Allicin: Chemistry and Biological Properties
Abstract
:1. Introduction
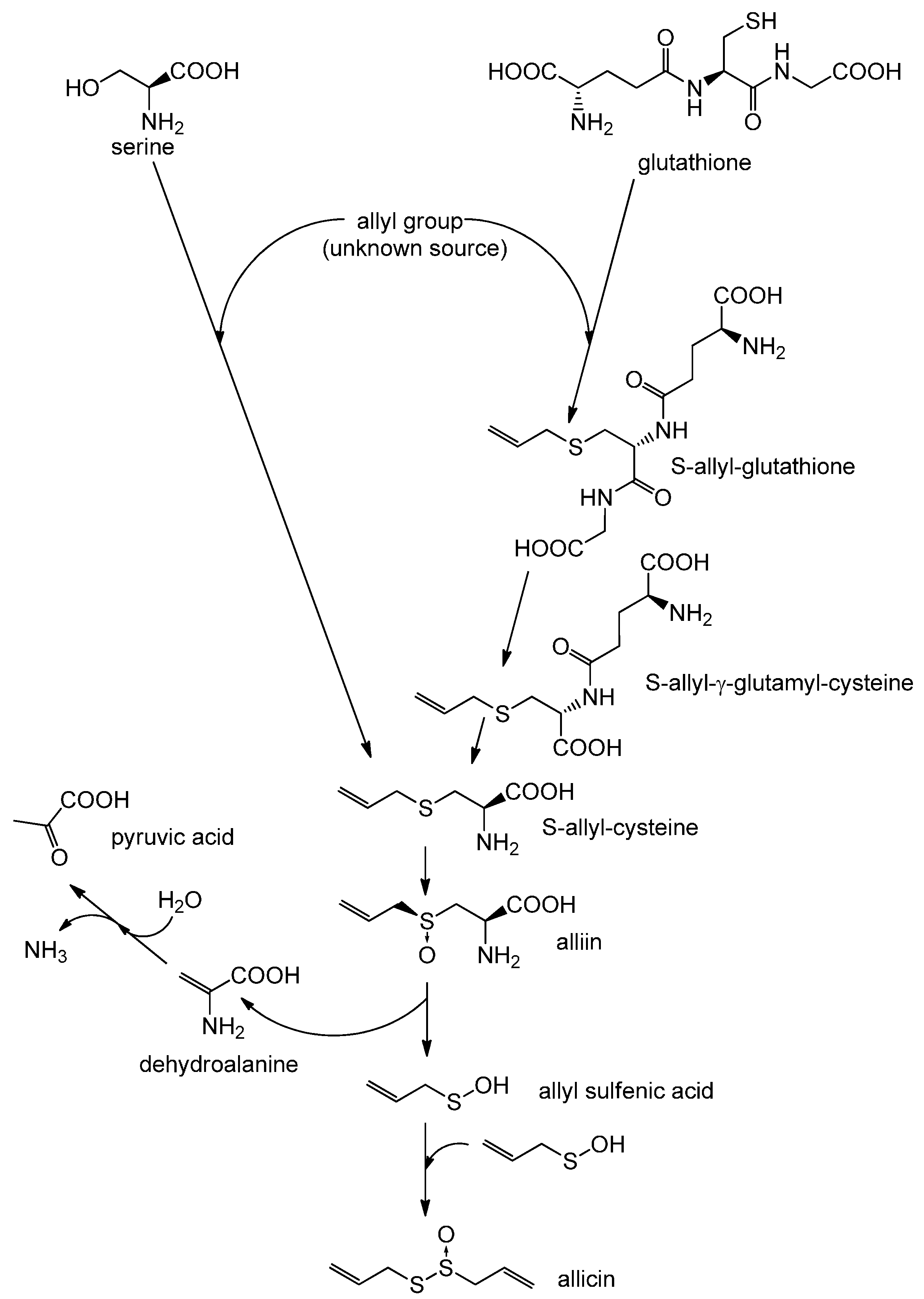
2. Biosynthesis of Allicin
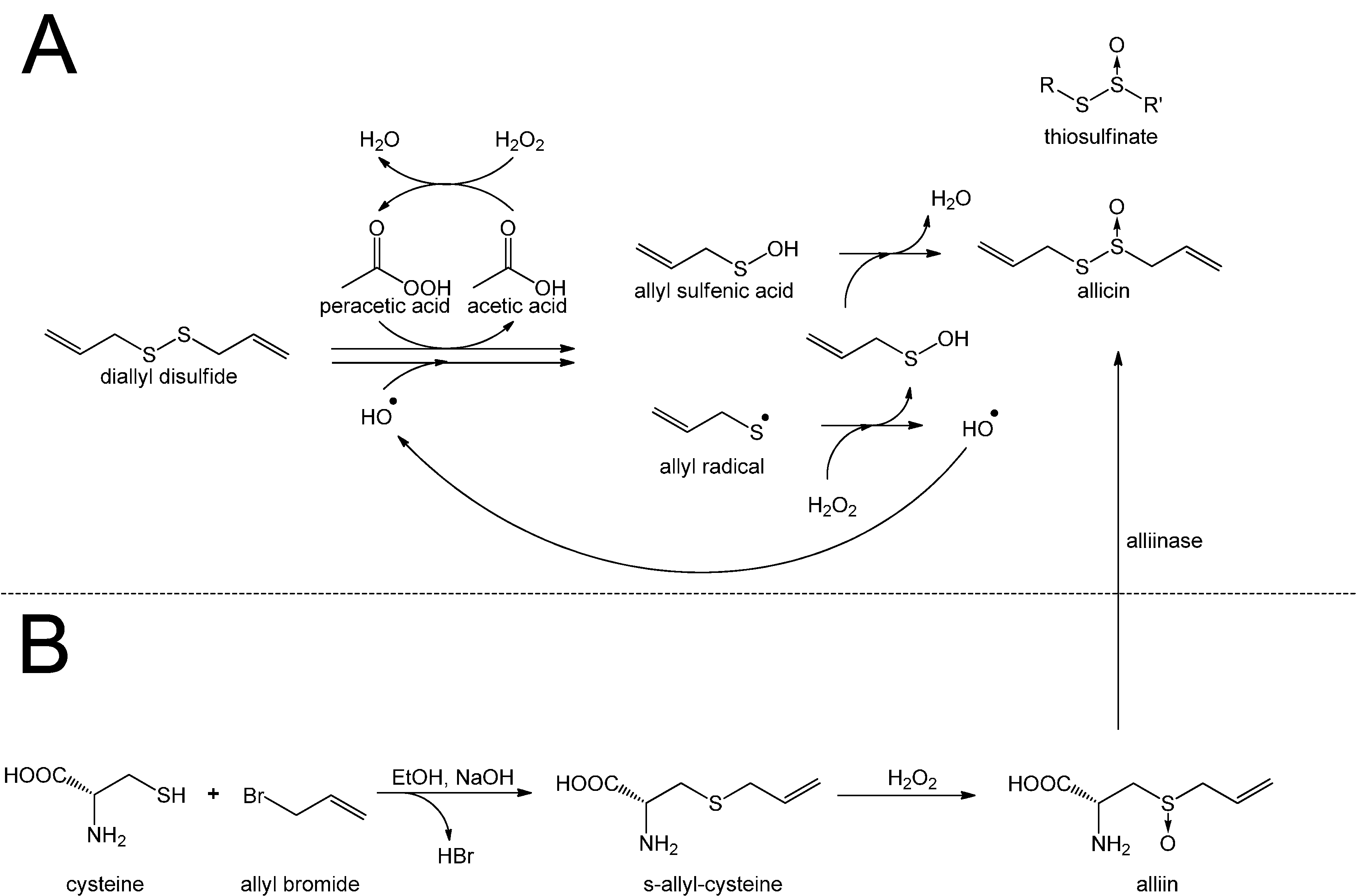
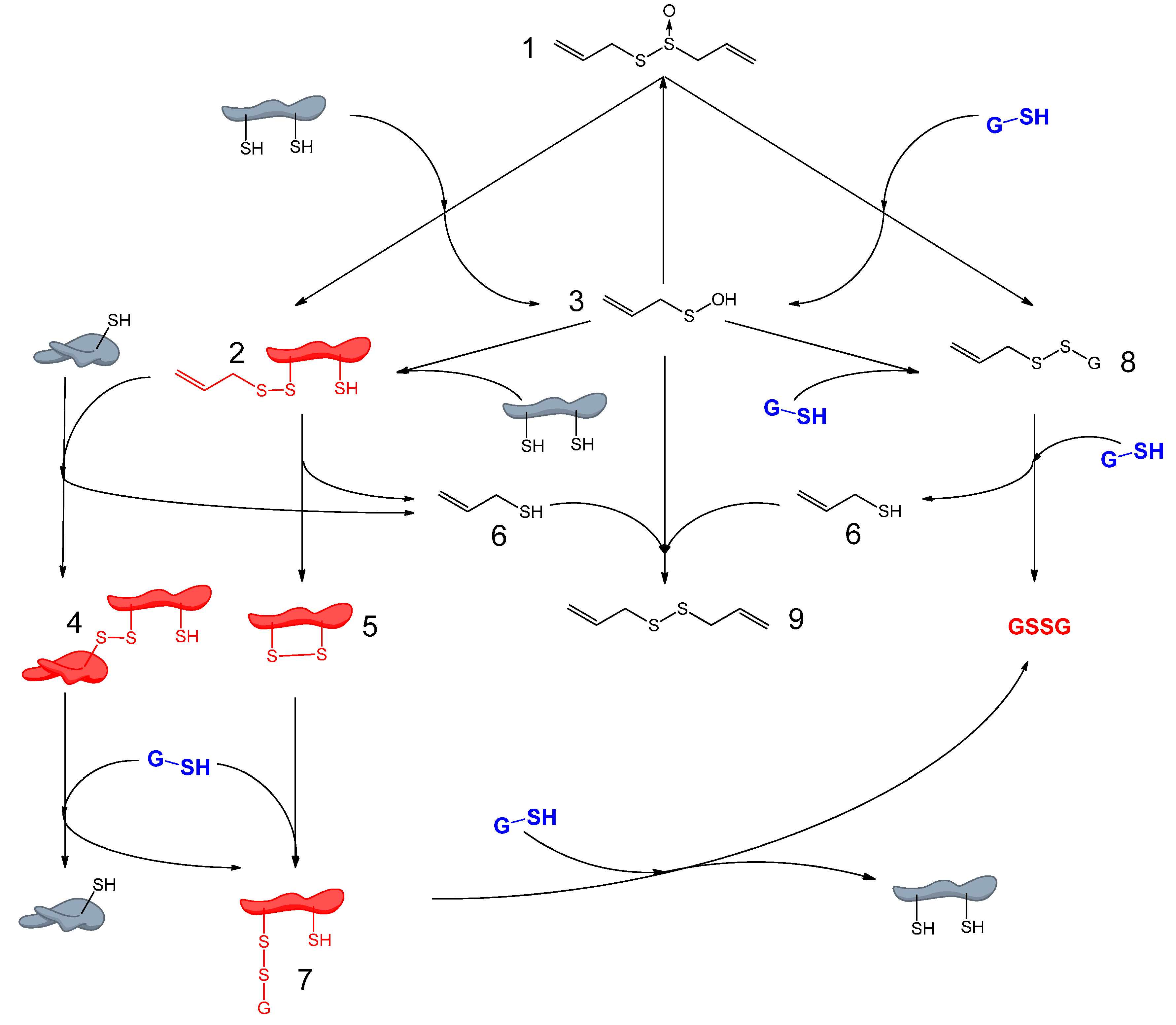
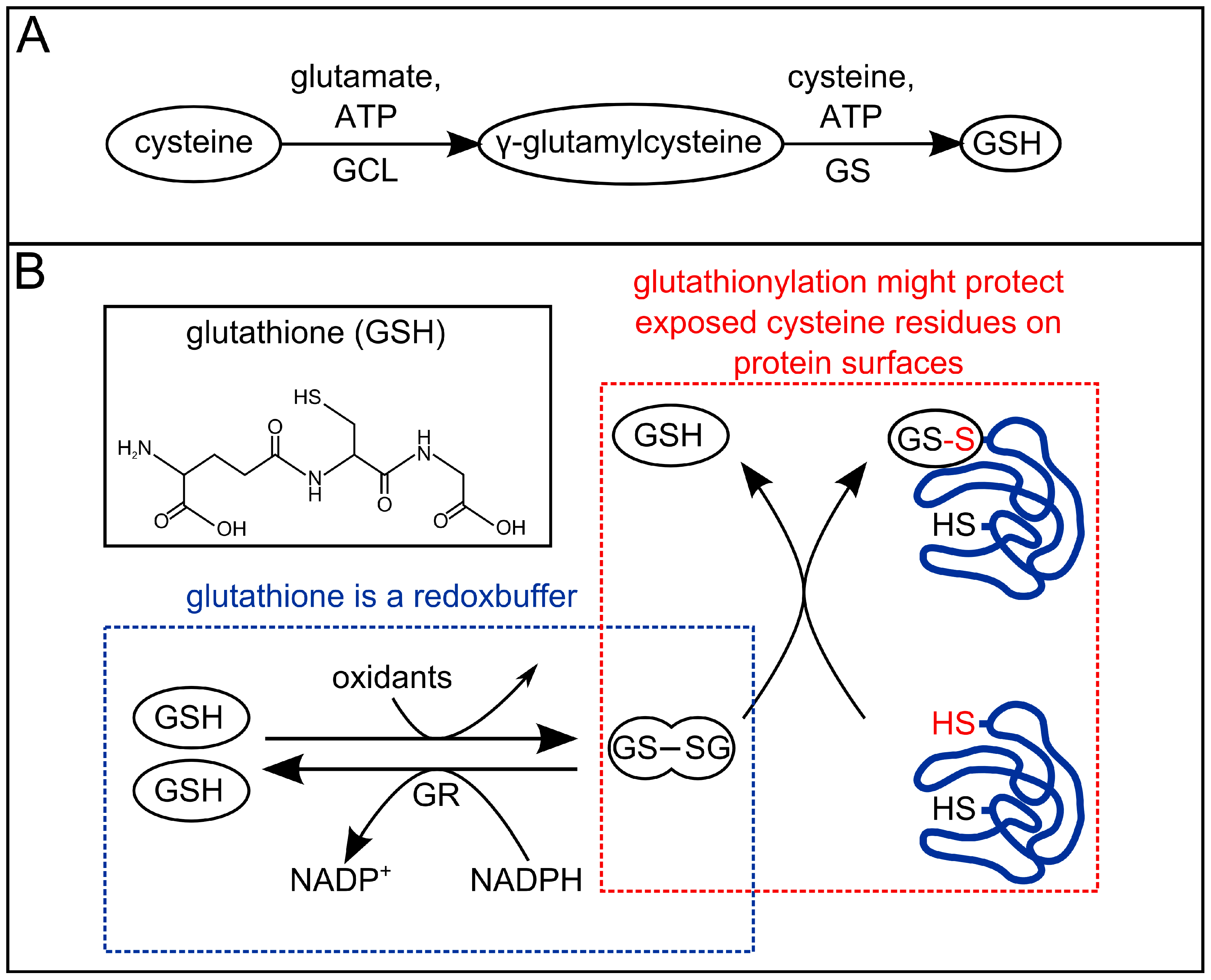
3. Redox-Chemistry Pertaining to Allicin
4. Antimicrobial Activity of Allicin
4.1. Allicin Is almost Exclusively Responsible for the Antimicrobial Activity of Freshly Crushed Garlic

4.2. A Closer Look at the Basis for Antimicrobial Activity of Allicin
| Bacteria | Source of Allicin | Amount of Allicin | Experimental System a,b,c,d | Reference | |
|---|---|---|---|---|---|
| Gram positive | |||||
| Bacillus spp. | pure allicin extracted from garlic | 80 µM | a | [9] | |
| synthetic | 30–150 µM | a | [38] | ||
| Streptococcus spp. | pure allicin extracted from garlic | 80 µM | a | [9] | |
| synthetic | ranging from 60 µM to 200 µM | a | [38] | ||
| methicillin sensitive Staphylococcus aureus NBRC 12732 | synthetic and garlic extract | 2.2 × 10−3–0.92 µmol | b, c | [40] | |
| methicillin resistant Staphylococcus aureus (clinical isolates) | garlic extract | 0.04–0.62 µmol | b, e | [41] | |
| Gram negative | |||||
| Salmonella typhimurium | pure allicin extracted from garlic | 80 µM | a | [9] | |
| enzymatically synthesized from alliin | ranging from 200 µM to 500 µM | a | [46] | ||
| Agrobacterium tumefaciens | garlic extract | 1.72 µmol | b, d | [39] | |
| Escherichia coli K12 | garlic extract | 0.52–1.72 µmol | b, c | [39] | |
| Pseudomonas syringae (various pathovars) | garlic extract | 1.72 µmol | b, d | [39] | |
