1. Introduction
Hemoglobin-based oxygen carriers have been studied for many years as potential therapeutics [
1,
2,
3,
4,
5]. The hemoglobins required to produce candidate therapeutics have been obtained from outdated human blood, transgenic and other animal sources, and bacterial or yeast fermentation. Solutions of unmodified cell-free hemoglobin (Hb) exhibit renal toxicity due to dissociation of α
2β
2 tetramers into αβ dimers (which are small enough to be filtered in the kidneys). This toxicity can be prevented by covalent linking of the globin subunits using a variety of chemical crosslinking agents, or by gene fusion (reviewed in [
1,
2,
3,
4,
5]). Production of hemoglobin by recombinant DNA technology provides the ability to selectively alter the hemoglobin structure and function by genetic manipulation, and many point mutations which affect hemoglobin function have been identified and characterized [
6,
7]. For example, replacement of Asn108 with Lys in the β globins and covalent linkage of the two α globins in the human α
2β
2 tetramer by insertion of a single Gly codon between two α genes yields a hemoglobin, rHb1.1, which binds oxygen in a cooperative manner with a P
50 close to that of human blood [
8]. This example demonstrates that a relatively simple structural alteration (
i.e., linking the α globins) can yield a protein with improved
in vivo function.
Preclinical and clinical experiences with solutions of hemoglobin-based oxygen carriers (HBOCs) have identified several other severe side-effects, such as pulmonary and systemic vasoactivity, myocardial lesions, elevation of serum enzyme levels, and complement activation [
2,
3,
9]. Many of these side-effects are hypothesized to result from scavenging of nitric oxide (NO) by oxy-Hb following extravasation of the cell-free Hb into extravascular spaces [
9,
10]. NO scavenging activity can be modulated by site-directed mutagenesis of the heme binding pocket, and there is a direct correlation between the NO scavenging rates of the resulting mutant rHbs and vasoactivity in animal models [
11].
Another approach to the amelioration of increased vasoactivity is to reduce extravasation of the cell-free Hb by increasing its size. Crosslinked polymeric Hbs exhibit reduced extravasation rates and appear to elicit an attenuated pressor response when administered to conscious rats and cats [
12,
13,
14]. Thus, it appears that polymerized Hbs may have improved hemodynamic properties compared to the parent 64 kDa molecule. It is not clear whether an increase in hydrodynamic radius/molecular weight
per se, or chemical modification of certain surface residues in Hb is responsible for this observed reduction in vasoactivity, since a 64 kDa rHb1.1 that was modified by treatment with glutaraldehyde (but not polymerized) also showed reduced vasoactivity in rats [
12]. Nevertheless, polymeric Hbs are attractive targets in blood replacement therapy because they could be formulated as oxygen carrying therapeutics with equivalent oxygen binding capacity at lower oncotic pressures compared to a 64 kDa cell-free hemoglobin.
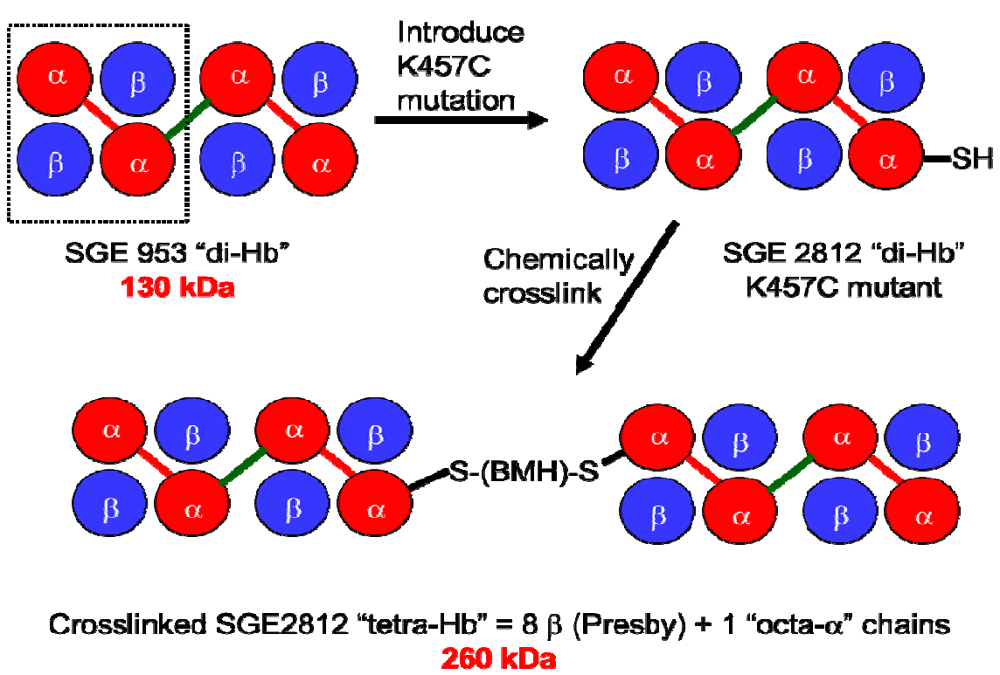
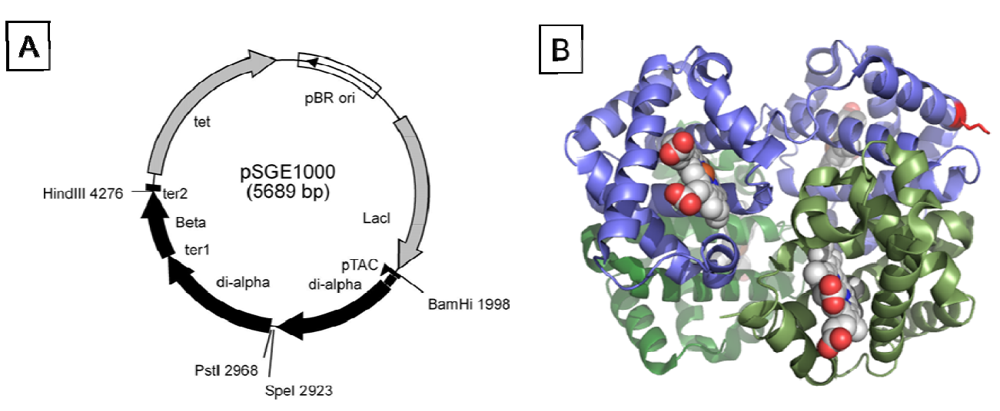
To address directly the effect of increased molecular size on the vasoactivity of rHb1.1, and to assess the feasibility of producing polymeric Hbs with defined oligomerization states, we produced a ~130 kDa “dihemoglobin”. “Dihemoglobin” as used herein refers to an oligomeric hemoglobin composed of four covalently linked α globin subunits which are noncovalently associated with four β globin subunits to yield a monodisperse 130 kDa protein complex (
Figure 1).
Figure 1.
