The Role of Mechanistic Target of Rapamycin (mTOR) Complexes Signaling in the Immune Responses
Abstract
:1. Introduction
2. mTOR Signaling Pathway
2.1. Upstream of mTOR
mTOR Multiple Signaling Components
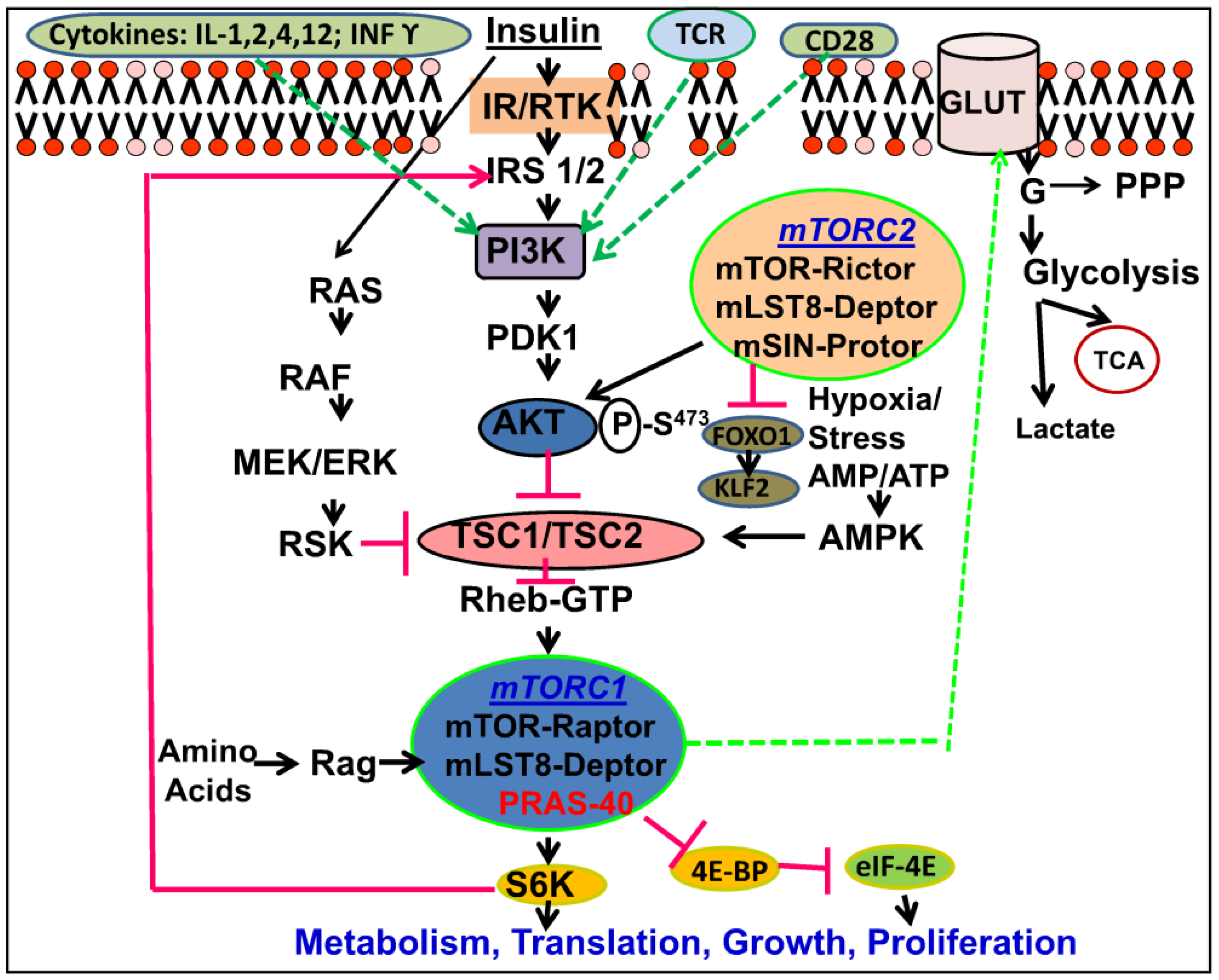
2.2. mTOR Complexes
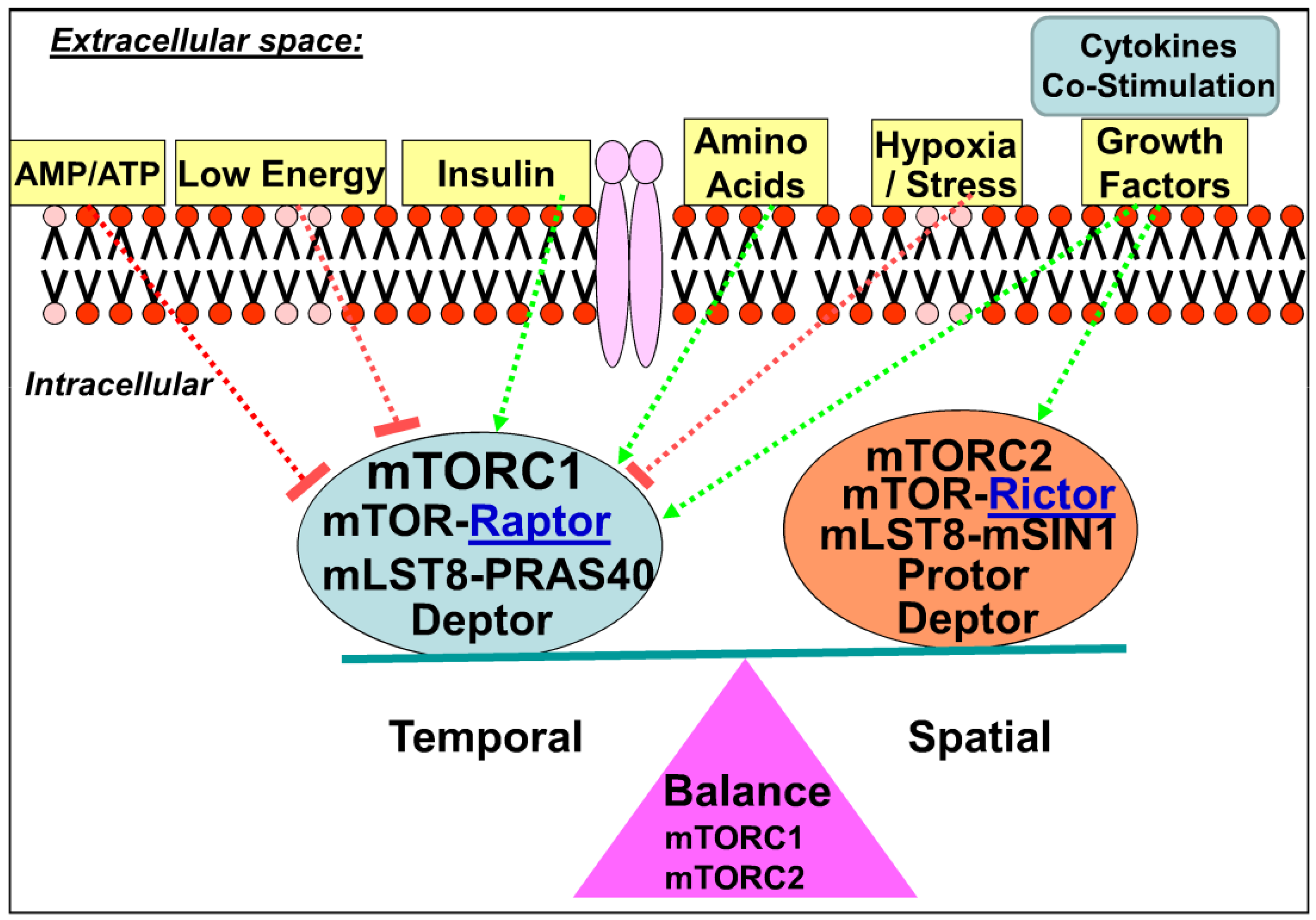
2.3. Downstream of mTOR
2.4. mTOR Pathways and Metabolism
2.4.1. mTOR Regulation of Anabolic and Catabolic Pathways
2.4.2. mTOR and Carbohydrate Metabolism
2.4.3. mTOR and Lipid Metabolism
2.4.4. mTOR and Protein Metabolism
3. mTOR and the Innate Immunity

4. mTOR and Adaptive Immunity
4.1. mTOR and Humoral Immunity (B Cells)
4.2. mTOR and Cell-Mediated Immunity
4.2.1. mTOR and Antigen Presenting Cells
4.2.2. mTOR and T Cell Functions
4.2.3. mTOR and CD4+ CD25+ FoxP3 Regulatory T Cells (Treg)
4.2.4. mTOR and Differentiation of T Helper Cells (T Cell Activation and T Cell Differentiation)
4.2.5. mTOR and Regulation of CD8+ T Effector Cells and CD8+ Memory Cells
4.2.6. mTOR and T Cell Anergy, Tolerance and Hyporesponsiveness
5. Pharmacological Inhibition of mTOR
6. Conclusions
Acknowledgment
Conflict of Interest
References
- Soliman, G.A. The integral role of mTOR in lipid metabolism. Cell Cycle 2011, 10, 861–862. [Google Scholar] [CrossRef]
- Laplante, M.; Sabatini, D.M. mTOR signaling at a glance. J. Cell Sci. 2009, 122, 3589–3594. [Google Scholar] [CrossRef]
- Howell, J.J.; Manning, B.D. mTOR couples cellular nutrient sensing to organismal metabolic homeostasis. Trends Endocrinol. Metab. 2011, 22, 94–102. [Google Scholar] [CrossRef]
- Yecies, J.L.; Manning, B.D. mTOR links oncogenic signaling to tumor cell metabolism. J. Mol. Med. 2011, 89, 221–228. [Google Scholar] [CrossRef]
- Soliman, G.A. The mammalian target of rapamycin signaling network and gene regulation. Curr. Opin. Lipidol. 2005, 16, 317–323. [Google Scholar] [CrossRef]
- Foster, K.G.; Fingar, D.C. Mammalian target of rapamycin (mTOR): Conducting the cellular signaling symphony. J. Biol. Chem. 2010, 285, 14071–14077. [Google Scholar] [CrossRef]
- Stan, R.; McLaughlin, M.M.; Cafferkey, R.; Johnson, R.K.; Rosenberg, M.; Livi, G.P. Interaction between FKBP12-rapamycin and TOR involves a conserved serine residue. J. Biol. Chem. 1994, 269, 32027–32030. [Google Scholar]
- Cafferkey, R.; McLaughlin, M.M.; Young, P.R.; Johnson, R.K.; Livi, G.P. Yeast TOR (DRR) proteins: Amino-acid sequence alignment and identification of structural motifs. Gene 1994, 141, 133–136. [Google Scholar] [CrossRef]
- Stewart, M.J.; Berry, C.O.; Zilberman, F.; Thomas, G.; Kozma, S.C. The Drosophila p70s6k homolog exhibits conserved regulatory elements and rapamycin sensitivity. Proc. Natl. Acad. Sci. USA 1996, 93, 10791–10796. [Google Scholar] [CrossRef]
- Polak, P.; Hall, M.N. mTORC2 caught in a SINful Akt. Dev. Cell 2006, 11, 433–434. [Google Scholar] [CrossRef]
- Wullschleger, S.; Loewith, R.; Hall, M.N. TOR signaling in growth and metabolism. Cell 2006, 124, 471–484. [Google Scholar] [CrossRef]
- Corradetti, M.N.; Guan, K.L. Upstream of the mammalian target of rapamycin: Do all roads pass through mTOR? Oncogene 2006, 25, 6347–6360. [Google Scholar] [CrossRef]
- Staschke, K.A.; Dey, S.; Zaborske, J.M.; Palam, L.R.; McClintick, J.N.; Pan, T.; Edenberg, H.J.; Wek, R.C. Integration of general amino acid control and target of rapamycin (TOR) regulatory pathways in nitrogen assimilation in yeast. J. Biol. Chem. 2010, 285, 16893–16911. [Google Scholar] [CrossRef]
- Schmidt, A.; Beck, T.; Koller, A.; Kunz, J.; Hall, M.N. The TOR nutrient signalling pathway phosphorylates NPR1 and inhibits turnover of the tryptophan permease. EMBO J. 1998, 17, 6924–6931. [Google Scholar] [CrossRef]
- Laplante, M.; Sabatini, D.M. An emerging role of mTOR in lipid biosynthesis. Curr. Biol. 2009, 19, R1046–R1052. [Google Scholar] [CrossRef]
- Duvel, K.; Yecies, J.L.; Menon, S.; Raman, P.; Lipovsky, A.I.; Souza, A.L.; Triantafellow, E.; Ma, Q.; Gorski, R.; Cleaver, S.; et al. Activation of a metabolic gene regulatory network downstream of mTOR complex 1. Mol. Cell 2010, 39, 171–183. [Google Scholar] [CrossRef]
- McMahon, G.; Weir, M.R.; Li, X.C.; Mandelbrot, D.A. The evolving role of mTOR inhibition in transplantation tolerance. J. Am. Soc. Nephrol. 2011, 22, 408–415. [Google Scholar] [CrossRef]
- Reidy-Lagunes, D.; Thornton, R. Pancreatic neuroendocrine and carcinoid tumors: What’s new, what’s old, and what’s different? Cur. Oncol. Rep. 2012, 14, 249–256. [Google Scholar] [CrossRef]
- Arva, N.C.; Pappas, J.G.; Bhatla, T.; Raetz, E.A.; Macari, M.; Ginsburg, H.B.; Hajdu, C.H. Well-differentiated pancreatic neuroendocrine carcinoma in tuberous sclerosis––Case report and review of the literature. Am. J. Surg. Pathol. 2012, 36, 149–153. [Google Scholar] [CrossRef]
- Aggarwal, D.; Fernandez, M.L.; Soliman, G.A. Rapamycin, an mTOR inhibitor, disrupts triglyceride metabolism in guinea pigs. Metab. Clin. Exp. 2006, 55, 794–802. [Google Scholar] [CrossRef]
- Chakrabarti, P.; English, T.; Shi, J.; Smas, C.M.; Kandror, K.V. Mammalian target of rapamycin complex 1 suppresses lipolysis, stimulates lipogenesis, and promotes fat storage. Diabetes 2010, 59, 775–781. [Google Scholar] [CrossRef]
- Ogmundsdottir, M.H.; Heublein, S.; Kazi, S.; Reynolds, B.; Visvalingam, S.M.; Shaw, M.K.; Goberdhan, D.C. Proton-assisted amino acid transporter PAT1 complexes with Rag GTPases and activates TORC1 on late endosomal and lysosomal membranes. PLoS One 2012, 7, e36616. [Google Scholar] [CrossRef]
- Cang, C.; Zhou, Y.; Navarro, B.; Seo, Y.J.; Aranda, K.; Shi, L.; Battaglia-Hsu, S.; Nissim, I.; Clapham, D.E.; Ren, D. mTOR regulates lysosomal ATP-sensitive two-pore Na(+) channels to adapt to metabolic state. Cell 2013, 152, 778–790. [Google Scholar] [CrossRef]
- Powell, J.D.; Pollizzi, K.N.; Heikamp, E.B.; Horton, M.R. Regulation of immune responses by mTOR. Ann. Rev. Immunol. 2012, 30, 39–68. [Google Scholar]
