EROD and MROD as Markers of Cytochrome P450 1A Activities in Hepatic Microsomes from Entire and Castrated Male Pigs
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals, reagents and standard solutions
2.2. Instrumentation and chromatographic conditions
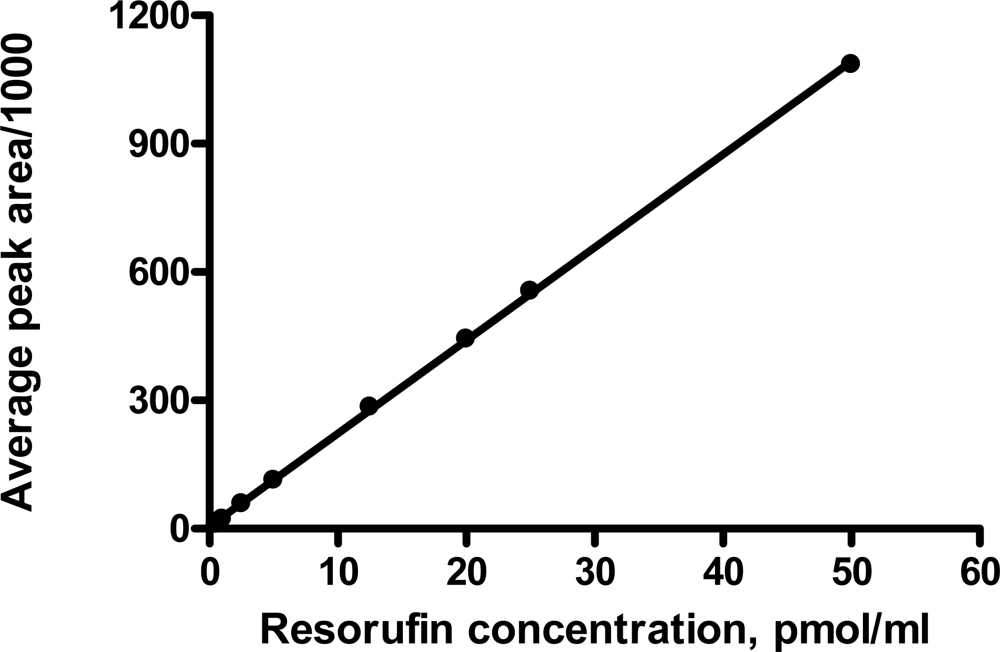
2.3. Standard curve
2.4. Porcine hepatic microsome preparation
2.5. EROD and MROD activity assays
2.6. Linearity with incubation time and protein content and stability
2.7. Recovery, intra- and inter-assay variations, limit of quantitation and stability
2.8. Enzyme kinetic analysis and inhibition constants
3. Results and Discussion
3.1. Fluorescence detection of resorufin
3.2. Validation of microsomal incubation
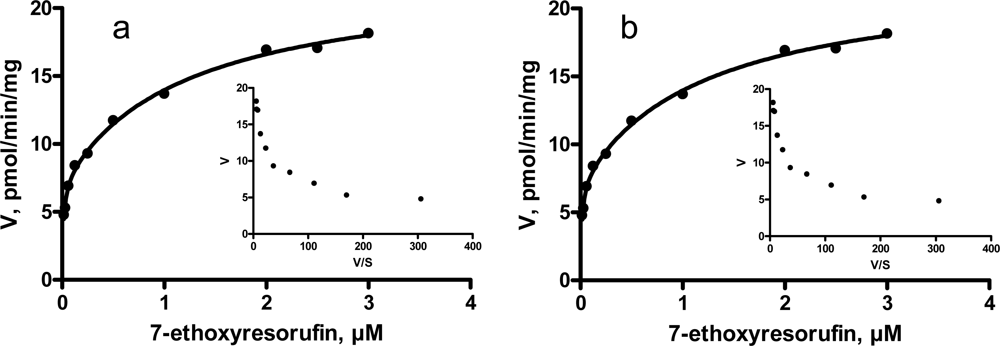
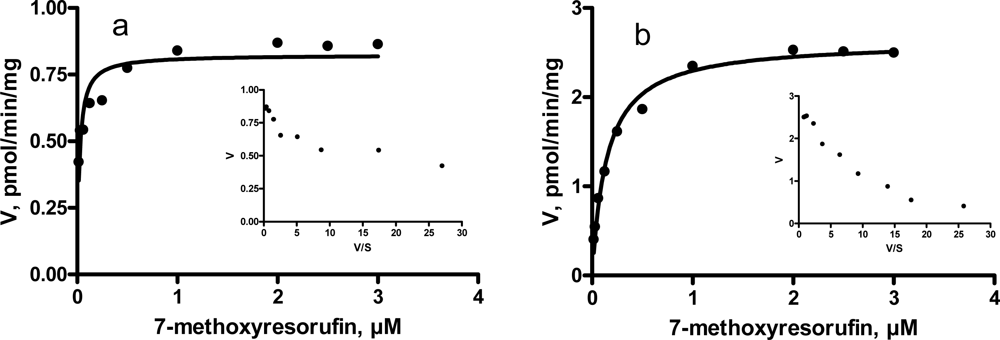
3.3. Kinetic characteristics of EROD and MROD activities
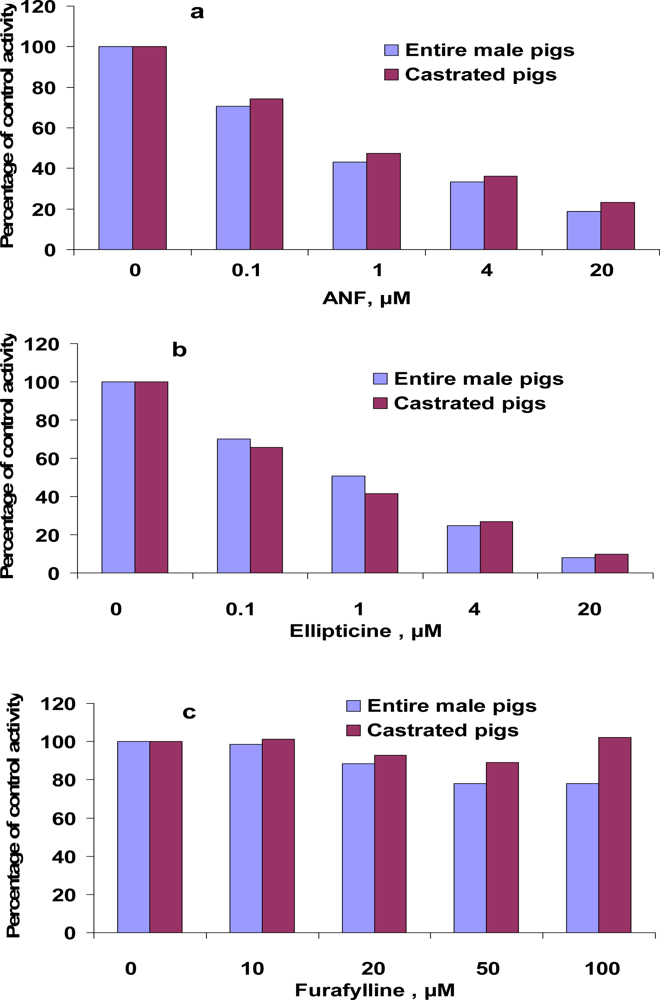
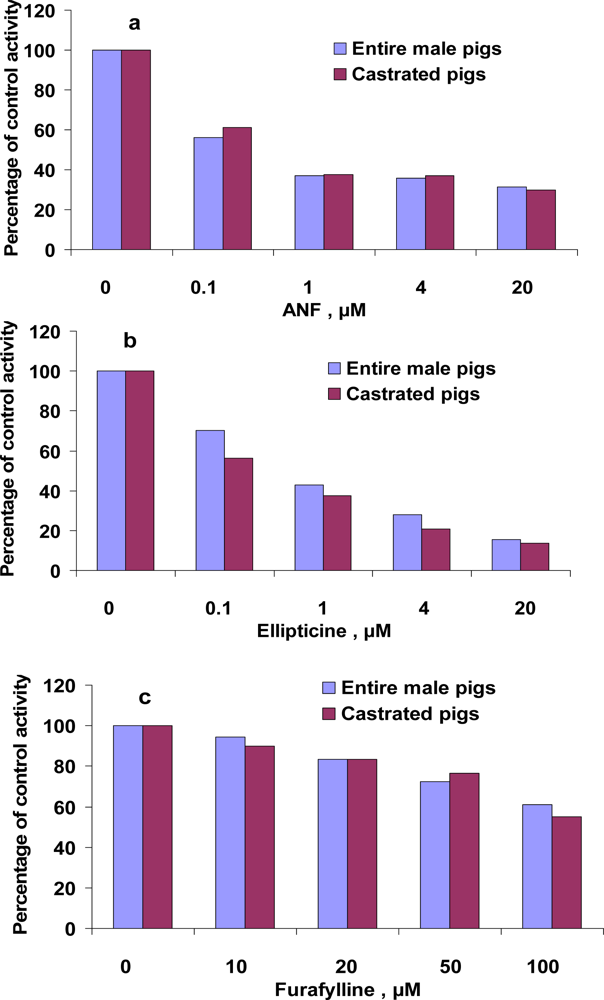
3.4. Inhibition of EROD and MROD activities by α-naphthoflavone (ANF), ellipticine, furafylline
4. Conclusions
Acknowledgments
References and Notes
- Havelkova, M.; Randak, T.; Zlabek, V.; Krijt, J.; Kroupova, H.; Pulkrabova, J.; Svobodova, Z. Biochemical Markers for Assessing Aquatic Contamination. Sensors 2007, 7, 2599–2611. [Google Scholar]
- Havelkova, M.; Blahova, J.; Kroupova, H.; Randak, T.; Slatinska, I.; Leontovycova, D.; Grabic, R.; Pospisil, R.; Svobodova, Z. Biomarkers of Contaminant Exposure in Chub (Leuciscus cephalus L.) – Biomonitoring of Major Rivers in the Czech Republic. Sensors 2008, 8, 2589–2603. [Google Scholar]
- Monshouwer, M.; Van’t Klooster, G.A.E.; Nijmeijer, S.M.; Witkamp, R.F.; Van Miert, A.S.J.P.A.M. Characterization of cytochrome P450 isoenzymes in primary cultures of pig hepatocytes. Toxicology In Vitro 1998, 12, 715–723. [Google Scholar]
