Whole Grain Intake and Glycaemic Control in Healthy Subjects: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Abstract
:1. Introduction
2. Methods
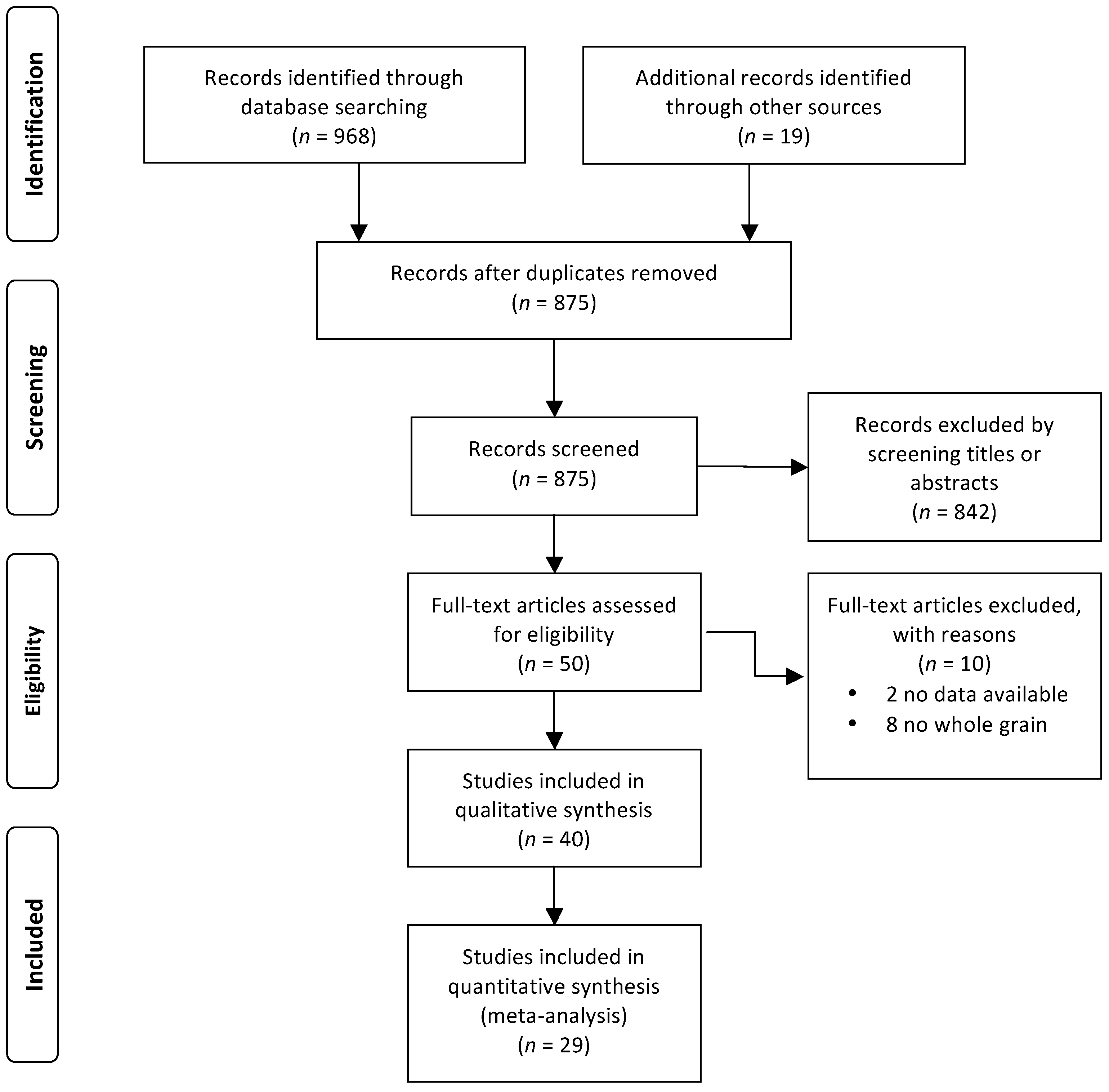
2.1. Search Strategy and Selection of the Studies
2.2. Data Extraction and Study Quality
2.3. Statistical Analysis
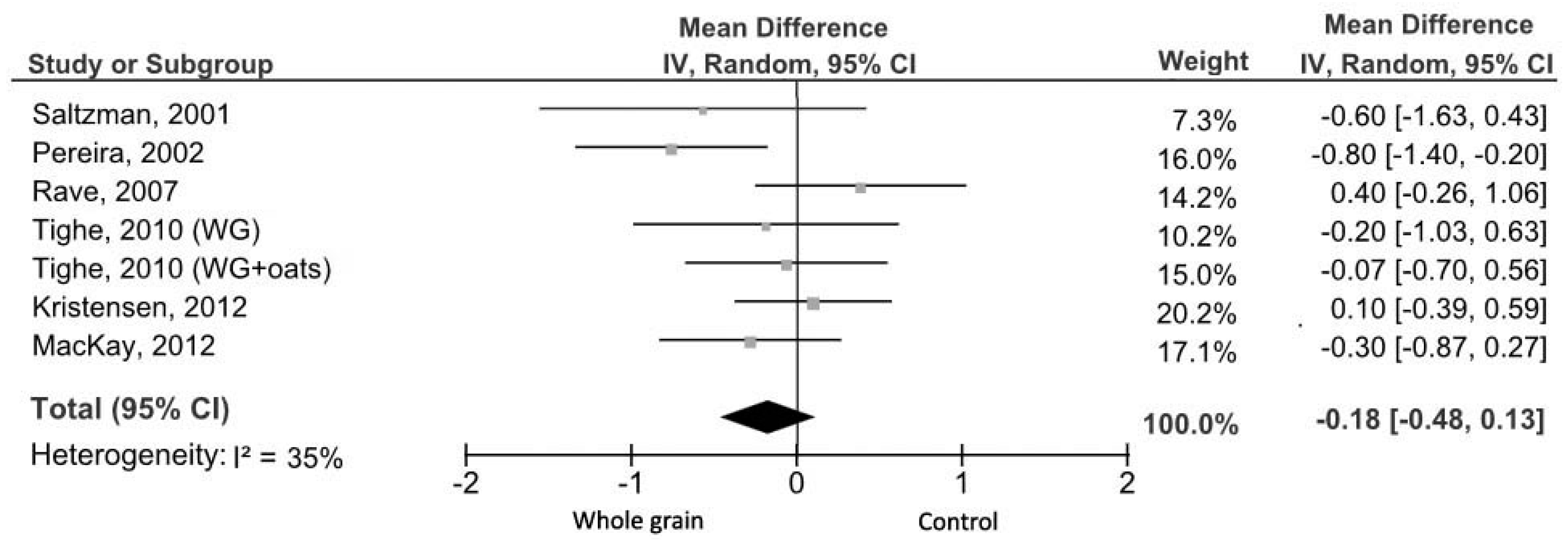
3. Results
3.1. Study Selection and Main Characteristics
3.2. Acute Studies
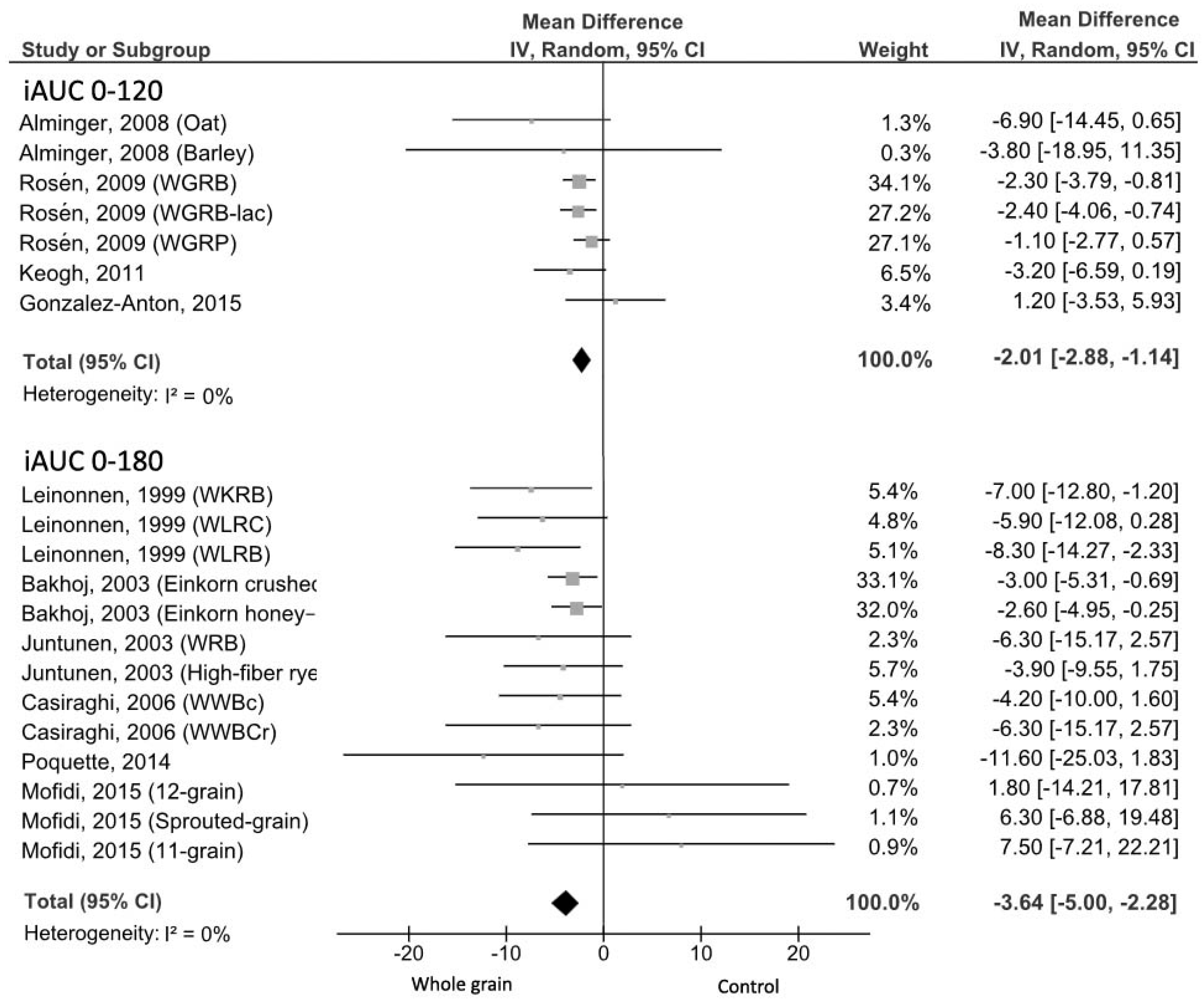
3.2.1. Glucose iAUC
3.2.2. Insulin iAUC
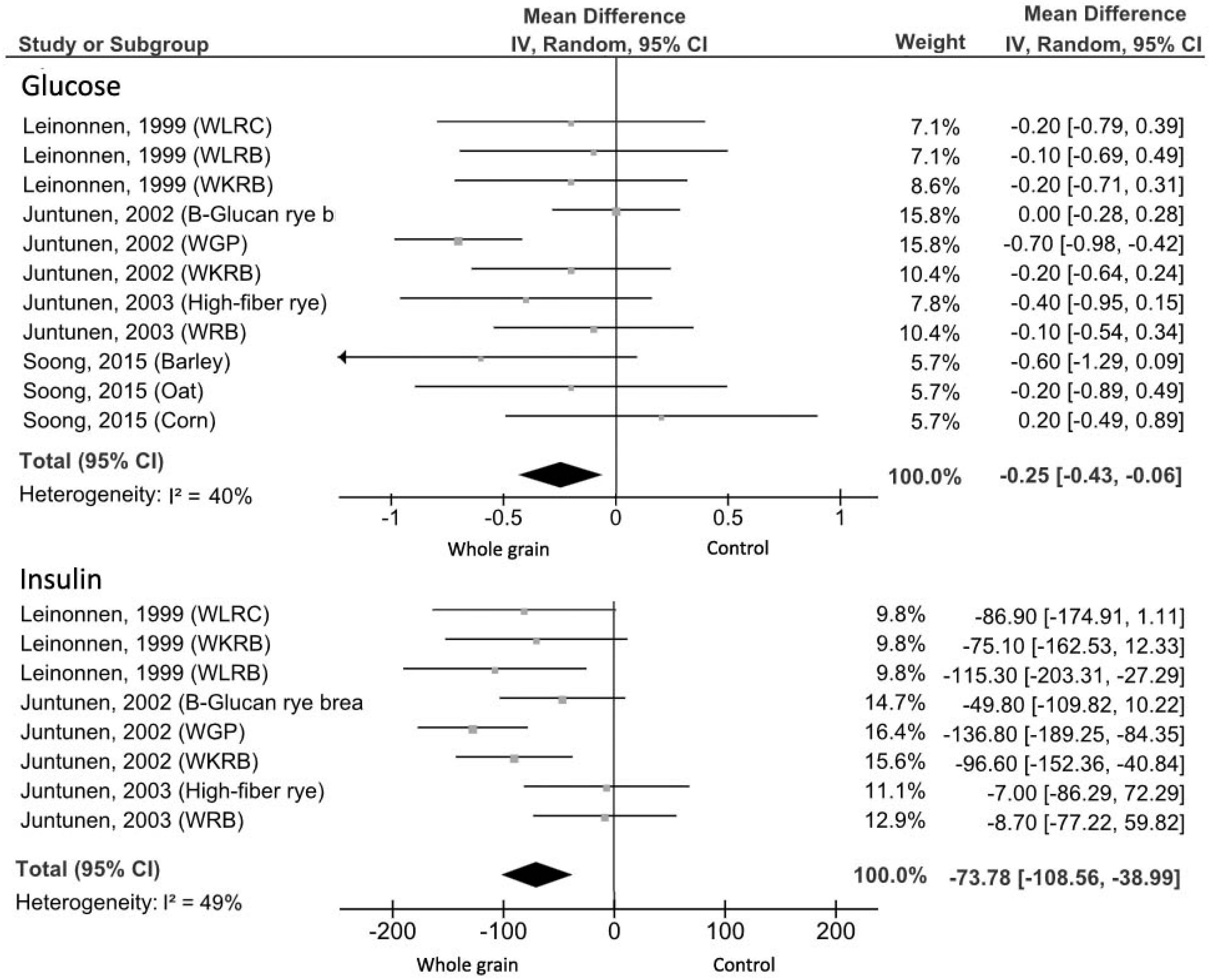
3.2.3. Maximal Glucose and Insulin Response
3.3. Medium- and Long-Term Studies
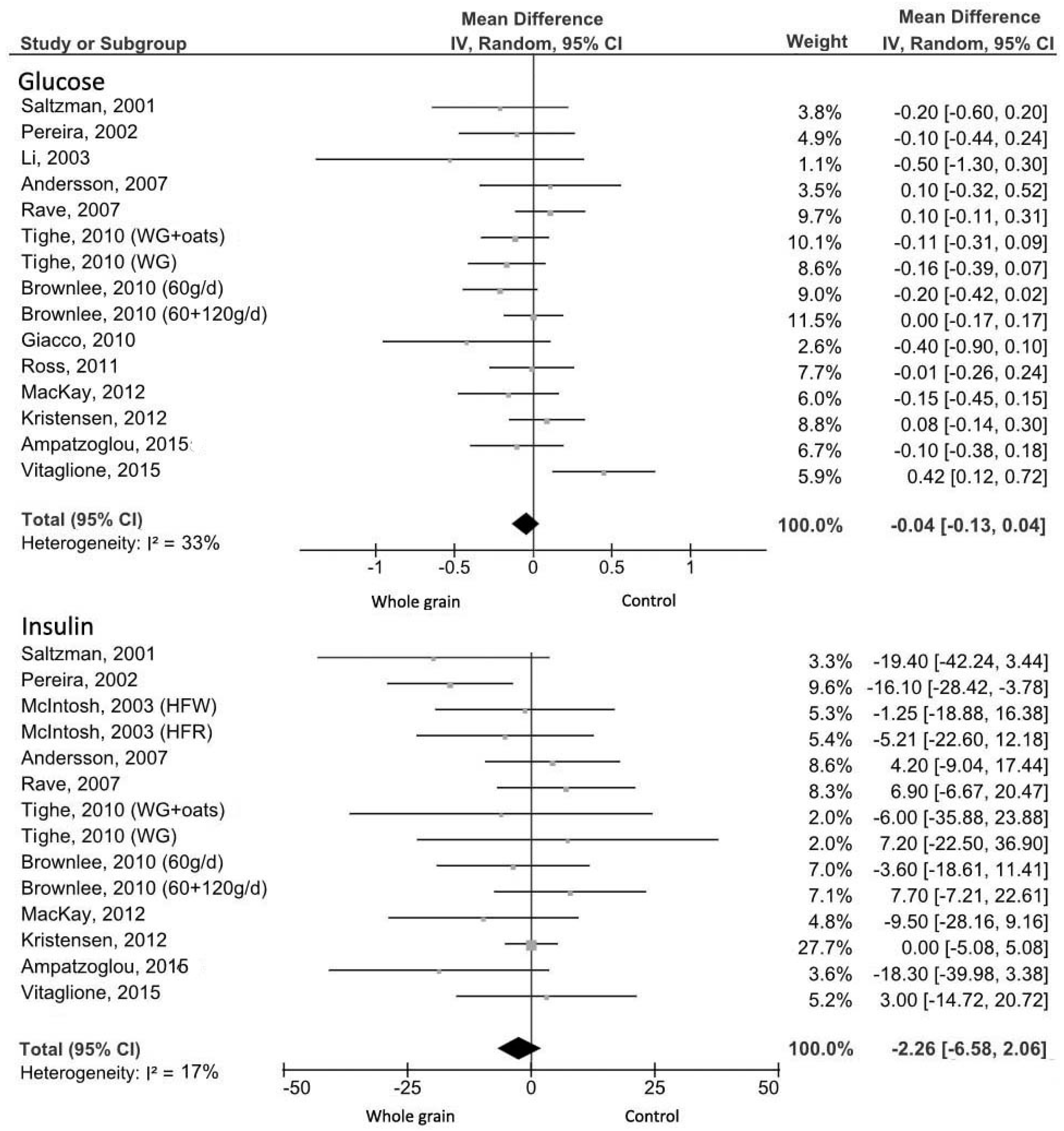
3.3.1. Fasting Glucose
3.3.2. Fasting Insulin
3.3.3. HOMA-IR
3.3.4. Subgroup Analyses
4. Discussion
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- World Health Organization. World Health Statistics; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Slavin, J.L. Why wholegrains are protective: Biological mechanisms. Proc. Nutr. Soc. 2003, 62, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Seal, C.J.; Brownlee, I.A. Whole-grain foods and chronic disease: Evidence from epidemiological and intervention studies. Proc. Nutr. Soc. 2015, 74, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Costabile, A.; Klinder, A.; Fava, F.; Napolitano, A.; Fogliano, V.; Leonard, C.; Gibson, G.R.; Tuohy, K.M. Whole-grain wheat breakfast cereal has a prebiotic effect on the human gut microbiota: A double-blind, placebo-controlled, crossover study. Br. J. Nutr. 2008, 99, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Giacco, R.; Clemente, G.; Cipriano, D.; Luongo, D.; Viscovo, D.; Patti, L.; Di Marino, L.; Giacco, A.; Naviglio, D.; Bianchi, M.A.; et al. Effects of the regular consumption of wholemeal wheat foods on cardiovascular risk factors in healthy people. Nutr. Metab. Cardiovasc. Dis. NMCD 2010, 20, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Juntunen, K.S.; Laaksonen, D.E.; Poutanen, K.S.; Niskanen, L.K.; Mykkanen, H.M. High-fiber rye bread and insulin secretion and sensitivity in healthy postmenopausal women. Am. J. Clin. Nutr. 2003, 77, 385–391. [Google Scholar] [PubMed]
- Kelly, S.A.; Summerbell, C.D.; Brynes, A.; Whittaker, V.; Frost, G. Wholegrain cereals for coronary heart disease. Cochrane Database Syst. Rev. 2007, 18, CD005051. [Google Scholar]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Whole grain and refined grain consumption and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Eur. J. Epidemiol. 2013, 28, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Ye, E.Q.; Chacko, S.A.; Chou, E.L.; Kugizaki, M.; Liu, S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. J. Nutr. 2012, 142, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Chan, D.S.; Lau, R.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Dietary fibre, whole grains, and risk of colorectal cancer: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2011, 343, d6617. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A. New hypotheses for the health-protective mechanisms of whole-grain cereals: What is beyond fibre? Nutr. Res. Rev. 2010, 23, 65–134. [Google Scholar] [CrossRef] [PubMed]
- Nettleton, J.A.; McKeown, N.M.; Kanoni, S.; Lemaitre, R.N.; Hivert, M.F.; Ngwa, J.; van Rooij, F.J.; Sonestedt, E.; Wojczynski, M.K.; Ye, Z.; et al. Interactions of dietary whole-grain intake with fasting glucose- and insulin-related genetic loci in individuals of european descent: A meta-analysis of 14 cohort studies. Diabetes Care 2010, 33, 2684–2691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (nos) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; Ottawa Health Research Institute: Ottawa, ON, Canada, 1999. [Google Scholar]