- Colombetti, S.; Basso, V.; Mueller, D.L.; Mondino, A. Prolonged TCR/CD28 engagement drives IL-2-independent T cell clonal expansion through signaling mediated by the mammalian target of rapamycin. J. Immunol. 2006, 176, 2730–2738. [Google Scholar]
- Powell, J.D.; Delgoffe, G.M. The mammalian target of rapamycin: Linking T cell differentiation, function, and metabolism. Immunity 2010, 33, 301–311. [Google Scholar] [CrossRef]
- Rao, R.R.; Li, Q.; Odunsi, K.; Shrikant, P.A. The mTOR kinase determines effector versus memory CD8+ T cell fate by regulating the expression of transcription factors T-bet and Eomesodermin. Immunity 2010, 32, 67–78. [Google Scholar] [CrossRef]
- Rao, R.R.; Li, Q.; Shrikant, P.A. Fine-tuning CD8(+) T cell functional responses: mTOR acts as a rheostat for regulating CD8(+) T cell proliferation, survival and differentiation? Cell Cycle 2010, 9, 2996–3001. [Google Scholar] [CrossRef]
- Gulen, M.F.; Kang, Z.; Bulek, K.; Youzhong, W.; Kim, T.W.; Chen, Y.; Altuntas, C.Z.; Sass Bak-Jensen, K.; McGeachy, M.J.; Do, J.S.; et al. The receptor SIGIRR suppresses Th17 cell proliferation via inhibition of the interleukin-1 receptor pathway and mTOR kinase activation. Immunity 2010, 32, 54–66. [Google Scholar] [CrossRef]
- Xiao, H.; Yin, W.; Khan, M.A.; Gulen, M.F.; Zhou, H.; Sham, H.P.; Jacobson, K.; Vallance, B.A.; Li, X. Loss of single immunoglobulin interlukin-1 receptor-related molecule leads to enhanced colonic polyposis in Apc(min) mice. Gastroenterology 2010, 139, 574–585. [Google Scholar] [CrossRef]
- Garami, A.; Zwartkruis, F.J.; Nobukuni, T.; Joaquin, M.; Roccio, M.; Stocker, H.; Kozma, S.C.; Hafen, E.; Bos, J.L.; Thomas, G. Insulin activation of Rheb, a mediator of mTOR/S6K/4E-BP signaling, is inhibited by TSC1 and 2. Mol. Cell 2003, 11, 1457–1466. [Google Scholar] [CrossRef]
- Stocker, H.; Radimerski, T.; Schindelholz, B.; Wittwer, F.; Belawat, P.; Daram, P.; Breuer, S.; Thomas, G.; Hafen, E. Rheb is an essential regulator of S6K in controlling cell growth in Drosophila. Nat. Cell Biol. 2003, 5, 559–565. [Google Scholar]
- Nobukini, T.; Thomas, G. The mTOR/S6K signalling pathway: The role of the TSC1/2 tumour suppressor complex and the proto-oncogene Rheb. Novartis Found. Symp. 2004, 262, 148–154; discussion 154–159, 265–268. [Google Scholar] [CrossRef]
- Kwiatkowski, D.J.; Zhang, H.; Bandura, J.L.; Heiberger, K.M.; Glogauer, M.; el-Hashemite, N.; Onda, H. A mouse model of TSC1 reveals sex-dependent lethality from liver hemangiomas, and up-regulation of p70S6 kinase activity in Tsc1 null cells. Hum. Mol. Genet. 2002, 11, 525–534. [Google Scholar] [CrossRef]
- Kwiatkowski, D.J.; Manning, B.D. Tuberous sclerosis: A GAP at the crossroads of multiple signaling pathways. Hum. Mol. Genet. 2005, 14, R251–R258. [Google Scholar] [CrossRef]
- Inoki, K.; Ouyang, H.; Zhu, T.; Lindvall, C.; Wang, Y.; Zhang, X.; Yang, Q.; Bennett, C.; Harada, Y.; Stankunas, K.; et al. TSC2 integrates Wnt and energy signals via a coordinated phosphorylation by AMPK and GSK3 to regulate cell growth. Cell 2006, 126, 955–968. [Google Scholar] [CrossRef]
- Yoshida, S.; Hong, S.; Suzuki, T.; Nada, S.; Mannan, A.M.; Wang, J.; Okada, M.; Guan, K.L.; Inoki, K. Redox regulates mammalian target of rapamycin complex 1 (mTORC1) activity by modulating the TSC1/TSC2-Rheb GTPase pathway. J. Biol. Chem. 2011, 286, 32651–32660. [Google Scholar] [CrossRef]
- Yang, Q.; Inoki, K.; Kim, E.; Guan, K.L. TSC1/TSC2 and Rheb have different effects on TORC1 and TORC2 activity. Proc. Natl. Acad. Sci. USA 2006, 103, 6811–6816. [Google Scholar] [CrossRef]
- Um, S.H.; Frigerio, F.; Watanabe, M.; Picard, F.; Joaquin, M.; Sticker, M.; Fumagalli, S.; Allegrini, P.R.; Kozma, S.C.; Auwerx, J.; et al. Absence of S6K1 protects against age- and diet-induced obesity while enhancing insulin sensitivity. Nature 2004, 431, 200–205. [Google Scholar] [CrossRef]
- Peterson, T.R.; Sengupta, S.S.; Harris, T.E.; Carmack, A.E.; Kang, S.A.; Balderas, E.; Guertin, D.A.; Madden, K.L.; Carpenter, A.E.; Finck, B.N.; et al. mTOR complex 1 regulates lipin 1 localization to control the SREBP pathway. Cell 2011, 146, 408–420. [Google Scholar] [CrossRef]
- Kim, S.G.; Hoffman, G.R.; Poulogiannis, G.; Buel, G.R.; Jang, Y.J.; Lee, K.W.; Kim, B.Y.; Erikson, R.L.; Cantley, L.C.; Choo, A.Y.; et al. Metabolic stress controls mTORC1 lysosomal localization and dimerization by regulating the TTT-RUVBL1/2 complex. Mol. Cell 2013, 49, 172–185. [Google Scholar]
- Sparks, C.A.; Guertin, D.A. Targeting mTOR: Prospects for mTOR complex 2 inhibitors in cancer therapy. Oncogene 2010, 29, 3733–3744. [Google Scholar] [CrossRef]
- Hresko, R.C.; Mueckler, M. mTOR.RICTOR is the Ser473 kinase for Akt/protein kinase B in 3T3-L1 adipocytes. J. Biol. Chem. 2005, 280, 40406–40416. [Google Scholar] [CrossRef]
- Yecies, J.L.; Manning, B.D. Transcriptional control of cellular metabolism by mTOR signaling. Cancer Res. 2011, 71, 2815–2820. [Google Scholar] [CrossRef]
- Cantor, J.R.; Sabatini, D.M. Cancer cell metabolism: One hallmark, many faces. Cancer Discov. 2012, 2, 881–898. [Google Scholar] [CrossRef]
- Warburg, O. The effect of hydrogen peroxide on cancer cells and on embryonic cells. Acta Unio Int. Contra Cancrum 1958, 14, 55–57. [Google Scholar]
- Zheng, Y.; Delgoffe, G.M.; Meyer, C.F.; Chan, W.; Powell, J.D. Anergic T cells are metabolically anergic. J. Immunol. 2009, 183, 6095–6101. [Google Scholar] [CrossRef]
- Delgoffe, G.M.; Powell, J.D. mTOR: Taking cues from the immune microenvironment. Immunology 2009, 127, 459–465. [Google Scholar] [CrossRef]
- Powell, J.D.; Lerner, C.G.; Schwartz, R.H. Inhibition of cell cycle progression by rapamycin induces T cell clonal anergy even in the presence of costimulation. J. Immunol. 1999, 162, 2775–2784. [Google Scholar]
- Allen, A.; Zheng, Y.; Gardner, L.; Safford, M.; Horton, M.R.; Powell, J.D. The novel cyclophilin binding compound, sanglifehrin A, disassociates G1 cell cycle arrest from tolerance induction. J. Immunol. 2004, 172, 4797–4803. [Google Scholar]
