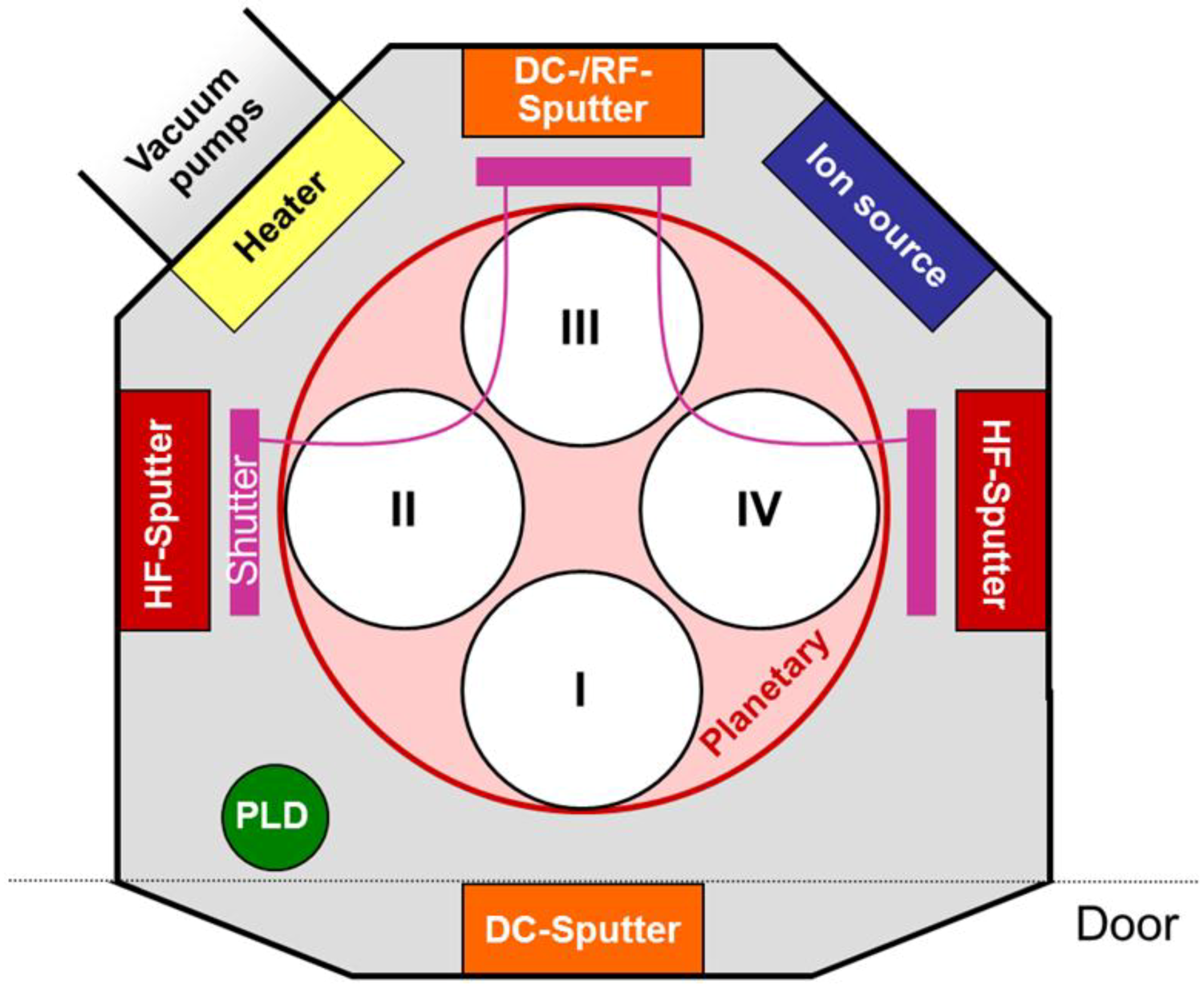
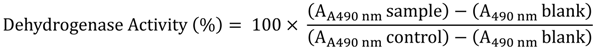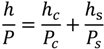3.1. Chemical Composition, Binding and Chemical Structure Analysis of the Films
The film materials, described above in
Section 2.2, were chosen from chemical elements with well-known non- or low cytotoxicity (C, O, N, Ti, Si) to minimize any possible negative effects on the biological interaction. This interaction to proteins and cells occurs on the topmost surface of the material, which is generally the oxide layer formed by oxidation in ambient atmosphere after vacuum film deposition. Proofing the chemistry of this surface was carried out by surface sensitive XPS investigations (1–3 nm analysis depth) without any sputtering of the oxide surface layer. Results are shown in
Table 2.
SiOx films are slightly substoichiometric with an O/Si ratio of 1.85 (SiO2:O:Si = 2.0) The oxygen contents of the other films were found to be between 6.5 and 32 at% leading to an O/C ratio between 0.07 and 0.53 with the highest values for the Ti doped DLC film (a-C:H:Ti) and the lower for pure, N and Si doped films. The ratio of (Si, Ti, N) dopant to carbon atoms is found to be Si/C = 0.02, N/C = 0.11, and Ti/C = 0.13 for a-C:H:Si, a-C:H:N, a-C:H:Ti films, respectively.
Table 2.
Results of the chemical X-ray photo-electron spectroscopy (XPS) analysis of thin film surfaces: Binding energies, calculated element concentrations and concentration ratios.
Table 2.