- The Journal of the American Medical Association. Author Instructions: Systeme International (si) Conversion Factors for Selected Laboratory Components; The Journal of the American Medical Association: Berlin, Germany, 2001. [Google Scholar]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (Updated March 2011); The Cochrane Collaboration: London, UK, 2011. [Google Scholar]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansson, D.P.; Lee, I.; Riserus, U.; Langton, M.; Landberg, R. Effects of unfermented and fermented whole grain rye crisp breads served as part of a standardized breakfast, on appetite and postprandial glucose and insulin responses: A randomized cross-over trial. PLoS ONE 2015, 10, e0122241. [Google Scholar] [CrossRef] [PubMed]
- Alminger, M.; Eklund-Jonsson, C. Whole-grain cereal products based on a high-fibre barley or oat genotype lower post-prandial glucose and insulin responses in healthy humans. Eur. J. Nutr. 2008, 47, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Hlebowicz, J.; Wickenberg, J.; Fahlstrom, R.; Bjorgell, O.; Almer, L.O.; Darwiche, G. Effect of commercial breakfast fibre cereals compared with corn flakes on postprandial blood glucose, gastric emptying and satiety in healthy subjects: A randomized blinded crossover trial. Nutr. J. 2007, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.C.; Ostman, E.M.; Granfeldt, Y.; Bjorck, I.M. Effect of cereal test breakfasts differing in glycemic index and content of indigestible carbohydrates on daylong glucose tolerance in healthy subjects. Am. J. Clin. Nutr. 2008, 87, 645–654. [Google Scholar] [PubMed]
- Rosen, L.A.; Silva, L.O.; Andersson, U.K.; Holm, C.; Ostman, E.M.; Bjorck, I.M. Endosperm and whole grain rye breads are characterized by low post-prandial insulin response and a beneficial blood glucose profile. Nutr. J. 2009, 8, 42. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.A.; Ostman, E.M.; Bjorck, I.M. Effects of cereal breakfasts on postprandial glucose, appetite regulation and voluntary energy intake at a subsequent standardized lunch; focusing on rye products. Nutr. J. 2011, 10, 7. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.; Tengblad, S.; Karlstrom, B.; Kamal-Eldin, A.; Landberg, R.; Basu, S.; Aman, P.; Vessby, B. Whole-grain foods do not affect insulin sensitivity or markers of lipid peroxidation and inflammation in healthy, moderately overweight subjects. J. Nutr. 2007, 137, 1401–1407. [Google Scholar] [PubMed]
- Hlebowicz, J.; Jonsson, J.M.; Lindstedt, S.; Bjorgell, O.; Darwich, G.; Almer, L.O. Effect of commercial rye whole-meal bread on postprandial blood glucose and gastric emptying in healthy subjects. Nutr. J. 2009, 8, 26. [Google Scholar] [CrossRef] [PubMed]
- Leinonen, K.; Liukkonen, K.; Poutanen, K.; Uusitupa, M.; Mykkanen, H. Rye bread decreases postprandial insulin response but does not alter glucose response in healthy finnish subjects. Eur. J. Clin. Nutr. 1999, 53, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Juntunen, K.S.; Niskanen, L.K.; Liukkonen, K.H.; Poutanen, K.S.; Holst, J.J.; Mykkanen, H.M. Postprandial glucose, insulin, and incretin responses to grain products in healthy subjects. Am. J. Clin. Nutr. 2002, 75, 254–262. [Google Scholar] [PubMed]
- Juntunen, K.S.; Laaksonen, D.E.; Autio, K.; Niskanen, L.K.; Holst, J.J.; Savolainen, K.E.; Liukkonen, K.H.; Poutanen, K.S.; Mykkanen, H.M. Structural differences between rye and wheat breads but not total fiber content may explain the lower postprandial insulin response to rye bread. Am. J. Clin. Nutr. 2003, 78, 957–964. [Google Scholar] [PubMed]
- Lappi, J.; Aura, A.M.; Katina, K.; Nordlund, E.; Kolehmainen, M.; Mykkanen, H.; Poutanen, K. Comparison of postprandial phenolic acid excretions and glucose responses after ingestion of breads with bioprocessed or native rye bran. Food Funct. 2013, 4, 972–981. [Google Scholar] [CrossRef] [PubMed]
- Moazzami, A.A.; Shrestha, A.; Morrison, D.A.; Poutanen, K.; Mykkanen, H. Metabolomics reveals differences in postprandial responses to breads and fasting metabolic characteristics associated with postprandial insulin demand in postmenopausal women. J. Nutr. 2014, 144, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Mofidi, A.; Ferraro, Z.M.; Stewart, K.A.; Tulk, H.M.; Robinson, L.E.; Duncan, A.M.; Graham, T.E. The acute impact of ingestion of sourdough and whole-grain breads on blood glucose, insulin, and incretins in overweight and obese men. J. Nutr. Metab. 2012, 2012, 184710. [Google Scholar] [CrossRef] [PubMed]