| Vibrio cholerae | pure allicin extracted from garlic | 80 µM | a | [9] | |
4.3. Does Allicin have an Effect on DNA-, RNA- and Protein Synthesis?
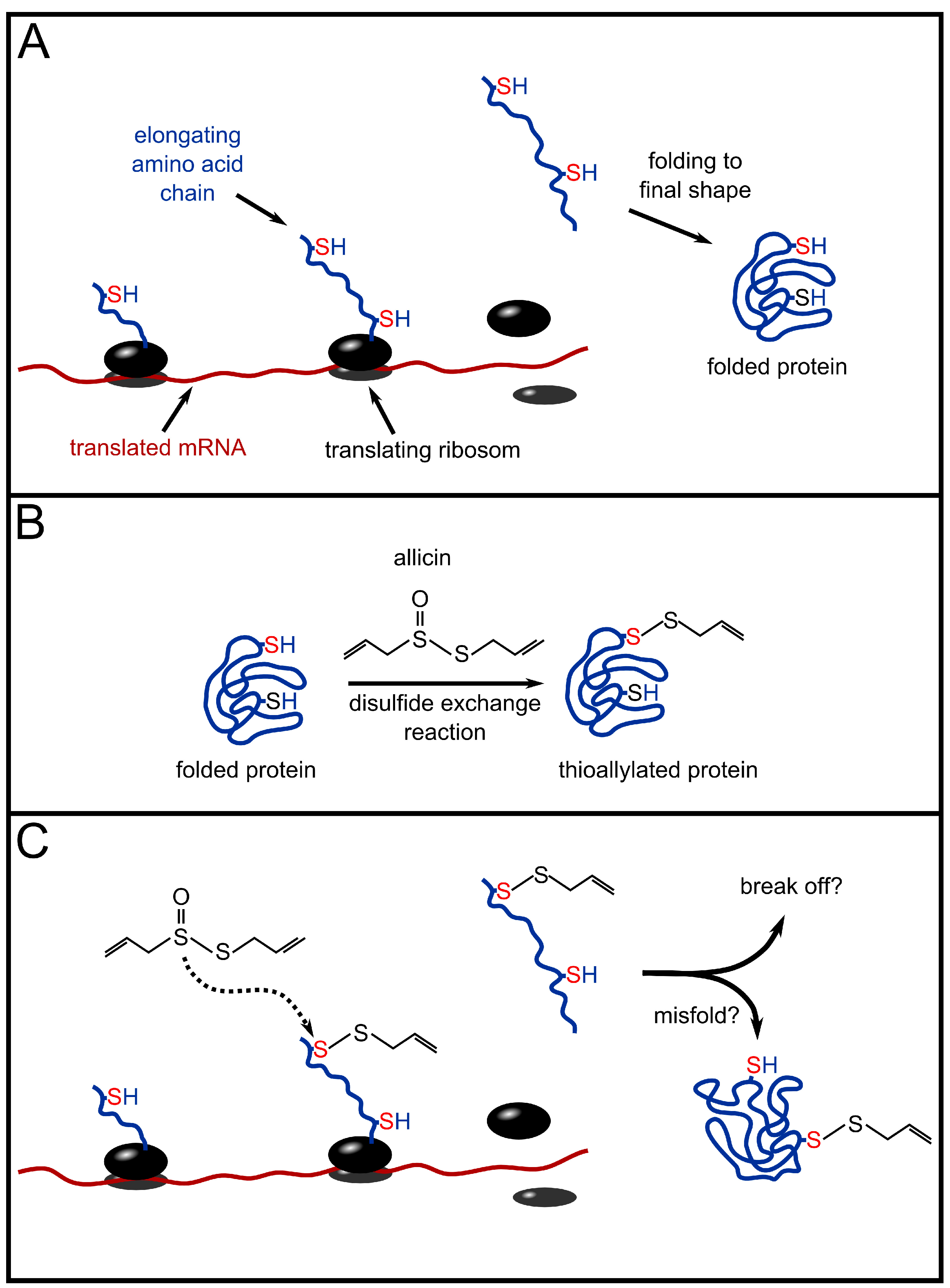
4.4. Effect of Allicin on Fungi
5. Allicin’s Effects on Animal and Human Cells
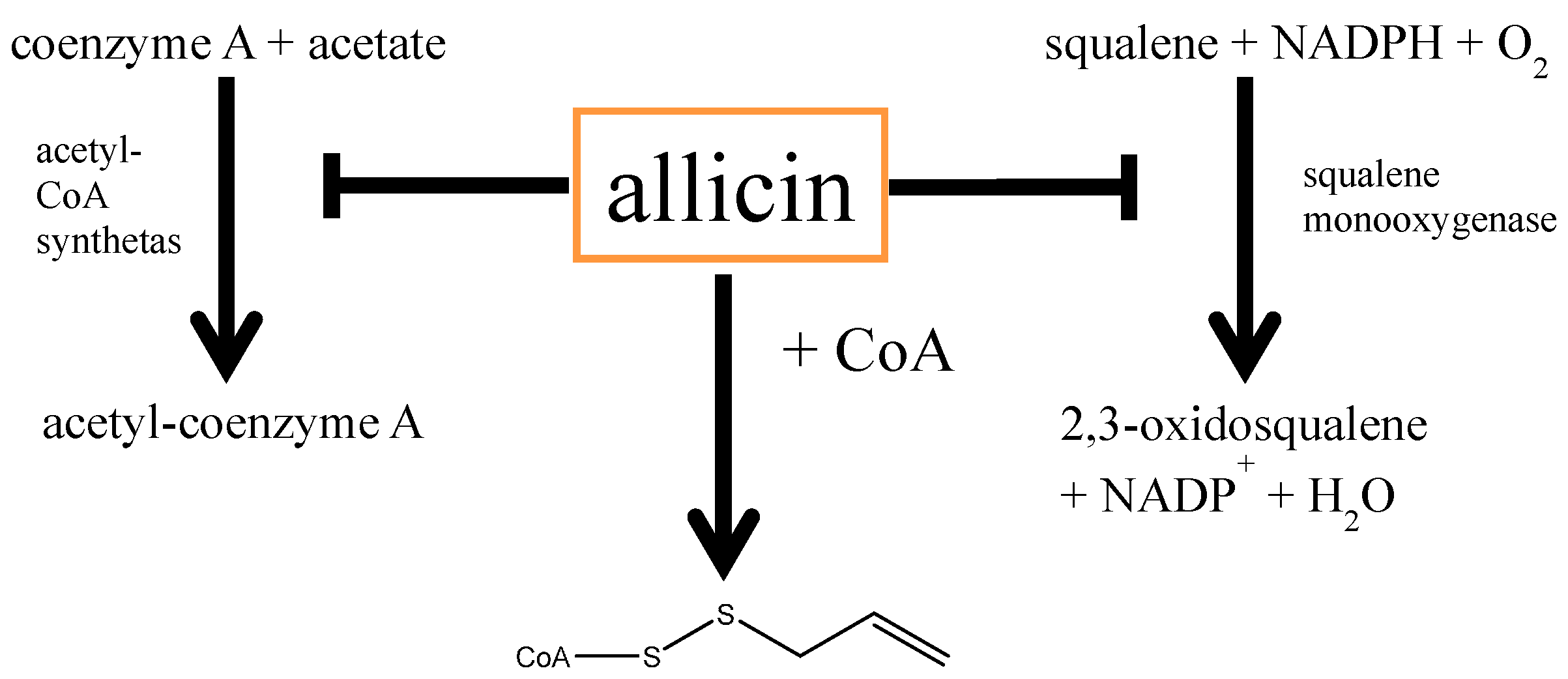
5.1. Allicin and Cardiovascular Diseases
5.2. Allicin’s Immune-Modulatory Activity

5.3. Allicin and Cancer
6. Effects of Allicin on Plants
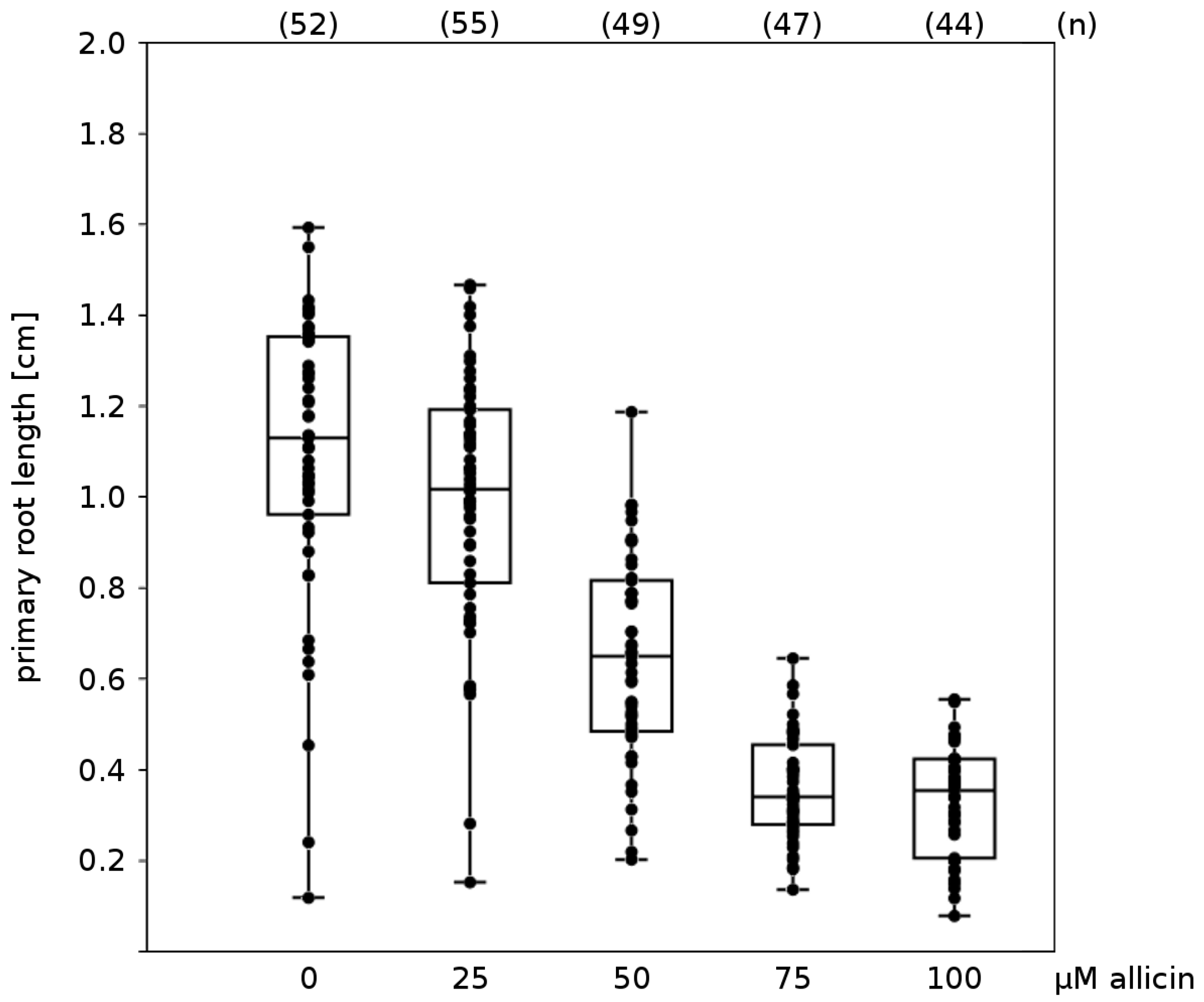
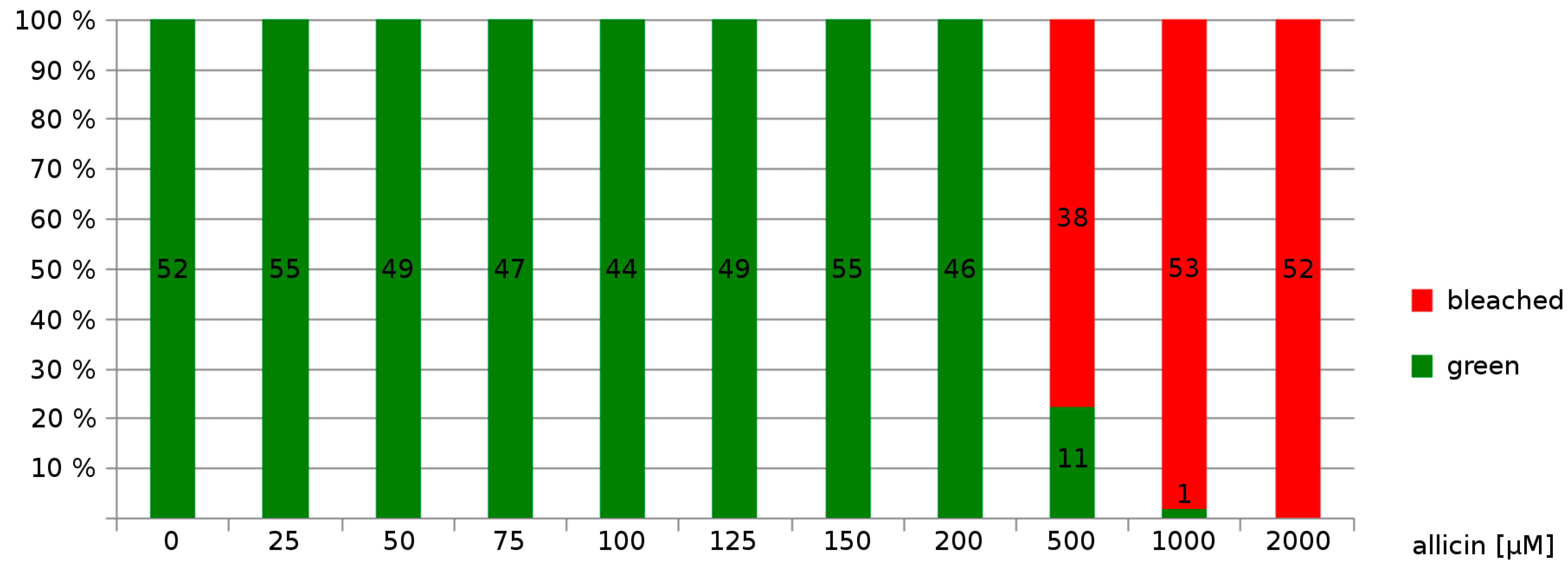
Effects on Germination, Root Growth and Viability on Arabidopsis thaliana

7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Slusarenko, A.J.; Patel, A.; Portz, D. Control of plant diseases by natural products: Allicin from garlic as a case study. Eur. J. Plant Pathol. 2008, 121, 313–322. [Google Scholar]
- Rishton, G.M. Natural products as a robust source of new drugs and drug leads: Past successes and present day issues. Am. J. Cardiol. 2008, 101, 43–49. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. The influence of natural products upon drug discovery (Antiquity to late 1999). Nat. Prod. Rep. 2000, 17, 215–234. [Google Scholar] [CrossRef]
- Block, E. Garlic and Other Alliums—The Lore and The Science; RSC publishing: Cambridge, UK, 2010. [Google Scholar]
- Koch, H.P.; Lawson, L.D. Garlic: The Science and Therapeutic Application of Allium sativum L. and Related Species; Williams & Wilkins: Baltimore, MD, USA, 1996. [Google Scholar]
- Castiglioni, A. A History of Medicine; Jason Aronson Inc.: New York, NY, USA, 1978. [Google Scholar]
- Brewster, J.L. Onions and Other Vegetable Alliums; CABI Publishing: Wallingford, UK, 2008. [Google Scholar]
- Rabinowitch, H.D.; Currah, L. Allium Crop Science: Recent Advances; CABI Publishing: Wallingford, UK, 2002. [Google Scholar]
- Cavallito, C.; Bailey, J. Allicin, the antibacterial principle of Allium sativum. I. Isolation, physical properties and antibacterial action. J. Am. Chem. Soc. 1944, 66, 1950–1951. [Google Scholar] [CrossRef]
- Josling, P. Allicin—The Heart of Garlic; NWI Publishing: Callahan Florida, FL, USA, 2007. [Google Scholar]
- Stoll, A.; Seebeck, E. Über Alliin, die genuine Muttersubstanz des Knoblauchöls. Helv. Chim. Acta 1948, 31, 189–210. [Google Scholar] [CrossRef]
- Cavallito, C.; Bailey, J.; Buck, J. The antibacterial principle of Allium sativum. III. Its precursor and “essential oil of garlic”. J. Am. Chem. Soc. 1945, 67, 1032–1033. [Google Scholar] [CrossRef]
- Granroth, B. Biosynthesis and decomposition of cysteine derivatives in onion and other Allium species. Ann. Acad. Sci. Fenn. Chem. 1970, 154, 4–71. [Google Scholar]
- Stoll, A.; Seebeck, E. Über die Spezifität der Alliinase und die Synthese mehrerer dem Alliin verwandter Verbindungen. Helv. Chim. Acta 1949, 32, 866–876. [Google Scholar] [CrossRef]
- Ilić, D.; Nikolić, V.; Nikolić, L.; Stanković, M.; Stanojević, L.; Cakić, M. Allicin and related compounds: Biosynthesis, synthesis and pharmacological activity. Facta Univ. Phys. Chem. Technol. 2011, 9, 9–20. [Google Scholar] [CrossRef]
- Virtanen, A.I.; Matikkala, E.J. The Isolation of S-methylcysteinesulphoxide and S-n-propenylcysteinesulphoxide from onion (Allium cepa) and the antibiotic activity of crushed onion. Acta Chem. Scand. 1959, 13, 1898–1900. [Google Scholar] [CrossRef]
- Stoll, A.; Seebeck, E. Über Alliin, die genuine Muttersubstanz des Knoblauchöls. Experientia 1947, 3, 114–115. [Google Scholar] [CrossRef]
- Cruz-Villalon, G. Synthesis of allicin and purification by solid-phase extraction. Anal. Biochem. 2001, 290, 376–378. [Google Scholar] [CrossRef]
- Vaidya, V.; Ingold, K.U.; Pratt, D. A garlic: Source of the ultimate antioxidants-sulfenic acids. Angew. Chem. Int. Ed. Engl. 2009, 48, 157–160. [Google Scholar] [CrossRef]
- Miron, T.; Bercovici, T.; Rabinkov, A.; Wilchek, M.; Mirelman, D. [3H]Allicin: Preparation and applications. Anal. Biochem. 2004, 331, 364–369. [Google Scholar] [CrossRef]
- Fry, F.H.; Okarter, N.; Baynton-Smith, C.; Kershaw, M.J.; Talbot, N.J.; Jacob, C. Use of a substrate/alliinase combination to generate antifungal activity in situ. J. Agric. Food Chem. 2005, 53, 574–580. [Google Scholar] [CrossRef]
