Combined System of Activated Sludge and Ozonation for the Treatment of Kraft E1 Effluent
Abstract
:1. Introduction
2. Materials and Methods
2.1. Paper Mill Effluent
2.2. Activated Sludge System
2.3. Ozonation Process
2.4. Analytical Control
3. Results and Discussion
4. Conclusions
Acknowledgments
References and Notes
- Pokhrel, D; Viraraghavan, T. Treatment of pulp and paper mill wastewater - a review. Sci. Total Environ 2004, 333, 37–38. [Google Scholar]
- Kansal, SK; Singh, M; Sud, D. Effluent quality at kraft/soda agro-based paper mills and its treatment using a heterogeneous photocatalytic system. Desalination 2008, 228, 183–190. [Google Scholar]
- Atlow, SC; Bonadonna-Aparo, L; Klibanov, AM. Dephenolization of industrial wastewaters catalyzed by polyphenol oxidade. Biotechnol. Bioeng 1984, 26, 599–603. [Google Scholar]
- Tsang, YF; Hua, FL; Chua, H; Sin, SN; Wang, YJ. Optimization of biological treatment of paper mill effluent in a sequencing batch reactor. Biochem. Eng. J 2007, 34, 193–199. [Google Scholar]
- Mvula, E; von Sonntag, C. Ozonolysis of phenols in aqueous solution. Org. Biomol. Chem 2003, 1, 1749–1753. [Google Scholar]
- Savant, DV; Abdul-Rahman, R; Ranade, DR. Anaerobic degradation of absorbable organic halides (AOX) from pulp and paper industry wastewater. Bioresource Technol 2006, 97, 1092–1104. [Google Scholar]
- Ali, M; Sreekrishnan, T.R. Aquatic toxicity from pulp and paper mill effluent. Adv. Environ. Res 2001, 5, 175–196. [Google Scholar]
- Karrasch, B; Parra, O; Cid, H; Mehrens, M; Pacheco, P; Urrutia, R; Valdovinos, C; Zaror, C. Effects of pulp and paper Mill effluents on the microplankton ans microbial self-purification capabilities of the Bibio River. Chile Sci. Total Environ 2006, 359, 194–208. [Google Scholar]
- Tortella, GR; Diez, MC; Durán, N. Fungal diversity and use in decomposition of environmental pollutants. Crit. Rev. Microbiol 2005, 31, 197–212. [Google Scholar]
- Freire, RS; Kunz, A; Durán, N. Remediation and toxicity removal from Kraft E1 paper mill effluent treatment with ozone. Environ. Technol 2000, 21, 717–721. [Google Scholar]
- Fontanier, V; Baig, S; Albet, J; Molinier, J. Comparation of conventional and catalytic ozonation for the treatment of pulp mill wastewater. Environ. Eng. Sci 2005, 22, 127–136. [Google Scholar]