- Kojima, M.; Morozumi, T. Cloning of six full-length cDNAs encoding pig cytochrome P450 enzymes and gene expression of these enzymes in the liver and kidney. Journal of Health Science 2004, 50, 518–529. [Google Scholar]
- Kojima, M.; Sekimoto, M.; Degawa, M. A novel gender-related difference in the constitutive expression of hepatic cytochrome P4501A subfamily enzymes in Meishan pigs. Biochemical Pharmacology 2008, 75, 1076–1082. [Google Scholar]
- Messina, A.; Chirulli, V.; Gervasi, P.G.; Longo, V. Purification, molecular cloning, heterologous expression and characterization of pig CYP1A2. Xenobiotica 2008, 38, 1453–1470. [Google Scholar]
- Burke, M.D.; Thompson, S.; Weaver, R.J.; Wolf, C.R.; Mayer, R.T. Cytochrome P450 specificities of alkoxyresorufin O-dealkylation in human and rat liver microsomes. Biochemical Pharmacology 1994, 48, 923–936. [Google Scholar]
- Machala, M.; Soucek, P.; Neca, J.; Ulrich, R.; Lamka, J.; Szotáková, B.; Skálová, L. Inter-species comparisons of hepatic cytochrome P450 enzyme levels in male ruminants. Archives of Toxicology 2003, 77, 555–560. [Google Scholar]
- Burke, M.; Mayer, R. Ethoxyresorufin: direct fluometric assay of a microsomal O-dealkylation which is preferentially inducible by 3-methylchloranthrene. Drug Metabolism and Dispostion 1974, 2, 583–588. [Google Scholar]
- Hanioka, N.; Tatarazako, N.; Jinno, H.; Arizono, K.; Ando, M. Determination of cytochrome P4501A activities in mammalian liver microsomes by high-performance liquid chromatography with fluorescence detection. Journal of Chromatography B 2000, 744, 399–406. [Google Scholar]