- Luhovyy, B.L.; Mollard, R.C.; Yurchenko, S.; Nunez, M.F.; Berengut, S.; Liu, T.T.; Smith, C.E.; Pelkman, C.L.; Anderson, G.H. The effects of whole grain high-amylose maize flour as a source of resistant starch on blood glucose, satiety, and food intake in young men. J. Food Sci. 2014, 79, H2550–H2556. [Google Scholar] [CrossRef] [PubMed]
- MacKay, K.A.; Tucker, A.J.; Duncan, A.M.; Graham, T.E.; Robinson, L.E. Whole grain wheat sourdough bread does not affect plasminogen activator inhibitor-1 in adults with normal or impaired carbohydrate metabolism. Nutr. Metab. Cardiovasc. Dis. NMCD 2012, 22, 704–711. [Google Scholar] [CrossRef] [PubMed]
- Najjar, A.M.; Parsons, P.M.; Duncan, A.M.; Robinson, L.E.; Yada, R.Y.; Graham, T.E. The acute impact of ingestion of breads of varying composition on blood glucose, insulin and incretins following first and second meals. Br. J. Nutr. 2009, 101, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Vuksan, V.; Jenkins, A.L.; Dias, A.G.; Lee, A.S.; Jovanovski, E.; Rogovik, A.L.; Hanna, A. Reduction in postprandial glucose excursion and prolongation of satiety: Possible explanation of the long-term effects of whole grain Salba (Salvia Hispanica L.). Eur. J. Clin. Nutr. 2010, 64, 436–438. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, I.A.; Moore, C.; Chatfield, M.; Richardson, D.P.; Ashby, P.; Kuznesof, S.A.; Jebb, S.A.; Seal, C.J. Markers of cardiovascular risk are not changed by increased whole-grain intake: The wholeheart study, a randomised, controlled dietary intervention. Br. J. Nutr. 2010, 104, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Ampatzoglou, A.; Atwal, K.K.; Maidens, C.M.; Williams, C.L.; Ross, A.B.; Thielecke, F.; Jonnalagadda, S.S.; Kennedy, O.B.; Yaqoob, P. Increased whole grain consumption does not affect blood biochemistry, body composition, or gut microbiology in healthy, low-habitual whole grain consumers. J. Nutr. 2015, 145, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Tighe, P.; Duthie, G.; Vaughan, N.; Brittenden, J.; Simpson, W.G.; Duthie, S.; Mutch, W.; Wahle, K.; Horgan, G.; Thies, F. Effect of increased consumption of whole-grain foods on blood pressure and other cardiovascular risk markers in healthy middle-aged persons: A randomized controlled trial. Am. J. Clin. Nutr. 2010, 92, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Ampatzoglou, A.; Williams, C.L.; Atwal, K.K.; Maidens, C.M.; Ross, A.B.; Thielecke, F.; Jonnalagadda, S.S.; Kennedy, O.B.; Yaqoob, P. Effects of increased wholegrain consumption on immune and inflammatory markers in healthy low habitual wholegrain consumers. Eur. J. Nutr. 2016, 55, 183–195. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, G.H.; Noakes, M.; Royle, P.J.; Foster, P.R. Whole-grain rye and wheat foods and markers of bowel health in overweight middle-aged men. Am. J. Clin. Nutr. 2003, 77, 967–974. [Google Scholar] [PubMed]
- Keogh, J.; Atkinson, F.; Eisenhauer, B.; Inamdar, A.; Brand-Miller, J. Food intake, postprandial glucose, insulin and subjective satiety responses to three different bread-based test meals. Appetite 2011, 57, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Stefoska-Needham, A.; Beck, E.J.; Johnson, S.K.; Chu, J.; Tapsell, L.C. Flaked sorghum biscuits increase postprandial glp-1 and gip levels and extend subjective satiety in healthy subjects. Mol. Nutr. Food Res. 2016, 60, 1118–1128. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, M.; Toubro, S.; Jensen, M.G.; Ross, A.B.; Riboldi, G.; Petronio, M.; Bugel, S.; Tetens, I.; Astrup, A. Whole grain compared with refined wheat decreases the percentage of body fat following a 12-week, energy-restricted dietary intervention in postmenopausal women. J. Nutr. 2012, 142, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Bakhoj, S.; Flint, A.; Holst, J.J.; Tetens, I. Lower glucose-dependent insulinotropic polypeptide (gip) response but similar glucagon-like peptide 1 (glp-1), glycaemic, and insulinaemic response to ancient wheat compared to modern wheat depends on processing. Eur. J. Clin. Nutr. 2003, 57, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, M.; Jensen, M.G.; Riboldi, G.; Petronio, M.; Bugel, S.; Toubro, S.; Tetens, I.; Astrup, A. Wholegrain vs. Refined wheat bread and pasta. Effect on postprandial glycemia, appetite, and subsequent ad libitum energy intake in young healthy adults. Appetite 2010, 54, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Vitaglione, P.; Mennella, I.; Ferracane, R.; Rivellese, A.A.; Giacco, R.; Ercolini, D.; Gibbons, S.M.; La Storia, A.; Gilbert, J.A.; Jonnalagadda, S.; et al. Whole-grain wheat consumption reduces inflammation in a randomized controlled trial on overweight and obese subjects with unhealthy dietary and lifestyle behaviors: Role of polyphenols bound to cereal dietary fiber. Am. J. Clin. Nutr. 2015, 101, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Casiraghi, M.C.; Garsetti, M.; Testolin, G.; Brighenti, F. Post-prandial responses to cereal products enriched with barley beta-glucan. J. Am. Coll. Nutr. 2006, 25, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Saltzman, E.; Das, S.K.; Lichtenstein, A.H.; Dallal, G.E.; Corrales, A.; Schaefer, E.J.; Greenberg, A.S.; Roberts, S.B. An oat-containing hypocaloric diet reduces systolic blood pressure and improves lipid profile beyond effects of weight loss in men and women. J. Nutr. 2001, 131, 1465–1470. [Google Scholar] [PubMed]
- Pereira, M.A.; Jacobs, D.R., Jr.; Pins, J.J.; Raatz, S.K.; Gross, M.D.; Slavin, J.L.; Seaquist, E.R. Effect of whole grains on insulin sensitivity in overweight hyperinsulinemic adults. Am. J. Clin. Nutr. 2002, 75, 848–855. [Google Scholar] [PubMed]
- Poquette, N.M.; Gu, X.; Lee, S.O. Grain sorghum muffin reduces glucose and insulin responses in men. Food Funct. 2014, 5, 894–899. [Google Scholar] [CrossRef] [PubMed]
- Rave, K.; Roggen, K.; Dellweg, S.; Heise, T.; Tom Dieck, H. Improvement of insulin resistance after diet with a whole-grain based dietary product: Results of a randomized, controlled cross-over study in obese subjects with elevated fasting blood glucose. Br. J. Nutr. 2007, 98, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kaneko, T.; Qin, L.Q.; Wang, J.; Wang, Y. Effects of barley intake on glucose tolerance, lipid metabolism, and bowel function in women. Nutrition 2003, 19, 926–929. [Google Scholar] [CrossRef]
- Zafar, T.A.; Al-Hassawi, F.; Al-Khulaifi, F.; Al-Rayyes, G.; Waslien, C.; Huffman, F.G. Organoleptic and glycemic properties of chickpea-wheat composite breads. J. Food Sci. Technol. 2015, 52, 2256–2263. [Google Scholar] [CrossRef] [PubMed]
- Soong, Y.Y.; Quek, R.Y.; Henry, C.J. Glycemic potency of muffins made with wheat, rice, corn, oat and barley flours: A comparative study between in vivo and in vitro. Eur. J. Nutr. 2015, 54, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Anton, C.; Rico, M.C.; Sanchez-Rodriguez, E.; Ruiz-Lopez, M.D.; Gil, A.; Mesa, M.D. Glycemic responses, appetite ratings and gastrointestinal hormone responses of most common breads consumed in spain. A randomized control trial in healthy humans. Nutrients 2015, 7, 4033–4053. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Bruce, S.J.; Blondel-Lubrano, A.; Oguey-Araymon, S.; Beaumont, M.; Bourgeois, A.; Nielsen-Moennoz, C.; Vigo, M.; Fay, L.B.; Kochhar, S.; et al. A whole-grain cereal-rich diet increases plasma betaine, and tends to decrease total and ldl-cholesterol compared with a refined-grain diet in healthy subjects. Br. J. Nutr. 2011, 105, 1492–1502. [Google Scholar] [CrossRef] [PubMed]