- Vanasek, T.L.; Khoruts, A.; Zell, T.; Mueller, D.L. Antagonistic roles for CTLA-4 and the mammalian target of rapamycin in the regulation of clonal anergy: Enhanced cell cycle progression promotes recall antigen responsiveness. J. Immunol. 2001, 167, 5636–5644. [Google Scholar]
- Hay, N.; Sonenberg, N. Upstream and downstream of mTOR. Genes Dev. 2004, 18, 1926–1945. [Google Scholar] [CrossRef]
- Lekmine, F.; Uddin, S.; Sassano, A.; Parmar, S.; Brachmann, S.M.; Majchrzak, B.; Sonenberg, N.; Hay, N.; Fish, E.N.; Platanias, L.C. Activation of the p70 S6 kinase and phosphorylation of the 4E-BP1 repressor of mRNA translation by type I interferons. J. Biol. Chem. 2003, 278, 27772–27780. [Google Scholar] [CrossRef]
- Gingras, A.C.; Kennedy, S.G.; O’Leary, M.A.; Sonenberg, N.; Hay, N. 4E-BP1, a repressor of mRNA translation, is phosphorylated and inactivated by the Akt(PKB) signaling pathway. Genes Dev. 1998, 12, 502–513. [Google Scholar] [CrossRef]
- Fingar, D.C.; Richardson, C.J.; Tee, A.R.; Cheatham, L.; Tsou, C.; Blenis, J. mTOR controls cell cycle progression through its cell growth effectors S6K1 and 4E-BP1/eukaryotic translation initiation factor 4E. Mol. Cell. Biol. 2004, 24, 200–216. [Google Scholar] [CrossRef]
- Fingar, D.C.; Blenis, J. Target of rapamycin (TOR): An integrator of nutrient and growth factor signals and coordinator of cell growth and cell cycle progression. Oncogene 2004, 23, 3151–3171. [Google Scholar] [CrossRef]
- Guertin, D.A.; Stevens, D.M.; Thoreen, C.C.; Burds, A.A.; Kalaany, N.Y.; Moffat, J.; Brown, M.; Fitzgerald, K.J.; Sabatini, D.M. Ablation in mice of the mTORC components raptor, rictor, or mLST8 reveals that mTORC2 is required for signaling to Akt-FOXO and PKCalpha, but not S6K1. Dev. Cell 2006, 11, 859–871. [Google Scholar] [CrossRef]
- Lazorchak, A.S.; Su, B. Perspectives on the role of mTORC2 in B lymphocyte development, immunity and tumorigenesis. Protein Cell 2011, 2, 523–530. [Google Scholar] [CrossRef]
- Yuan, M.; Pino, E.; Wu, L.; Kacergis, M.; Soukas, A.A. Identification of Akt-independent regulation of hepatic lipogenesis by mammalian target of rapamycin (mTOR) complex 2. J. Biol. Chem. 2012, 287, 29579–29588. [Google Scholar] [CrossRef]
- Mayer, C.; Grummt, I. Ribosome biogenesis and cell growth: mTOR coordinates transcription by all three classes of nuclear RNA polymerases. Oncogene 2006, 25, 6384–6391. [Google Scholar] [CrossRef]
- White, R.J.; Sharrocks, A.D. Coordinated control of the gene expression machinery. Trends Genet 2010, 26, 214–220. [Google Scholar] [CrossRef]
- Gingras, A.C.; Raught, B.; Sonenberg, N. mTOR signaling to translation. Curr. Top. Microbiol. Immunol. 2004, 279, 169–197. [Google Scholar]
- Proud, C.G. Role of mTOR signalling in the control of translation initiation and elongation by nutrients. Curr. Top. Microbiol. Immunol. 2004, 279, 215–244. [Google Scholar] [CrossRef]
- Proud, C.G. The multifaceted role of mTOR in cellular stress responses. DNA Repair 2004, 3, 927–934. [Google Scholar] [CrossRef]
- Desvergne, B.; Michalik, L.; Wahli, W. Transcriptional regulation of metabolism. Physiol. Rev. 2006, 86, 465–514. [Google Scholar] [CrossRef]
- Lian, J.; Yan, X.H.; Peng, J.; Jiang, S.W. The mammalian target of rapamycin pathway and its role in molecular nutrition regulation. Mol. Nutr. Food Res. 2008, 52, 393–399. [Google Scholar] [CrossRef]
- Peng, T.; Golub, T.R.; Sabatini, D.M. The immunosuppressant rapamycin mimics a starvation-like signal distinct from amino acid and glucose deprivation. Mol. Cell. Biol. 2002, 22, 5575–5584. [Google Scholar] [CrossRef]
- Brown, N.F.; Stefanovic-Racic, M.; Sipula, I.J.; Perdomo, G. The mammalian target of rapamycin regulates lipid metabolism in primary cultures of rat hepatocytes. Metabolism 2007, 56, 1500–1507. [Google Scholar] [CrossRef]
- Porstmann, T.; Santos, C.R.; Griffiths, B.; Cully, M.; Wu, M.; Leevers, S.; Griffiths, J.R.; Chung, Y.L.; Schulze, A. SREBP activity is regulated by mTORC1 and contributes to Akt-dependent cell growth. Cell Metab. 2008, 8, 224–236. [Google Scholar] [CrossRef]
- Mauvoisin, D.; Rocque, G.; Arfa, O.; Radenne, A.; Boissier, P.; Mounier, C. Role of the PI3-kinase/mTor pathway in the regulation of the stearoyl CoA desaturase (SCD1) gene expression by insulin in liver. J. Cell Commun. Signal. 2007, 1, 113–125. [Google Scholar] [CrossRef]
- Kumar, A.; Lawrence, J.C., Jr.; Jung, D.Y.; Ko, H.J.; Keller, S.R.; Kim, J.K.; Magnuson, M.A.; Harris, T.E. Fat cell-specific ablation of rictor in mice impairs insulin-regulated fat cell and whole-body glucose and lipid metabolism. Diabetes 2010, 59, 1397–1406. [Google Scholar] [CrossRef]
- Meijer, A.J.; Codogno, P. Regulation and role of autophagy in mammalian cells. Int. J. Biochem. Cell Biol. 2004, 36, 2445–2462. [Google Scholar] [CrossRef]
- Iwamaru, A.; Kondo, Y.; Iwado, E.; Aoki, H.; Fujiwara, K.; Yokoyama, T.; Mills, G.B.; Kondo, S. Silencing mammalian target of rapamycin signaling by small interfering RNA enhances rapamycin-induced autophagy in malignant glioma cells. Oncogene 2007, 26, 1840–1851. [Google Scholar] [CrossRef]
- Saltiel, A.R.; Kahn, C.R. Insulin signalling and the regulation of glucose and lipid metabolism. Nature 2001, 414, 799–806. [Google Scholar] [CrossRef]
- Zhong, H.; Chiles, K.; Feldser, D.; Laughner, E.; Hanrahan, C.; Georgescu, M.M.; Simons, J.W.; Semenza, G.L. Modulation of hypoxia-inducible factor 1alpha expression by the epidermal growth factor/phosphatidylinositol 3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells: Implications for tumor angiogenesis and therapeutics. Cancer Res. 2000, 60, 1541–1545. [Google Scholar]
- Hudson, C.C.; Liu, M.; Chiang, G.G.; Otterness, D.M.; Loomis, D.C.; Kaper, F.; Giaccia, A.J.; Abraham, R.T. Regulation of hypoxia-inducible factor 1alpha expression and function by the mammalian target of rapamycin. Mol. Cell. Biol. 2002, 22, 7004–7014. [Google Scholar] [CrossRef]
