Effects of Varied Cleaning Methods on Ni-5% W Substrate for Dip-Coating of Water-based Buffer Layers: An X-ray Photoelectron Spectroscopy Study
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemical Cleaning with Etching Process
2.2. Thermal Cleaning Process
2.3. XPS Characterization
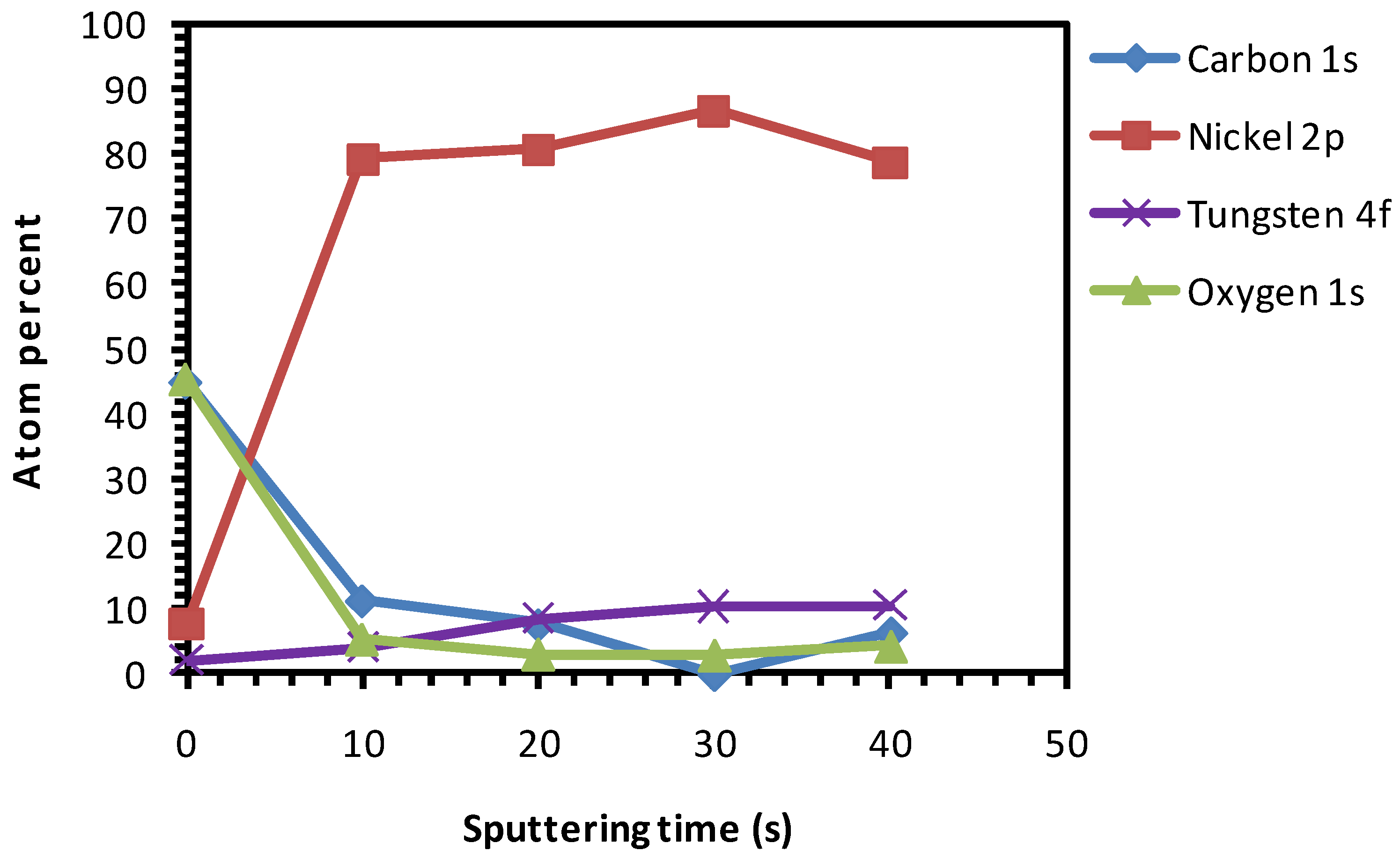
3. Results and Discussion
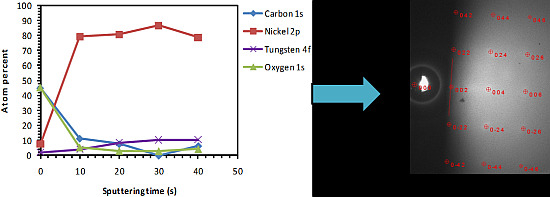
3.1. Uncleaned Ni-5% W Substrate
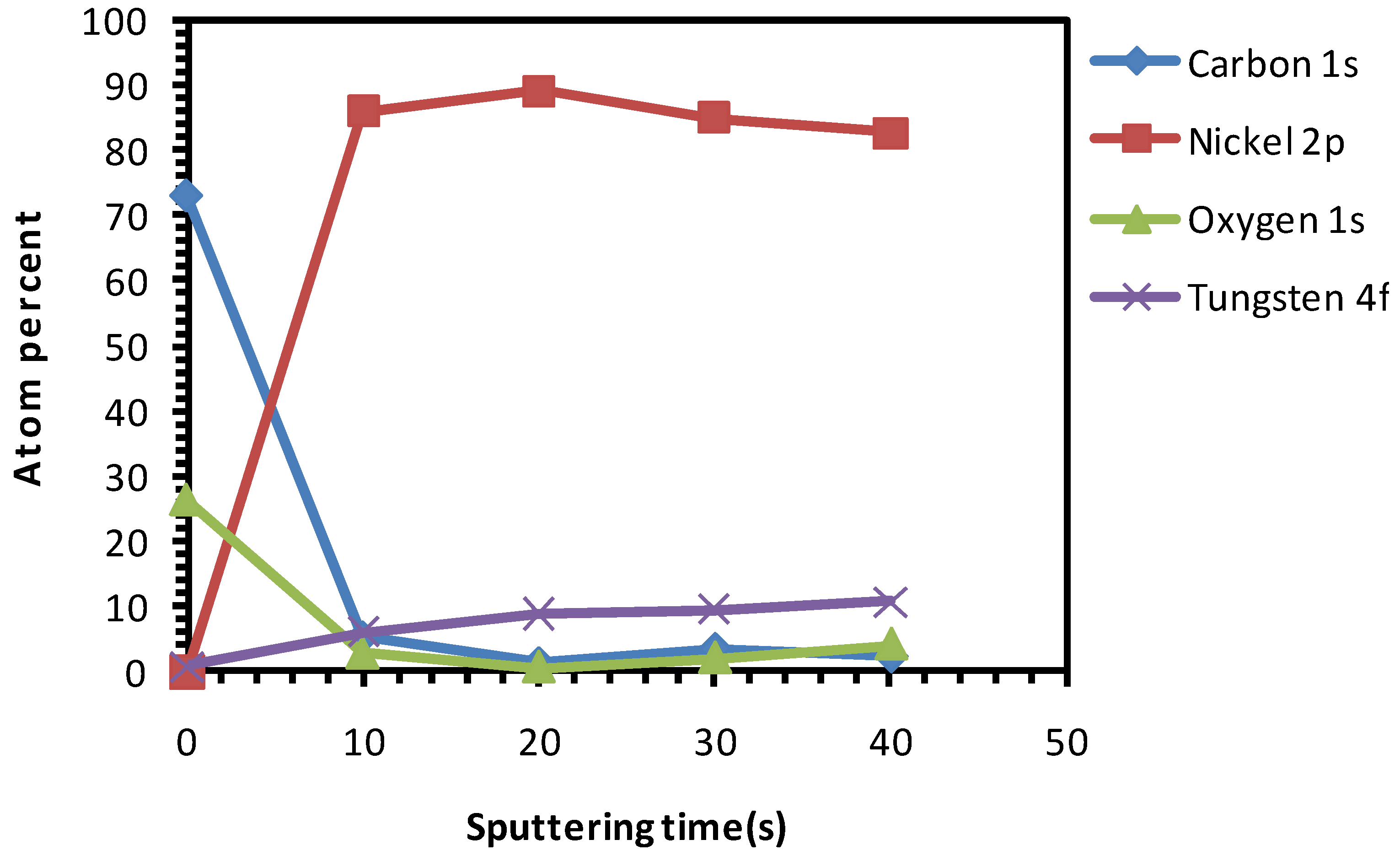
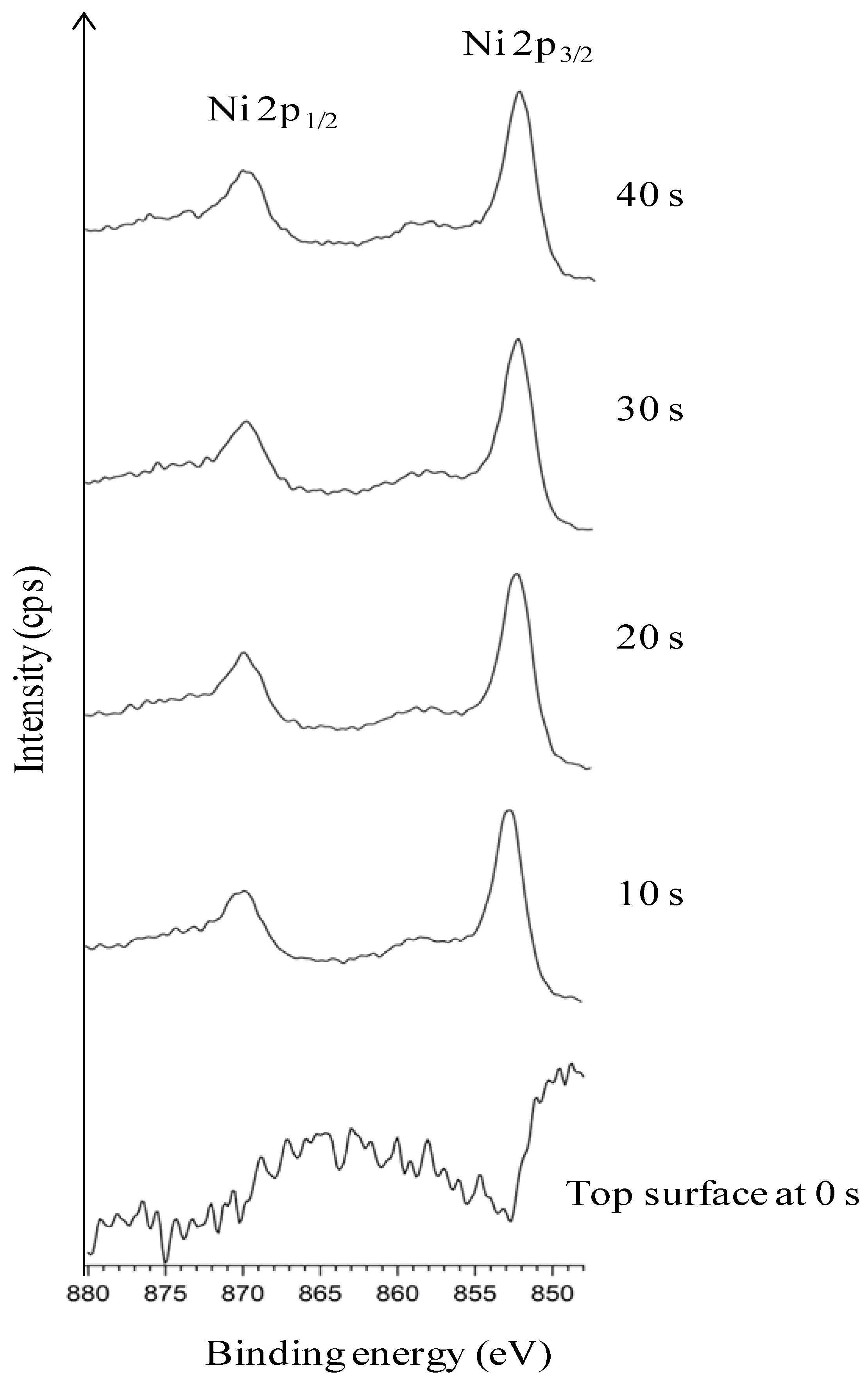

3.2. Chemical Cleaning with Etching for 15 min
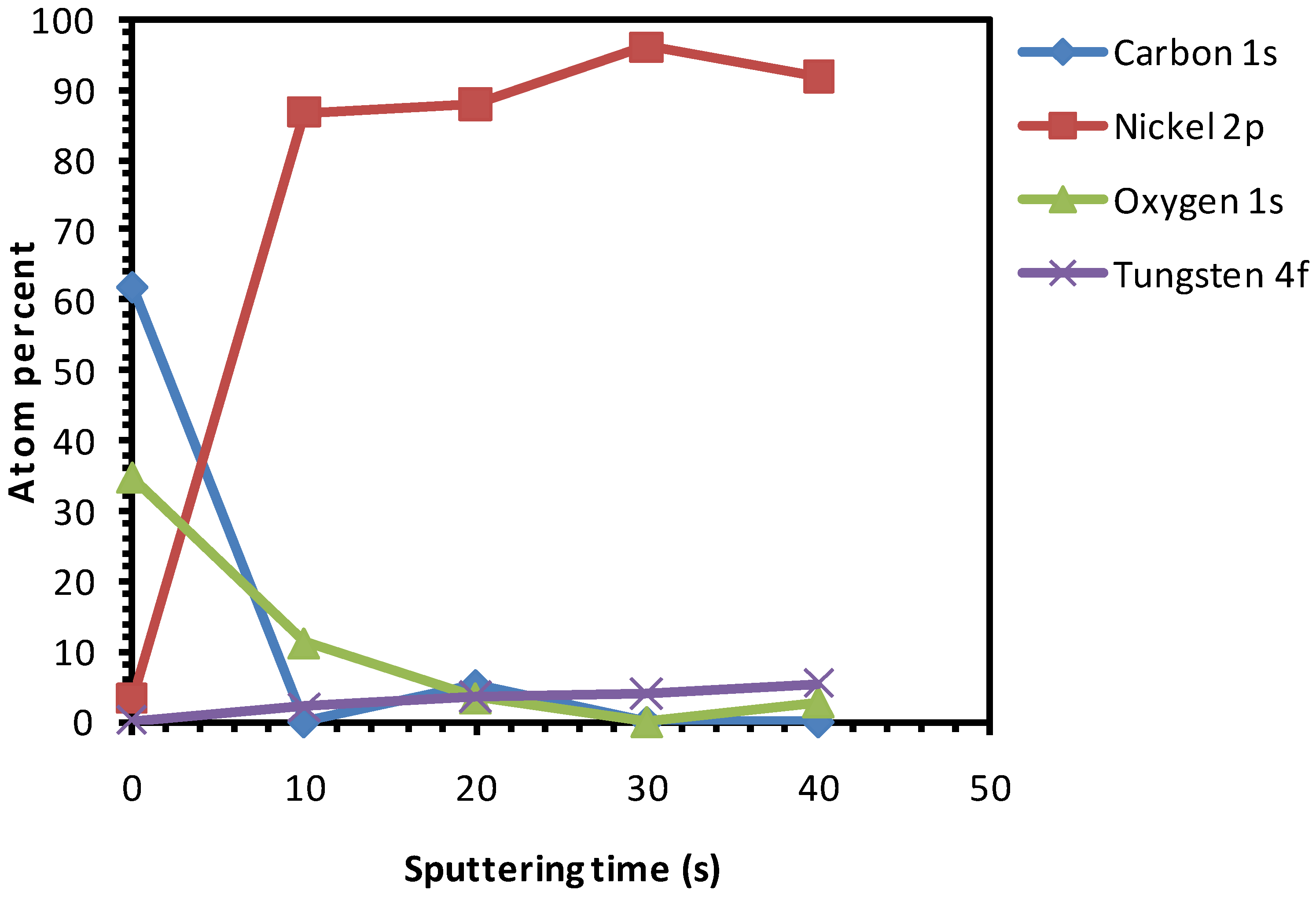
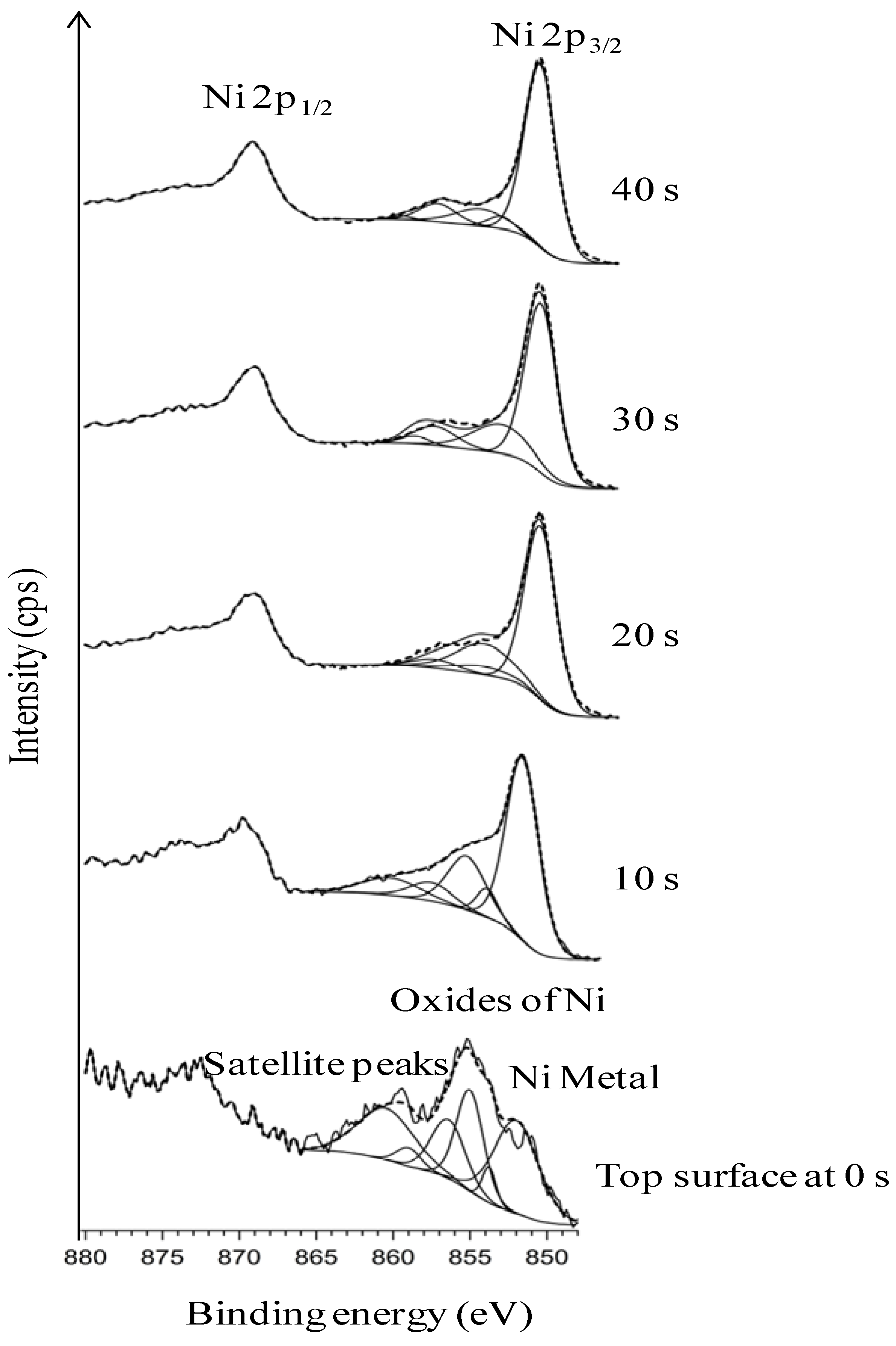

3.3. Thermal Cleaning
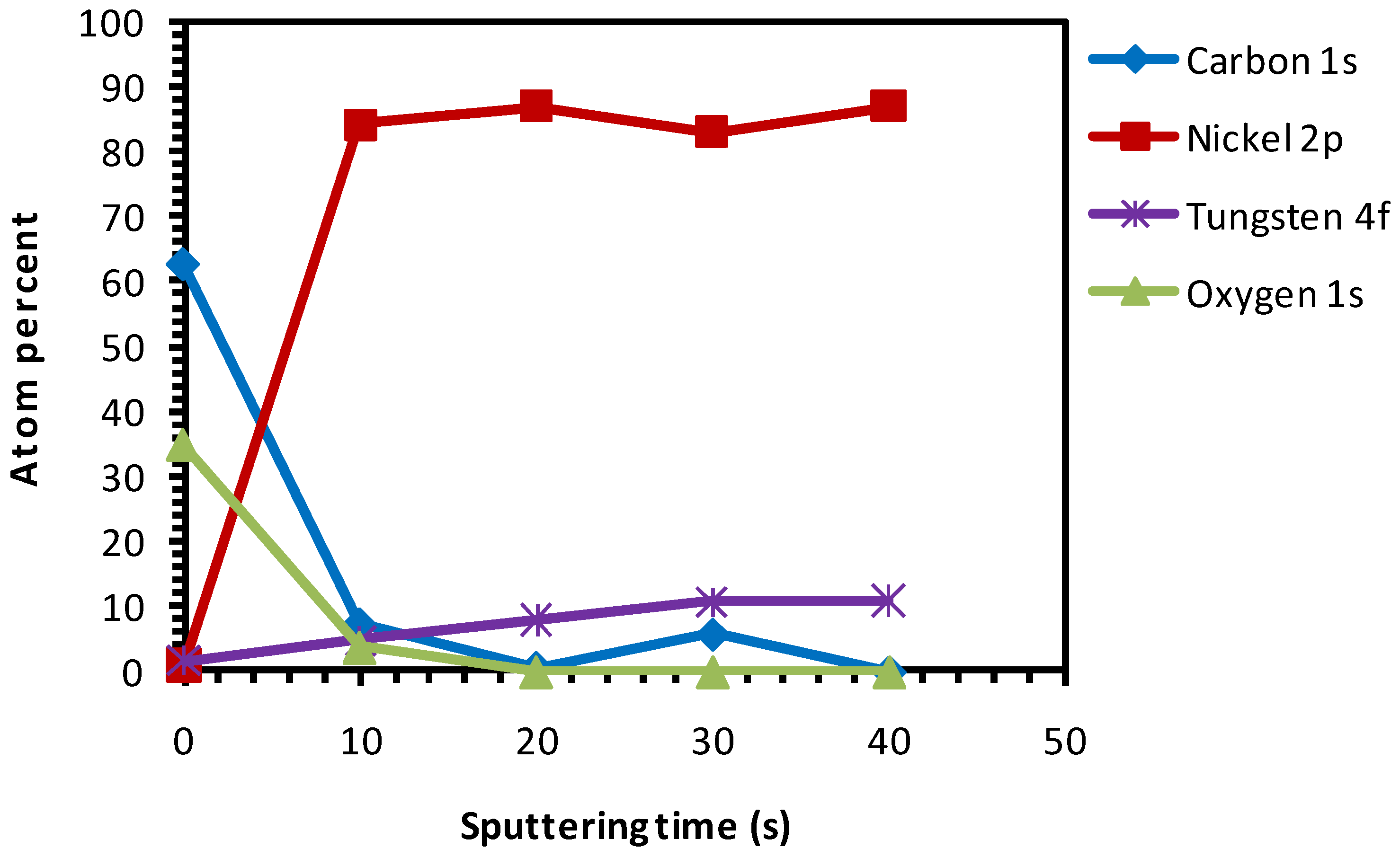

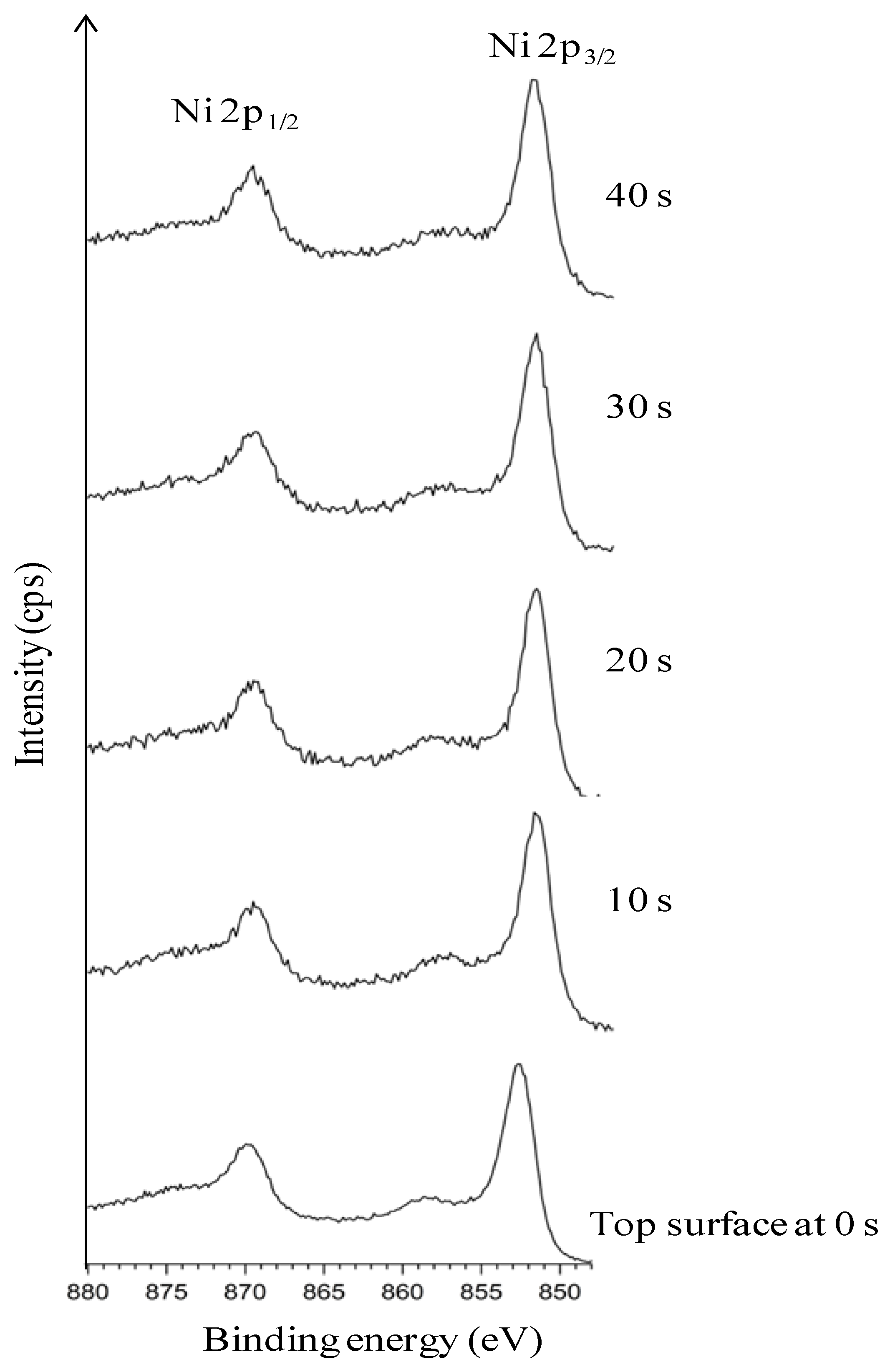
3.4. Thermal Cleaning Followed by Chemical Cleaning with Etching for 15 min
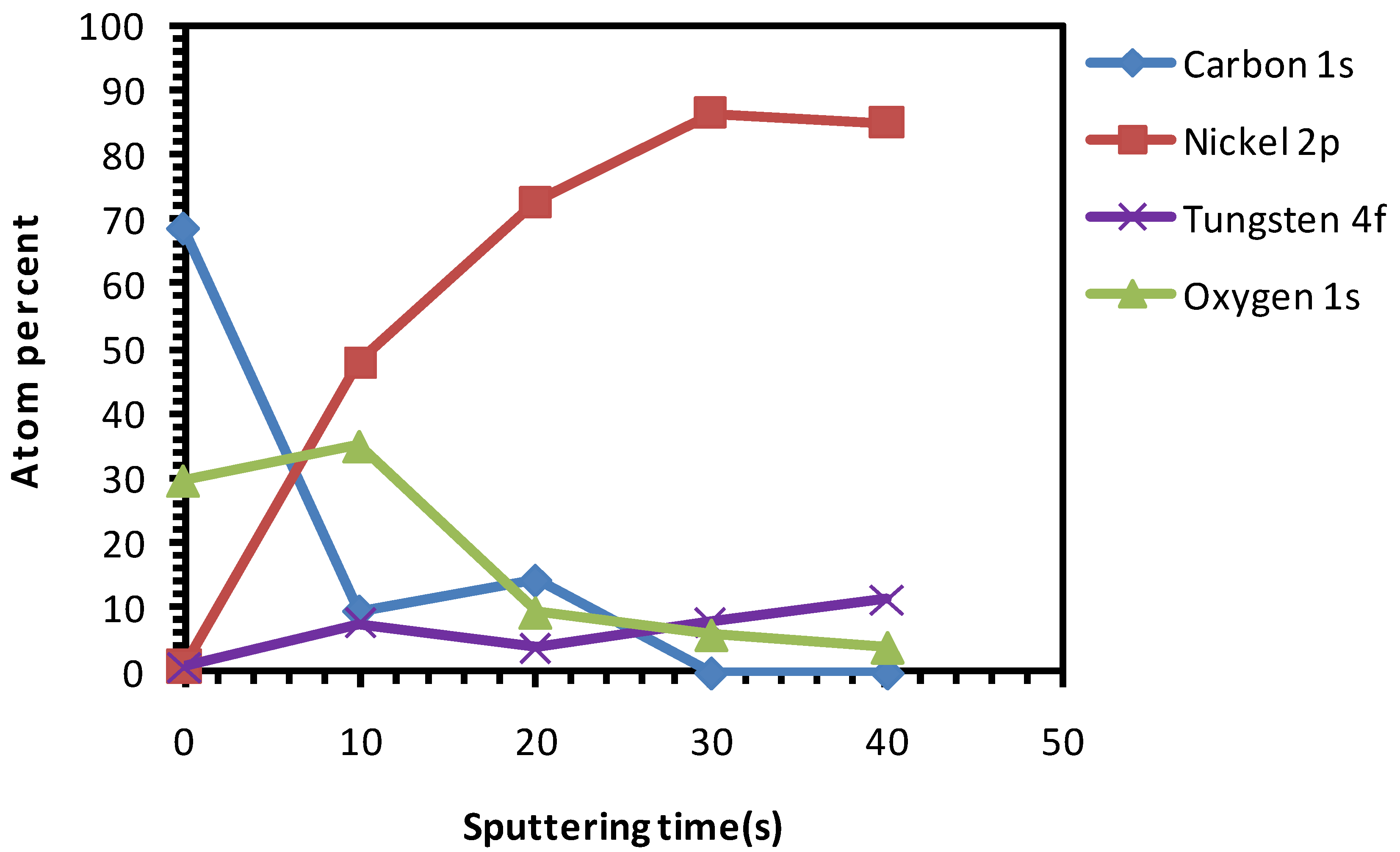