- Granfeldt, Y.; Wu, X.; Bjorck, I. Determination of glycaemic index; some methodological aspects related to the analysis of carbohydrate load and characteristics of the previous evening meal. Eur. J. Clin. Nutr. 2006, 60, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Ardisson Korat, A.V.; Willett, W.C.; Hu, F.B. Diet, lifestyle, and genetic risk factors for type 2 diabetes: A review from the nurses’ health study, nurses’ health study 2, and health professionals’ follow-up study. Curr. Nutr. Rep. 2014, 3, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Sahyoun, N.R.; Jacques, P.F.; Zhang, X.L.; Juan, W.; McKeown, N.N. Whole-grain intake is inversely associated with the metabolic syndrome and mortality in older adults. Am. J. Clin. Nutr. 2006, 83, 124–131. [Google Scholar] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.; Manson, J.; Liu, S. Glycemic index, glycemic load, and risk of type 2 diabetes. Am. J. Clin. Nutr. 2002, 76, 274S–280S. [Google Scholar] [PubMed]
- Lopez-Alarcon, M.; Perichart-Perera, O.; Flores-Huerta, S.; Inda-Icaza, P.; Rodriguez-Cruz, M.; Armenta-Alvarez, A.; Bram-Falcon, M.T.; Mayorga-Ochoa, M. Excessive refined carbohydrates and scarce micronutrients intakes increase inflammatory mediators and insulin resistance in prepubertal and pubertal obese children independently of obesity. Mediat. Inflamm. 2014, 2014, 849031. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.D.; Roach, A.K.; Sparks, K.C.; Marquart, L.; D’Agostino, R.B., Jr.; Mayer-Davis, E.J. Whole-grain intake and insulin sensitivity: The insulin resistance atherosclerosis study. Am. J. Clin. Nutr. 2003, 78, 965–971. [Google Scholar] [PubMed]
- Meynier, A.; Goux, A.; Atkinson, F.; Brack, O.; Vinoy, S. Postprandial glycaemic response: How is it influenced by characteristics of cereal products? Br. J. Nutr. 2015, 113, 1931–1939. [Google Scholar] [CrossRef] [PubMed]
- Steffen, L.M.; Jacobs, D.R., Jr.; Murtaugh, M.A.; Moran, A.; Steinberger, J.; Hong, C.P.; Sinaiko, A.R. Whole grain intake is associated with lower body mass and greater insulin sensitivity among adolescents. Am. J. Epidemiol. 2003, 158, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.P. Chronic oxidative stress as a central mechanism for glucose toxicity in pancreatic islet beta cells in diabetes. J. Biol. Chem. 2004, 8, 42351–42354. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, M.; Matuo, T.; Tsuno, T.; Hosoda, A.; Nomura, E.; Taniguchi, H.; Sasaki, H.; Morishita, H. Antioxidant activity and hypoglycemic effect of ferulic acid in stz-induced diabetic mice and kk-ay mice. BioFactors 2004, 21, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Marventano, S.; Kolacz, P.; Castellano, S.; Galvano, F.; Buscemi, S.; Mistretta, A.; Grosso, G. A review of recent evidence in human studies of n-3 and n-6 pufa intake on cardiovascular disease, cancer, and depressive disorders: Does the ratio really matter? Int. J. Food Sci. Nutr. 2015, 66, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations (FAOSTAT). Cereal Production; Food and Agriculture Organization of the United Nations: Rome, Italy, 2016. [Google Scholar]

| Author, Year (Reference) | Country | Design (Washout or Arms) | Participants, Age, Year | BMI, kg/m2 | Test Meals | Matching | Duration | Outcomes Evaluated | Main Results | Study Quality |
|---|---|---|---|---|---|---|---|---|---|---|
| Acute Effect | ||||||||||
| Stefoska-Needham, 2016 | Australia | C (3d) | 40 (20M/20F), 29.3 | 23.4 | I: Whole sorghum biscuits C: Wheat biscuits | none | - | Glucose and insulin iAUC | Greater insulin response (iAUC 4 h) after the red sorghum biscuit | No data |
| Gonzalez-Anton, 2015 | Spain | C (1w) | 23 (13M/10F), 25 ± 1 | 23.3 ± 0.5 | I: Wholemeal C: WWB | 50 g of available carbohydrates | - | Glucose and insulin iAUC | There were no differences in glucose and insulin iAUC | |
| Johansson, 2015 | Sweden | C (6d) | 23 (7M/16F), 60.1 ± 12.1 | 23.8 ± 3.4 | I: uRCB I2: RCB C: WCB | none | - | Glucose and insulin iAUC | Insulin response was lower for RCB (10%) and uRCB (21%) compared with WCB | TOTAL AUC |