- Hu, C.J.; Wang, L.Y.; Chodosh, L.A.; Keith, B.; Simon, M.C. Differential roles of hypoxia-inducible factor 1α (HIF-1α) and HIF-2α in hypoxic gene regulation. Mol. Cell. Biol. 2003, 23, 9361–9374. [Google Scholar] [CrossRef]
- Liu, H.; Remedi, M.S.; Pappan, K.L.; Kwon, G.; Rohatgi, N.; Marshall, C.A.; McDaniel, M.L. Glycogen synthase kinase-3 and mammalian target of rapamycin pathways contribute to DNA synthesis, cell cycle progression, and proliferation in human islets. Diabetes 2009, 58, 663–672. [Google Scholar]
- Wagle, A.; Jivraj, S.; Garlock, G.L.; Stapleton, S.R. Insulin regulation of glucose-6-phosphate dehydrogenase gene expression is rapamycin-sensitive and requires phosphatidylinositol 3-kinase. J. Biol. Chem. 1998, 273, 14968–14974. [Google Scholar] [CrossRef]
- Dunlop, E.A.; Tee, A.R. Mammalian target of rapamycin complex 1: Signalling inputs, substrates and feedback mechanisms. Cell Signal. 2009, 21, 827–835. [Google Scholar] [CrossRef]
- Vila-Bedmar, R.; Lorenzo, M.; Fernandez-Veledo, S. Adenosine 5′-monophosphate-activated protein kinase-mammalian target of rapamycin cross talk regulates brown adipocyte differentiation. Endocrinology 2010, 151, 980–992. [Google Scholar] [CrossRef]
- Ekim, B.; Magnuson, B.; Acosta-Jaquez, H.A.; Keller, J.A.; Feener, E.P.; Fingar, D.C. mTOR kinase domain phosphorylation promotes mTORC1 signaling, cell growth, and cell cycle progression. Mol. Cell. Biol. 2011, 31, 2787–2801. [Google Scholar] [CrossRef]
- Festuccia, W.T.; Laplante, M.; Brule, S.; Houde, V.P.; Achouba, A.; Lachance, D.; Pedrosa, M.L.; Silva, M.E.; Guerra-Sa, R.; Couet, J.; et al. Rosiglitazone-induced heart remodelling is associated with enhanced turnover of myofibrillar protein and mTOR activation. J. Mol. Cell. Cardiol. 2009, 47, 85–95. [Google Scholar] [CrossRef]
- Powers, T. Cell growth control: mTOR takes on fat. Mol. Cell 2008, 31, 775–776. [Google Scholar] [CrossRef]
- Soliman, G.A.; Acosta-Jaquez, H.A.; Fingar, D.C. mTORC1 inhibition via rapamycin promotes triacylglycerol lipolysis and release of free fatty acids in 3T3-L1 adipocytes. Lipids 2010, 45, 1089–1100. [Google Scholar] [CrossRef]
- Fraenkel, M.; Ketzinel-Gilad, M.; Ariav, Y.; Pappo, O.; Karaca, M.; Castel, J.; Berthault, M.F.; Magnan, C.; Cerasi, E.; Kaiser, N.; et al. mTOR inhibition by rapamycin prevents β-cell adaptation to hyperglycemia and exacerbates the metabolic state in type 2 diabetes. Diabetes 2008, 57, 945–957. [Google Scholar] [CrossRef]
- Leibowitz, G.; Kaiser, N.; Cerasi, E. Balancing needs and means: The dilemma of the beta-cell in the modern world. Diabetes Obes. Metab. 2009, 11 (Suppl. 4), 1–9. [Google Scholar] [CrossRef]
- Kimball, S.R. Regulation of translation initiation by amino acids in eukaryotic cells. Prog. Mol. Subcell Biol. 2001, 26, 155–184. [Google Scholar] [CrossRef]
- Roh, C.; Han, J.; Tzatsos, A.; Kandror, K.V. Nutrient-sensing mTOR-mediated pathway regulates leptin production in isolated rat adipocytes. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E322–E330. [Google Scholar]
- Tokunaga, C.; Yoshino, K.; Yonezawa, K. mTOR integrates amino acid- and energy-sensing pathways. Biochem. Biophys. Res. Commun. 2004, 313, 443–446. [Google Scholar] [CrossRef]
- Kim, D.H.; Sabatini, D.M. Raptor and mTOR: Subunits of a nutrient-sensitive complex. Curr. Top. Microbiol. Immunol. 2004, 279, 259–270. [Google Scholar] [CrossRef]
- Kimball, S.R.; Jefferson, L.S. Molecular mechanisms through which amino acids mediate signaling through the mammalian target of rapamycin. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 39–44. [Google Scholar] [CrossRef]
- Matsumura, T.; Morinaga, Y.; Fujitani, S.; Takehana, K.; Nishitani, S.; Sonaka, I. Oral administration of branched-chain amino acids activates the mTOR signal in cirrhotic rat liver. Hepatol. Res. 2005, 33, 27–32. [Google Scholar] [CrossRef]
- Nishitani, S.; Ijichi, C.; Takehana, K.; Fujitani, S.; Sonaka, I. Pharmacological activities of branched-chain amino acids: Specificity of tissue and signal transduction. Biochem. Biophys. Res. Commun. 2004, 313, 387–389. [Google Scholar] [CrossRef]
- Roscic, A.; Baldo, B.; Crochemore, C.; Marcellin, D.; Paganetti, P. Induction of autophagy with catalytic mTOR inhibitors reduces huntingtin aggregates in a neuronal cell model. J. Neurochem. 2011, 119, 398–407. [Google Scholar] [CrossRef]
- Delves, P.J.; Maitin, S.; Burton, D.R.; Roitt, I.M. Roitt’s Essential Immunology, 12th ed; Wiley-Blackwell: Hoboken, NJ, USA, 2011. [Google Scholar]
- Chi, H. Regulation and function of mTOR signalling in T cell fate decisions. Nat. Rev. Immunol. 2012, 12, 325–338. [Google Scholar]
- Iwasaki, A.; Medzhitov, R. Regulation of adaptive immunity by the innate immune system. Science 2010, 327, 291–295. [Google Scholar] [CrossRef]
- Haidinger, M.; Poglitsch, M.; Geyeregger, R.; Kasturi, S.; Zeyda, M.; Zlabinger, G.J.; Pulendran, B.; Horl, W.H.; Saemann, M.D.; Weichhart, T. A versatile role of mammalian target of rapamycin in human dendritic cell function and differentiation. J. Immunol. 2010, 185, 3919–3931. [Google Scholar] [CrossRef]
- Katholnig, K.; Kaltenecker, C.C.; Hayakawa, H.; Rosner, M.; Lassnig, C.; Zlabinger, G.J.; Gaestel, M.; Muller, M.; Hengstschlager, M.; Horl, W.H.; et al. p38alpha senses environmental stress to control innate immune responses via mechanistic target of rapamycin. J. Immunol. 2013, 190, 1519–1527. [Google Scholar] [CrossRef]
- Haidinger, M.; Werzowa, J.; Weichhart, T.; Saemann, M.D. Targeting the dysregulated mammalian target of rapamycin pathway in organ transplantation: Killing 2 birds with 1 stone. Transplant. Rev. 2011, 25, 145–153. [Google Scholar] [CrossRef]
