Adverse Effects of a Clinically Relevant Dose of Hydroxyurea Used for the Treatment of Sickle Cell Disease on Male Fertility Endpoints
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
Experiment 1
2.2. Stored Sperm Density
2.3. Progressive Sperm Motility
2.4. Sperm Morphology
2.5. Testosterone Radioimmunoassay (RIA)
Experiment 2
2.6. Histopathology
2.7. Stored Sperm Progressive Motility and Density
2.8. Statistical Analyses
3. Results
4. Discussion
Acknowledgments
References
- Smiley, D; Dagogo-Jack, S; Umpierrez, G. Therapy insight: metabolic and endocrine disorders in sickle cell disease. Nat. Clin. Pract. Metab 2008, 4, 102–109. [Google Scholar]
- Singhal, A; Thomas, P; Cook, R; Wierenga, K; Serjeant, G. Delayed adolescent growth in homozygous sickle cell disease. Arch. Dis. Child 1994, 71, 404–408. [Google Scholar]
- Modebe, O; Ifeno, SA. Growth retardation in homozygous sickle cell disease: role of calorie intake and possible gender related differences. Am. J. Hematol 1993, 44, 149–154. [Google Scholar]
- Rogers, ZR. Priapism in sickle cell disease. Hematol. Oncol. Clin. North Am 2005, 19, 917–928. [Google Scholar]
- Li, M; Forgarty, J; Whitney, KD; Stone, P. Repeated testicular infarction in a patient with sickle cell disease: a possible mechanism for testicular failure. Urology 2003, 62, 551. [Google Scholar]
- Modebe, O; Eze, U. Effect of age on testicular function in adult males with sickle cell anemia. Fertil. Steril 1995, 63, 907–912. [Google Scholar]
- King, BS. A role for nitric oxide in hydroxyurea-mediated fetal hemoglobin induction. J. Clin. Invest 2003, 111, 171–172. [Google Scholar]
- Charache, S; Terrin, ML; Moore, RD; Dover, GJ; Barton, FB; Eckert, SV; McMahon, RP; Bonds, DR. Effect of hydroxyurea on the frequency of painful crises in sickle cell anemia. N. Engl. J. Med 1995, 332, 1317–1322. [Google Scholar]
- Charache, S; Barton, FB; Moore, RD; Terrin, ML; Steinberg, MH; Dover, GJ; Ballas, SK; McMahon, RP; Castro, O; Orringer, EP. Hydroxyurea and sickle cell anemia: clinical utility of a myelosuppressive “switching” agent. Medicine 1996, 75, 300–326. [Google Scholar]
- Cokic, VP; Smith, RD; Beleslin-Cokic, BB; Njoroge, JM; Miller, JL; Gladwin, MT; Schechter, AN. Hydroxyurea induces fetal hemoglobin by the nitric oxide-dependent activation of soluble guanylyl cyclase. J. Clin. Invest 2003, 111, 171–172. [Google Scholar]
- Yarbro, JW. Mechanism of action of hydroxyurea. Semin. Oncol 1992, 19, 1–10. [Google Scholar]
- Fiscor, G; Ginsberg, LC. The effect of hydroxyurea and mitomycin C on sperm mobility in mice. Mutat. Res 1980, 70, 383–387. [Google Scholar]
- Evenson, DP; Jost, LK. Hydroxyurea alters mouse testicular kinetics and sperm chromatin structure. Cell Prolif 1993, 26, 147–159. [Google Scholar]
- Wiger, R; Hongslo, JK; Evenson, DP; de Angelis, P; Schwarze, PE; Holme, JA. Effects of acetaminophen and hydroxyurea on spermatogenesis and sperm chromatin structure in laboratory mice. Reprod. Toxicol 1995, 9, 21–33. [Google Scholar]
- Lu, CC; Meistrich, ML. Cytotoxic effects of chemotherapeutic drugs on mouse testis cells. Cancer Res 1979, 39, 3575–3582. [Google Scholar]