| Mofidi, 2015 | Canada | C (1w) | 12M, 54.9 ± 2.0 | 29.1 ± 1.1 | I1: 11-grain I2: Sprouted-grain I3: 12-grain C: WBB | 50 g of available carbohydrates | - | Insulin iAUC | Only sprouted-grain improved postprandial glucose and insulin response | |
| Soong, 2015 | Singapore | C (na) | 12 (4M/8F), 26.2 ± 5.3 | 20.2 ± 1.7 | I: WG barley flour I2: WG oat flour I3: WG yellow corn flour C: Refined wheat flour | 50 g of available carbohydrates | - | Glucose peak and iAUC | Improved postprandial glucose response for I1 and I2 but not for I3 | |
| Zafar, 2015 | Kuwait | C (na) | 13F, 21.4 ± 2.3 | 23.6 ± 2.4 | I: WGB C: WWB | 25 g available carbohydrate | - | Glucose peak and iAUC | Lower glucose peak and iAUC | |
| Luhovyy, 2014 | Canada | C (na) | 30M, 22.9 ± 0.6 | 22.6 ± 0.3 | I1: WG maize (high) I2: WG maize (low) C: Cookies | none | - | Glucose iAUC | Reduction in postprandial glucose response | |
| Moazzami, 2014 | Finland | C (1–2w) | 20F, 61.0 ± 4.8 | 26.0 ± 2.5 | I: WRB C: WWB | 50 g of available carbohydrates | - | Glucose and insulin iAUC | Improved postprandial insulin response but not glucose response | 15 |
| Poquette, 2014 | USA | C (1w) | 10M, 25.1 ± 4.0 | 24.2 ± 2.8 | I: Sorghum flour C: Wheat flour | 50 g of total starch | - | Glucose and insulin iAUC | Improved postprandial glucose and insulin response | 15 |
| Lappi, 2013 | Finland | C (3d) | 15 (6M/9F), 57 | 26 | I: WRB C: WWB | 50 g of available carbohydrates | - | Glucose and insulin iAUC | Improved postprandial insulin response but not glucose response | 15 |
| Keogh, 2011 | Australia | C (2d) | 10F, 29.4 | 21.8 | I: WGB C: WWB | none | - | Glucose and insulin iAUC | Improved postprandial glucose and insulin response | |
| Vuksan, 2010 | Canada | C (2d) | 11 (6M/5F), 30 ± 3.6 | 22.3 ± 2.8 | I1: WG low I2: WG intermediate I3: WG high C: WWB | 50 g of available carbohydrates | - | Glucose iAUC | Reduction in postprandial glucose response | |
| Rosén, 2011 | Sweden | C (1w) | 10 (5M/5F), 26.0 ± 1.1 | 22.6 ± 0.4 | I1: WGRB I2: WGRB-lac I3: RK I4: WK C: WWB | 50 g of available carbohydrates | - | Glucose and insulin total AUC, incremental glucose and insulin peak | Lower early glucose responses (0–60 min), insulin response and incremental glucose and insulin peak | 15 |
| Kristensen, 2010 | Denmark | C (na) | 16 (6M/10F), 24.1 ± 3.8 | 21.7 ± 2.2 | I1: WGB I2: WGP C: WWB and pasta | 50 g of available carbohydrates | - | Glucose iAUC | No differences between any WG product and R product | 15 |
| Hlebowicz, 2009 | Sweden | C (1w) | 10 (3M/7F), 26 ± 1 | 24.1 ± 0.8 | I: WRB C: WWB | 50 g of available carbohydrates | - | Glucose iAUC | No differences between WG product and R product | 15 |
| Najjar, 2009 | Canada | C (>1w) | 10M, 59 ± 2.41 | 30.8 ± 0.95 | I1: WGB I2: WG barley C: WWB | 50 g of available carbohydrates | - | Glucose and insulin AUC | No differences between WG product and R product | 15 |
| Rosén, 2009 | Sweden | C (1w) | 12 (9M/3F), 25.3 ± 0.8 | 23.1 ± 0.6 | I1: WGRB I2: WGRB-lac I3: WGRP C: WWB | 40 g of available carbohydrates | - | Glucose and insulin iAUC | Improved postprandial glucose and insulin response for WG products | 15 |
| Alminger, 2008 | Sweden | C (1w) | 13 (9F/4M), 56 ± 13.2 | 24.4 ± 2.6 | I1: Oat I2: Barley C: Glucose load | 25 g of available carbohydrates | - | Glucose and insulin iAUC | Improved postprandial glucose and insulin response for WG products | 15 |
| Nilsson, 2008 | Sweden | C (>3d) | 12 (7M/5F), 28.3 ± 5.1 | 22.1 ± 2.0 | I1: WK I2: RK I3: Oat kernels I4: Barley kernels I5: WG barley porridge C: WWB | 50 g of available carbohydrates | - | Glucose iAUC | Improved postprandial glucose response for RK and barley kernels consumption | 15 |
| Hlebowicz, 2007 | Sweden | C (>1w) | 12 (6M/6F), 28 ± 4 | 22 ± 2 | I1: WG oat flakes C: Cornflakes | None | - | Glucose iAUC | No differences between WG product and R product | 15 |
| Casiraghi, 2006 | Italy | C (2w) | 10 (5M/5F), 25.4 ± 0.5 | 22.6 ± 0.7 | I1: WWBCr I2: WWBc I3: BCr I4: BC C: WWB | 40 g of available carbohydrates | - | Glucose and insulin iAUC | Improved postprandial glucose and insulin response for WG products | 15 |