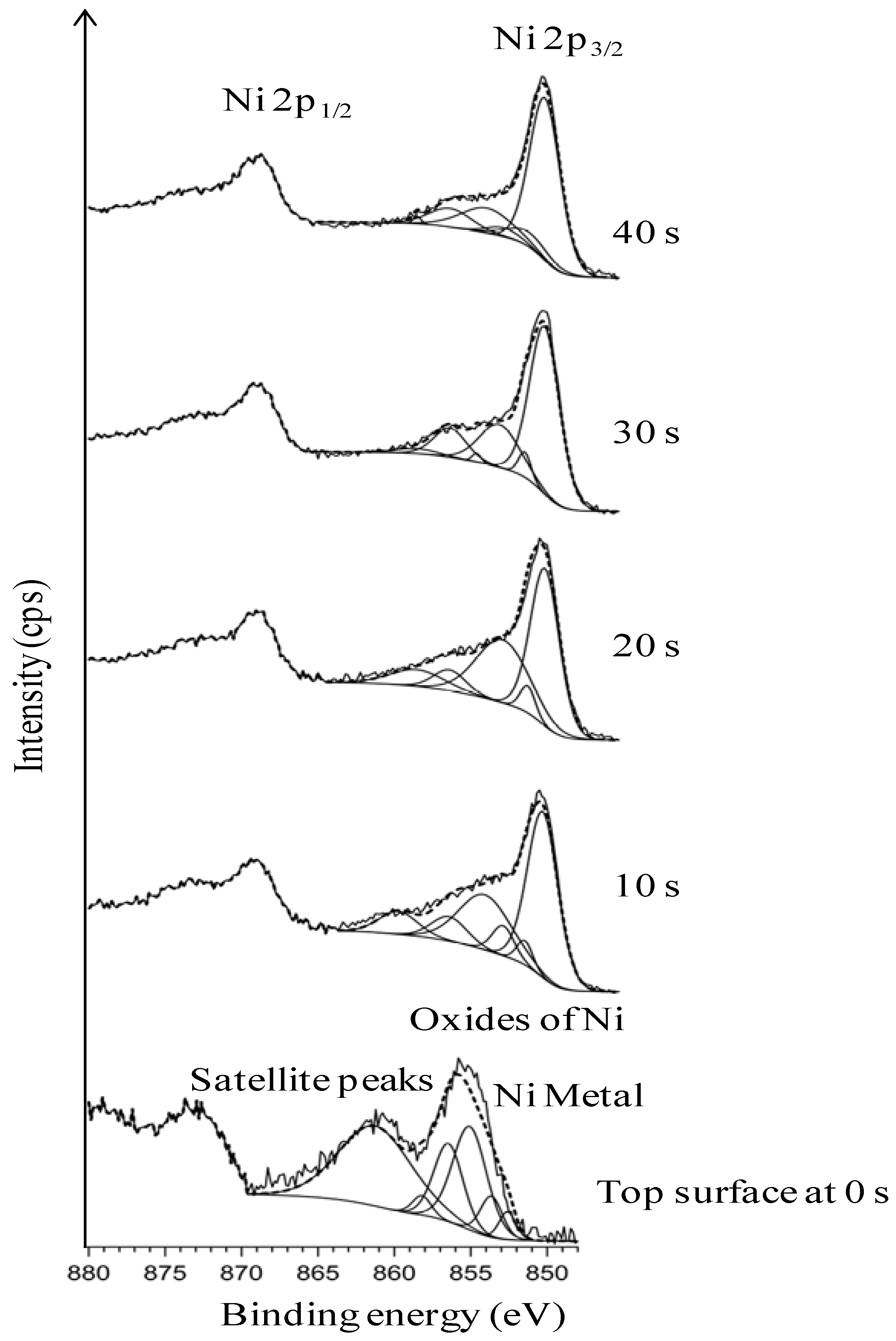
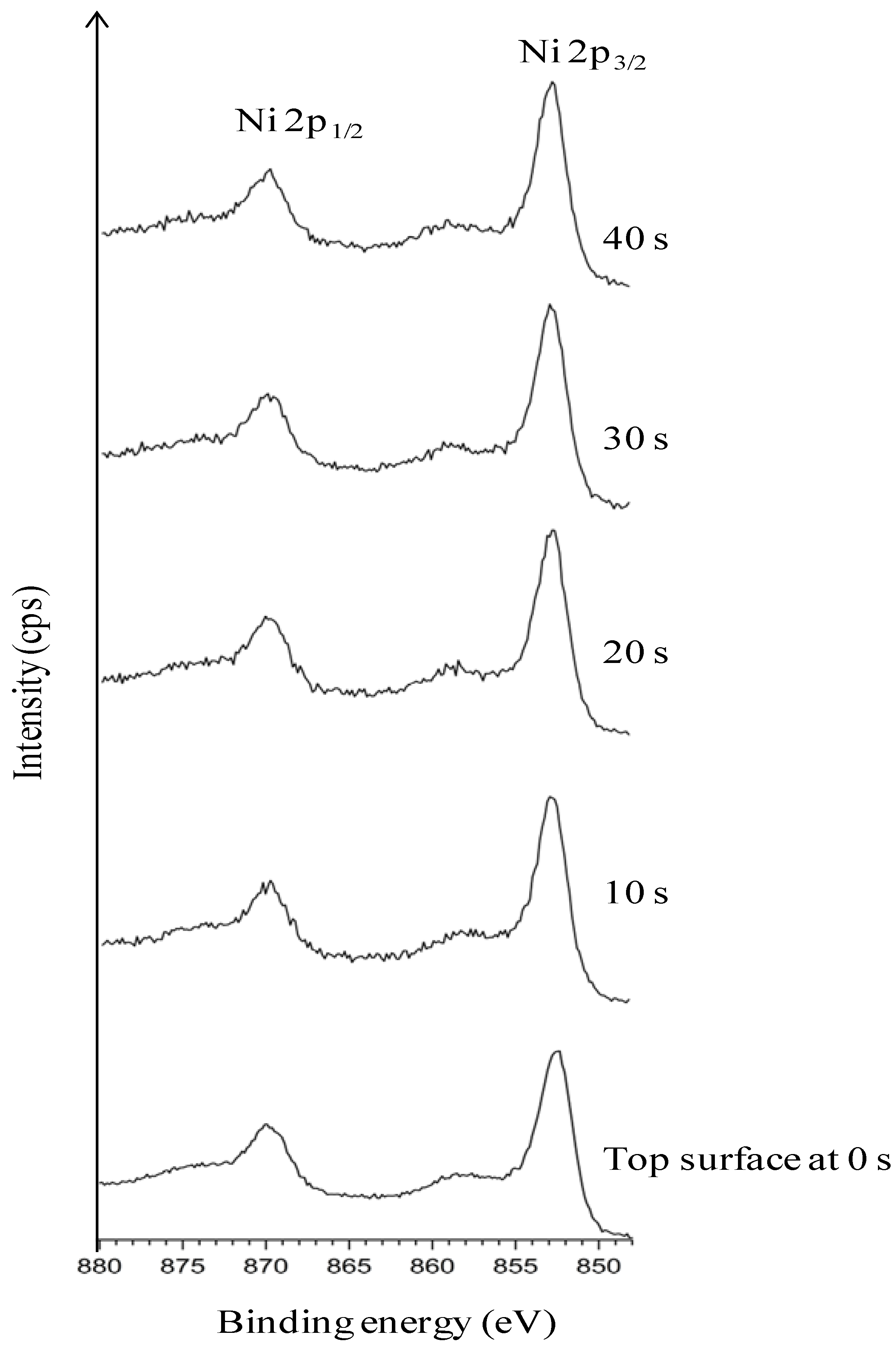
3.5. Chemical Cleaning with Etching for 15 min Followed by Thermal Cleaning
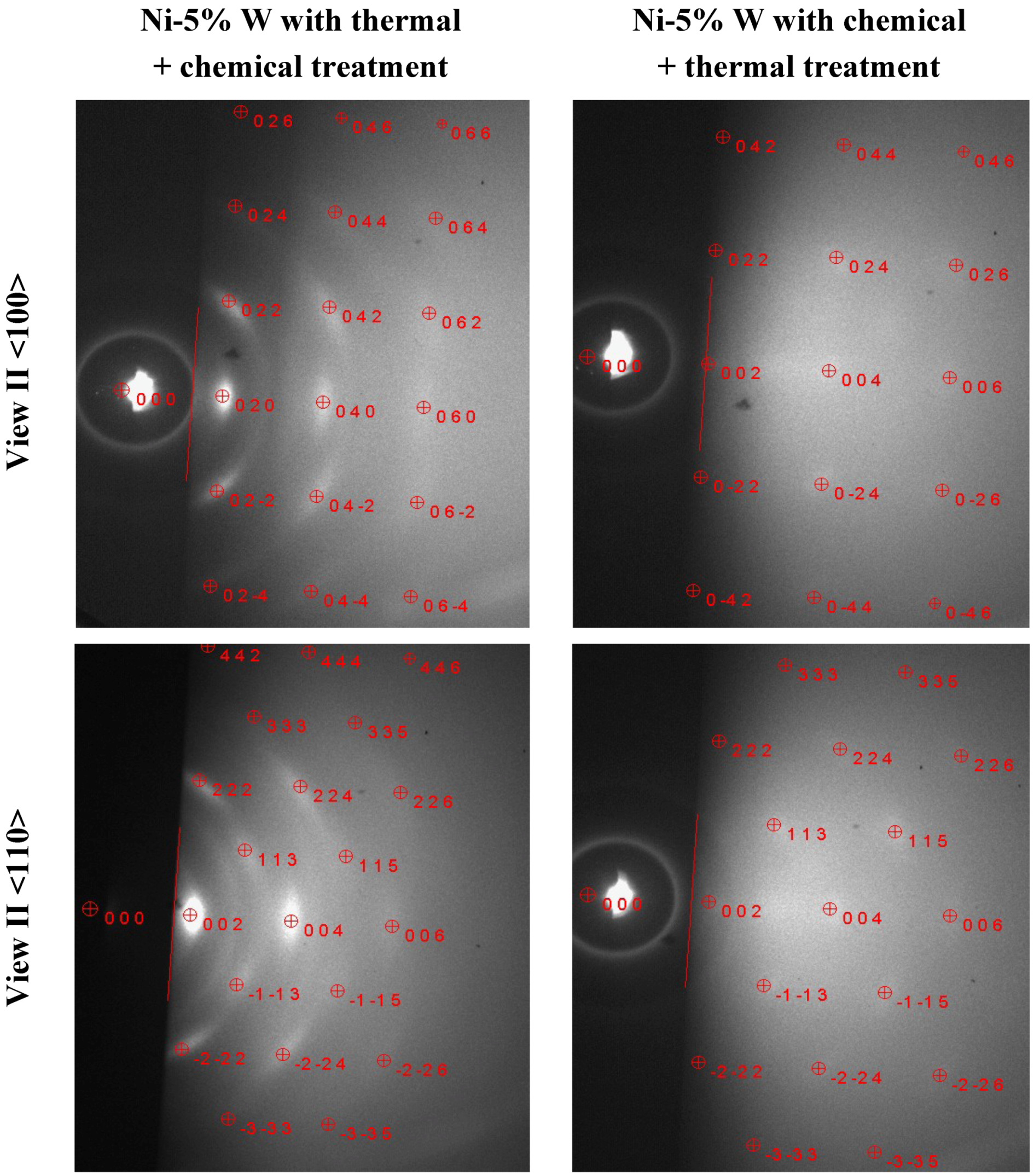
3.6. RHEED Analysis

4. Conclusions
Acknowledgments
References
- Goyal, A.; Norton, D.P.; Christen, D.K.; Specht, E.D.; Paranthaman, M.; Kroeger, D.M.; Budai, J.D.; He, Q.; List, F.A.; Feenstra, R.; Kerchner, H.R.; Lee, D.F.; Hatfield, E.; Martin, P.M.; Mathis, J.; Park, C. Epitaxial superconductors on rolling-assisted biaxially-textured substrates (RABiTS): A route towards high critical density wire. Appl. Supercond. 1996, 4, 403–427. [Google Scholar] [CrossRef]
- Chirayil, T.G.; Paranthaman, M.; Beach, D.B.; Lee, D.F.; Goyal, A.; Williams, R.K.; Cui, X.; Kroeger, D.M.; Feenstra, R.; Verebelyi, D.T.; Christen, D.K. Epitaxial growth of La2Zr2O7 thin films on rolled Ni-substrates by sol-gel process for high Tc superconducting tapes. Phys. C 2000, 336, 63–69. [Google Scholar] [CrossRef]