- Singh, H; Taylor, C. Effects of thiotepa and hydroxyurea on sperm production in Lakeview hamsters. J. Toxicol. Environ. Health 1981, 8, 307–316. [Google Scholar]
- Wyrobek, AJ; Bruce, WR. Chemical induction of sperm abnormalities in mice. Proc. Natl. Acad. Sci. U.S.A 1975, 72, 4425–4429. [Google Scholar]
- Shin, JH; Mori, C; Shiota, K. Involvement of germ cell apoptosis in the induction of testicular toxicity following hydroxyurea treatment. Toxicol. Appl. Pharmacol 1999, 155, 139–149. [Google Scholar]
- Garozzo, G; Disca, S; Fidone, C; Bonomo, P. Azoospermia in a patient with sickle cell disease treated with hydroxyurea. Haematologica 2000, 85, 1216–1218. [Google Scholar]
- Grigg, A. Effect of hydroxyurea on sperm count, motility and morphology in adult men with sickle cell or myeloproliferative disease. Intern. Med. J 2007, 37, 190–192. [Google Scholar]
- Berthaut, I; Guignedoux, G; Kirsch-Noir, F; de Larouziere, V; Ravel, C; Bachir, D; Galactéros, F; Ancel, PY; Kunstmann, JM; Levy, L; Jouannet, P; Girot, R; Mandelbaum, J. Influence of sickle cell disease and treatment with hydroxyurea on sperm parameters and fertility of human males. Haematologica 2008, 93, 988–993. [Google Scholar]
- Kopsombut, P; Mukherjee, S; Roa, DP; Turner, EA; Powell, A; Ademoyero, AA; Das, SK; Archibong, AE. Hydroxyurea and indices of male fertility. Adv. Reprod 2000, 4, 9–32. [Google Scholar]
- Popp, RA; Popp, DM; Shinpock, SG; Yang, MY; Mural, JG; Aguinaga, MP; Kopsombut, P; Roa, PD; Turner, EA; Rubin, EM. A transgenic mouse model of hemoglobin s Antilles disease. Blood 1997, 89, 4204–4212. [Google Scholar]
- Rubin, EM; Witkowska, HE. Hypoxia-induced in vitro sickling of transgenic mouse red cells. J. Clin. Invest 1991, 87, 639–647. [Google Scholar]
- Archibong, AE; Ramesh, A; Niaz, MS; Brook, CM; Roberson, SI; Lunstra, DD. Effects of Benzo(a)pyrene on intra-testicular function in F-344 rats. Int. J. Environ. Res. Public Health 2008, 5, 32–40. [Google Scholar]
- Whiten, WK. Nutrient requirements for the culture of preimplantation mouse embryos in vitro. Adv. Biosci 1971, 6, 129–139. [Google Scholar]
- Filler, R. Methods for evaluation of rat epididymal sperm morphology. In Male Reproductive Toxicology, Part A; Heindel, JJ, Chapin, RE, Eds.; Academic Press: New York, USA, 1993; Volume 3, pp. 334–343. [Google Scholar]
- Inyang, F; Ramesh, A; Kopsombut, P; Niaz, MS; Hood, DB; Nyanda, AM; Archibong, AE. Disruption of testicular steroidogenesis and epididymal function by inhaled benzo(a)pyrene. Reprod. Toxicol 2003, 17, 527–537. [Google Scholar]
- Rugh, R. The mouse: its reproduction and development; Burgess Publishing Co: Minneapolis Minn., USA, 1968; pp. 7–43. [Google Scholar]
- Lunstra, DD; Wise, TH; Ford, JJ. Sertoli cells in the boar testis: Changes during development and compensatory hypertrophy after hemicastration at different ages. Biol. Reprod 2003, 68, 140–150. [Google Scholar]
- Steel, RGD; Torrie, JH. Principles and procedures of statistics: A biometrical approach, 2nd Ed ed; McGraw-Hill Book Co.: New York, NY, USA, 1980; pp. 87–343. [Google Scholar]
- Lipshultz, LI. Witt M.A. Infertility in the male. In Infertility, a practical guide for the physician, 3rd Ed; Hammond, MG, Talbert, LM, Eds.; Blackwell Scientific Publications: Boston, MA, USA; pp. 26–55.