- Freeman, F.; Kodera, Y. Garlic chemistry: Stability of S-(2-propenyl)-2-propene-1-sulfinothioate (allicin) in blood, solvents, and simulated physiological fluids. J. Agric. Food Chem. 1995, 43, 2332–2338. [Google Scholar] [CrossRef]
- Gruhlke, M.C.H.; Slusarenko, A.J. The biology of reactive sulfur species (RSS). Plant Physiol. Biochem. 2012, 59, 98–107. [Google Scholar]
- Gruhlke, M.; Slusarenko, A.J. The Cellular “Thiolstat” as an Emerging Potential Target of Some Secondary Metabolites. In Recent Advances in Redox Active Plant and Microbial Products; Jacob, C., Ed.; Springer: Dordrecht, The Netherlands, 2014. [Google Scholar]
- Nagy, P. Kinetics and mechanisms of thiol-disulfide exchange covering direct substitution and thiol oxidation-mediated pathways. Antioxid. Redox Signal. 2013, 18, 1023–1041. [Google Scholar]
- Morgan, B.; Ezeriņa, D.; Amoako, T. Multiple glutathione disulfide removal pathways mediate cytosolic redox homeostasis. Nat. Chem. Biol. 2012, 9, 119–125. [Google Scholar]
- De Rey-Pailhade, J. Sur un corps d’origine organique hydrogenant le soufre a froid. CR Acad. Sci. 1888, 106, 1683–1684. [Google Scholar]
- Hopkins, F. On an autoxidisable constituent of the cell. Biochem. J. 1921, 15, 286–305. [Google Scholar]
- Harington, C.; Mead, T. Synthesis of glutathione. Biochem. J. 1935, 29, 1602–1611. [Google Scholar]
- Conn, E.; Vennesland, B. Glutathione reductase of wheat germ. J. Biol. Chem. 1951, 192, 17–28. [Google Scholar]
- Conn, E.; Vennesland, B. Enzymatic reduction of glutathione by triphosphopyridine nucleotide (co-enzyme II). Nature 1951, 167, 976–977. [Google Scholar] [CrossRef]
- Grant, C.M.; Collinson, L.P.; Roe, J.H.; Dawes, I.W. Yeast glutathione reductase is required for protection against oxidative stress and is a target gene for YAP-1 transcriptional regulation. Mol. Microbiol. 1996, 21, 171–179. [Google Scholar]
- Muller, E.G. A glutathione reductase mutant of yeast accumulates high levels of oxidized glutathione and requires thioredoxin for growth. Mol. Biol. Cell 1996, 7, 1805–1813. [Google Scholar] [CrossRef]
- Tada, Y.; Spoel, S.H.; Pajerowska-Mukhtar, K.; Mou, Z.; Song, J.; Wang, C.; Zuo, J.; Dong, X. Plant immunity requires conformational changes [corrected] of NPR1 via S-nitrosylation and thioredoxins. Science 2008, 321, 952–956. [Google Scholar] [CrossRef]
- Kuge, S.; Arita, M.; Murayama, A. Regulation of the yeast Yap1p nuclear export signal is mediated by redox signal-induced reversible disulfide bond formation. Mol. Cell. Biol. 2001, 21, 6139–6150. [Google Scholar] [CrossRef]
- Buelna-Chontal, M.; Zazueta, C. Redox activation of Nrf2 & NF-κB: A double end sword? Cell Signal. 2013, 25, 2548–2557. [Google Scholar] [CrossRef]
- Pasteur, L. Memoire sur la fermentation appelee lactique. Mem. Soc. Imp. Sci. Agric. Lille 1858, 5, 13–26. [Google Scholar]
- Small, L.D.; Bailey, J.H.; Cavallito, C.J. Alkyl Thiosulfinates. J. Am. Chem. Soc. 1947, 69, 1710–1713. [Google Scholar] [CrossRef]
- Curtis, H.; Noll, U.; Störmann, J.; Slusarenko, A.J. Broad-spectrum activity of the volatile phytoanticipin allicin in extracts of garlic (Allium sativum L.) against plant pathogenic bacteria, fungi and Oomycetes. Physiol. Mol. Plant Pathol. 2004, 65, 79–89. [Google Scholar] [CrossRef]
- Fujisawa, H.; Watanabe, K.; Suma, K.; Origuchi, K.; Matsufuji, H.; Seki, T.; Ariga, T. Antibacterial potential of garlic-derived allicin and its cancellation by sulfhydryl compounds. Biosci. Biotechnol. Biochem. 2009, 73, 1948–1955. [Google Scholar] [CrossRef]
- Cutler, R.R.; Wilson, P. Antibacterial activity of a new, stable, aqueous extract of allicin against methicillin-resistant Staphylococcus aureus. Br. J. Biomed. Sci. 2004, 61, 71–74. [Google Scholar]
- Cañizares, P.; Gracia, I.; Gómez, L.A.; García, A.; de Argila, C.M.; Boixeda, D.; de Rafael, L. Thermal degradation of allicin in garlic extracts and its implication on the inhibition of the in vitro growth of helicobacter pylori. Biotechnol. Prog. 2004, 20, 32–37. [Google Scholar]
- Hughes, B.; Lawson, L. Antimicrobial effects of Allium sativum L. (garlic), Allium ampeloprasum L. (elephant garlic), and Allium cepa L. (onion), garlic compounds and commercial garlic supplement products. Phyther. Res. 1991, 5, 154–158. [Google Scholar] [CrossRef]
