato beetle on a servosphere. Arthropod-Plant Interact. 2007, 1, 27–35. [Google Scholar]
- Hare, J.D. Bioassay methods with terrestrial invertebrates. In Methods in Chemical Ecology; Haynes, K.F., Millar, J.G., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998; Volume 2, pp. 212–270. [Google Scholar]
- Levinson, H.Z.; Levinson, A.R.; Jen, T.L. Sex recognition by a pheromone in the Colorado beetle. Naturwissenschaften 1979, 66, 472–473. [Google Scholar] [CrossRef]
- Jermy, T.; Butt, B.A. Method for screening females sex-pheromone extracts of the Colorado potato beetle. Entomol. Exp. Appl. 1991, 59, 75–78. [Google Scholar] [CrossRef]
- Edwards, M.A.; Seabrook, W.D. Evidence for an airborne sex pheromone in the Colorado potato beetle, Leptinotarsa decemlineata. Can. Entomol. 1997, 129, 667–672. [Google Scholar] [CrossRef]
- Dubis, E.; Malinski, E.; Dubis, A.; Szafranek, J.; Nawrot, J.; Poplawski, J.; Wrobel, J.T. Sex-dependent composition of cuticular hydrocarbons of the Colorado beetle, Leptinotarsa decemlineata Say. Comp. Biochem. Physiol. A Physiol. 1987, 87, 839–843. [Google Scholar] [CrossRef]
- Dickens, J.C. Sexual contact influences orientation to plant attractant in Colorado potato beetle, Leptinotarsa decemlineata Say (Coleoptera : Chrysomelidae). Naturwissenschaften 2007, 94, 847–852. [Google Scholar] [CrossRef]
- Dickens, J.C.; Oliver, J.E.; Hollister, B.; Davis, J.C.; Klun, J.A. Breaking a paradigm: male-produced aggregation pheromone for the Colorado potato beetle. J. Exp. Biol. 2002, 205, 1925–1933. [Google Scholar]
- Hsiao, T.H.; Fraenkel, G. Properties of leptinotarsin: A toxic hemolymph protein from the Colorado potato beetle. Toxicon 1969, 7, 119–128. [Google Scholar] [CrossRef]
- Bortels, A. A Cation Selective Channel Induced by Two Protein Fractions from the Larval Hemolymph of the Colorado Potato Beetle, Leptinotarsa decemlineata (Say) An Electrophysiological Characterization on Guinea-pig Ventricular Myocytes and on Artificial Membranes; K.U.Leuven: Leuven, Belgium, 1994. [Google Scholar]
- Chiou, S.J.; Cerstiaens, A.; Kotanen, S.P.; De Loof, A.; Schoofs, L. Insect larvae contain substances toxic to adults: the discovery of paralysins. J. Insect Physiol. 1998, 44, 405–411. [Google Scholar] [CrossRef]
- Armer, C.A.; Rao, S.J.Y.; Berry, R.E. Insect cellular and chemical limitations to pathogen development: the Colorado potato beetle, the nematode Heterorhabditis marelatus, and its symbiotic bacteria. J. Invertebr. Pathol. 2004, 87, 114–122. [Google Scholar] [CrossRef]
- Daloze, D.; Braekman, J.C.; Pasteels, J.M. A toxic dipeptide from the defense glands of the Colorado beetle. Science 1986, 233, 221–223. [Google Scholar]
- Hough-Goldstein, J.A.; Geiger, J.; Chang, D.; Saylor, W. Palatability and toxicity of the Colorado potato beetle (Coleoptera, Chrysomelidae) to domestic chickens. Ann. Entomol. Soc. Am. 1993, 86, 158–164. [Google Scholar]
- Dickens, J.C. Predator-prey interactions: olfactory adaptations of generalist and specialist predators. Agric. For. Entomol. 1999, 1, 47–54. [Google Scholar] [CrossRef]
- van Loon, J.J.A.; de Vos, E.W.; Dicke, M. Orientation behaviour of the predatory hemipteran Perillus bioculatus to plant and prey odours. Entomol. Exp. Appl. 2000, 96, 51–58. [Google Scholar]
- Weissbecker, B.; Van Loon, J.J.A.; Dicke, M. Electroantennogram responses of a predator, Perillus bioculatus, and its prey, Leptinotarsa decemlineata, to plant volatiles. J. Chem. Ecol. 1999, 25, 2313–2325. [Google Scholar] [CrossRef]