- Jones, R. Sperm survival versus degradation in mammalian epididymis: A hypothesis. Biol. Reprod 2004, 71, 1412–1418. [Google Scholar]
- Pham, PT; Pham, PC; Wilkinson, AH; Lew, SQ. Renal abnormalities in sickle cell disease. Kidney Int 2000, 57, 1–8. [Google Scholar]
- Bauer, TW; Moore, GW; Hutchins, GM. The liver in sickle cell disease. A chlinicopathologic study of 70 patients. Am. J. Med 1980, 69, 833–837. [Google Scholar]
- Johnson, CS; Omata, M; Tong, MJ; Simmons, JF, Jr; Weiner, J; Tatter, D. Liver involvement in sickle cell disease. Medicine 1985, 64, 349–356. [Google Scholar]
- Sikka, SC. Role of oxidative stress and antioxidants in andrology and assisted reproductive technology. J. Androl 2004, 25, 5–18. [Google Scholar]
- Donnelly, ET; Lewis, SE; McNally, JA; Thompson, W. In vitro fertilization and pregnancy rates: The influence of sperm motility and morphology on IVF outcome. Fertil. Steril 1998, 70, 305–314. [Google Scholar]
- Jouannet, P; Ducot, B; Feneux, D; Spira, A. Male factors and the likelihood of pregnancy in infertile couples. I. Study of sperm characteristics. Int. J. Androl 1988, 11, 379–384. [Google Scholar]
- Perreault, SD. The mature spermatozoa as a target for reproductive toxicants. In Reproductive and Endocrine Toxicology; Boekelheide, K, Chapin, RE, Hoyer, PB, Harris, C, Eds.; Comprehensive Toxicology, Pergamon Press: New York, NY, USA, 1997; Volume 10, pp. 165–179. [Google Scholar]
- Fibach, E; Rachmilewitz, E. The role of oxidative stress in hemolytic anemia. Curr. Mol. Med 2008, 8, 609–619. [Google Scholar]
- Yu, LK; Gengaro, PE; Niederberger, M; Burke, TJ; Schrier, RW. Nitric oxide: a mediator in rat tubular hypoxia/reoxygenation injury. Proc. Natl. Acad. Sci. U.S.A 1994, 91, 1691–1695. [Google Scholar]
- Wildhirt, SM; Suzuki, H; Wolf, WP; Dudek, R; Horstman, D; Weismueller, S; Reichart, B. S-Methylisothiourea Inhibits Inducible Nitric Oxide Synthase and Improves Left Ventricular Performance after Acute Myocardial Infarction. Biochem. Biophys. Res. Commun 1996, 227, 328–333. [Google Scholar]