- De Lancey Pulcini, E. Bacterial biofilms: A review of current research. Nephrologie 2001, 22, 439–441. [Google Scholar]
- Miron, T.; Rabinkov, A.; Mirelman, D.; Wilchek, M.; Weiner, L. The mode of action of allicin: Its ready permeability through phospholipid membranes may contribute to its biological activity. Biochim. Biophys. Acta Biomembr. 2000, 1463, 20–30. [Google Scholar] [CrossRef]
- Feldberg, R.; Chang, S. In vitro mechanism of inhibition of bacterial cell growth by allicin. Antimicrob. Agents Chemother. 1988, 32, 1763–1768. [Google Scholar] [CrossRef]
- Cavallito, C.J.; Bailey, J.H. Preliminary note on the inactivation of antibiotics. Science 1944, 100, 390–390. [Google Scholar]
- Cavallito, C.J. Relationship of thiol structures to reaction with antibiotics. J. Biol. Chem. 1946, 164, 29–34. [Google Scholar]
- Roos, G.; Foloppe, N.; Messens, J. Understanding the pKa of redox cysteines: The key role of hydrogen bonding. Antioxid. Redox Signal. 2013, 18, 94–127. [Google Scholar] [CrossRef]
- Focke, M.; Feld, A.; Lichtenthaler, H.K. Allicin, a naturally occurring antibiotic from garlic, specifically inhibits acetyl-CoA synthetase. FEBS Lett. 1990, 261, 106–108. [Google Scholar] [CrossRef]
- Rabinkov, A.; Miron, T.; Konstantinovski, L.; Wilchek, M.; Mirelman, D.; Weiner, L. The mode of action of allicin: Trapping of radicals and interaction with thiol containing proteins. Biochim. Biophys. Acta 1998, 1379, 233–244. [Google Scholar] [CrossRef]
- Wills, E.D. Enzyme inhibition by allicin, the active principle of garlic. Biochem. J. 1956, 63, 514–520. [Google Scholar]
- Van Etten, H.D.; Mansfield, J.W.; Bailey, J.A.; Farmer, E.E. Two classes of plant antibiotics: Phytoalexins versus “phytoanticipins”. Plant Cell 1994, 6, 1191–1192. [Google Scholar] [CrossRef]
- Auger, J.; Arnault, I.; Diwo-Allain, S.; Ravier, N.; Molia, F.; Pettiti, M. Insecticidal and fungicidal potential of Allium substances as biofumigants. Agroindustria 2004, 3, 5–8. [Google Scholar]
- Khodavandi, A.; Alizadeh, F.; Harmal, N.S.; Sidik, S.M.; Othman, F.; Sekawi, Z.; Jahromi, M.A.F.; Ng, K.P.; Chong, P.P. Comparison between efficacy of allicin and fluconazole against Candida albicans in vitro and in a systemic candidiasis mouse model. FEMS Microbiol. Lett. 2011, 315, 87–93. [Google Scholar] [CrossRef]
- Perelló, A.; Gruhlke, M.; Slusarenko, A.J. Effect of garlic extract on seed germination, seedling health, and vigour of pathogen-infested wheat. J. Plant Prot. Res. 2013, 53, 317–323. [Google Scholar]
- Yamada, Y.; Azuma, K. Evaluation of the in vitro antifungal activity of allicin. Antimicrob. Agents Chemother. 1977, 11, 743–749. [Google Scholar] [CrossRef]
- Shadkchan, Y.; Shemesh, E.; Mirelman, D.; Miron, T.; Rabinkov, A.; Wilchek, M.; Osherov, N. Efficacy of allicin, the reactive molecule of garlic, in inhibiting Aspergillus spp. in vitro, and in a murine model of disseminated aspergillosis. J. Antimicrob. Chemother. 2004, 53, 832–836. [Google Scholar] [CrossRef]
- Ogita, A.; Hirooka, K.; Yamamoto, Y.; Tsutsui, N.; Fujita, K.-I.; Taniguchi, M.; Tanaka, T. Synergistic fungicidal activity of Cu2+ and allicin, an allyl sulfur compound from garlic, and its relation to the role of alkyl hydroperoxide reductase 1 as a cell surface defense in Saccharomyces cerevisiae. Toxicology 2005, 215, 205–213. [Google Scholar] [CrossRef]
- Ogita, A.; Fujita, K.-I.; Taniguchi, M.; Tanaka, T. Dependence of synergistic fungicidal activity of Cu2+ and allicin, an allyl sulfur compound from garlic, on selective accumulation of the ion in the plasma membrane fraction via allicin-mediated phospholipid peroxidation. Planta Med. 2006, 72, 875–880. [Google Scholar] [CrossRef]
- Ogita, A.; Fujita, K.; Taniguchi, M.; Tanaka, T. Enhancement of the fungicidal activity of amphotericin B by allicin, an allyl-sulfur compound from garlic, against the yeast Saccharomyces cerevisiae as a model system. Planta Med. 2006, 72, 1247–1250. [Google Scholar] [CrossRef]