- Weissbecker, B.; Van Loon, J.J.A.; Posthumus, M.A.; Bouwmeester, H.J.; Dicke, M. Identification of volatile potato sesquiterpenoids and their olfactory detection by the two-spotted stinkbug Perillus bioculatus. J. Chem. Ecol. 2000, 26, 1433–1445. [Google Scholar] [CrossRef]
- Sant'Ana, J.; Da Silva, R.F.P.; Dickens, J.C. Olfactory reception of conspecific aggregation pheromone and plant odors by nymphs of the predator, Podisus maculiventris. J. Chem. Ecol. 1999, 25, 1813–1826. [Google Scholar] [CrossRef]
- Lynch, M.E.; Kaplan, I.; Dively, G.P.; Denno, R.F. Host-plant-mediated competition via induced resistance: Interactions between pest herbivores on potatoes. Ecol. Appl. 2006, 16, 855–864. [Google Scholar] [CrossRef]
- Gosset, V.; Harmel, N.; Gobel, C.; Francis, F.; Haubruge, E.; Wathelet, J.P.; du Jardin, P.; Feussner, I.; Fauconnier, M.L. Attacks by a piercing-sucking insect (Myzus persicae Sultzer) or a chewing insect (Leptinotarsa decemlineata Say) on potato plants (Solanum tuberosum L.) induce differential changes in volatile compound release and oxylipin synthesis. J. Exp. Bot. 2009, 60, 1231–1240. [Google Scholar]
- Brunissen, L.; Vincent, C.; Le Roux, V.; Giordanengo, P. Effects of systemic potato response to wounding and jasmonate on the aphid Macrosiphum euphorbiae (Sternorryncha:Aphididae). J. Appl. Entomol. 2010, 134, 562–571. [Google Scholar]
- Panasiuk, O. Response of Colorado potato beetles, Leptinotarsa decemlineata (Say), to volatile components of tansy, Tanacetum vulgare. J. Chem. Ecol. 1984, 10, 1325–1333. [Google Scholar] [CrossRef]
- Matthews, D.L.; Michalak, P.S.; MacRae, R.J. The Effect of Traditional Insect Repellent Plants on Insect Numbers in a Mixed Planting System. In Proceedings of the 4th International Federation of Organic Agricultural Movements Conference, Resource conserving, environmentally sound agricultural alternatives, Cambridge, MA, USA, 18–20 August 1982.
- Szendrei, Z.; Rodriguez-Saona, C. A meta-analysis of insect pest behavioral manipulation with plant volatiles. Entomol. Exp. Appl. 2010, 134, 201–210. [Google Scholar] [CrossRef]
- Dickens, J.C. Orientation of Colorado potato beetle to natural and synthetic blends of volatiles emitted by potato plants. Agric. For. Entomol. 2000, 2, 167–172. [Google Scholar] [CrossRef]
- Martel, J.W.; Alford, A.R.; Dickens, J.C. Laboratory and greenhouse evaluation of a synthetic host volatile attractant for Colorado potato beetle, Leptinotarsa decemlineata (Say). Agric. For. Entomol. 2005, 7, 71–78. [Google Scholar] [CrossRef]
- Martel, J.W.; Alford, A.R.; Dickens, J.C. Synthetic host volatiles increase efficacy of trap cropping for management of Colorado potato beetle, Leptinotarsa decemlineata (Say). Agric. For. Entomol. 2005, 7, 79–86. [Google Scholar] [CrossRef]
- Kuhar, T.P.; Mori, K.; Dickens, J.C. Potential of a synthetic aggregation pheromone for integrated pest management of Colorado potato beetle. Agric. For. Entomol. 2006, 8, 77–81. [Google Scholar] [CrossRef]
- Dickens, J.C. Plant volatiles moderate response to aggregation pheromone in Colorado potato beetle. J. Appl. Entomol. 2006, 130, 26–31. [Google Scholar] [CrossRef]
- Martel, J.W.; Alford, A.R.; Dickens, J.C. Evaluation of a novel host plant volatile-based attracticide for management of Colorado potato beetle, Leptinotarsa decemlineata (Say). Crop. Prot. 2007, 26, 822–827. [Google Scholar]
- Ragsdale, D.W.; Radcliffe, E.B. Colorado potato beetle management. Available online: http://ipmworld.umn.edu/chapters/ragsdale.htm (accessed on 14 September 2011).