- Isobe, M; Katsuramaki, T; Hirata, K; Kimura, H; Nagayama, M; Matsuno, T. Beneficial effects of inducible nitric oxide synthase inhibitor on reperfusion injury in the pig liver. Transplantation 1999, 68, 803–813. [Google Scholar]
- Isobe, M; Katsuramaki, T; Kimura, H; Nagayama, M; Matsuno, T; Yagihashi, A; Hirata, K. Role of inducible nitric oxide synthase on hepatic ischemia and reperfusion injury. Transplant. Proc 2000, 32, 1650–1652. [Google Scholar]
- Farombi, EO; Ugwuezunmba, MC; Ezenwadu, TT; Oyeyemi, MO; Ekor, M. Tetracycline-induced reproductive toxicity in male rats: Effects of vitamin C and N-acetylcysteine. Exp. Toxicol. Pathol 2008, 60, 77–85. [Google Scholar]
- Aitken, RJ; Harkiss, D; Buckingham, D. Relationship between iron-catalyzed lipid peroxidation and human sperm function. J. Reprod. Fertil 1993, 98, 257–265. [Google Scholar]
- Klinefelter, GR. The epididymis as a target for toxicants. In Reproductive and Endocrine Toxicology; Boekelheide, K, Chapin, RE, Hoyer, PB, Harris, C, Eds.; Comprehensive Toxicology, Pergamon Press: New York, NY, USA, 1997; Volume 10, pp. 151–163. [Google Scholar]
- Guthrie, HD; Welch, GR; Long, JA. Mitochondrial function and reactive oxygen species action in relation to boar motility. Theriogenology 2008, 70, 1209–1215. [Google Scholar]
- Miller, RP. Degradation of spermatozoa in the epididymis of a seasonally breeding mammal, the rock hyrax, Procavia capensis. J. Reprod. Fertil 1972, 30, 447–450. [Google Scholar]
- El Hamzi, TH.J; Bahakim, HM; Al-Fawaz, I. Endocrine functions in sickle cell anemia patients. J. Trop. Pediatr 1992, 38, 307–313. [Google Scholar]
- Roger, ZR. Hydroxyurea for diverse pediatric populations with sickle cell disease. Semin. Hematol 1997, 34, 42–47. [Google Scholar]
- Hackney, AC; Hezier, W; Gulledge, TP; Jones, S; Strayhorn, D; Busby, M; Hoffman, E; Orringer, EP. Effects of hydroxyurea administration on the body weight, body composition and exercise performance of patients with sickle cell anemia. Clin. Sci. (Lond) 1997, 92, 481–486. [Google Scholar]