- Wanwimolruk, S.; Wanwimolruk, P. Characterization of CYP1A enzyme in Adélie penguin liver. Comparative Biochemistry and Physiology Part C: Toxicology and Pharmacology 2006, 144, 148–154. [Google Scholar]
- Pegolo, S.; Merlanti, R.; Giantin, M.; Dacasto, M.; Montesissa, C.; Capolongo, F. High performance liquid chromatography determination of cytochrome P450 1A and 2C activities in bovine liver microsomes. The Veterinary Journal 2009, (in press; doi: 10.1016/j.tvjl.2008.08.006).. [Google Scholar]
- Tassaneeyakul, W.; Birkett, D.J.; Veronese, M.E.; McManus, M.E.; Tukey, R.H.; Quattrochi, L.C.; Gelboin, H.V.; Miners, J.O. Specificity of substrate and inhibitor probes for human cytochromes P450 1A1 and 1A2. Journal of Pharmacology And Experimental Therapeutics 1993, 265, 401–407. [Google Scholar]
- Zamaratskaia, G.; Zlabek, V.; Chen, G.; Madej, A. Modulation of porcine cytochrome P450 enzyme activities by surgical castration and immunocastration. Animal 2009, (in press).. [Google Scholar]
- Zamaratskaia, G.; Andersson, H.K.; Chen, G.; Andersson, K.; Madej, A.; Lundström, K. Effect of a gonadotropin-releasing hormone vaccine ImprovacTM on steroid hormones, boar taint and performance in entire male pigs. Reproduction in Domestic Animals 2008, 43, 351–359. [Google Scholar]
- Nicolau-Solano, S.I.; McGivan, J.D.; Whittington, F.M.; Nieuwhof, G.J.; Wood, J.D.; Doran, O. Relationship between the expression of hepatic but not testicular 3beta-hydroxysteroid dehydrogenase with androstenone deposition in pig adipose tissue. Journal of Animal Science 2006, 84, 2809–2817. [Google Scholar]