Results of the chemical X-ray photo-electron spectroscopy (XPS) analysis of thin film surfaces: Binding energies, calculated element concentrations and concentration ratios.
| Binding state | SiOx | a-C:H | a-C:H:N | a-C:H:Si | a-C:H:Ti |
|---|
| Binding energy [eV] | Content [at%] | Binding energy [eV] | Content [at%] | Binding energy [eV] | Content [at%] | Binding energy [eV] | Content [at%] | Binding energy [eV] | Content [at%] |
|---|
| C1s | 282.80 | 8.27 | | | | | | | | |
| 284.80 | 17.74 | 284.60 | 78.41 | 284.60 | 41.65 | 284.60 | 58.11 | 284.60 | 44.21 |
| | 285.54 | 10.59 | 285.57 | 18.69 | 285.62 | 7.71 | 285.90 | 8.50 |
| | 286.84 | 3.20 | 286.51 | 9.64 | 286.47 | 11.28 | 286.98 | 4.35 |
| | | | 287.70 | 4.87 | 287.64 | 5.64 | | |
| | 288.31 | 1.30 | | | | | 288.41 | 3.24 |
| | | | 288.84 | 2.78 | 288.79 | 2.99 | | |
| | | | 290.50 | 0.60 | | | | |
| ΣC | | 26.02 | | 93.50 | | 78.23 | | 85.73 | | 60.29 |
| Si2p | 100.88 | 4.57 | | | | | | | | |
| | | | | | 101.16 | 0.35 | | |
| | | | | | 102.53 | 1.38 | | |
| 103.43 | 21.40 | | | | | | | | |
| ΣSi | | 25.97 | | | | | | 1.73 | | |
| Ti2p | | | | | | | | | 458.49 | 6.88 |
| | | | | | | | 471.54 | 0.81 |
| ΣTi | | | | | | | | | | 7.69 |
| N1s | | | | | 398.23 | 1.09 | | | | |
| | | | 398.93 | 3.58 | | | | |
| | | | 400.01 | 3.48 | | | | |
| | | | 401.23 | 0.70 | | | | |
| | | | 402.81 | 0.30 | | | | |
| ΣN | | | | | | 9.15 | | | | |
| O1s | 529.16 | 0.76 | | | | | | | 529.92 | 16.44 |
| 530.64 | 36.90 | | | | | | | | |
| | 531.14 | 1.20 | 531.11 | 3.78 | 531.16 | 4.49 | 531.23 | 7.69 |
| 532.16 | 10.34 | 532.13 | 3.40 | 532.25 | 5.37 | 532.2 | 1.50 | 532.14 | 5.67 |
| | 533.11 | 1.90 | 533.38 | 3.48 | 533.2 | 6.56 | 533.15 | 2.23 |
| ΣO | | 48.01 | | 6.50 | | 12.62 | | 12.54 | | 32.02 |
| x/C | | | | | N/C | 0.11 | Si/C | 0.02 | Ti/C | 0.13 |
| O/C | | 1.85 | | 0.07 | | 0.16 | | 0.14 | | 0.53 |
| O/x | O/Si | 1.85 | | | | | O/Si | 7.26 | O/Ti | 4.16 |
Comparing the peak positions of the binding energy spectra with literature data for AlKα radiation [
49] allows conclusions for the formed chemical compounds: Standard bond energies for carbon (C1s) are between 280.8 and 283.0 eV for metal carbides, 284.5 eV for graphite, 285.2–288.4 eV for C–N, and 286.1–291.5 eV for C–O compounds. Thus, the films contain a high fraction of C–C bonds and only small fractions of C–O and C–N bonds. Metal carbide bonds are rather missing (e.g., being at 281.6 eV for TiC). Binding energy for Si2p is between 98.8 and 99.5 eV for pure Si, between 102.3 and 103.8 eV for SiO
2, and in between these thresholds for substoichiometric compounds. Thus, the SiO
x and a-C:H:Si film surfaces contain mixtures of Si and SiO
2 bonds with contributions of carbidic bonds in the doped DLC coating (standard binding energies for SiC: 99.9–100.9 eV). The high bond energy of the Ti2p peak indicates TiC-type bonds for the a-C:H:Ti film [
50]. N1s binding energies between 398 and 403 eV give hints for N incorporation in organic matrix (399–401 eV). Low binding energies for O1s in a-C:H:Ti (528.2–531.1 eV) indicate metal oxides (e.g., TiO
2: 529.9 eV), high energies in a-C:H:Si and SiO
x (532.5–533.5 eV) SiO
2.
The structural analysis of all carbon coatings was performed by Raman spectroscopy with much lower surface sensitivity (>10 nm analysis depth) than XPS. Basis for this analysis is the frequency shift of the incident light by Raman active molecules (Stokes scattering): Amorphous carbon has five frequency shifts (bands) between 800 and 2000 cm
−1, of which two enable conclusions on the chemical structure: The G band around 1550 cm
−1 is due to longitudinal oscillations of C atoms in sp
2 hybridization in rings and chains. Conversely, the D band around 1350 cm
−1 is due to centric oscillations of C atoms in rings. The intensity ratio of these two bands (
ID/
IG) provides the basis for all estimations of the sp
2 bond content in ring structures [
50]: Lower values are found for C atoms in chain structures, higher values for C in aromatic rings. If the D band is missing, no sp
2 hybridization of C in rings is present. Detailed information of the elemental bonds is gathered by FWHM analysis of the G band [
51], which is influenced by the structural disorder (bond lengths and angles). High FWHM (G) is found for a-C:H with max. 30 at% H (non-polymeric films) with high C–C sp
3 hybridization.
Figure 4 shows the overview of the taken Raman spectra in the range of the significant shifts for carbon atoms,
Table 3 the results of Gauss-Lorentz peak fitting (peak position FWHM,
ID/
IG ratios). The shown curves reveal an amorphous C structure for the a-C:H, a-C:H:N, and a-C:H:Ti films. However, the a-C:H:Si film is rather weak in Raman activity, which could be the reason for the high Si:C ratio. The G band is found for undoped a-C:H around 1534 cm
−1 and increases by N and Ti doping to 1543–1544 cm
−1, with highest FWHM (G) found for a-C:H:N. The positions of the D band are between 1340 and 1380 cm
−1 for a-C:H and a-C:H:N, but only at 1280 cm
−1 for a-C:H:Ti films. No tendency in relation to the film material is found for the FWHM (being generally between 300 and 350 cm
−1). The smallest I
D/I
G ratio was evaluated for the a-C:H:Ti film and increases for a-C:H and a-C:H:N films, revealing a shift of carbon binding from chain to aromatic ring structures. The higher FWHM (G) of a-C:H:N leads to expect a higher sp
3 content in C–C bonds.