- Sathyamurthy, S.; Paranthaman, M.P.; Zhai, H.Y.; Kang, S.K.; Christen, H.M.; Cantoni, C.; Goyal, A.; Martin, P.M. Solution processing of lanthanum zirconate films as single buffer layers for high Ic YBCO coated conductors. IEEE Trans. Appl. Supercond. 2003, 13, 2658–2660. [Google Scholar] [CrossRef]
- Bhuiyan, M.S.; Paranthaman, M.; Salama, K. Solution-derived textured oxide thin films: A review. Supercond. Sci. Technol. 2006, 19, R1–R21. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, S.F.; Zhao, S.Q.; Zhou, Y.L. Epitaxial growth of CeO2/YSZ/CeO2 buffer layers on textured Ni substrates for YBCO conductors. J. Supercond. 2005, 18, 537–540. [Google Scholar] [CrossRef]
- Knoth, K.; Huhne, R.; Oswald, S.; Schultz, L.; Holzapfel, B. Detailed investigations on La2Zr2O7 buffer layers for YBCO-coated conductors prepared by chemical solution deposition. Acta Mater. 2007, 55, 517–529. [Google Scholar]
- Van de Velde, N.; van de Vyver, D.; Brunkahl, O.; Hoste, S.; Bruneel, E.; van Driessche, I. CeO2 buffer layers for HTSC via an aqueous sol-gel method: Chemistry and microstructure. Eur. J. Inorg. Chem. 2010, 2, 233–241. [Google Scholar]
- Van de Velde, N.; Bruggeman, T.; Stove, L.; Pollefeyt, G.; Brunkahl, O.; van Driessche, I. Influence of morphology and texture of CeO2 on YBCO growth and BaCeO3 formation in solution-derived synthesis. Eur. J. Inorg. Chem. 2012, 8, 1186–1194. [Google Scholar]
- Narayanan, V.; Lommens, P.; de Buysser, K.; Huhne, R.; van Driessche, I. Thick lanthanum zirconate buffer layers from water-based precursor solutions on Ni-5% W substrates. J. Solid State Chem. 2011, 184, 2887–2896. [Google Scholar]