- Florensa, R; Bachs, O; Agell, N. ATM/ATR-independent inhibition of cyclin B accumulation in response to hydroxyurea in nontransformed cell lines is altered in tumor cell lines. Oncogene 2003, 13, 8283–8292. [Google Scholar]
- Lamanna, C; Assisi, L; Vittoria, A; Botte, V; Di Fiore, MM. d-Aspartic acid and nitric oxide as regulators of androgen production in boar testis. Theriogenology 2006, 67, 249–254. [Google Scholar]
- Huang, J; Yakubu, M; Kim-Shapiro, DB; King, B. Rat liver-mediated metabolism of hydroxyurea to nitric oxide. Free Radical Biol. Med 2006, 40, 1675–1681. [Google Scholar]
- Reiter, CD; Gladwin, MT. An emerging role for nitric oxide in sickle cell disease vascular homeostasis and therapy. Curr. Opin. Hematol 2003, 10, 99–107. [Google Scholar]
- Reiter, CD; Wang, J; Tanus-Santos, XE; Hogg, N; Cannon, RO; Schechter, AN; Gladwin, MT. Cell-free hemoglobin limits nitric oxide bioavailability in sickle cell disease. Nat. Med 2002, 8, 1383–1389. [Google Scholar]
- Aslan, M; Ryan, TM; Adler, B; Townes, TM; Parks, DA; Thompson, JA; Tousson, A; Gladwin, MT; Patel, RP; Tarpey, MM; White, RC; Freeman, RC. Oxygen radical inhibition of nitric oxide-dependent vascular function in sickle cell disease. Proc. Natl. Acad. Sci 2001, 98, 15215–15220. [Google Scholar]
- Cokic, VP; Smith, RD; Beleslin-Cokic, BB; Njoroge, JM; Miller, JL; Gladwin, MT; Schechter, AN. Hydroxyurea induces fetal hemoglobin by the nitric oxide-dependent activation of soluble guanylyl cyclase. J. Clin. Invest 2003, 111, 231–239. [Google Scholar]
- Singh, H; Tate, F. Antispermatogenic effects of ethylmethane sulfonate and benzo(a)pyrene on PD4 Lakeview hamsters. J. Toxicol. Environ. Health 1981, 8, 929–937. [Google Scholar]