Table 3.
Calculated wave numbers of peak maxima and FWHM (full width at half maximum) values of the D and G bands as well as ID/IG ratio of all diamond-like carbon based materials (DLC) films with measurable peaks (a-C:H, a-C:H:N, a-C:H:Ti).
Table 3.
Calculated wave numbers of peak maxima and FWHM (full width at half maximum) values of the D and G bands as well as ID/IG ratio of all diamond-like carbon based materials (DLC) films with measurable peaks (a-C:H, a-C:H:N, a-C:H:Ti).
| Film type | D [cm−1] | FWHM (D) [cm−1] | G [cm−1] | FWHM (G) [cm−1] | ID/IG [1] |
|---|
| a-C:H | 1345.26 | 324.25 | 1534.46 | 194.48 | 0.42 |
| a-C:H:N | 1377.09 | 332.49 | 1543.53 | 172.99 | 0.76 |
| a-C:H:Ti | 1277.10 | 291.01 | 1542.76 | 180.40 | 0.31 |
Figure 4.
Raman shifts of the carbon based thin films (a-C:H, a-C:H:N, a-C:H:Si, a-C:H:Ti) in the characteristic wave number range for the D (1350 cm−1) and G band (~1550 cm−1) of carbon.
Figure 4.
Raman shifts of the carbon based thin films (a-C:H, a-C:H:N, a-C:H:Si, a-C:H:Ti) in the characteristic wave number range for the D (1350 cm−1) and G band (~1550 cm−1) of carbon.
3.3. Film Topography on the Micro- and Nanometer Scale
Nano- and micro-sized defects in the films have especially a decisive influence on the gas barrier behavior. Consequently, strong attention was paid to topographical imaging: Light microscopy images in
Figure 6 show the topography on the micrometer scale of the films on PEEK as well as of the uncoated PEEK as reference. In contrast to the homogenous appearance of the PEEK substrate surface under the light microscope in reflected-light mode (
Figure 6a), the deposited films (
Figure 6b–f) possess distinct topographical features: This black-white contrast, indicating different intensity of reflected light by variously aligned surfaces, is significantly larger in the case of DLC based materials (a-C:H, a-C:H:N, a-C:H:Si, and a-C:H:Ti) than for SiO
x. Furthermore, these topographical structures are finer for ion source deposited than for sputtered films. Microdefects like pinholes, particulates, droplets,
etc. are missing rather: Counting of light microscopically visible defects (>1 µm diameter) on 1 mm
2 large areas reveals 5 ± 1 mm
−2 for a-C:H:Ti, 6 ± 1 mm
−2 for a-C:H:Si, 10 ± 1 mm
−2 for a-C:H:N, 19 ± 2 mm
−2 for SiO
x, and 21 ± 2 mm
−2 for a-C:H films. In contrast to our expectations, the magnetron sputtered films contain significantly less defects than the ion source deposited.
Figure 6.
Light microscopical images of (a) uncoated polyetheretherketone (PEEK) and thin films of 26 ± 3 nm on PEEK: (b) SiOx; (c) a-C:H; (d) a-C:H:N; (e) a-C:H:Si; and (f) a-C:H:Ti.
Figure 6.
Light microscopical images of (a) uncoated polyetheretherketone (PEEK) and thin films of 26 ± 3 nm on PEEK: (b) SiOx; (c) a-C:H; (d) a-C:H:N; (e) a-C:H:Si; and (f) a-C:H:Ti.
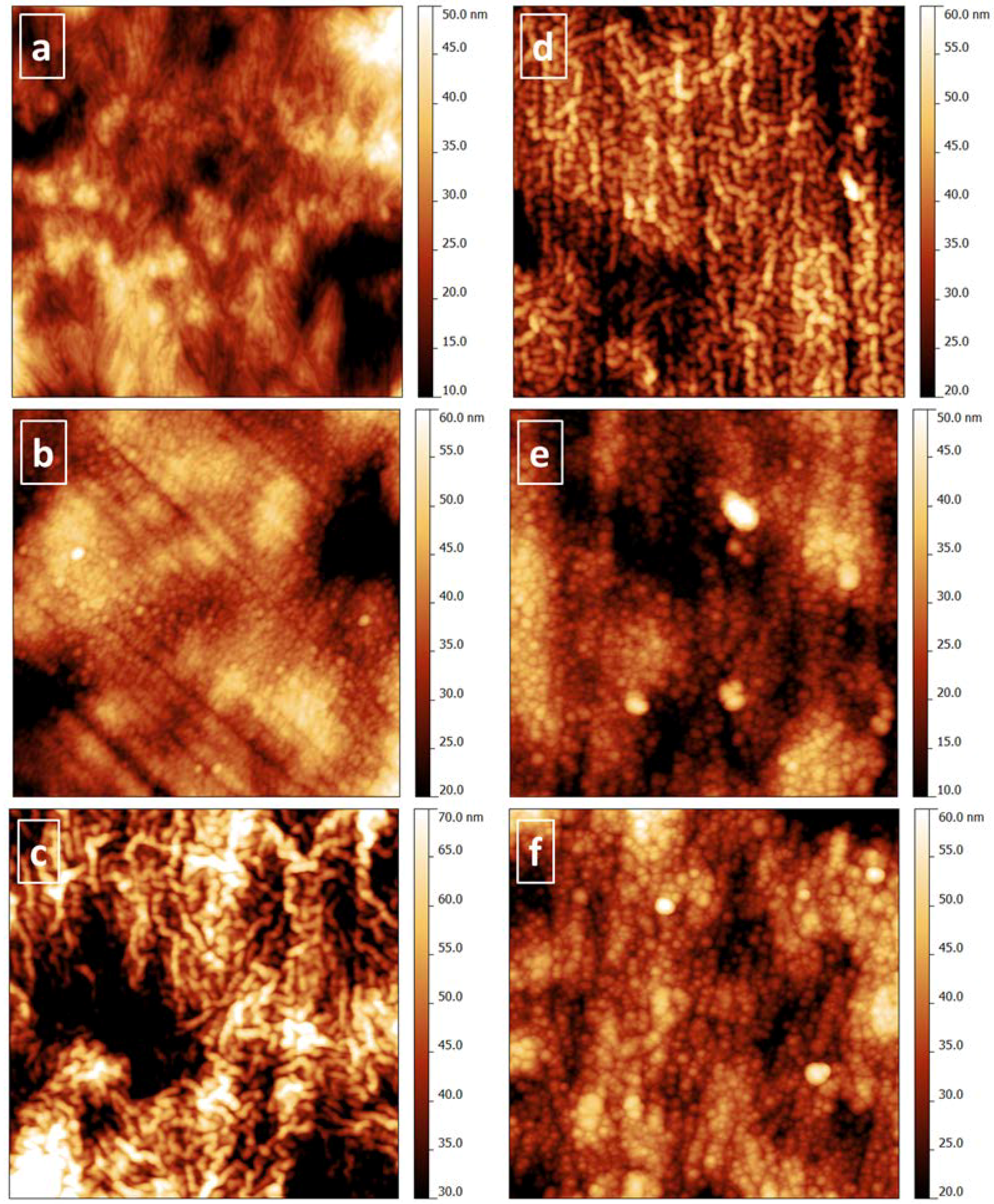
Detailed investigations of the surface nanotopography were carried out by AFM: Images are shown in
Figure 7 for films on PEEK and for comparison and for discussion of the occurring growth phenomena in
Figure 8 for films on glass substrates. Calculated RMS roughness, lateral correlation length (LCL), and Hurst parameter values are given in