- Ogita, A.; Fujita, K.; Tanaka, T. Enhancement of the fungicidal activity of amphotericin B by allicin: Effects on intracellular ergosterol trafficking. Planta Med. 2009, 75, 222–226. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Kim, K.S.; Han, I.; Kim, M.-H.; Jung, M.H.; Park, H.K. Quantitative and qualitative analysis of the antifungal activity of allicin alone and in combination with antifungal drugs. PLoS One 2012, 7, e38242. [Google Scholar]
- Ogita, A.; Nagao, Y.; Fujita, K.-I.; Tanaka, T. Amplification of vacuole-targeting fungicidal activity of antibacterial antibiotic polymyxin B by allicin, an allyl sulfur compound from garlic. J. Antibiot. (Tokyo) 2007, 60, 511–518. [Google Scholar] [CrossRef]
- Ogita, A.; Yutani, M.; Fujita, K.; Tanaka, T. Dependence of vacuole disruption and indipendence of potassium ion efflux in fungicidal activity induced by combination of amphotericin B and allicin against Saccharomyces cerevisiae. J. Antibiot. (Tokyo) 2010, 63, 689–692. [Google Scholar] [CrossRef]
- Carmona-Gutierrez, D.; Eisenberg, T.; Büttner, S.; Meisinger, C.; Kroemer, G.; Madeo, F. Apoptosis in yeast: Triggers, pathways, subroutines. Cell Death Differ. 2010, 17, 763–773. [Google Scholar] [CrossRef]
- Schafer, F.Q.; Buettner, G.R. Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple. Free Radic. Biol. Med. 2001, 30, 1191–1212. [Google Scholar] [CrossRef]
- Gruhlke, M.C.H.; Portz, D.; Stitz, M.; Anwar, A.; Schneider, T.; Jacob, C.; Schlaich, N.L.; Slusarenko, A.J. Allicin disrupts the cell’s electrochemical potential and induces apoptosis in yeast. Free Radic. Biol. Med. 2010, 49, 1916–1924. [Google Scholar] [CrossRef]
- Yu, L.; Guo, N.; Meng, R.; Liu, B.; Tang, X.; Jin, J.; Cui, Y.; Deng, X. Allicin-induced global gene expression profile of Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 2010, 88, 219–229. [Google Scholar] [CrossRef]
- Kleijnen, J.; Knipschild, P.; ter Riet, G. Garlic, onions and cardiovascular risk factors. A review of the evidence from human experiments with emphasis on commercially available preparations. Br. J. Clin. Pharmacol. 1989, 28, 535–544. [Google Scholar] [CrossRef]
- Ried, K.; Toben, C.; Fakler, P. Effect of garlic on serum lipids: An updated meta-analysis. Nutr. Rev. 2013, 71, 282–299. [Google Scholar] [CrossRef]
- Warshafsky, S.; Kamer, R.S.; Sivak, S.L. Effect of garlic on total serum cholesterol. A meta-analysis. Ann. Intern. Med. 1993, 119, 599–605. [Google Scholar]
- Kris-Etherton, P.M.; Hecker, K.D.; Bonanome, A.; Coval, S.M.; Binkoski, A.E.; Hilpert, K.F.; Griel, A.E.; Etherton, T.D. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002, 113, 71–88. [Google Scholar] [CrossRef]
- Ngo, S.N.T.; Williams, D.B.; Cobiac, L.; Head, R.J. Does garlic reduce risk of colorectal cancer? A systematic review. J. Nutr. 2007, 137, 2264–2269. [Google Scholar]
- Isaacsohn, J.L.; Moser, M.; Stein, E.A.; Dudley, K.; Davey, J.A.; Liskov, E.; Black, H.R. Garlic powder and plasma lipids and lipoproteins: A multicenter, randomized, placebo-controlled trial. Arch. Intern. Med. 1998, 158, 1189–1194. [Google Scholar] [CrossRef]
- Rahman, K.; Lowe, G. Garlic and cardiovascular disease: A critical review. J. Nutr. 2006, 136, 736–740. [Google Scholar]
- Kita, T.; Kume, N.; Minami, M.; Hayashida, K.; Murayama, T.; Sano, H.; Moriwaki, H.; Kataoka, H.; Nishi, E.; Horiuchi, H.; et al. Role of oxidized LDL in atherosclerosis. Ann. N. Y. Acad. Sci. 2001, 947, 199–206. [Google Scholar]
- Gruhlke, M.C.H.; Nwachukwu, I.; Arbach, M.; Anwar, A.; Noll, U.; Slusarenko, A.J. Allicin from garlic, effective in controlling several plant diseases, ia a reactive sulfur species (RSS) that pushes cells into apoptosis. In Modern Fungicides and Antifungal Compounds VI; DPG Publishers: Braunschweig, Gemany, 2011; pp. 325–334. [Google Scholar]
- Li, X.H.; Li, C.Y.; Xiang, Z.G.; Hu, J.J.; Lu, J.M.; Tian, R.B.; Jia, W. Allicin ameliorates cardiac hypertrophy and fibrosis trought enhancing of Nrf2 antioxidant signaling pathways. Cardiovasc. Drugs Ther. 2012, 26, 457–465. [Google Scholar] [CrossRef]
- Li, X.-H.; Li, C.-Y.; Lu, J.-M.; Tian, R.-B.; Wei, J. Allicin ameliorates cognitive deficites ageing-induced learning and memory deficites trought enhancing of Nrf2 antioxidant signaling pathways. Neurosci. Lett. 2012, 514, 46–50. [Google Scholar] [CrossRef]