- Assalin, MR; Almeida, ES; Rosa, MA; Moraes, SG; Durán, N. Application of Ozonization Process in Industrial Wastewaters: Textile, Kraft E1 and Whey Effluents. Environ. Technol 2004a, 25, 867–872. [Google Scholar]
- Assalin, MR; Rosa, MA; Durán, N. Remediation of Kraft effluent by ozonation: effect of applied ozone concentration and initial pH. Ozone-Sci. Eng 2004b, 26, 317–322. [Google Scholar]
- Belmont, M; Xavier, C; Decap, J; Martinez, M; Sierra-Alvarez, P; Vidal, G. Improved aerobic biodegradation of a abietic acid in ECF bleached Kraft mill effluent due to biomass adaptation. J. Hazard. Mater. B 2006, 135, 256–263. [Google Scholar]
- Buzzini, AP; Pires, EC. Evaluation of an upflow anaerobic sludge blanket reactor with partial recirculation of effluent used to treat wastewater from pulp and paper plants. Bioresource Thecnol 2007, 98, 1838–1848. [Google Scholar]
- Malavyva, P; Rathore, V.S. Biorremediation of pulp and paper mill effluent by a novel fungal consortium isolated from polluted soil. Bioresource Technol 2007, 98, 3647–3651. [Google Scholar]
- Nair, IC; Jayachandran, K; Shashidhar, S. Treatment of paper factory effluent using a phenol degrading Alcaligenes sp under free and immobilized condition. Bioresource Technol 2007, 98, 714–716. [Google Scholar]
- Ma, H; Wang, Y. Application of molybdenum and phosphate modified kaolin in electrochemical treatment of paper mill wastewater. J. Hazard. Mater 2007, 145, 417–423. [Google Scholar]
- Rodrigues, AC; Boroski, M; Shimada, NS; Garcia, JC; Nozaki, J; Hioka, N. Treatment of paper pulp and paper Mill by coagulation-flocculation followed by heterogeneous photocatalysis. J. Photochem. Photobiol. A 2008, 194, 1–10. [Google Scholar]
- Thompson, G; Swain, J; Kay, M; Forster, C.F. The treatment of pulp and paper mill effluent: a review. Bioresource Technol 2001, 77, 275–286. [Google Scholar]
- Manttari, M; Kuosa, M; Kallas, J; Nystroma, M. Membrane filtration and ozone treatment of biologically treated effluents from the pulp and paper industry. J. Membrane Sci 2008, 309, 112. [Google Scholar]
- Beltran, FJ; Alvarez, PM; Rodriguez, EM; Rivas, J. Incidence of an ozonation stage on the treatment of cherry stillage by activated sludge. Ozone-Sci. Eng 2004, 26, 257–266. [Google Scholar]