- Cloet, V.; Feys, J.; Huhne, R.; Hoste, S.; van Driessche, I. Thin La2Zr2O7 films made from a water-based solution. J. Solid State Chem. 2009, 182, 37–42. [Google Scholar] [CrossRef]
- Penneman, G.; van Driessche, I.; Bruneel, E.; Hoste, S. Deposition of CeO2 buffer layers and YBa2Cu3O7−δ superconducting layers using an aqueous sol-gel method. Key Eng. Mater. 2004, 264-268, 501–504. [Google Scholar] [CrossRef]
- Van Driessche, I.; Penneman, G.; Abell, J.S.; Bruneel, E.; Hoste, S. Chemical approach to the deposition of textured CeO2 buffer layers based on sol gel dip coating. Mater. Sci. Forum 2003, 426, 3517–3522. [Google Scholar] [CrossRef]
- Narayanan, V.; Lommens, P.; de Buysser, K.; Hühne, R.; Vanpoucke, D.; Molina, L.; van Tendeloo, G.; van der Voort, P.; van Driessche, I. Aqueous CSD approach for the growth of lattice-tuned novel LaxCe1-xOδ epitaxial layers. J. Mater. Chem. 2012, 22, 8476–8483. [Google Scholar]
- Van Driessche, I.; Penneman, G.; Bruneel, E.; Hoste, S. Nonvacuum-based deposition techniques for superconducting ceramic coatings. Pure Appl. Chem. 2002, 74, 2101–2109. [Google Scholar] [CrossRef]
- Mouganie, T.; Moram, M.A.; Sumner, J.; Glowacki, B.A.; Schoofs, B.; van Driessche, I.; Hoste, S. Chemical and physical analysis of acetate-oxide sol-gel processing routes for the Y-Ba-Cu-O system. J. Sol Gel Sci. Technol. 2005, 36, 87–94. [Google Scholar] [CrossRef]
- Sathyamurthy, S.; Salama, K. Chemical cleaning treatment of textured nickel for the deposition of epitaxial thin films. Supercond. Sci. Technol. 2001, 14, 643–645. [Google Scholar] [CrossRef]
- Narayanan, V.; de Buysser, K.; Bruneel, E.; van Driessche, I. XPS depth profiling for evaluation of LZO buffer layer capacity. Materials 2012, 5, 364–376. [Google Scholar] [CrossRef] [Green Version]