- Saemann, M.D.; Haidinger, M.; Hecking, M.; Horl, W.H.; Weichhart, T. The multifunctional role of mTOR in innate immunity: Implications for transplant immunity. Am. J. Transplant. 2009, 9, 2655–2661. [Google Scholar] [CrossRef]
- Weichhart, T.; Saemann, M.D. The PI3K/Akt/mTOR pathway in innate immune cells: Emerging therapeutic applications. Ann. Rheum. Dis. 2008, 67 (Suppl. 3), iii70–iii74. [Google Scholar] [CrossRef]
- Hackstein, H.; Taner, T.; Zahorchak, A.F.; Morelli, A.E.; Logar, A.J.; Gessner, A.; Thomson, A.W. Rapamycin inhibits IL-4––Induced dendritic cell maturation in vitro and dendritic cell mobilization and function in vivo. Blood 2003, 101, 4457–4463. [Google Scholar] [CrossRef]
- Hackstein, H.; Taner, T.; Logar, A.J.; Thomson, A.W. Rapamycin inhibits macropinocytosis and mannose receptor-mediated endocytosis by bone marrow-derived dendritic cells. Blood 2002, 100, 1084–1087. [Google Scholar] [CrossRef]
- Monti, P.; Mercalli, A.; Leone, B.E.; Valerio, D.C.; Allavena, P.; Piemonti, L. Rapamycin impairs antigen uptake of human dendritic cells. Transplantation 2003, 75, 137–145. [Google Scholar] [CrossRef]
- Sathaliyawala, T.; O’Gorman, W.E.; Greter, M.; Bogunovic, M.; Konjufca, V.; Hou, Z.E.; Nolan, G.P.; Miller, M.J.; Merad, M.; Reizis, B. Mammalian target of rapamycin controls dendritic cell development downstream of Flt3 ligand signaling. Immunity 2010, 33, 597–606. [Google Scholar] [CrossRef]
- Turnquist, H.R.; Raimondi, G.; Zahorchak, A.F.; Fischer, R.T.; Wang, Z.; Thomson, A.W. Rapamycin-conditioned dendritic cells are poor stimulators of allogeneic CD4+ T cells, but enrich for antigen-specific Foxp3+ T regulatory cells and promote organ transplant tolerance. J. Immunol. 2007, 178, 7018–7031. [Google Scholar]
- Ohtani, M.; Hoshii, T.; Fujii, H.; Koyasu, S.; Hirao, A.; Matsuda, S. Cutting edge: mTORC1 in intestinal CD11c+ CD11b+ dendritic cells regulates intestinal homeostasis by promoting IL-10 production. J. Immunol. 2012, 188, 4736–4740. [Google Scholar] [CrossRef]
- Weichhart, T.; Costantino, G.; Poglitsch, M.; Rosner, M.; Zeyda, M.; Stuhlmeier, K.M.; Kolbe, T.; Stulnig, T.M.; Horl, W.H.; Hengstschlager, M.; et al. The TSC-mTOR signaling pathway regulates the innate inflammatory response. Immunity 2008, 29, 565–577. [Google Scholar] [CrossRef]
- Neudoerfl, C.; Mueller, B.J.; Blume, C.; Daemen, K.; Stevanovic-Meyer, M.; Keil, J.; Lehner, F.; Haller, H.; Falk, C.S. The peripheral NK cell repertoire after kidney transplantation is modulated by different immunosuppressive drugs. Front. Immunol. 2013, 4, 46. [Google Scholar] [CrossRef]
- Weichhart, T. Mammalian target of rapamycin: A signaling kinase for every aspect of cellular life. Methods Mol. Biol. 2012, 821, 1–14. [Google Scholar] [CrossRef]
- Thomson, A.W.; Turnquist, H.R.; Raimondi, G. Immunoregulatory functions of mTOR inhibition. Nat. Rev. Immunol. 2009, 9, 324–337. [Google Scholar] [CrossRef]
- Rosborough, B.R.; Raich-Regue, D.; Matta, B.M.; Lee, K.; Gan, B.; Depinho, R.A.; Hackstein, H.; Boothby, M.; Turnquist, H.R.; Thomson, A.W. Murine dendritic cell rapamycin-resistant and rictor-independent mTOR controls IL-10, B7-H1 and regulatory T cell induction. Blood 2013. [Google Scholar] [CrossRef]
- Schmitz, F.; Heit, A.; Dreher, S.; Eisenacher, K.; Mages, J.; Haas, T.; Krug, A.; Janssen, K.P.; Kirschning, C.J.; Wagner, H. Mammalian target of rapamycin (mTOR) orchestrates the defense program of innate immune cells. Eur. J. Immunol. 2008, 38, 2981–2992. [Google Scholar] [CrossRef]
- Kirsch, A.H.; Riegelbauer, V.; Tagwerker, A.; Rudnicki, M.; Rosenkranz, A.R.; Eller, K. The mTOR-inhibitor rapamycin mediates proteinuria in nephrotoxic serum nephritis by activating the innate immune response. Am. J. Physiol. Ren. Physiol. 2012, 303, F569–F575. [Google Scholar] [CrossRef]
- Wicker, L.S.; Boltz, R.C., Jr.; Matt, V.; Nichols, E.A.; Peterson, L.B.; Sigal, N.H. Suppression of B cell activation by cyclosporin A, FK506 and rapamycin. Eur. J. Immunol. 1990, 20, 2277–2283. [Google Scholar] [CrossRef]
- Kay, J.E.; Kromwel, L.; Doe, S.E.; Denyer, M. Inhibition of T and B lymphocyte proliferation by rapamycin. Immunology 1991, 72, 544–549. [Google Scholar]
- Aagaard-Tillery, K.M.; Jelinek, D.F. Inhibition of human B lymphocyte cell cycle progression and differentiation by rapamycin. Cell. Immunol. 1994, 156, 493–507. [Google Scholar] [CrossRef]
- Sakata, A.; Kuwahara, K.; Ohmura, T.; Inui, S.; Sakaguchi, N. Involvement of a rapamycin-sensitive pathway in CD40-mediated activation of murine B cells in vitro. Immunol. Lett. 1999, 68, 301–309. [Google Scholar] [CrossRef]
- Donahue, A.C.; Fruman, D.A. Distinct signaling mechanisms activate the target of rapamycin in response to different B-cell stimuli. Eur. J. Immunol. 2007, 37, 2923–2936. [Google Scholar] [CrossRef]
- Zhang, S.; Readinger, J.A.; DuBois, W.; Janka-Junttila, M.; Robinson, R.; Pruitt, M.; Bliskovsky, V.; Wu, J.Z.; Sakakibara, K.; Patel, J.; et al. Constitutive reductions in mTOR alter cell size, immune cell development, and antibody production. Blood 2011, 117, 1228–1238. [Google Scholar] [CrossRef]
- Benhamron, S.; Tirosh, B. Direct activation of mTOR in B lymphocytes confers impairment in B-cell maturation andloss of marginal zone B cells. Eur. J. Immunol. 2011, 41, 2390–2396. [Google Scholar] [CrossRef]
- Llorian, M.; Stamataki, Z.; Hill, S.; Turner, M.; Martensson, I.L. The PI3K p110delta is required for down-regulation of RAG expression in immature B cells. J. Immunol. 2007, 178, 1981–1985. [Google Scholar]
- Donahue, A.C.; Fruman, D.A. Proliferation and survival of activated B cells requires sustained antigen receptor engagement and phosphoinositide 3-kinase activation. J. Immunol. 2003, 170, 5851–5860. [Google Scholar]
- Hess, K.L.; Donahue, A.C.; Ng, K.L.; Moore, T.I.; Oak, J.; Fruman, D.A. Frontline: The p85alpha isoform of phosphoinositide 3-kinase is essential for a subset of B cell receptor-initiated signaling responses. Eur. J. Immunol. 2004, 34, 2968–2976. [Google Scholar] [CrossRef]