- Brown, M.S.; Goldstein, J.L. How LDL receptors influence colesterol and arterosclerosis. Sci. Am. 1984, 251, 58–66. [Google Scholar] [CrossRef]
- Grobbee, D.E.; Bots, M.L. Statin treatment and progression of artherosclerotic plaque burden. Drugs 2003, 63, 893–911. [Google Scholar] [CrossRef]
- Eilat, S.; Oestraicher, Y.; Rabinkov, A.; Ohad, D.; Mirelman, D.; Battler, A.; Eldar, M.; Vered, Z. Alteration of lipid profile in hyperlipidemic rabbits by allicin, an active constituent of garlic. Coron. Arter. Dis. 1995, 6, 985–990. [Google Scholar]
- Gebhardt, R.; Beck, H.; Wagner, K.G. Inhibition of cholesterol biosynthesis by allicin and ajoene in rat hepatocytes and HepG2 cells. Biochim. Biophys. Acta 1994, 1213, 57–62. [Google Scholar] [CrossRef]
- Gupta, N.; Porter, T. Garlic and garlic-derived compounds inhibit human squalene monooxygenase. J. Nutr. 2001, 131, 1662–1667. [Google Scholar]
- Yip, J.; Shen, Y.; Berndt, M.C.; Andrews, R.K. Primary platelet adhesion receptors. IUBMB Life 2005, 57, 103–108. [Google Scholar] [CrossRef]
- Briggs, W.H.; Xiao, H.; Parkin, K.L.; Shen, C.; Goldman, I.L. Differential inhibition of human platelet aggregation by selected Allium thiosulfinates. J. Agric. Food Chem. 2000, 48, 5731–5735. [Google Scholar] [CrossRef]
- Benavides, G.A; Squadrito, G.L.; Mills, R.W.; Patel, H.D.; Isbell, T.S.; Patel, R.P.; Darley-Usmar, V.M.; Doeller, J.E.; Kraus, D.W. Hydrogen sulfide mediates the vasoactivity of garlic. Proc. Natl. Acad. Sci. USA 2007, 104, 17977–17982. [Google Scholar] [CrossRef]
- Zoccali, C.; Catalano, C.; Rastelli, S. Blood pressure control: Hydrogen sulfide, a new gasotransmitter, takes stage. Nephrol. Dial. Transplant. 2009, 24, 1394–1396. [Google Scholar] [CrossRef]
- Epstein, F.H. Cardiovascular disease epidemiology: A journey from the past into the future. Circulation 1996, 93, 1755–1764. [Google Scholar] [CrossRef]
- Hobauer, R.; Frass, M.; Gmeiner, B.; Kaye, A.D.; Frost, E.A. Garlic extract (Allium sativum) reduces migration of neutrophils trought endothelial cell monolayers. Middle East J. Anesthesiol. 2000, 15, 649–650. [Google Scholar]
- Gu, X.; Wu, H.; Fu, P. Allicin attenuates inflammation and suppresses HLA-B27 protein expression in ankylosing spondylitis mice. Biomed Res. Int. 2013, 171573:1–171573:6. [Google Scholar]
- Sela, U.R.I.; Ganor, S.; Hecht, I.; Brill, A.; Miron, T.; Rabinkov, A.; Sela, U. Allicin inhibits SDF-1 a -induced T cell interactions with fibronectin and endothelial cells by down-regulating cytoskeleton rearrangement , Pyk-2 phosphorylation and VLA-4 expression. Immunology 2004, 111, 391–399. [Google Scholar] [CrossRef]
- Prager-Khoutorsky, M.; Goncharov, I.; Rabinkov, A.; Mirelman, D.; Geiger, B.; Bershadsky, A.D. Allicin inhibits cell polarization, migration and division via its direct effect on microtubules. Cell Motil. Cytoskeleton 2007, 64, 321–337. [Google Scholar] [CrossRef]
- Tocque, B.; Delumeau, I.; Parker, F.; Maurier, F.; Multon, M.C.; Schweighoffer, F. Ras-GTPase activating protein (GAP): A putative effector for Ras. Cell Signal. 1997, 9, 153–158. [Google Scholar] [CrossRef]
- Patya, M. Allicin stimulates lymphocytes and elicits an antitumor effect: A possible role of p21ras. Int. Immunol. 2004, 16, 275–281. [Google Scholar] [CrossRef]
- Ellerin, T.; Rubin, R.H.; Weinblatt, M.E. Infections and anti-tumor necrosis factor alpha therapy. Arthritis Rheum. 2003, 48, 3013–3022. [Google Scholar] [CrossRef]
- Lang, A.; Lahav, M.; Sakhnini, E.; Barshack, I.; Fidder, H.H.; Avidan, B.; Bardan, E.; Hershkoviz, R.; Bar-Meir, S.; Chowers, Y. Allicin inhibits spontaneous and TNF-alpha induced secretion of proinflammatory cytokines and chemokines from intestinal epithelial cells. Clin. Nutr. 2004, 23, 1199–1208. [Google Scholar] [CrossRef]
- Parameswaran, N.; Patial, S. Tumor necrosis factor-α signaling in macrophages. Crit. Rev. Eukaryot. 2010, 20, 87–103. [Google Scholar] [CrossRef]
- Haase, H.; Hieke, N.; Plum, L.M.; Gruhlke, M.C.H.; Slusarenko, A.J.; Rink, L. Impact of allicin on macrophage activity. Food Chem. 2012, 134, 141–148. [Google Scholar] [CrossRef]