Table 4. RMS roughness, a measure of the vertical roughness, provides height (amplitude) information of the topographical features, while LCL data indicates lateral frequency of the roughness features (lateral distance, across which heights of the surface are correlated).
Generally, the several times smoother glass substrate results in smaller RMS roughness, standard deviation of RMS, and LCL of the thereon deposited films compared to films on PEEK. Furthermore, no significant differences are found for RMS and LCL data obtained from 2 × 2 and 16 × 16 µm
2 images on glass. However, the image size strongly influences these values for coated and uncoated PEEK. In contrast, a significant influence of the Hurst parameter, describing the “jaggedness” of the surface, is missing for differently sized images obtained from coated PEEK, but found for films on glass substrates. Generally, films deposited on glass substrates exhibited a smoother topography, resulting in smaller RMS. The smoothest film is found to be the ion source deposited a-C:H, the roughest the sputtered SiO
x and a-C:H:Si. For the latter film, the LCL is also quite high, which is due to some large features influencing the data analysis. The images (
Figure 8b–f) reveal preferentially filling of the valleys in between the roughness tips of the uncoated glass substrate (
Figure 8a) with material during deposition by diffusion. Due to room temperature deposition, diffusion is scarcely thermally activated and needs additional driving force, provided by higher energetic species from the plasma. While the ion energy during (non-biased) DC pulsed or RF sputtering is generally lower than 20 eV, ions and kinetic atoms in the fragmented ethyne precursor plasma have several 100 eV of energy (medium energy ~400 eV). Reasonably, the ion source deposited a-C:H and a-C:H:N films are decisively smoother than all others. This is visible in
Figure 8c,d which also lack the dot-like features of about 20 nm lateral size and 30–50 nm distance (LCR) , which are found for a-C:H:Si, a-C:H:Ti and SiO
x (
Figure 8b,e,f). Such features have been widely studied for crystalline thin film growth and are due to the preferred growth of some crystallographically well aligned nano-grains. For preponderant amorphous films on not ultra-smooth substrates, this process is influenced by the substrate topography.
Figure 7.
Atomic Force Microscopy (AFM) surface topography images of (a) PEEK substrate, and (b) SiOx, (c) a-C:H, (d) a-C:H:N, (e) a-C:H:Si, and (f) a-C:H:Ti on PEEK substrates. (image size: 2 × 2 µm2, height scale: 100 nm)
Figure 7.
Atomic Force Microscopy (AFM) surface topography images of (a) PEEK substrate, and (b) SiOx, (c) a-C:H, (d) a-C:H:N, (e) a-C:H:Si, and (f) a-C:H:Ti on PEEK substrates. (image size: 2 × 2 µm2, height scale: 100 nm)
Figure 8.
AFM surface topography images of (a) glass substrate, and (b) SiOx, (c) a-C:H, (d) a-C:H:N, (e) a-C:H:Si, and (f) a-C:H:Ti on glass substrate. (image size: 2 × 2 µm2, height scale: 30 nm)
Figure 8.
AFM surface topography images of (a) glass substrate, and (b) SiOx, (c) a-C:H, (d) a-C:H:N, (e) a-C:H:Si, and (f) a-C:H:Ti on glass substrate. (image size: 2 × 2 µm2, height scale: 30 nm)
Table 4.
Root-mean-squared (RMS) roughness, lateral correlation length and Hurst parameter of uncoated substrates (PEEK, glass) and ~25 nm thin films thereon.
Table 4.