- Ioannidis, P.M.; Grafius, E.; Whalon, M.E. Patterns of insecticide resistance to azinphosmethyl, carbofuran, and permethrin in the Colorado potato beetle (Coleoptera, Chrysomelidae). J. Econ. Entomol. 1991, 84, 1417–1423. [Google Scholar]
- Otálora-Luna, F.; Dickens, J.C. Multimodal stimulation of Colorado potato beetle reveals modulation of pheromone response by yellow light. PLoS ONE 2011, 6, e20990. [Google Scholar]
- Otálora-Luna, F.; Dickens, J.C. Spectral preference and temporal modulation of photic orientation by Colorado potato beetle on a servosphere. Entomol. Exp. Appl. 2011, 138, 93–103. [Google Scholar]
- Heuskin, S.; Verheggen, F.J.; Haubruge, E.; Wathelet, J.P.; Lognay, G. The use of semiochemical slow-release devices in integrated pest management strategies. Biotechnol. Agron. Soc. Environ. 2011, 15, 459–470. [Google Scholar]
- Hsiao, T.H.; Fraenkel, G. The role of secondary plant substances in the food specificity of the Colorado potato beetle. Ann. Entomol. Soc. Am. 1968, 61, 485–493. [Google Scholar]
- Jermy, T.; Butt, B.A.; McDonough, L.; Dreyer, D.L.; Rose, A.F. Antifeedants for the Colorado potato beetle - I. Antifeeding constituents of some plants from the sagebrush community. Insect Sci. Appl. 1981, 1, 237–242. [Google Scholar]
- Hare, J.D.; Logan, P.A.; Wright, R.J. Suppression of Colorado potato beetle, Leptinotarsa decemlineata (Say), (Coleoptera: Chrysomelidae) populations with antifeedant fungicides. Environ. Entomol. 1983, 12, 1470–1477. [Google Scholar]
- Drummond, F.A.; Casagrande, R.A. Effect of white oak extracts on feeding by the Colorado potato beetle (Coleoptera: Chrysomelidae). J. Econ. Entomol. 1985, 78, 1272–1274. [Google Scholar]
- Alford, A.R.; Cullen, J.A.; Storch, R.H.; Bentley, M.D. Antifeedant activity of limonin against the Colorado potato beetle (Coleoptera, Chrysomelidae). J. Econ. Entomol. 1987, 80, 575–578. [Google Scholar]
- Kim, M.S.; Lan, Q. Larvicidal activity of α-mangostin in the Colorado potato beetle, Leptinotarsa decemlineata. Journal of Pesticide Science 2011, 36, 370–375. [Google Scholar]
- Gonzalezcoloma, A.; Reina, M.; Cabrera, R.; Castanera, P.; Gutierrez, C. Antifeedant and toxic effects of sesquiterpenes from Senecio-palmensis to Colorado potato beetle. J. Chem. Ecol. 1995, 21, 1255–1270. [Google Scholar]
- Szczepanik, N.; Dams, I.; Wawrzenczyk, C. Feeding deterrent activity of terpenoid lactones with the p-menthane system against the Colorado potato beetle (Coleoptera:Chrysomelidae). Environ. Entomol. 2005, 34, 1433–1440. [Google Scholar]
- Gokce, A.; Isaacs, R.; Whalon, M.E. Behavioural response of Colorado potato beetle (Leptinotarsa decemlineata) larvae to selected plant extracts. Pest Manag. Sci. 2006, 62, 1052–1057. [Google Scholar]
- Pavela, R.; Sajfrtova, M.; Sovova, H.; Karban, J.; Barnet, M. The effects of extracts obtained by supercritical fluid extraction and traditional extraction techniques on larvae Leptinotarsa decemlineata SAY. J. Essent. Oil Res. 2009, 21, 367–373. [Google Scholar]