- Dirsch, V.M.; Kiemer, A.K.; Wagner, H.; Vollmar, A.M. Effect of allicin and ajoene, two compounds of garlic, on inducible nitric oxide synthase. Atherosclerosis 1998, 139, 333–339. [Google Scholar] [CrossRef]
- Dipaolo, J.A.; Carruthers, C. The effect of allicin from garlic on tumor growth. Cancer Res. 1960, 20, 431–434. [Google Scholar]
- Miron, T.; Wilchek, M.; Sharp, A.; Nakagawa, Y.; Naoi, M.; Nozawa, Y.; Akao, Y. Allicin inhibits cell growth and induces apoptosis through the mitochondrial pathway in HL60 and U937 cells. J. Nutr. Biochem. 2008, 19, 524–535. [Google Scholar] [CrossRef]
- Oommen, S.; Anto, R.J.; Srinivas, G.; Karunagaran, D. Allicin (from garlic) induces caspase-mediated apoptosis in cancer cells. Eur. J. Pharmacol. 2004, 485, 97–103. [Google Scholar] [CrossRef]
- Park, S.-Y.; Cho, S.-J.; Kwon, H.-C.; Lee, K.-R.; Rhee, D.-K.; Pyo, S. Caspase-independent cell death by allicin in human epithelial carcinoma cells: involvement of PKA. Cancer Lett. 2005, 224, 123–132. [Google Scholar] [CrossRef]
- Bat-Chen, W.; Golan, T.; Peri, I.; Ludmer, Z.; Schwartz, B. Allicin purified from fresh garlic cloves induces apoptosis in colon cancer cells via Nrf2. Nutr. Cancer 2010, 62, 947–957. [Google Scholar] [CrossRef]
- Loboda, A.; Was, H.; Jozkowicz, A.; Dulak, J. Janus face of Nrf2-HO-1 axis in cancer—Friend in chemoprevention, foe in anticancer therapy. Lung Cancer 2008, 60, 1–3. [Google Scholar] [CrossRef]
- Niture, S.K.; Jaiswal, A.K. Nrf2 protein up-regulates antiapoptotic protein Bcl-2 and prevents cellular apoptosis. J. Biol. Chem. 2012, 287, 9873–9886. [Google Scholar] [CrossRef]
- Niture, S.K.; Jaiswal, A.K. Nrf2-induced antiapoptotic Bcl-xL protein enhances cell survival and drug resistance. Free Radic. Biol. Med. 2013, 57, 119–131. [Google Scholar] [CrossRef]
- Cha, J.H.; Choi, Y.J.; Cha, S.H.; Choi, C.H.; Cho, W.H. Allicin inhibits cell growth and induces apoptosis in U87MG human glioblastoma cells through an ERK-dependent pathway. Oncol. Rep. 2012, 28, 41–48. [Google Scholar]
- Arditti, F.D.; Rabinkov, A.; Miron, T.; Reisner, Y.; Berrebi, A.; Wilchek, M.; Mirelman, D. Apoptotic killing of B-chronic lymphocytic leukemia tumor cells by allicin generated in situ using a rituximab-alliinase conjugate Apoptotic killing of B-chronic lymphocytic leukemia tumor cells by allicin generated in situ using a rituximab-alliinase con. Mol. Cancer Ther. 2005, 4, 325–332. [Google Scholar]
- Parisy, V.; Poinssot, B.; Owsianowski, L.; Buchala, A.; Glazebrook, J.; Mauch, F. Identification of PAD2 as a gamma-glutamylcysteine synthetase highlights the importance of glutathione in disease resistance of Arabidopsis. Plant J. 2007, 49, 159–172. [Google Scholar]
- Rabinkov, A.; Miron, T.; Mirelman, D.; Wilchek, M.; Glozman, S.; Yavin, E.; Weiner, L. S-Allylmercaptoglutathione: The reaction product of allicin with glutathione possesses SH-modifying and antioxidant properties. Biochim. Biophys. Acta 2000, 1499, 144–153. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9–18. [Google Scholar]
- Noctor, G.; Gomez, L.; Vanacker, H.; Foyer, C.H. Interactions between biosynthesis, compartmentation and transport in the control of glutathione homeostasis and signalling. J. Exp. Bot. 2002, 53, 1283–1304. [Google Scholar] [CrossRef]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Borlinghaus, J.; Albrecht, F.; Gruhlke, M.C.H.; Nwachukwu, I.D.; Slusarenko, A.J. Allicin: Chemistry and Biological Properties. Molecules 2014, 19, 12591-12618. https://doi.org/10.3390/molecules190812591
Borlinghaus J, Albrecht F, Gruhlke MCH, Nwachukwu ID, Slusarenko AJ. Allicin: Chemistry and Biological Properties. Molecules. 2014; 19(8):12591-12618. https://doi.org/10.3390/molecules190812591
Chicago/Turabian StyleBorlinghaus, Jan, Frank Albrecht, Martin C. H. Gruhlke, Ifeanyi D. Nwachukwu, and Alan J. Slusarenko. 2014. "Allicin: Chemistry and Biological Properties" Molecules 19, no. 8: 12591-12618. https://doi.org/10.3390/molecules190812591
APA StyleBorlinghaus, J., Albrecht, F., Gruhlke, M. C. H., Nwachukwu, I. D., & Slusarenko, A. J. (2014). Allicin: Chemistry and Biological Properties. Molecules, 19(8), 12591-12618. https://doi.org/10.3390/molecules190812591