- Chang, T.K.H.; Gonzalez, F.J.; Waxman, D.J. Evaluation of Triacetyloleandomycin, α-naphthoflavone and diethyldithiocarbamate as selective chemical probes for inhibition of human cytochrome P450. Archives of Biochemistry and Biophysics 1994, 311, 437–442. [Google Scholar]
- Takahashi, N.; Miranda, C.L.; Henderson, M.C.; Buhler, D.R.; Williams, D.E.; Bailey, G.S. Inhibition of in-vitro aflatoxin b-1-DNA binding in rainbow trout by CYP1A inhibitors—alpha-naphthoflavone, beta-naphthoflavone, and trout CYP1A peptide antibody. Comparative Biochemistry and Physiology C 1995, 110, 273–280. [Google Scholar]
- Sesardic, D.; Boobis, A.R.; Murray, B.P.; Murray, S.; Segura, J.; De La Torre, R.; Davies, D.S. Furafylline is a potent and selective inhibitor of cytochrome P4501A2 in man. British Journal of Pharmacology 1990, 29, 651–663. [Google Scholar]
- Ueng, Y.F.; Jan, W.C.; Lin, L.C.; Chen, T.L.; Guengerich, F.P.; Chen, C.F. The alkaloid rutaecarpine is a selective inhibitor of cytochrome P450 1A in mouse and human liver microsomes. Drug Metabolism and Disposition 2002, 30, 349–353. [Google Scholar]
- Schulz, T.G.; Neubert, D.; Davies, D.S.; Edwards, R. J. Inducibility of cytochromes P-450 by dioxin in liver and extrahepatic tissues of the marmoset monkey (Callithrix jacchus). Biochimica et Biophysica Acta 1996, 1298, 131–140. [Google Scholar]
| Added concentration of resorufin, pmol/mL | Concentration of resorufin measured in blank incubations, pmol/mL (recovered, %) | Concentration of resorufin measured in microsomal incubations, pmol/mL (recovered, %) |
|---|---|---|
| 0.5 | 0.45 (90.4) | 0.39 (77.8) |
| 10 | 10.3 (103.5) | 10.7 (107.1) |
| 50 | 49.2 (98.3) | 47.7 (95.3) |
| Sample | EROD | MROD | ||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 1 | 2 | 3 | |