- Revel, A; Roanani, H; Younglai, E; Xu, J; Han, R; Savouret, J-F; Casper, RF. Resveratrol, a natural aryl hydrocarbon receptor antagonist, protects sperm from DNA damage and apoptosis caused by benzo(a)pyrene. Reprod. Toxicol 2001, 15, 479–486. [Google Scholar]
- Plant, TM; Marshall, GR. The functional significance of FSH in spermatogenesis and the control of its secretion in male primates. Endocr. Rev 2001, 22, 764–786. [Google Scholar]
- Balercia, G; Moretti, S; Vignini, A; Magagnini, M; Mantero, F; Boscaro, M; Ricciardo-Lamonica, G; Mazzanti, L. Role of nitric oxide concentrations on human sperm motility. J. Androl 2004, 25, 245–249. [Google Scholar]
- Li, X; Nokkala, E; Yan, W; Streng, T; Saarinen, N; Warri, A; Huhtaniemi, I; Santti, R; Makela, N; Poutanen, M. Altered structure and function of reproductive organs in transgenic male mice overexpressing human aromatase. Endocrinology 2001, 142, 2435–2442. [Google Scholar]
- Dobashi, M; Fujisawa, M; Yamazaki, T; Okuda, Y; Kanzaki, M; Tatsumi, N; Tsuji, T; Okada, H; Kamidono, S. Inhibition of steroidogenesis in Leydig cells by exogenous nitric oxide occurs independently of steroidogenic acute regulatory protein (StAR) mRNA. Arch. Androl 2001, 47, 203–209. [Google Scholar]
- Kostic, TS; Andric, SA; Maric, D; Stojilkovic, SS; Kovacevic, R. Involvement of inducible nitric oxide synthase in stress-impaired testicular steroidogenesis. J. Endocrinol 1999, 163, 409–416. [Google Scholar]
- Pomerantz, DK; Petelka, V. Nitric oxide is a mediator of the inhibitory effect of activated macrophages on production of androgen by the Leydig cell of the mouse. Endocrinology 1998, 139, 922–931. [Google Scholar]

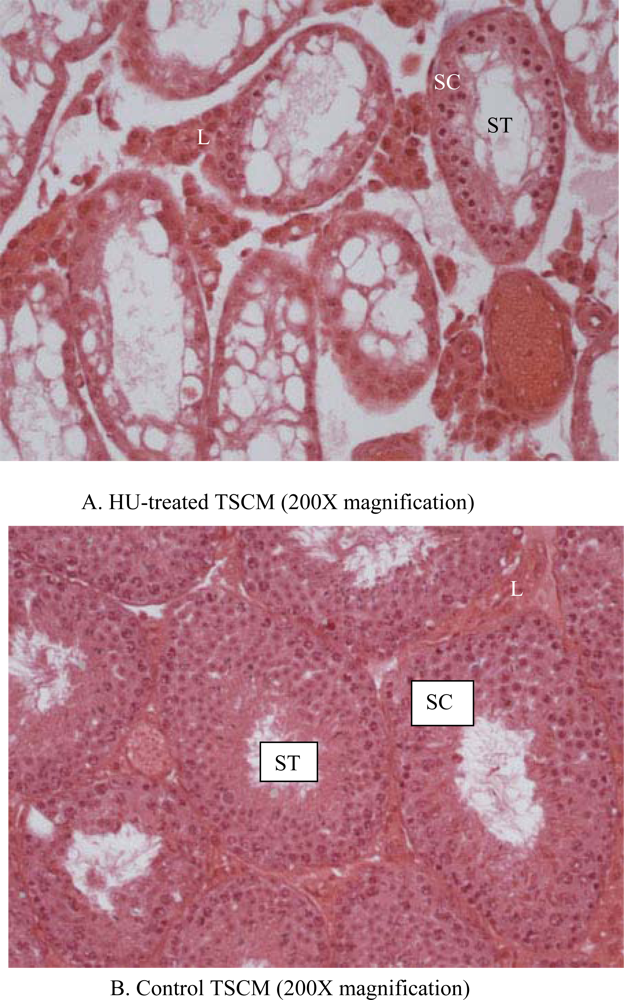
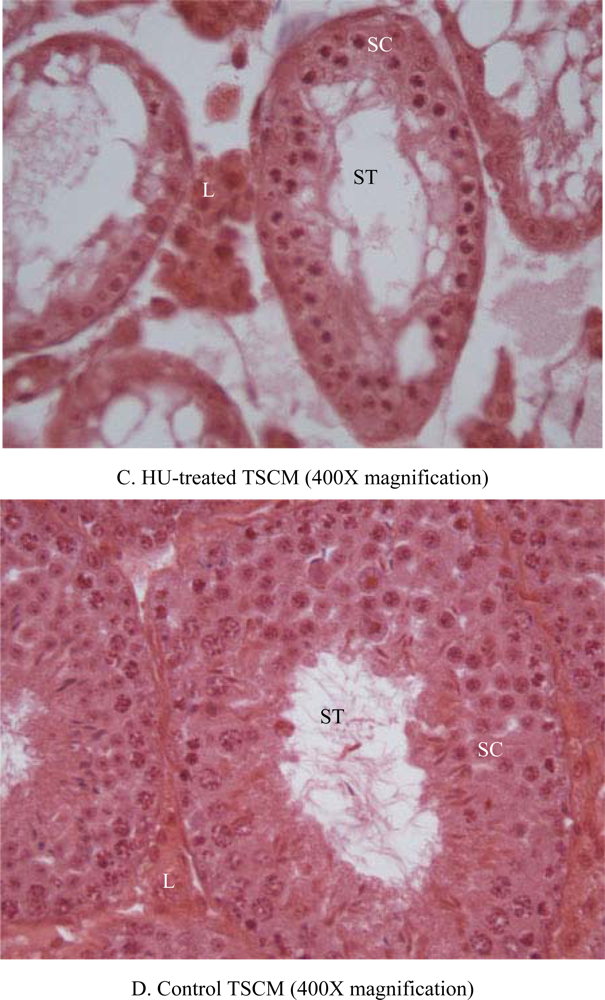



| Item | TSCM | ICRM |
|---|---|---|
| Testis wt (g) | 0.15 ± 0.01** | 0.25 ± 0.01 |
| Stored sperm density (x 106) | 14 ± 3.6*** | 40.3 ± 3.6 |
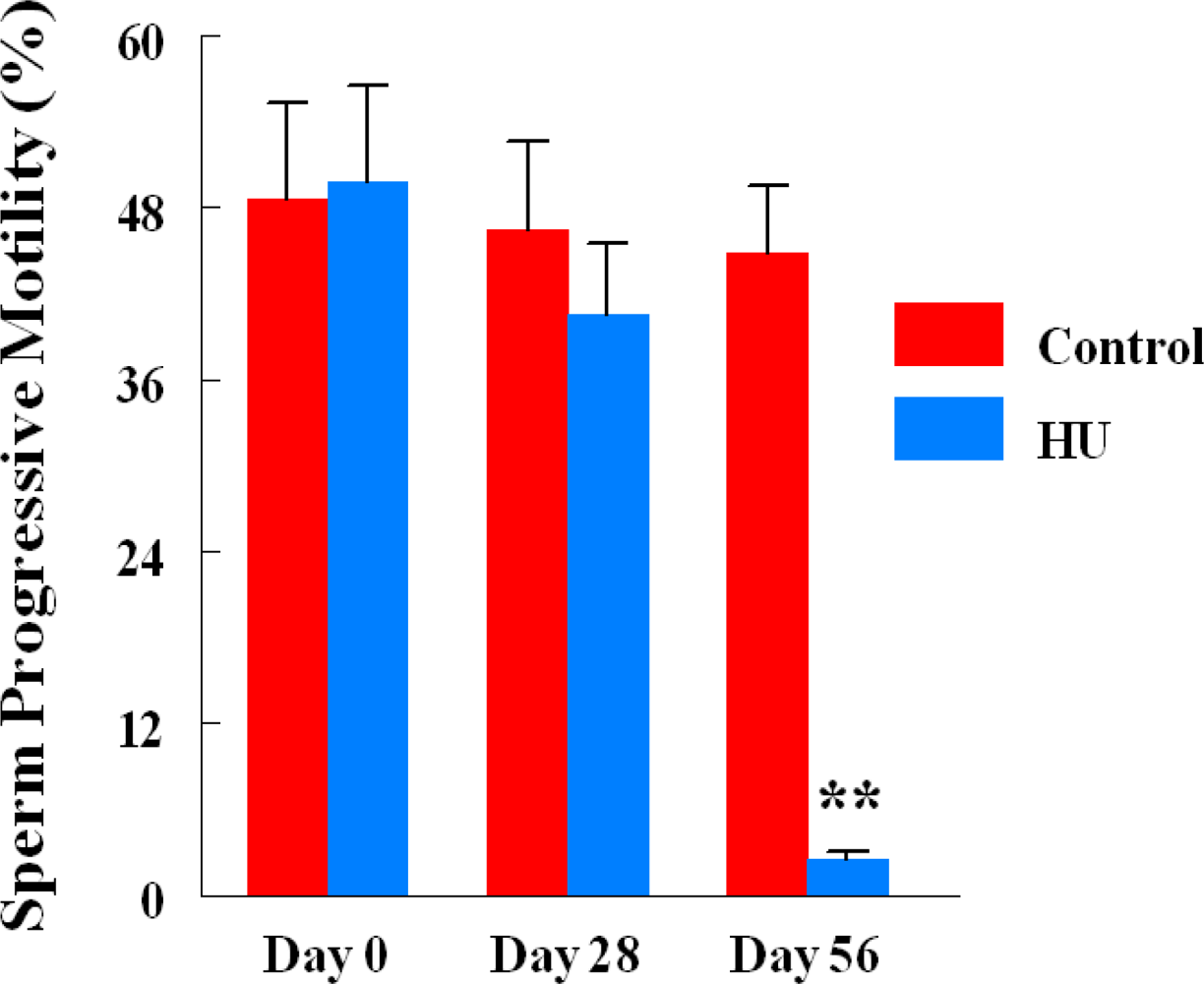
| Sperm progressive motility (%) | 39.0 + 8.7*** | 65.13 + 2.3 |
| Sperm with normal morphology (%) | 14.7 ± 2.3*** | 48.0 ± 5.1 |
| Plasma testosterone concentrations (ng/mL) | 2.4 ± 0.72* | 7.0 ± 0.75 |