- Pelletier, Y.; Dutheil, J. Behavioural responses of the Colorado potato beetle to trichomes and leaf surface chemicals of Solanum tarijense. Entomol. Exp. Appl. 2006, 120, 125–130. [Google Scholar]
- Yencho, G.C.; Renwick, J.A.A.; Steffens, J.C.; Tingey, W.M. Leaf surface extracts of Solanum-berthaultii Hawkes deter Colorado potato beetle feeding. J. Chem. Ecol. 1994, 20, 991–1007. [Google Scholar] [CrossRef]
- Liu, Y.B.; Alford, A.R.; Bentley, M.D. Effects of epilimonol and starvation on feeding and oviposition by Leptinotarsa-decemlineata. Entomol. Exp. Appl. 1989, 53, 39–44. [Google Scholar] [CrossRef]
- Murray, K.D.; Groden, E.; Drummond, F.A.; Alford, A.R.; Conley, S.; Storch, R.H.; Bentley, M.D. Citrus limonoid effects on Colorado potato beetle (Coleoptera:Chrysomelidae) colonization and oviposition. Environ. Entomol. 1995, 24, 1275–1283. [Google Scholar]
- Szafranek, B.; Synak, E.; Waligora, D.; Szafranek, J.; Nawrot, J. Leaf surface compounds of the potato (Solanum tuberosum) and their influence on Colorado potato beetle (Leptinotarsa decemlineata) feeding. Chemoecology 2008, 18, 205–216. [Google Scholar] [CrossRef]
- Jermy, T. Prospects of antifeedant approach to pest-control - a critical review. J. Chem. Ecol. 1990, 16, 3151–3166. [Google Scholar] [CrossRef]
- Szentesi, A. Antifeedant-treated potato plants as egg-laying traps for the Colorado beetle (Leptinotarsa decemlineata Say, Col., Chrysomelidae). Acta Phytopathol. Hun. 1981, 16, 203–209. [Google Scholar]
- Hare, J.D. Suppression of the Colorado potato beetle, Leptinotarsa decemlineata (Say) (Coleoptera:Chrysomelidae), on solanaceous crops with a copper-based fungicide. Environ. Entomol. 1984, 13, 1010–1014. [Google Scholar]
- Zehnder, G.; Warthen, J.D. Feeding inhibition and mortality effects of neem-seed extract on the Colorado potato beetle (Coleoptera, Chrysomelidae). J. Econ. Entomol. 1988, 81, 1040–1044. [Google Scholar]
- Moreau, T.L.; Warman, P.R.; Hoyle, J. An evaluation of companion planting and botanical extracts as alternative pest controls for the Colorado potato beetle. Biol. Agric. Hortic. 2006, 23, 351–370. [Google Scholar] [CrossRef]
- Marčić, D.; Perić, P. Field evaluation of natural and synthetic insecticides against Leptinotarsa decemlineata say. Acta Hortic. 2009, 830, 391–396. [Google Scholar]
- Alford, R. Deployment strategies for antifeedants in management of Colorado potato beetle. In Advances in Potato Pest Biology and Management; ehnder, G.W., Powelson, M.L., Jansson, R.K., Raman, K.V., Eds.; APS Press: St Paul, MI, USA, 1994; pp. 342–356. [Google Scholar]
- Murray, K.D.; Groden, E.; Drummond, F.A.; Alford, A.R.; Storch, R.H.; Bentley, M.D. Citrus limonoid effects on Colorado potato beetle larval survival and development. Entomol. Exp. Appl. 1996, 80, 503–510. [Google Scholar] [CrossRef]
- Sinden, S.L. Potato plants make their own insect repellent. Agric. Res. USA 1988, 36, 15. [Google Scholar]
