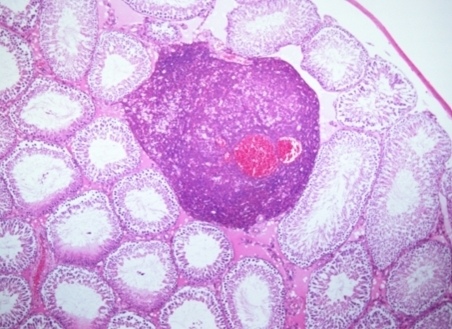
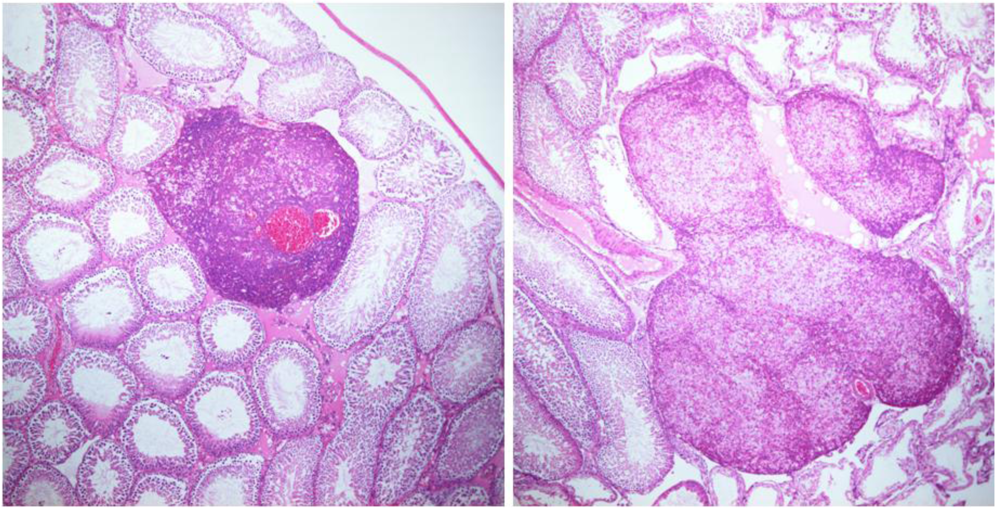
Comments on “Ochratoxin A: In utero Exposure in Mice Induces Adducts in Testicular DNA. Toxins 2010, 2, 1428–1444”—Mis-Citation of Rat Literature to Justify a Hypothetical Role for Ochratoxin A in Testicular Cancer
Abstract
:
References
- Mantle, P.G.; Nolan, C.C. Pathological outcomes in kidney and brain of male Fischer rats given dietary ochratoxin A, commencing at one year of age. Toxins 2010, 2, 1100–1110. [Google Scholar]
- Mantle, P.; Kulinskaya, E.; Nestler, S. Renal tumourigenesis in male rats in response to chronic dietary ochratoxin A. Food Addit. Contam. 2005, 22 (Suppl. 1), 58–64. [Google Scholar] [CrossRef] [PubMed]
- Boorman, G.A. Toxicology and Carcinogenesis Studies of Ochratoxin A (CAS No. 303-47-9) in F344/N Rats (Gavage Studies); National Toxicology Program Technical Report No. 358; National Institutes of Health: Research Triangle Park, NC, USA, 1989. [Google Scholar]
- Bendele, A.M.; Carlton, W.W.; Krogh, P.; Lillehoj, E.B. Ochratoxin A carcinogenesis in the (C57BL/6J x C3H)F1 mouse. J. Natl. Cancer Inst. 1985, 75, 733–742. [Google Scholar]
- Jennings-Gee, J.E.; Tozlovanu, M.; Manderville, R.; Miller, M.S.; Pfohl-Leszkowicz, A.; Schwartz, G.G. Ochratoxin A: In utero exposure in mice induces adducts in testicular DNA. Toxins 2010, 2, 1428–1444. [Google Scholar]
- Toxicology and carcinogenesis studies of fumonisin B1 in F344/N rats and B6C3F1 mice (feed studies). NTP Technical Report No. 496. p. 112. Available online: http://ntp.niehs.nih.gov/ntp/htdocs/LT_rpts/tr496.pdf (accessed on 26 July 2010).
- Mantle, P.G.; Faucet-Marquis, V.; Manderville, R.A.; Squillaci, B.; Pfohl-Leszkowicz, A. Structures of covalent adducts between DNA and ochratoxin A: a new factor in debate about genotoxicity and human risk assessment. Chem. Res. Toxicol. 2010, 23, 89–98. [Google Scholar]
- Abouzied, M.M.; Horvath, A.D.; Podlesny, P.M.; Regina, N.P.; Metodiev, V.D.; Kamenova-Tozeva, R.M.; Niagolova, N.D.; Stein, A.D.; Petropoulos, E.A.; Ganev, V.S. Ochratoxin A concentrations in food and feed from a region with Balkan Endemic Nephropathy. Food Addit. Contam. 2002, 19, 755–764. [Google Scholar]
- Schwartz, G.G. Hypothesis: Does ochratoxin A cause testicular cancer? Cancer Causes Control 2002, 13, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Mantle, P.G. Interpretation of pharmacodynamics of ochratoxin A in blood plasma of rats, during and after acute or chronic ingestion. Food Chem. Toxicol. 2008, 46, 1808–1816. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mantle, P.G. Comments on “Ochratoxin A: In utero Exposure in Mice Induces Adducts in Testicular DNA. Toxins 2010, 2, 1428–1444”—Mis-Citation of Rat Literature to Justify a Hypothetical Role for Ochratoxin A in Testicular Cancer. Toxins 2010, 2, 2333-2336. https://doi.org/10.3390/toxins2102333
Mantle PG. Comments on “Ochratoxin A: In utero Exposure in Mice Induces Adducts in Testicular DNA. Toxins 2010, 2, 1428–1444”—Mis-Citation of Rat Literature to Justify a Hypothetical Role for Ochratoxin A in Testicular Cancer. Toxins. 2010; 2(10):2333-2336. https://doi.org/10.3390/toxins2102333
Chicago/Turabian StyleMantle, Peter G. 2010. "Comments on “Ochratoxin A: In utero Exposure in Mice Induces Adducts in Testicular DNA. Toxins 2010, 2, 1428–1444”—Mis-Citation of Rat Literature to Justify a Hypothetical Role for Ochratoxin A in Testicular Cancer" Toxins 2, no. 10: 2333-2336. https://doi.org/10.3390/toxins2102333