- Hostachy, JC; Lenon, G; Pisicchio, JL; Coste, C; Legay, C. Reduction of pulp and paper mill pollution by ozone treatment. Water Sci. Techol 1997, 35, 262–268. [Google Scholar]
- Freire, RS; Kubota, LS; Duran, N. Remediation and toxicity removal from Kraft E1 paper mill effluent by ozonation. Environ. Technol 2001, 22, 897–904. [Google Scholar]
- Kunz, A; Mansila, H; Duran, N. A degradation and toxicity study of three textile reactive dyes by ozone. Environ. Technol 2002, 23, 911–918. [Google Scholar]
- Pereira, WS; Freire, RS. Ferro zero-Uma nova abordagem para o tratamento de águas contaminadas com compostos orgânicos poluentes. Quim. Nova 2005, 28, 130–136. [Google Scholar]
- Chang, EE; Hsing, HJ; Chiang, PC; Chen, MY; Shyng, JY. The chemical and biological characteristics of coke-oven wastewater by ozonation. J. Hazard. Mater 2008, 156, 560–557. [Google Scholar]
- Morais, ADA; Mounteer, AH; Silveira, DSA. Improvement of eucalyptus bleached kraft pulp effluent treatment through combined ozone-biological treatment. Tappi J 2008, 7, 26–32. [Google Scholar]
- Baig, S; Liechti, P.A. Ozone treatment for biorefractory COD removal. Water Sci. Technol 2001, 43, 197–204. [Google Scholar]
- Bijan, L; Moheseni, M. Integrate ozone and biotreatment of pulp mill effluent and changes in biodegradability and molecular weight distribution of organic compounds. Water Res 2005, 39, 3763–3772. [Google Scholar]
- Diez, MC; Inostroza, L; Ramirez, J. Efficiency evaluation of activated sludge treatment of wastewater from fiber board manufacturing. Biotechnol. Lett 1996, 18, 187–192. [Google Scholar]
- Diez, MC; Castillo, G; Aguilar, L; Vidal, G; Mora, M.L. Operational factors and nutrient effects on activated sludge treatment of Pinus radiata kraft mill wastewater. Bioresource Technol 2002, 83, 131–138. [Google Scholar]
- Kunz, A; Freire, RS; Rohwedder, J.J.R.; Durán, N; Mansilla, H; Rodriguez, J. Assembly and optimization of a system for ozone utilization in laboratory scale. Quim. Nova 1999, 22, 425–429. [Google Scholar]
- Kunz, A; Freire, RS; Rohwedder, JJR; Gutierrrez, JPR; Durán, N. Design and assembly of an ozonation system to production and utilization of ozone in a bench scale. Brazilian Patent PI 9802076-5, 1998. [Google Scholar]