- Biesinger, M.C.; Payne, B.P.; Lau, L.W.M.; Gerson, A.; Smart, R.St.C. X-ray photoelectron spectroscopic chemical state quantification of mixed nickel metal, oxide and hydroxide systems. Surf. Interface Anal. 2009, 41, 324–332. [Google Scholar] [CrossRef]
- Wörz, B.; Heinrich, A.; Stritzker, B. Epitaxial NiO buffer layer by chemical enhanced surface oxidation epitaxy on Ni-5% W RABiTSfor YBCO coated conductors. Phys. C 2005, 418, 107–120. [Google Scholar] [CrossRef]
- Kursumovic, A.; Hühne, R.; Tomov, R.; Holzapfel, B.; Glowacki, B.A.; Evetts, J.E. Investigation of the growth and stability of (1 0 0)[0 0 1] NiO films grown by thermal oxidation of textured(1 0 0)[0 0 1] Ni tapes for coated conductor applications during oxygen exposure from 700 to 1400 °C. Acta Mater. 2003, 51, 3759–3768. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Narayanan, V.; Bruneel, E.; Hühne, R.; Van Driessche, I. Effects of Varied Cleaning Methods on Ni-5% W Substrate for Dip-Coating of Water-based Buffer Layers: An X-ray Photoelectron Spectroscopy Study. Nanomaterials 2012, 2, 251-267. https://doi.org/10.3390/nano2030251
Narayanan V, Bruneel E, Hühne R, Van Driessche I. Effects of Varied Cleaning Methods on Ni-5% W Substrate for Dip-Coating of Water-based Buffer Layers: An X-ray Photoelectron Spectroscopy Study. Nanomaterials. 2012; 2(3):251-267. https://doi.org/10.3390/nano2030251
Chicago/Turabian StyleNarayanan, Vyshnavi, Els Bruneel, Ruben Hühne, and Isabel Van Driessche. 2012. "Effects of Varied Cleaning Methods on Ni-5% W Substrate for Dip-Coating of Water-based Buffer Layers: An X-ray Photoelectron Spectroscopy Study" Nanomaterials 2, no. 3: 251-267. https://doi.org/10.3390/nano2030251