- Goldfinger, M.; Shmuel, M.; Benhamron, S.; Tirosh, B. Protein synthesis in plasma cells is regulated by crosstalk between endoplasmic reticulum stress and mTOR signaling. Eur. J. Immunol. 2011, 41, 491–502. [Google Scholar] [CrossRef]
- Waickman, A.T.; Powell, J.D. Mammalian target of rapamycin integrates diverse inputs to guide the outcome of antigen recognition in T cells. J. Immunol. 2012, 188, 4721–4729. [Google Scholar] [CrossRef]
- Katzman, S.D.; O’Gorman, W.E.; Villarino, A.V.; Gallo, E.; Friedman, R.S.; Krummel, M.F.; Nolan, G.P.; Abbas, A.K. Duration of antigen receptor signaling determines T-cell tolerance or activation. Proc. Natl. Acad. Sci. USA 2010, 107, 18085–18090. [Google Scholar] [CrossRef]
- Hackstein, H. Rapamycin and dendritic cells: Keep on movin’. Transplantation 2006, 82, 739–740. [Google Scholar] [CrossRef]
- Yang, K.; Neale, G.; Green, D.R.; He, W.; Chi, H. The tumor suppressor Tsc1 enforces quiescence of naive T cells to promote immune homeostasis and function. Nat. Immunol. 2011, 12, 888–897. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, H.; Li, L.; Xiao, Y.; Rao, E.; Miao, Z.; Chen, H.; Sun, L.; Li, H.; Liu, G.; et al. TSC1/2 signaling complex is essential for peripheral naive CD8+ T cell survival and homeostasis in mice. PLoS One 2012, 7, e30592. [Google Scholar] [CrossRef]
- Gao, N.; Zhang, Z.; Jiang, B.H.; Shi, X. Role of PI3K/AKT/mTOR signaling in the cell cycle progression of human prostate cancer. Biochem. Biophys. Res. Commun. 2003, 310, 1124–1132. [Google Scholar] [CrossRef]
- Gao, N.; Flynn, D.C.; Zhang, Z.; Zhong, X.S.; Walker, V.; Liu, K.J.; Shi, X.; Jiang, B.H. G1 cell cycle progression and the expression of G1 cyclins are regulated by PI3K/AKT/mTOR/p70S6K1 signaling in human ovarian cancer cells. Am. J. Physiol. Cell Physiol. 2004, 287, C281–C291. [Google Scholar] [CrossRef]
- Jones, R.G.; Thompson, C.B. Revving the engine: Signal transduction fuels T cell activation. Immunity 2007, 27, 173–178. [Google Scholar] [CrossRef]
- De Zoeten, E.F.; Lee, I.; Wang, L.; Chen, C.; Ge, G.; Wells, A.D.; Hancock, W.W.; Ozkaynak, E. Foxp3 processing by proprotein convertases and control of regulatory T cell function. J. Biol. Chem. 2009, 284, 5709–5716. [Google Scholar]
- Mercer, F.; Unutmaz, D. The biology of FoxP3: A key player in immune suppression during infections, autoimmune diseases and cancer. Adv. Exp. Med. Biol. 2009, 665, 47–59. [Google Scholar] [CrossRef]
- Battaglia, M.; Stabilini, A.; Roncarolo, M.G. Rapamycin selectively expands CD4+CD25+FoxP3+ regulatory T cells. Blood 2005, 105, 4743–4748. [Google Scholar] [CrossRef]
- Battaglia, M.; Stabilini, A.; Migliavacca, B.; Horejs-Hoeck, J.; Kaupper, T.; Roncarolo, M.G. Rapamycin promotes expansion of functional CD4+CD25+FOXP3+ regulatory T cells of both healthy subjects and type 1 diabetic patients. J. Immunol. 2006, 177, 8338–8347. [Google Scholar]
- Monti, P.; Scirpoli, M.; Maffi, P.; Piemonti, L.; Secchi, A.; Bonifacio, E.; Roncarolo, M.G.; Battaglia, M. Rapamycin monotherapy in patients with type 1 diabetes modifies CD4+CD25+FOXP3+ regulatory T-cells. Diabetes 2008, 57, 2341–2347. [Google Scholar] [CrossRef]
- Kang, J.; Huddleston, S.J.; Fraser, J.M.; Khoruts, A. De novo induction of antigen-specific CD4+CD25+Foxp3+ regulatory T cells in vivo following systemic antigen administration accompanied by blockade of mTOR. J. Leukoc. Biol. 2008, 83, 1230–1239. [Google Scholar] [CrossRef]
- Kopf, H.; de la Rosa, G.M.; Howard, O.M.; Chen, X. Rapamycin inhibits differentiation of Th17 cells and promotes generation of FoxP3+ T regulatory cells. Int. Immunopharmacol. 2007, 7, 1819–1824. [Google Scholar] [CrossRef]
- Zeiser, R.; Leveson-Gower, D.B.; Zambricki, E.A.; Kambham, N.; Beilhack, A.; Loh, J.; Hou, J.Z.; Negrin, R.S. Differential impact of mammalian target of rapamycin inhibition on CD4+CD25+Foxp3+ regulatory T cells compared with conventional CD4+ T cells. Blood 2008, 111, 453–462. [Google Scholar] [CrossRef]
- Delgoffe, G.M.; Pollizzi, K.N.; Waickman, A.T.; Heikamp, E.; Meyers, D.J.; Horton, M.R.; Xiao, B.; Worley, P.F.; Powell, J.D. The kinase mTOR regulates the differentiation of helper T cells through the selective activation of signaling by mTORC1 and mTORC2. Nat. Immunol. 2011, 12, 295–303. [Google Scholar]
- Shi, L.Z.; Wang, R.; Huang, G.; Vogel, P.; Neale, G.; Green, D.R.; Chi, H. HIF1alpha-dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. J. Exp. Med. 2011, 208, 1367–1376. [Google Scholar] [CrossRef]
- Liu, Q.; Xu, C.; Kirubakaran, S.; Zhang, X.; Hur, W.; Liu, Y.; Kwiatkowski, N.P.; Wang, J.; Westover, K.D.; Gao, P.; et al. Characterization of Torin2, an ATP-competitive inhibitor of mTOR, ATM and ATR. Cancer Res. 2013, 73, 2574–2586. [Google Scholar] [CrossRef]
- Cobbold, S.P.; Adams, E.; Farquhar, C.A.; Nolan, K.F.; Howie, D.; Lui, K.O.; Fairchild, P.J.; Mellor, A.L.; Ron, D.; Waldmann, H. Infectious tolerance via the consumption of essential amino acids and mTOR signaling. Proc. Natl. Acad. Sci. USA 2009, 106, 12055–12060. [Google Scholar] [CrossRef]
- Peter, C.; Waldmann, H.; Cobbold, S.P. mTOR signalling and metabolic regulation of T cell differentiation. Curr. Opin. Immunol. 2010, 22, 655–661. [Google Scholar] [CrossRef]
- Merkenschlager, M.; von Boehmer, H. PI3 kinase signalling blocks Foxp3 expression by sequestering Foxo factors. J. Exp. Med. 2010, 207, 1347–1350. [Google Scholar] [CrossRef]
- Sauer, S.; Bruno, L.; Hertweck, A.; Finlay, D.; Leleu, M.; Spivakov, M.; Knight, Z.A.; Cobb, B.S.; Cantrell, D.; O’Connor, E.; et al. T cell receptor signaling controls Foxp3 expression via PI3K, Akt, and mTOR. Proc. Natl. Acad. Sci. USA 2008, 105, 7797–7802. [Google Scholar] [CrossRef]
- Delgoffe, G.M.; Kole, T.P.; Zheng, Y.; Zarek, P.E.; Matthews, K.L.; Xiao, B.; Worley, P.F.; Kozma, S.C.; Powell, J.D. The mTOR kinase differentially regulates effector and regulatory T cell lineage commitment. Immunity 2009, 30, 832–844. [Google Scholar] [CrossRef]