| Mean, pmol/min/mg | 14.4 | 42.4 | 55.6 | 5.1 | 7.8 | 14.5 |
| SD | 1.3 | 1.5 | 8.5 | 0.57 | 0.88 | 0.69 |
| RSD,% | 9.3 | 3.6 | 15.4 | 11.1 | 6.1 | 8.8 |
| Kinetic parameter | Incubations
| |||
|---|---|---|---|---|
| Entire male pigs | Castrated male pigs | |||
| EROD | High-affinity component | Km, μM (CI) | 0.01 (0.005 – 0.022) | 0.01 (0.001 – 0.020) |
| Vmax, pmol/min/mg (CI) | 5.9 (3.1 – 8.6) | 7.3 (5.5 – 9.0) | ||
| Low-affinity component | Km, μM (CI) | 0.54 (0.10 – 0.99) | 1.25 (0.37 – 2.14) | |
| Vmax, pmol/min/mg (CI) | 11.7 (9.6 – 13.8) | 15.2 (12.9 – 17.6) | ||
| MROD | Km, μM (CI) | 0.02 (0.75 – 0.89) | 0.14 (0.11 – 0.18) | |
| Vmax, pmol/min/mg (CI) | 0.8 (0.7 – 0.9) | 2.6 (2.5 – 2.8) | ||
| Inhibitor | Ki, μM
| |||
|---|---|---|---|---|
| EROD | MROD | |||
| Entire male pigs | Castrated pigs | Entire male pigs | Castrated pigs | |
| ANF | 0.031 | 0.096 | 0.014 | 0.018 |
| Ellipticine | 0.017 | 0.021 | 0.024 | 0.019 |
| Furafylline | no* | no* | 102.7 | 37.6 |
© 2009 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zamaratskaia, G.; Zlabek, V. EROD and MROD as Markers of Cytochrome P450 1A Activities in Hepatic Microsomes from Entire and Castrated Male Pigs. Sensors 2009, 9, 2134-2147. https://doi.org/10.3390/s90302134
Zamaratskaia G, Zlabek V. EROD and MROD as Markers of Cytochrome P450 1A Activities in Hepatic Microsomes from Entire and Castrated Male Pigs. Sensors. 2009; 9(3):2134-2147. https://doi.org/10.3390/s90302134
Chicago/Turabian StyleZamaratskaia, Galia, and Vladimir Zlabek. 2009. "EROD and MROD as Markers of Cytochrome P450 1A Activities in Hepatic Microsomes from Entire and Castrated Male Pigs" Sensors 9, no. 3: 2134-2147. https://doi.org/10.3390/s90302134