| Bakhoj, 2003 | Denmark | C (1w) | 11M, 25 ± 2 | 23 ± 4 | I1: Einkorn honey–salt I2: Einkorn crushed C: Wheat | 50 g of available carbohydrates | - | Glucose and insulin total AUC | No differences between WG product and R product | 15 |
| Juntunen, 2003 | Finland | C (1–2w) | 19F, 61 ± 4.8 | 26 ± 2.5 | I1: WRB I2: High-fibre rye bread C: WWB | 50 g of available carbohydrates | - | Glucose and insulin iAUC, maximal glucose and insulin response | Improved postprandial insulin response for WRB intake and maximal insulin response for both WRB and High-fibre rye bread. No differences for glucose iAUC and maximal response for any WG products | 15 |
| Juntunen, 2002 | Finland | C (1–2w) | 20 (10M/10F), 28.5 ± 1.8 | 22.9 ± 1 | I1: WKRB I2: β-glucan rye bread I3: WGP C: WWB | 50 g of available carbohydrates | - | Maximal glucose and insulin response | Improved maximal glucose response for WGP and improved maximal insulin response for all the WG meals | 15 |
| Leinonnen, 1999 | Finland | C (na) | 20 (10M/10F), M 32 ± 3 F 27 ± 5 | M 24.5 ± 2.2; F 20.3 ± 1.1 | I1: WKRB I2: WRB I3: WRC C: WWB | 50 g of available carbohydrates | - | Glucose and insulin iAUC, maximal glucose and insulin response | Improved insulin iAUC and maximal response for WKRB intake. No differences for glucose iAUC and maximal response for any WG products | 15 |
| Medium-long term effect | ||||||||||
| Ampatzoglou, 2015a | UK | C (4w) | 33 (12M/21F), 48.8 ± 1.1 | 27.9 ± 0.7 | I: WG pasta, rice, snacks, breakfast cereals C: RG pasta, rice, snacks, breakfast cereals | isoenergetic (2000 kcal/day) | 6w | Fasting glucose | Fasting glucose did not differ between groups | 14 |
| Ampatzoglou, 2015b | UK | C (4w) | 33 (12M/21F), 48.8 ± 1.1 | 27.9 ± 0.7 | I: WG pasta, rice, snacks, breakfast cereals C: RG pasta, rice, snacks, breakfast cereals | isoenergetic (2000 kcal/day) | 6w | Fasting insulin | Fasting insulin did not differ between groups | 14 |
| Vitaglione, 2015 | Italy | P (2 arms) | 68 (23M/45F), I: 40 ± 2 C: 37 ± 2 | I: 30.0 ± 0.5 C: 29.5 ± 0.4 | I: 3 WG biscuits C: 1 package of crackers and 3 slices of toasted bread | isoenergetic (1500 kcal/day) | 8w | Fasting glucose and insulin | Fasting glucose and insulin did not differ between groups | 13 |
| Kristensen, 2012 | Denmark | P (2 arms) | 72F, I: 60.3 ± 5.3 C: 59.1 ± 5.6 | I: 30.4 ± 0.6 C: 30.0 ± 0.4 | I: WG pasta, bread and biscuits C: RG pasta, bread and biscuits | hypocaloric (300–1200 kcal/day) | 12w | Fasting glucose and insulin, HOMA and HbA1c | HbA1c, fasting glucose and insulin, and HOMA did not differ between groups | 14 |
| MacKay, 2012 | Canada | C (4–5w) | 14 (10M/4F), 53 ± 6.0 | 26.5 ± 2.9 | I: WG sourdough bread C: WWB | isoenergetic | 6w | Fasting glucose and insulin and HOMA | Fasting glucose and insulin and HOMA did not differ between groups | 13 |
| Ross, 2011 | Switzerland | C (5–7w) | 17 (6M/11F), M 36.5 ± 4.2 F 34.1 ± 3.0 | M 24.5 ± 0.6 F 23.1 ± 0.8 | I: WG pasta, rice, snacks and breakfast cereals C: RG pasta, rice, snacks and breakfast cereals | isoenergetic (2000 kcal/day) | 2w | Fasting glucose | Fasting glucose did not differ between groups | 13 |
| Brownlee, 2010 | UK | P (3 arms) | 266 (132M/134F), 45.7 ± 10 | I: 30.0 ± 3.7 C:30.3 ± 4.5 | I: WG pasta, rice, snacks and breakfast cereals C: RG pasta, rice, snacks and breakfast cereals | none | 16w | Fasting glucose and insulin | Fasting glucose did not differ between groups | 14 |
| Giacco, 2010 | Italy | C (none) | 15 (12M/3F), 54.5 ± 7.6 | 27.4 ± 3.0 | I: WG bread, pasta, rusks and crackers C: RG bread, pasta, rusks and crackers | isoenergetic (2000 kcal/day) | 3w | Fasting glucose and insulin and HOMA | Fasting glucose and insulin and HOMA did not differ between groups | 14 |
| Tighe, 2010 | UK | P (3 arms) | 206 (104M/102F), I1: 51.6 ± 0.8 I2: 52.1 ± 0.9 C: 51.8 ± 0.83 | I1: 28.0 ± 0.5; I2: 27.0 ± 0.4 C: 28.0 ± 0.5 | I1: WG bread and WG cereals I2: WG wheat food plus oat C: RG bread and cereals | isoenergetic (2100 kcal/day) | 16w | Fasting glucose and insulin and HOMA | Fasting glucose and insulin and HOMA did not differ between groups | 13 |