- Waickman, A.T.; Powell, J.D. mTOR, metabolism, and the regulation of T-cell differentiation and function. Immunol. Rev. 2012, 249, 43–58. [Google Scholar] [CrossRef]
- Lee, K.; Gudapati, P.; Dragovic, S.; Spencer, C.; Joyce, S.; Killeen, N.; Magnuson, M.A.; Boothby, M. Mammalian target of rapamycin protein complex 2 regulates differentiation of Th1 and Th2 cell subsets via distinct signaling pathways. Immunity 2010, 32, 743–753. [Google Scholar] [CrossRef]
- Delgoffe, G.M.; Powell, J.D. Exploring functional in vivo consequences of the selective genetic ablation of mTOR signaling in T helper lymphocytes. Methods Mol. Biol. 2012, 821, 317–327. [Google Scholar] [CrossRef]
- Yamada, T.; Gierach, K.; Lee, P.H.; Wang, X.; Lacorazza, H.D. Cutting edge: Expression of the transcription factor E74-like factor 4 is regulated by the mammalian target of rapamycin pathway in CD8+ T cells. J. Immunol. 2010, 185, 3824–3828. [Google Scholar] [CrossRef]
- Araki, K.; Youngblood, B.; Ahmed, R. The role of mTOR in memory CD8 T-cell differentiation. Immunol. Rev. 2010, 235, 234–243. [Google Scholar] [CrossRef]
- Li, Q.; Rao, R.R.; Araki, K.; Pollizzi, K.; Odunsi, K.; Powell, J.D.; Shrikant, P.A. A central role for mTOR kinase in homeostatic proliferation induced CD8+ T cell memory and tumor immunity. Immunity 2011, 34, 541–553. [Google Scholar] [CrossRef]
- Finlay, D.K. mTORC1 regulates CD8+ T-cell glucose metabolism and function independently of PI3K and PKB. Biochem. Soc. Trans. 2013, 41, 681–686. [Google Scholar] [CrossRef]
- Finlay, D.; Cantrell, D. Phosphoinositide 3-kinase and the mammalian target of rapamycin pathways control T cell migration. Ann. N. Y. Acad. Sci. 2010, 1183, 149–157. [Google Scholar]
- Finlay, D.K.; Rosenzweig, E.; Sinclair, L.V.; Feijoo-Carnero, C.; Hukelmann, J.L.; Rolf, J.; Panteleyev, A.A.; Okkenhaug, K.; Cantrell, D.A. PDK1 regulation of mTOR and hypoxia-inducible factor 1 integrate metabolism and migration of CD8+ T cells. J. Exp. Med. 2012, 209, 2441–2453. [Google Scholar] [CrossRef]
- Kurebayashi, Y.; Nagai, S.; Ikejiri, A.; Koyasu, S. Recent advances in understanding the molecular mechanisms of the development and function of Th17 cells. Genes Cells Devoted Mol. Cell. Mech. 2013, 18, 247–265. [Google Scholar] [CrossRef]
- Ikejiri, A.; Nagai, S.; Goda, N.; Kurebayashi, Y.; Osada-Oka, M.; Takubo, K.; Suda, T.; Koyasu, S. Dynamic regulation of Th17 differentiation by oxygen concentrations. Int. Immunol. 2012, 24, 137–146. [Google Scholar]
- Sengupta, S.; Peterson, T.R.; Sabatini, D.M. Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress. Mol. Cell 2010, 40, 310–322. [Google Scholar] [CrossRef]
- Majumder, P.K.; Febbo, P.G.; Bikoff, R.; Berger, R.; Xue, Q.; McMahon, L.M.; Manola, J.; Brugarolas, J.; McDonnell, T.J.; Golub, T.R.; et al. mTOR inhibition reverses Akt-dependent prostate intraepithelial neoplasia through regulation of apoptotic and HIF-1-dependent pathways. Nat. Med. 2004, 10, 594–601. [Google Scholar] [CrossRef]
- Brugarolas, J.; Lei, K.; Hurley, R.L.; Manning, B.D.; Reiling, J.H.; Hafen, E.; Witters, L.A.; Ellisen, L.W.; Kaelin, W.G., Jr. Regulation of mTOR function in response to hypoxia by REDD1 and the TSC1/TSC2 tumor suppressor complex. Genes Dev. 2004, 18, 2893–2904. [Google Scholar] [CrossRef]
- Araki, K.; Turner, A.P.; Shaffer, V.O.; Gangappa, S.; Keller, S.A.; Bachmann, M.F.; Larsen, C.P.; Ahmed, R. mTOR regulates memory CD8 T-cell differentiation. Nature 2009, 460, 108–112. [Google Scholar] [CrossRef]
- He, S.; Kato, K.; Jiang, J.; Wahl, D.R.; Mineishi, S.; Fisher, E.M.; Murasko, D.M.; Glick, G.D.; Zhang, Y. Characterization of the metabolic phenotype of rapamycin-treated CD8+ T cells with augmented ability to generate long-lasting memory cells. PLoS One 2011, 6, e20107. [Google Scholar] [CrossRef]
- Pearce, E.L.; Walsh, M.C.; Cejas, P.J.; Harms, G.M.; Shen, H.; Wang, L.S.; Jones, R.G.; Choi, Y. Enhancing CD8 T-cell memory by modulating fatty acid metabolism. Nature 2009, 460, 103–107. [Google Scholar] [CrossRef]
- Hand, T.W.; Cui, W.; Jung, Y.W.; Sefik, E.; Joshi, N.S.; Chandele, A.; Liu, Y.; Kaech, S.M. Differential effects of STAT5 and PI3K/AKT signaling on effector and memory CD8 T-cell survival. Proc. Natl. Acad. Sci. USA 2010, 107, 16601–16606. [Google Scholar]
- Wang, Y.; Wang, X.Y.; Subjeck, J.R.; Shrikant, P.A.; Kim, H.L. Temsirolimus, an mTOR inhibitor, enhances anti-tumour effects of heat shock protein cancer vaccines. Br. J. Cancer 2011, 104, 643–652. [Google Scholar] [CrossRef]
- Turner, A.P.; Shaffer, V.O.; Araki, K.; Martens, C.; Turner, P.L.; Gangappa, S.; Ford, M.L.; Ahmed, R.; Kirk, A.D.; Larsen, C.P. Sirolimus enhances the magnitude and quality of viral-specific CD8+ T-cell responses to vaccinia virus vaccination in rhesus macaques. Am. J. Transplant. 2011, 11, 613–618. [Google Scholar] [CrossRef]
- El Essawy, B.; Putheti, P.; Gao, W.; Strom, T.B. Rapamycin generates graft-homing murine suppressor CD8(+) T cells that confer donor-specific graft protection. Cell Transplant. 2011, 20, 1759–1769. [Google Scholar] [CrossRef]
- Ferrer, I.R.; Wagener, M.E.; Robertson, J.M.; Turner, A.P.; Araki, K.; Ahmed, R.; Kirk, A.D.; Larsen, C.P.; Ford, M.L. Cutting edge: Rapamycin augments pathogen-specific but not graft-reactive CD8+ T cell responses. J. Immunol. 2010, 185, 2004–2008. [Google Scholar] [CrossRef]
- Zheng, Y.; Collins, S.L.; Lutz, M.A.; Allen, A.N.; Kole, T.P.; Zarek, P.E.; Powell, J.D. A role for mammalian target of rapamycin in regulating T cell activation versus anergy. J. Immunol. 2007, 178, 2163–2170. [Google Scholar]
- Taner, T.; Hackstein, H.; Wang, Z.; Morelli, A.E.; Thomson, A.W. Rapamycin-treated, alloantigen-pulsed host dendritic cells induce ag-specific T cell regulation and prolong graft survival. Am. J. Transplant. 2005, 5, 228–236. [Google Scholar] [CrossRef]