Root-mean-squared (RMS) roughness, lateral correlation length and Hurst parameter of uncoated substrates (PEEK, glass) and ~25 nm thin films thereon.
| Material | Substrate | SiOx | a-C:H | a-C:H:N | a-C:H:Si | a-C:H:Ti |
|---|
| PEEK | RMS roughness [nm] | 16 × 16 µm2 scan size | 26.70 ± 4.85 | 22.85 ± 2.70 | 25.70 ± 1.05 | 25.15 ± 3.45 | 19.05 ± 1.65 | 24.05 ± 1.45 |
| 2 × 2 µm2 scan size | 9.50 ± 1.05 | 8.60 ± 0.70 | 13.65 ± 2.35 | 8.70 ± 0.60 | 7.50 ± 0.65 | 9.35 ± 1.85 |
| Lateral correlation length ξ [nm] | 16x16 µm2 scan size | 1200 ± 600 | 1100 ± 150 | 1200 ± 250 | 1600 ± 250 | 1100 ± 150 | 745 ± 85 |
| 2 × 2 µm2 scan size | 220 ± 40 | 260 ± 50 | 170 ± 45 | 130 ± 45 | 200 ± 60 | 220 ± 60 |
| Hurst parameter α [1] | 16 × 16 µm2 scan size | 0.55 ± 0.10 | 0.60 ± 0.05 | 0.50 ± 0.00 | 0.50 ± 0.05 | 0.50 ± 0.05 | 0.50 ± 0.00 |
| 2 × 2 µm2 scan size | 0.80 ± 0.20 | 0.70 ± 0.10 | 0.50 ± 0.05 | 0.50 ± 0.00 | 0.60 ± 0.10 | 0.55 ± 0.05 |
| Glass | RMS roughness [nm] | 16 × 16 µm2 scan size | 4.55 ± 1.20 | 2.75 ± 0.20 | 1.15 ± 0.25 | 1.95 ± 0.40 | 2.10 ± 1.25 | 2.10 ± 0.10 |
| 2 × 2 µm2 scan size | 3.85 ± 0.55 | 2.75 ± 0.15 | 0.80 ± 0.10 | 1.80 ± 0.20 | 3.40 ± 2.40 | 2.10 ± 0.15 |
| Lateral correlation length ξ [nm] | 16 × 16 µm2 scan size | 80 ± 55 | 80 ± 25 | 155 ± 120 | 125 ± 65 | 160 ± 65 | 40 ± 5 |
| 2 × 2 µm2 scan size | 45 ± 5 | 30 ± 10 | 35 ± 5 | 40 ± 5 | 140 ± 100 | 20 ± 1 |
| Hurst parameter α [1] | 16 × 16 µm2 scan size | 0.65 ± 0.10 | 0.50 ± 0.00 | 0.50 ± 0.00 | 0.55 ± 0.05 | 0.55 ± 0.05 | 0.65 ± 0.05 |
| 2 × 2 µm2 scan size | 0.85 ± 0.05 | 0.70 ± 0.05 | 0.80 ± 0.10 | 0.75 ± 0.05 | 0.60 ± 0.10 | 0.85 ± 0.05 |
On PEEK substrates, the film formation mode appears to be very different especially for a-C:H and a-C:H:N, if comparing to layers on glass substrate: Apart from the overall rougher topography of PEEK, the growth mode of sputtered films appears to be similar to glass with the formation of small dots. Compared to the substrate, the RMS and LCL are not significantly influenced. a-C:H and a-C:H:N films form a different type of topography, consisting of worm-like features. Such a film formation process (“nano-wrinkling”) was described by the authors for other soft polymer substrate types [
54,
55,
56] and is mechanically influenced: The higher the energy of the deposited species, the higher is the tendency to implantation of plasma species into the growing films. Such a several nanometer deep implantation increases the intrinsic stresses of the films. Soft substrates like polymers are able to deform elastically to decrease the intrinsic stress, which is generally compressive under the applied deposition conditions. The result of this stress relaxation is the tensile straining of the surface, resulting in a wavy topography. Stress relaxation occurs stepwise in continuing film growth: Thicker films on polymers are stiffer and, thus, wrinkle to reduce the internal stress at larger wavelengths. Finally, a hierarchically self-structured surface is formed. While a 25 nm film thickness is sufficient on a soft PU substrate (
E < 0.5 GPa) for the introduction of a second, larger hierarchical wrinkle structure, such topography is not incisive on PEEK (
E ~ 2.5 GPa).
3.4. Mechanical Behavior of Films—Elastic Modulus and Hardness
Indentation tests were used to obtain the mechanical properties of the thin films. To exclude influences of soft substrate deflection under the applied measurement loads, >500 nm thick films on silicon substrates were applied with >1 mN normal forces on the Berkovich indenter to guarantee a maximum indentation depth of 10% of the film thickness and to measure intrinsic film properties. However, this excludes the compound properties and, thus, the influences of wrinkling on elastic properties. Generally, wrinkles smooth out under tensile stresses, providing higher total substrate strain to film failure [
57]. However, studies which included such substrate material behavior (thus, giving information about the compliance of substrate and film) were previously published by the authors [
58]).
Calculated hardness and elastic modulus data from thick films, based on Oliver-Pharr [
36] theory, are shown in
Table 5. Although these mechanical values were gathered from measurements on silicon substrates, they provide the basis for a qualitative discussion and allow predictions of the behavior of coated polymers. Elastic deformability is of general importance in the field of “stiff” and dense gas permeation barrier of thin film materials on soft, deformable polymers: Cracks under tensile stresses, which develop during bending of the very thin foil substrates, must also be prevented. Elastic modulus and hardness give measures of the dependency of stress and strain in the elastic regime and for the ultimate strength before plastic deformation (the end of the elastic regime), respectively. In combination, the ratio
H/
E (elasticity ratio) provides a factor to compare materials [
59,
60]: A high elasticity ratio indicates high elastic deformability before fracture. As visible in
Table 5, non-wrinkled a-C:H:Si as well as SiO
x thin films on stiff Si substrates would be the best candidates based on the H/E ratio. Values for fracture toughness (
Kc) based on crack propagation after Vicker indentation [
37,
38] are also given in
Table 5, which give an indication of the plastic deformability of the material before the onset of fracture: High values are present for nanocrystalline a-C:H:Ti films.
Table 5.
Hardness (H), elastic modulus (E), fracture toughness (Kc) and elasticity ratio (H/E) of >500 nm thick coatings on Si substrates.
Table 5.