| Findings | Control TSCM | HU Treated TSCM | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| #1 | #2 | #3 | #4 | #5 | Mean | #1 | #2 | #3 | #4 | #5 | Mean | |
| Testis dimensions (L X W; sq mm) | 12.0 | 7.5 | 12.0 | 10.5 | 12.0 | 10.8 ± 0.9 | 5.0 | 5.0 | 5.0 | 5.0 | 6.0 | 5.2 ± 0.2*** |
| Seminiferous tubular atrophy/degeneration(Score) | Min. (2.1) | None (1.1) | None (1.1) | None (1.1) | None (1.1) | 1.3 ± 0.2 | Mod. (3.1) | Mod. (3.1) | Mod. (3.1) | Mod. (3.1) | Min. (2.1) | 2.9 ± 0.2*** |
| Leydig cell prominence (Score) | Min. (2.1) | WNL (1.1) | WNL (1.1) | WNL (1.1) | WNL (1.1) | 1.3 + 0.2 | Mod. (3.1) | Mod. (3.1) | Min. (2.1) | Min. (2.1) | WNL (1.1) | 2.3 + 0.4* |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jones, K.M.; Niaz, M.S.; Brooks, C.M.; Roberson, S.I.; Aguinaga, M.P.; Hills, E.R.; Rice, V.M.; Bourne, P.; Bruce, D.; Archibong, A.E. Adverse Effects of a Clinically Relevant Dose of Hydroxyurea Used for the Treatment of Sickle Cell Disease on Male Fertility Endpoints. Int. J. Environ. Res. Public Health 2009, 6, 1124-1144. https://doi.org/10.3390/ijerph6031124
Jones KM, Niaz MS, Brooks CM, Roberson SI, Aguinaga MP, Hills ER, Rice VM, Bourne P, Bruce D, Archibong AE. Adverse Effects of a Clinically Relevant Dose of Hydroxyurea Used for the Treatment of Sickle Cell Disease on Male Fertility Endpoints. International Journal of Environmental Research and Public Health. 2009; 6(3):1124-1144. https://doi.org/10.3390/ijerph6031124
Chicago/Turabian StyleJones, Kea M., Mohammad S. Niaz, Cynthia M. Brooks, Shannon I. Roberson, Maria P. Aguinaga, Edward R. Hills, Valerie Montgomery Rice, Phillip Bourne, Donald Bruce, and Anthony E. Archibong. 2009. "Adverse Effects of a Clinically Relevant Dose of Hydroxyurea Used for the Treatment of Sickle Cell Disease on Male Fertility Endpoints" International Journal of Environmental Research and Public Health 6, no. 3: 1124-1144. https://doi.org/10.3390/ijerph6031124