| Costabile, 2008 | UK | C (2w) | 31 (15M/16F), 25 | 20–30 range | I: WG cereals C: Wheat bran cereals | 48 g/day portion | 3w | Fasting glucose and insulin | No significant differences were observed | |
| Anderson, 2007 | Sweden | C (6–8w) | 30 (8M/22F), 59 ± 5 | 28.3 ± 2.0 30.0 ± 4.0 C; | I1: WG bread, crispbread, muesli, pasta, pancakes, scones, pie, pizza C: RG bread, crispbread, muesli, pasta, pancakes, scones, pie, pizza | isoenergetic (2100 kcal/day) | 6w | Fasting glucose and insulin | Fasting glucose and insulin did not differ between groups | 12 |
| Rave, 2007 | Germany | C (2w) | 31 (13M/18F), 51 ± 13 | 33.9 ± 2.3 | I: WG C: MR | isoenergetic (1700 kcal/day) | 4w | Fasting glucose and insulin, HOMA | HOMA, fasting glucose and insulin did not differ between groups | |
| Li, 2003 | Japan | C (4w) | 10F, 20.4 ± 1.3 | 19.2 ± 2.0 | I: Barley diet C: standard diet | isoenergetic (1900 kcal/day) | 4w | Fasting glucose and HbA1 | Fasting glucose and HbA1 did not differ between group | 15 |
| McIntosh, 2003 | Australia | C (none) | 28M, range 40-65 | 30 ± 0.9 | I1: WG bread, crispbread and breakfast cereal I2: WG rye bread, rye crispbread and rye breakfast cereal C: WWB, RG crispbread and rice cereal | isoenergetic (2300 Kcal/day) | 4w | Fasting glucose and insulin | Fasting glucose and insulin were lower in the WG groups | 13 |
| Pereira, 2002 | USA | C (6–9w) | 11 (5M-6F), 41.6 ± 2.67 | 30.2 ± 1.01 | I: WG pasta, rice, snacks, breakfast cereals C: RG pasta, rice, snacks, breakfast cereals | isoenergetic (2000 kcal/day) | 6w | Fasting glucose and insulin and HOMA | Fasting insulin and HOMA were significantly lower in the WG group | 13 |
| Saltzman, 2001 | USA | P (2 arms) | 43 (20M/23F), I: 45.1 ± 22.7 C: 44.1 ± 21.3 | I: 26.1 ± 3.4 C: 26.7 ± 3.2 | I: Standard diet plus WG oat C: Standard diet | hypocaloric (1900 kcal/day) | 6w | Fasting glucose and insulin and HOMA | Fasting glucose and insulin and HOMA did not differ between groups | 15 |
| Glucose | Insulin | |||||||
|---|---|---|---|---|---|---|---|---|
| No. of Datasets | OR (95% CI) | Heterogeneity | No. of Datasets | OR (95% CI) | Heterogeneity | |||
| I2 (%) | p | I2 (%) | p | |||||
| Study design | ||||||||
| Parallel | 8 | −0.03 (−0.17, 0.11) | 58 | 0.03 | 7 | −0.14 (−4.40, 4.12) | 0 | 0.61 |
| Crossover | 8 | −0.04 (−0.15, 0.07) | 0 | 0.49 | 6 | −4.76 (−12.37, 2.86) | 38 | 0.14 |
| Geographical area | ||||||||
| Europe | 11 | −0.02 (−0.12, 0.08) | 44 | 0.06 | 8 | 0.78 (−3.09, 4.66) | 0 | 0.80 |
| Other | 4 | −0.17 (−0.36, 0.02) | 0 | 0.84 | 5 | −10.71 (−18.19, −3.24) | 0 | 0.59 |
| BMI category | ||||||||
| Normal/overweight | 9 | −0.12 (−0.22, −0.02) | 0 | 0.83 | 5 | −3.15 (−12.14, 5.84) | 2 | 0.40 |
| Obese | 5 | 0.04 (−0.11, 0.19) | 58 | 0.04 | 8 | −1.17 (−6.21, 3.86) | 23 | 0.25 |
| Duration | ||||||||
| ≤6 weeks | 10 | −0.05 (−0.16, 0.05) | 0 | 0.54 | 8 | −5.93 (−13.31, 1.44) | 13 | 0.38 |
| >6 weeks | 5 | −0.01 (−0.16, 0.13) | 64 | 0.02 | 6 | 0.56 (−3.78, 5.89) | 0 | 0.89 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marventano, S.; Vetrani, C.; Vitale, M.; Godos, J.; Riccardi, G.; Grosso, G. Whole Grain Intake and Glycaemic Control in Healthy Subjects: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2017, 9, 769. https://doi.org/10.3390/nu9070769
Marventano S, Vetrani C, Vitale M, Godos J, Riccardi G, Grosso G. Whole Grain Intake and Glycaemic Control in Healthy Subjects: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. 2017; 9(7):769. https://doi.org/10.3390/nu9070769
Chicago/Turabian StyleMarventano, Stefano, Claudia Vetrani, Marilena Vitale, Justyna Godos, Gabriele Riccardi, and Giuseppe Grosso. 2017. "Whole Grain Intake and Glycaemic Control in Healthy Subjects: A Systematic Review and Meta-Analysis of Randomized Controlled Trials" Nutrients 9, no. 7: 769. https://doi.org/10.3390/nu9070769