Hardness (H), elastic modulus (E), fracture toughness (Kc) and elasticity ratio (H/E) of >500 nm thick coatings on Si substrates.
| Coating type | H [GPa] | E [GPa] | KC [MPa m0.5] | H/E [1] |
|---|
| a-C:H | 18.5 ± 2.2 | 206 ± 16 | 0.9 ± 0.1 | 0.0898 |
| a-C:H:N | 14.2 ± 1.9 | 163 ± 8 | 1.1 ± 0.1 | 0.0871 |
| a-C:H:Si | 18.0 ± 2.1 | 170 ± 6 | 1.6 ± 0.1 | 0.1059 |
| a-C:H:Ti | 18.6 ± 2.2 | 260 ± 15 | 3.9 ± 0.2 | 0.0715 |
| SiOx | 7.9 ± 1.3 | 75 ± 3 | 0.7 ± 0.1 | 0.1056 |
3.5. Wetting Characteristics and Surface Energies of the Films
The wetting behavior as well as the surface energy of the coatings was characterized by contact angle measurements with 1 µL droplet size, followed by Kaelble plotting for calculating the polar and dispersive contributions to the surface energy (
Table 6). Basically all films as well as the PEEK substrate are hydrophobic with the lowest water contact angle found for SiO
x. While the PEEK substrate and the carbon based films have a high dispersive contribution to the total surface energy, the polar contribution is significantly higher for the SiO
x films. The highest total surface energy is present for a-C:H:Ti, followed by SiO
x films, while all other materials show rather similar, lower values. Comparing these results with the chemical composition on the surface, the high oxygen content on the a-C:H:Ti film by the atmospheric oxidation reaction of Ti seems to be the cause of this behavior.
Table 6.
Contact angles and surface energies (total, polar, dispersive) of PEEK foils and thin films on PEEK.
Table 6.
Contact angles and surface energies (total, polar, dispersive) of PEEK foils and thin films on PEEK.
| Material | PEEK | SiOx | a-C:H | a-C:H:N | a-C:H:Si | a-C:H:Ti |
|---|
| H2Odeionized contact angle [°] | 85.2 ±3.0 | 48.8 ± 2.8 | 92.9 ± 1.0 | 95.3 ± 0.8 | 88.3 ± 1.6 | 108.5 ± 0.8 |
| Polar contribution to surface energy σps [mJ m−2] | 3.5 | 37.9 | 1.3 | 0.7 | 3.1 | 1.0 |
| Dispersive contribution to surface energy σds [mJ m−2] | 33.3 | 11.3 | 36.2 | 39.3 | 33.8 | 57.5 |
| Total surface energy σs [mJ m−2] | 36.8 | 48.98 | 37.5 | 40.0 | 36.9 | 58.5 |
Contact angle measurements with about one order of magnitude smaller droplet size (one picoliter, average lateral droplet diameter in µm range) revealed in earlier investigations a significant influence of wrinkling nanotopography: While thicker films with 100 nm are highly hydrophobic (contact angle of a-C:H and titanium nitride on polyurethane ~80°), such films of 20 and 23 nm thickness with only first order wrinkles (~26 nm RMS, similar to results of this work) are significantly higher hydrophilic (water contact angle ~50°).
3.6. Gas Permeation Behavior
The permeation of O
2 through coated as well as uncoated PEEK was measured by optochemical analysis. Presented data for the OTR in
Table 7 indicates very high oxygen permeation through the 50 µm thick PEEK foil (>200 cm
3 m
−2 d
−1 bar
−1, being out of the range of the highly accurate analysis technique). Even 26 nm thin films reduce the oxygen permeation by nearly two orders of magnitude with high reproducibility (low standard deviation in measurements): Lowest OTR values are found for the films grown by direct ion source deposition (a-C:H, a-C:H:N), while sputtered films enable higher gas permeation. The barrier effect is higher for a-C:H:Si than for the SiO
x and a:C:H:Ti films, whereby the level of permeation is comparable to published data of sputtered oxide coatings of similar thickness [
61].
Table 7.
Oxygen transmission rates (OTR) of uncoated and 25 nm thin coating on PEEK.
Table 7.
Oxygen transmission rates (OTR) of uncoated and 25 nm thin coating on PEEK.
| Material | PEEK-Substrate | SiOx | a-C:H | a-C:H:N | a-C:H:Si | a-C:H:Ti |
|---|
| OTR [cm3 m−2 d−1 bar−1] | >200 | 7.91 ± 0.011 | 5.0 ± 0.005 | 3.16 ± 0.006 | 6.59 ± 0.016 | 9.74 ± 0.021 |
Gas permeation can be theoretically described based on the equation
in which
h is the thickness and
P the permeability of the film (c), substrate (s) and the whole system [
62]. Generally, permeation in thin film materials is defect controlled, while the intrinsic permeation rates of the material has inferior contribution. Different effects occur for micro and nano scale defects: Pores (pinholes) and cracks with lateral size >1 µm belong to the group of micro defects [
63,
64], while the group of nano defects contain specific film growth effects like the porosity around the tapered grains [
65]. Although the nuclei density at the beginning of film growth is about 2 nm
−2, the preferential growth of some crystals as well as the mechanical stress relaxation by wrinkling, results in the structures shown in the AFM images above (
Figure 7 and
Figure 8). The effect of nano defects is much higher than that of micro defects due to their extremely high density over the whole surface. This explains the advantageous behavior of the films, deposited directly from the ion source plasma, compared to the sputtered types, although especially the a-C:H films contain a larger density of micro defects (see light microscopy results above). The OTR values for a-C:H:Si, a-C:H:Ti, and SiO
x films with lower defect density, but domed surfaces are significantly higher. A positive influence seems to occur with wrinkling, which increases the mechanical tensile strain of fracture: Before fracture, wrinkles will smooth out and form a flat surface, providing up to 5% elasticity [
57]. The highest OTR value for a-C:H:Ti can both be influenced by the low elasticity index of this material and the quite porous structure (see above).
3.7. In Vitro Biocompatibility Testing
Although all materials in the investigations were selected from the group of non-toxic elements, biocompatibility tests were performed to finally check the interaction of the films with biological material.