- Sinden, S.L.; Sanford, L.L.; Osman, S.F. Glycoalkaloids and resistance to the Colorado potato beetle in Solanum chacoense Bitter. Am. Potato J. 1980, 57, 331–343. [Google Scholar] [CrossRef]
- Wegorek, W.; Dubniak, H. Reaction of potato leaves to the eggs of the Colorado beetle (Leptinotarsa decemlineata Say). Biuletyn Instytutu Ochrony Roslin 1972, 52, 409–413. [Google Scholar]
- Balbyshev, N.F.; Lorenzen, J.H. Hypersensitivity and egg drop: A novel mechanism of host plant resistance to Colorado potato beetle (Coleoptera: Chrysomelidae). J. Econ. Entomol. 1997, 90, 652–657. [Google Scholar]
- Guenthner, J.F. Consumer acceptance of genetically modified potaoes. Am. J. Potato Res. 2002, 79, 309–316. [Google Scholar] [CrossRef]
- Grapputo, A.; Boman, S.; Lindstrom, L.; Lyytinen, A.; Mappes, J. The voyage of an invasive species across continents: genetic diversity of North American and European Colorado potato beetle populations. Mol. Ecol. 2005, 14, 4207–4219. [Google Scholar] [CrossRef]
- Clark, J.M.; Lee, S.H.; Kim, H.J.; Yoon, K.S.; Zhang, A.G. DNA-based genotyping techniques for the detection of point mutations associated with insecticide resistance in Colorado potato beetle Leptinotarsa decemlineata. Pest Manag. Sci. 2001, 57, 968–974. [Google Scholar] [CrossRef]
- Boman, S.; Grapputo, A.; Lindstrom, L.; Lyytinen, A.; Mappes, J. Quantitative genetic approach for assessing invasiveness: geographic and genetic variation in life-history traits. Biol. Invasions 2008, 10, 1135–1145. [Google Scholar] [CrossRef]
- Forister, M.L.; Ehmer, A.G.; Futuyma, D.J. The genetic architecture of a niche: variation and covariation in host use traits in the Colorado potato beetle. J. Evol. Biol. 2007, 20, 985–996. [Google Scholar]
- Pelletier, Y.; Tai, G.C.C. Genotypic variability and mode of action of Colorado potato beetle (Coleoptera : Chrysomelidae) resistance in seven Solanum species. J. Econ. Entomol. 2001, 94, 572–578. [Google Scholar] [CrossRef]
- Visser, J.H. Differential sensory perception of plant compounds by insects. ACS symposiums series 208. In Plant Resistance to Insects; Hedin, P.A., Ed.; American chemical society: Washington, DC, USA, 1983; pp. 215–230. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sablon, L.; Dickens, J.C.; Haubruge, É.; Verheggen, F.J. Chemical Ecology of the Colorado Potato Beetle, Leptinotarsa decemlineata (Say) (Coleoptera: Chrysomelidae), and Potential for Alternative Control Methods. Insects 2013, 4, 31-54. https://doi.org/10.3390/insects4010031
Sablon L, Dickens JC, Haubruge É, Verheggen FJ. Chemical Ecology of the Colorado Potato Beetle, Leptinotarsa decemlineata (Say) (Coleoptera: Chrysomelidae), and Potential for Alternative Control Methods. Insects. 2013; 4(1):31-54. https://doi.org/10.3390/insects4010031
Chicago/Turabian StyleSablon, Ludovic, Joseph C. Dickens, Éric Haubruge, and François J. Verheggen. 2013. "Chemical Ecology of the Colorado Potato Beetle, Leptinotarsa decemlineata (Say) (Coleoptera: Chrysomelidae), and Potential for Alternative Control Methods" Insects 4, no. 1: 31-54. https://doi.org/10.3390/insects4010031