- Reichardt, W.; Durr, C.; von Elverfeldt, D.; Juttner, E.; Gerlach, U.V.; Yamada, M.; Smith, B.; Negrin, R.S.; Zeiser, R. Impact of mammalian target of rapamycin inhibition on lymphoid homing and tolerogenic function of nanoparticle-labeled dendritic cells following allogeneic hematopoietic cell transplantation. J. Immunol. 2008, 181, 4770–4779. [Google Scholar]
- Gabardi, S.; Baroletti, S.A. Everolimus: A proliferation signal inhibitor with clinical applications in organ transplantation, oncology, and cardiology. Pharmacotherapy 2010, 30, 1044–1056. [Google Scholar] [CrossRef]
- Dantal, J.; Berthoux, F.; Moal, M.C.; Rostaing, L.; Legendre, C.; Genin, R.; Toupance, O.; Moulin, B.; Merville, P.; Rerolle, J.P.; et al. Efficacy and safety of de novo or early everolimus with low cyclosporine in deceased-donor kidney transplant recipients at specified risk of delayed graft function: 12-Month results of a randomized, multicenter trial. Transpl. Int. 2010, 23, 1084–1093. [Google Scholar] [CrossRef]
- Kahan, B.D. Fifteen years of clinical studies and clinical practice in renal transplantation: Reviewing outcomes with de novo use of sirolimus in combination with cyclosporine. Transpl. Proc. 2008, 40 (Suppl. 10), S17–S20. [Google Scholar] [CrossRef]
- Rosner, D.; McCarthy, N.; Bennett, M. Rapamycin inhibits human in stent restenosis vascular smooth muscle cells independently of pRB phosphorylation and p53. Cardiovasc. Res. 2005, 66, 601–610. [Google Scholar] [CrossRef]
- Sampson, J.R. Therapeutic targeting of mTOR in tuberous sclerosis. Biochem. Soc. Trans. 2009, 37, 259–264. [Google Scholar] [CrossRef]
- Hoogeveen, R.C.; Ballantyne, C.M.; Pownall, H.J.; Opekun, A.R.; Hachey, D.L.; Jaffe, J.S.; Oppermann, S.; Kahan, B.D.; Morrisett, J.D. Effect of sirolimus on the metabolism of apoB100-containing lipoproteins in renal transplant patients. Transplantation 2001, 72, 1244–1250. [Google Scholar] [CrossRef]
- Kahan, B.D.; Napoli, K.L.; Kelly, P.A.; Podbielski, J.; Hussein, I.; Urbauer, D.L.; Katz, S.H.; van Buren, C.T. Therapeutic drug monitoring of sirolimus: Correlations with efficacy and toxicity. Clin. Transplant. 2000, 14, 97–109. [Google Scholar] [CrossRef]
- Chueh, S.C.; Kahan, B.D. Dyslipidemia in renal transplant recipients treated with a sirolimus and cyclosporine-based immunosuppressive regimen: Incidence, risk factors, progression, and prognosis. Transplantation 2003, 76, 375–382. [Google Scholar] [CrossRef]
- Kovarik, J.M.; Kaplan, B.; Tedesco Silva, H.; Kahan, B.D.; Dantal, J.; Vitko, S.; Boger, R.; Rordorf, C. Exposure-response relationships for everolimus in de novo kidney transplantation: Defining a therapeutic range. Transplantation 2002, 73, 920–925. [Google Scholar] [CrossRef]
- McGaha, T.L.; Huang, L.; Lemos, H.; Metz, R.; Mautino, M.; Prendergast, G.C.; Mellor, A.L. Amino acid catabolism: A pivotal regulator of innate and adaptive immunity. Immunol. Rev. 2012, 249, 135–157. [Google Scholar] [CrossRef]
- Morrisett, J.D.; Abdel-Fattah, G.; Hoogeveen, R.; Mitchell, E.; Ballantyne, C.M.; Pownall, H.J.; Opekun, A.R.; Jaffe, J.S.; Oppermann, S.; Kahan, B.D. Effects of sirolimus on plasma lipids, lipoprotein levels, and fatty acid metabolism in renal transplant patients. J. Lipid Res. 2002, 43, 1170–1180. [Google Scholar]
- Meiser, B.; Buchholz, S.; Kaczmarek, I. De-novo calcineurin-inhibitor-free immunosuppression with sirolimus and mycophenolate mofetil after heart transplantation: 5-Year results. Curr. Opin. Organ Transplant. 2011, 16, 522–528. [Google Scholar] [CrossRef]
- Guertin, D.A.; Stevens, D.M.; Saitoh, M.; Kinkel, S.; Crosby, K.; Sheen, J.H.; Mullholland, D.J.; Magnuson, M.A.; Wu, H.; Sabatini, D.M. mTOR complex 2 is required for the development of prostate cancer induced by Pten loss in mice. Cancer Cell 2009, 15, 148–159. [Google Scholar] [CrossRef]
- Guertin, D.A.; Sabatini, D.M. The pharmacology of mTOR inhibition. Sci. Signal. 2009, 2, pe24. [Google Scholar] [CrossRef]
- Guertin, D.A.; Sabatini, D.M. Defining the role of mTOR in cancer. Cancer Cell 2007, 12, 9–22. [Google Scholar] [CrossRef]
- Kordes, S.; Richel, D.J.; Klumpen, H.J.; Weterman, M.J.; Stevens, A.J.; Wilmink, J.W. A phase I/II, non-randomized, feasibility/safety and efficacy study of the combination of everolimus, cetuximab and capecitabine in patients with advanced pancreatic cancer. Investig. New Drugs 2013, 31, 85–91. [Google Scholar] [CrossRef]
- Dong, M.; Phan, A.T.; Yao, J.C. New strategies for advanced neuroendocrine tumors in the era of targeted therapy. Clin. Cancer Res. 2012, 18, 1830–1836. [Google Scholar] [CrossRef]
- Dong, M.; Yao, J.C. mTOR inhibition, a potential novel approach for bronchial carcinoids. Endocr. Relat. Cancer 2011, 18, C15–C18. [Google Scholar] [CrossRef]
- Peng, L.; Schwarz, R.E. Pancreatic neuroendocrine tumors: Signal pathways and targeted therapies. Curr. Mol. Med. 2013, 13, 333–339. [Google Scholar]
- Huang, J.; Manning, B.D. A complex interplay between Akt, TSC2 and the two mTOR complexes. Biochem. Soc. Trans. 2009, 37, 217–222. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Soliman, G.A. The Role of Mechanistic Target of Rapamycin (mTOR) Complexes Signaling in the Immune Responses. Nutrients 2013, 5, 2231-2257. https://doi.org/10.3390/nu5062231
Soliman GA. The Role of Mechanistic Target of Rapamycin (mTOR) Complexes Signaling in the Immune Responses. Nutrients. 2013; 5(6):2231-2257. https://doi.org/10.3390/nu5062231
Chicago/Turabian StyleSoliman, Ghada A. 2013. "The Role of Mechanistic Target of Rapamycin (mTOR) Complexes Signaling in the Immune Responses" Nutrients 5, no. 6: 2231-2257. https://doi.org/10.3390/nu5062231
APA StyleSoliman, G. A. (2013). The Role of Mechanistic Target of Rapamycin (mTOR) Complexes Signaling in the Immune Responses. Nutrients, 5(6), 2231-2257. https://doi.org/10.3390/nu5062231