In vitro testing of biomaterials based on ISO 10993 [
66] requires detection of bacterial contamination and the identification of possible cellular damage as the initial steps in proof of the biocompatibility. Endotoxin from gram-negative bacteria is the most common cause of toxic reactions resulting from the contamination of pharmaceutical products with pyrogens. Their pyrogenic activity is much higher than that of most other pyrogenic substances. In spite of some pyrogens with different structures, the absence of bacterial endotoxins in a product implies the absence of pyrogenic components, being a justified conclusion of such investigations. For the detection of endotoxin the increase in the rectal temperature of white rabbits after intravenous injection of samples extracts can be used. An alternative
in vivo assay, the Limulus Amebocyte Lysate (LAL) assay, has replaced this assay and is recommended by the European Medicines Agency to evaluate medical devices for bacterial endotoxins [
67]. The LAL test is based on the activation of coagulase in the blood cells (amoebocytes) of the horseshoe crab and was described as equivalent to the rabbit pyrogen test by Ronneberger [
68]. In order to perform the bacterial endotoxins test, an aqueous extract of the sample is used as the test solution [
47].
As the second step,
in vitro cytotoxicity screening is recommended to determine the cytotoxic potential of new materials or formulations for possible use in medical applications. This is needed because any local leakage of toxic substances can result in cell damage and death, which will result in the recruitment of a variety of cells to the site of implantation. Continuous or prolonged leakage of toxic substances will induce persistent inflammation and, thereby, interfere with the successful performance of the implant. Cytotoxicity testing on cell lines shows in many cases good correlation with animal assays. It is frequently more sensitive than animal studies and equally predictive for acute toxicity in humans to rodent
in vivo studies [
69]. According to ISO 10993-5, cytotoxicity testing should be performed by eluate testing and by direct contact testing [
70]. Cytotoxicity screening tests of the eluates are based on the determination of the total amount of proteins, DNA, cell number or enzymatic activity of a cell population [
71]. The use of formazan bioreduction for the assessment of cell viability is a generally accepted technique [
72].
The evaluation of the direct contact between cells and biomedical devices such as implants identifies the presence of potential leachables, which may induce toxic results
in vitro. The detection of cell damage is performed by cell staining using colorimetric dyes. The cell morphology and cell detachment are recorded after 24 h exposure [
45].
In pilot experiments, biocompatibility of the bare PEEK was assessed. Eluates from the foil did not decrease cell viability compared to cells treated with cell culture medium only. Viabilities were 103% ± 6% after 24 h, 102% ± 2% after 48 h, and 100% ± 9% after 72 h. Thereafter, PEEK with films were evaluated. All test sample extracts did not induce clotting of amoebocyte lysate. This indicates endotoxin levels below the detection threshold of the assay (0.06 EU/mL). Such low levels of endotoxin are regarded as non-pyrogenic according to the guidelines of the FDA and EMA [
73]. Testing of eluates/extracts decreased cell viability only in the positive controls (20 nm plain polystyrene particles) but not in the samples (
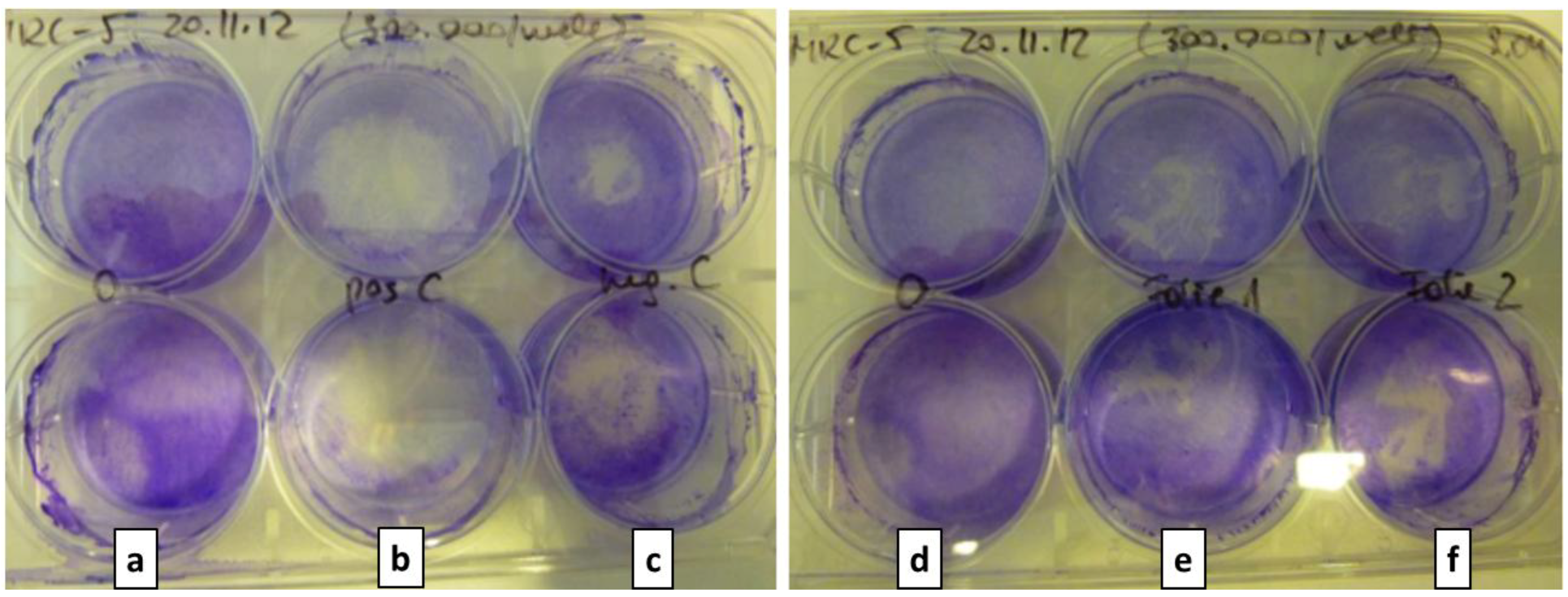
Figure 9, reaction of MRC-5 is shown as example). No time dependent decrease in viability was seen. Cells detached from the plastic support after exposure to the positive control (copper foil) (
Figure 10, MRC-5 cells shown). Negative control and test samples showed cell loss only beneath the contact zone with the test samples. This effect is usually explained by mechanical damage. Since cells and samples are only in loose contact, additional effects by lack of oxygen or medium supply can be disregarded.