- American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 19th Ed. ed; American Public Health Association: New York, USA, 1995. [Google Scholar]
- Cecen, F. Investigation of substrate degradable and nonbiodegradable portion in several pulp bleaching wastes. Water Sci. Technol 1999, 40, 305–312. [Google Scholar]
- Bjapai, P. Microbial degradation of pollutants in pulp mill effluents. Adv. Appl. Microbial 2001, 48, 79–134. [Google Scholar]
- Orhon, D; Dulkadiroglu, H; Droguel, S; Kabdasli, I; Sozen, S; Babuna, F.G. Ozonation application in activated sludge systems for a textile mill effluent. Water Sci. Technol 2002, 45, 305–313. [Google Scholar]
- Ramalho, RS. Introduction to wastewater treatment process, 2nd Ed ed; Academic Press: New York, USA, 1983. [Google Scholar]
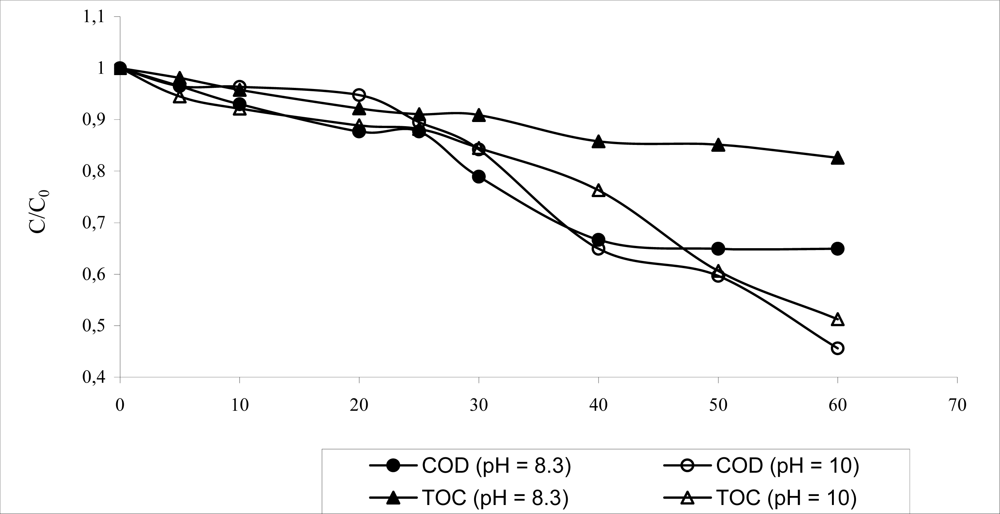
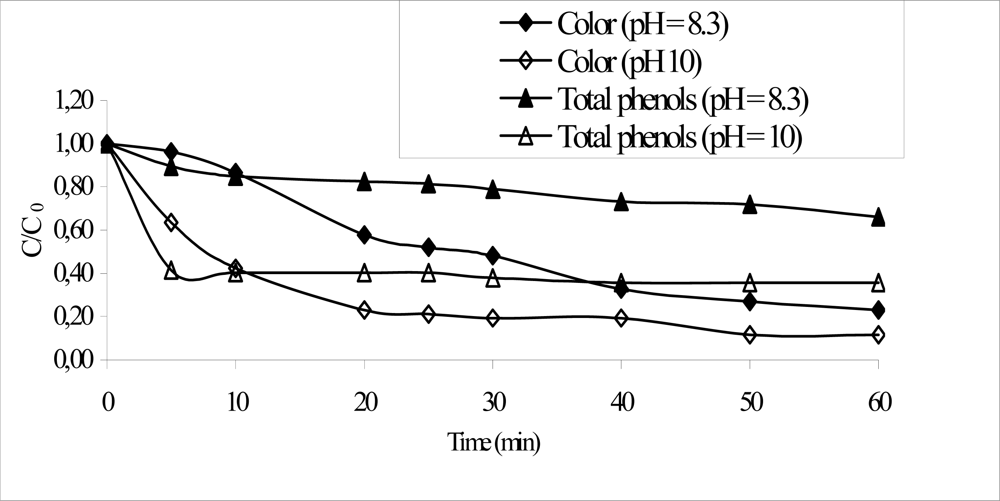
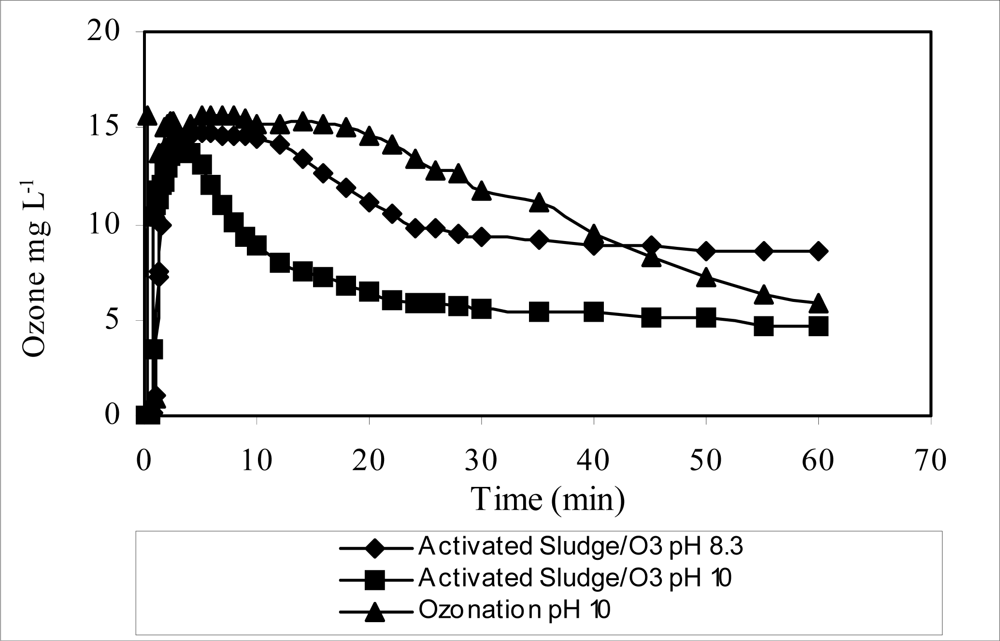
| COD (mg L–1) | 1,500–2,500 |
| TOC (mg L–1) | 500–1,000 |
| Color (at 465 nm) | 0.15–0.30 |
| pH | 9.8–10.2 |
| Total phenol (mg L−1) | 10–30 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Assalin, M.R.; Almeida, E.d.S.; Durán, N. Combined System of Activated Sludge and Ozonation for the Treatment of Kraft E1 Effluent. Int. J. Environ. Res. Public Health 2009, 6, 1145-1154. https://doi.org/10.3390/ijerph6031145
Assalin MR, Almeida EdS, Durán N. Combined System of Activated Sludge and Ozonation for the Treatment of Kraft E1 Effluent. International Journal of Environmental Research and Public Health. 2009; 6(3):1145-1154. https://doi.org/10.3390/ijerph6031145
Chicago/Turabian StyleAssalin, Marcia Regina, Edna dos Santos Almeida, and Nelson Durán. 2009. "Combined System of Activated Sludge and Ozonation for the Treatment of Kraft E1 Effluent" International Journal of Environmental Research and Public Health 6, no. 3: 1145-1154. https://doi.org/10.3390/ijerph6031145
APA StyleAssalin, M. R., Almeida, E. d. S., & Durán, N. (2009). Combined System of Activated Sludge and Ozonation for the Treatment of Kraft E1 Effluent. International Journal of Environmental Research and Public Health, 6(3), 1145-1154. https://doi.org/10.3390/ijerph6031145