Figure 11 and
Figure 12 show the different morphological features of MRC-5 and L929 cells. MRC-5 cells show the typical elongated, spindle-shaped morphology of differentiated fibroblasts (
Figure 11), while the shape of L929 cells is more epitheloid (
Figure 12). Both cell lines reacted to the exposure with the test samples in a similar way. In cells exposed to negative control and test samples (a-C:H foil is shown as example), the typical morphology of the cells is maintained (
Figure 11a,b,e,f and
Figure 12a,b,e,f), while cells exposed to positive control have an altered morphology with rounding and partial detachment of cells and loss of the typical nuclear architecture (
Figure 11c,d and
Figure 12c,d). To find out whether cellular adherence and growth pattern on a-C:H film on PEEK, on PVC foil (non-toxic negative control), and on cell culture plates were similar, fluorescence of Calcein staining was documented and quantified, and total cell number were determined. Cells on all substrates showed strong fluorescence (
Figure 13). Calcein fluorescence of cells grown on a-C:H film on PEEK and on PCV foil were 104.5% and 70.8% of control, respectively. Cell numbers on a-C:H film on PEEK and on PCV foil were 79.1% and 50.1% of control, respectively.
Figure 9.
Viability of human (MRC-5) fibroblasts according to formazan bioreduction after 24 h, 48 h and 72 h in the presence of positive control (PC) and test samples (pure eluates). Marked decrease in viability is seen after exposure to the positive control (20 nm plain polystyrene particles), while no decrease in viability occurred after exposure to eluates of test samples. Cells treated with medium only are set as 100% viability.
Figure 9.
Viability of human (MRC-5) fibroblasts according to formazan bioreduction after 24 h, 48 h and 72 h in the presence of positive control (PC) and test samples (pure eluates). Marked decrease in viability is seen after exposure to the positive control (20 nm plain polystyrene particles), while no decrease in viability occurred after exposure to eluates of test samples. Cells treated with medium only are set as 100% viability.
Figure 10.
Crystal violet staining of MRC-5 cells (a,d) without exposure (growth control) and (b) after exposure to positive control, (c) negative control, PEEK foils with (e) a-C:H films and (f) a-C:H:N films.
Figure 10.
Crystal violet staining of MRC-5 cells (a,d) without exposure (growth control) and (b) after exposure to positive control, (c) negative control, PEEK foils with (e) a-C:H films and (f) a-C:H:N films.
Figure 11.
Human (MRC-5) fibroblasts exposed to (a,b) negative control (PVC foil), (c,d) positive control (copper foil) and (e,f) a-C:H film on PEEK. Healthy MRC-5 cells are spindle-shaped and nuclei usually containing 1–2 darker nucleoli are clearly visible (arrowheads). No morphological alterations and no detached cells are seen in cells exposed to negative control (a,b) and test sample (e,f). Cells after exposure to the positive control show rounded appearance and no normal nucleus can be discerned (c,d).
Figure 11.
Human (MRC-5) fibroblasts exposed to (a,b) negative control (PVC foil), (c,d) positive control (copper foil) and (e,f) a-C:H film on PEEK. Healthy MRC-5 cells are spindle-shaped and nuclei usually containing 1–2 darker nucleoli are clearly visible (arrowheads). No morphological alterations and no detached cells are seen in cells exposed to negative control (a,b) and test sample (e,f). Cells after exposure to the positive control show rounded appearance and no normal nucleus can be discerned (c,d).
Figure 12.
Murine (L929) fibroblasts exposed to (a,b) negative control (PVC foil), (c,d) positive control (copper foil) and (e,f) a-C:H film on PEEK. Healthy L929 cells at low cell density have an elongated shape and at higher densities are cuboid. Nuclei containing several nucleoli are clearly visible (arrowheads). No morphological alterations and no detached cells are seen in cultures exposed to negative control (a,b) and test sample (e,f) and occasional dividing cells are seen (arrows). Cells after exposure to the positive control show rounded appearance and no normal nucleus can be discerned (c,d).
Figure 12.
Murine (L929) fibroblasts exposed to (a,b) negative control (PVC foil), (c,d) positive control (copper foil) and (e,f) a-C:H film on PEEK. Healthy L929 cells at low cell density have an elongated shape and at higher densities are cuboid. Nuclei containing several nucleoli are clearly visible (arrowheads). No morphological alterations and no detached cells are seen in cultures exposed to negative control (a,b) and test sample (e,f) and occasional dividing cells are seen (arrows). Cells after exposure to the positive control show rounded appearance and no normal nucleus can be discerned (c,d).
Figure 13.
MRC-5 fibroblasts cultured on cell culture plate (a), negative control (PVC foil, b) and a-C:H film on PEEK (c) and stained with Calcein AM dye (green). Scale bar: 50 µm.
Figure 13.
MRC-5 fibroblasts cultured on cell culture plate (a), negative control (PVC foil, b) and a-C:H film on PEEK (c) and stained with Calcein AM dye (green). Scale bar: 50 µm.
L929 cells were one of the first cells to be established in continuous culture. The line was established already in 1940 and is still the standard for cytotoxicity testing according to ISO 10993 guidelines [
44]. MRC-5 cells are of human origin and were established around 30 years later [
43]. Several publications suggest that human and rodent primary cells and cell lines derived from both species are equally suitable for the prediction of lethality [
74,
75]. Our data are in accordance with this assumption because all samples produced similar effects in both murine and human cell lines. It is, therefore, unlikely that these foils cause cell damage
in vivo.