Covalent crosslinking of diα-globins (red) by gene fusion using a peptide linker (green) yields dihemoglobin (“di-Hb”). Introduction of a Cys residue into di-Hb, followed by chemical crosslinking with bismaleimidohexane (BMH), yields “tetra-Hb”. The subunit composition of rHb1.1 is shown within the dashed-line rectangle.
Figure 1.
Covalent crosslinking of diα-globins (red) by gene fusion using a peptide linker (green) yields dihemoglobin (“di-Hb”). Introduction of a Cys residue into di-Hb, followed by chemical crosslinking with bismaleimidohexane (BMH), yields “tetra-Hb”. The subunit composition of rHb1.1 is shown within the dashed-line rectangle.
Given previous success with genetic fusion of α-globins [
8], we pursued a strategy of generating tandem repeats of diα-globins as the basis for polymerization of the 64 kDa rHb1.1 molecules. A simple computer model suggested that a minimum of seven amino acids in an extended conformation would be required to link two rHb1.1 diα-globins without distortion of the terminal structures or overlap of the van der Waals surfaces .
A priori a longer flexible linker might be best for minimizing effects on function (by imposing fewer steric constraints on the movement of the globin subunits during R- to T-state transitions), whereas a shorter linker might be better protected from proteolysis if the linker sequence proved to be a substrate for bacterial (or serum) proteases. Accordingly, dihemoglobins with 7aa (SGE 946) and 16aa (SGE 953) linkers were constructed to determine the effect of linker length on protein function and recovery.
The use of peptide linkers to create functional fusions of identical [
4,
15,
16,
17] or different [
18,
19,
20,
21] protein domains is well known, and a survey of linker regions listed in the Brookhaven protein database suggests that Thr, Ser, Gly and Ala are the most desirable constituent amino acids for a linker [
22]. Thus, we opted for flexible linkers composed of Ser and Gly residues. One potential drawback to this simple approach is that larger polymers or aggregates might form as the result of misfolding of the tetra-α globin. At the outset of this study it was not known whether α globin sequences with extensions at
both termini would fold correctly, or associate properly with the correct number of β-globins to yield a fully functional polymeric Hb.
To demonstrate that a 130 kDa polymeric hemoglobin with functional properties suitable for an oxygen carrying therapeutic can be produced solely by recombinant technology, we have produced and characterized two dihemoglobins, which differ only in the length of the linker between the second and third α globin subunits. To explore further the effects of increased molecular size on the vasoactivity of hemoglobin solutions, we also produced a Cys-containing dihemoglobin that can be chemically crosslinked to produce a monodisperse “tetrahemoglobin” of ~260 kDa (
Figure 1). Both dihemoglobin and tetrahemoglobin show reduced vasoactivity compared to rHb1.1 in conscious rats.
3. Results and Discussion
Polymeric hemoglobins have potential advantages over 64 kDa hemoglobins as cell-free injectable therapeutics because larger MW hemoglobins could be formulated as oxygen carrying therapeutics with equivalent (or greater) oxygen binding capacities at lower oncotic pressures compared to a 64 kDa cell-free hemoglobin [
1,
3]. Initially, Somatogen approached the generation of a polymeric hemoglobin solution via chemical crosslinking of rHb1.1 with glutaraldehyde [
12]. This was undertaken to determine the effects of increased molecular size on vasoactivity, as others had reported minimal vasoactivity with crosslinked Hbs [
28]. Solutions of chemically crosslinked Hbs contain multiple polymer species (see
Figure 3). To better define the relationship between polymer size and vasoactivity, we sought to take advantage of the power of recombinant DNA technology to generate polymeric rHbs of defined molecular weight.
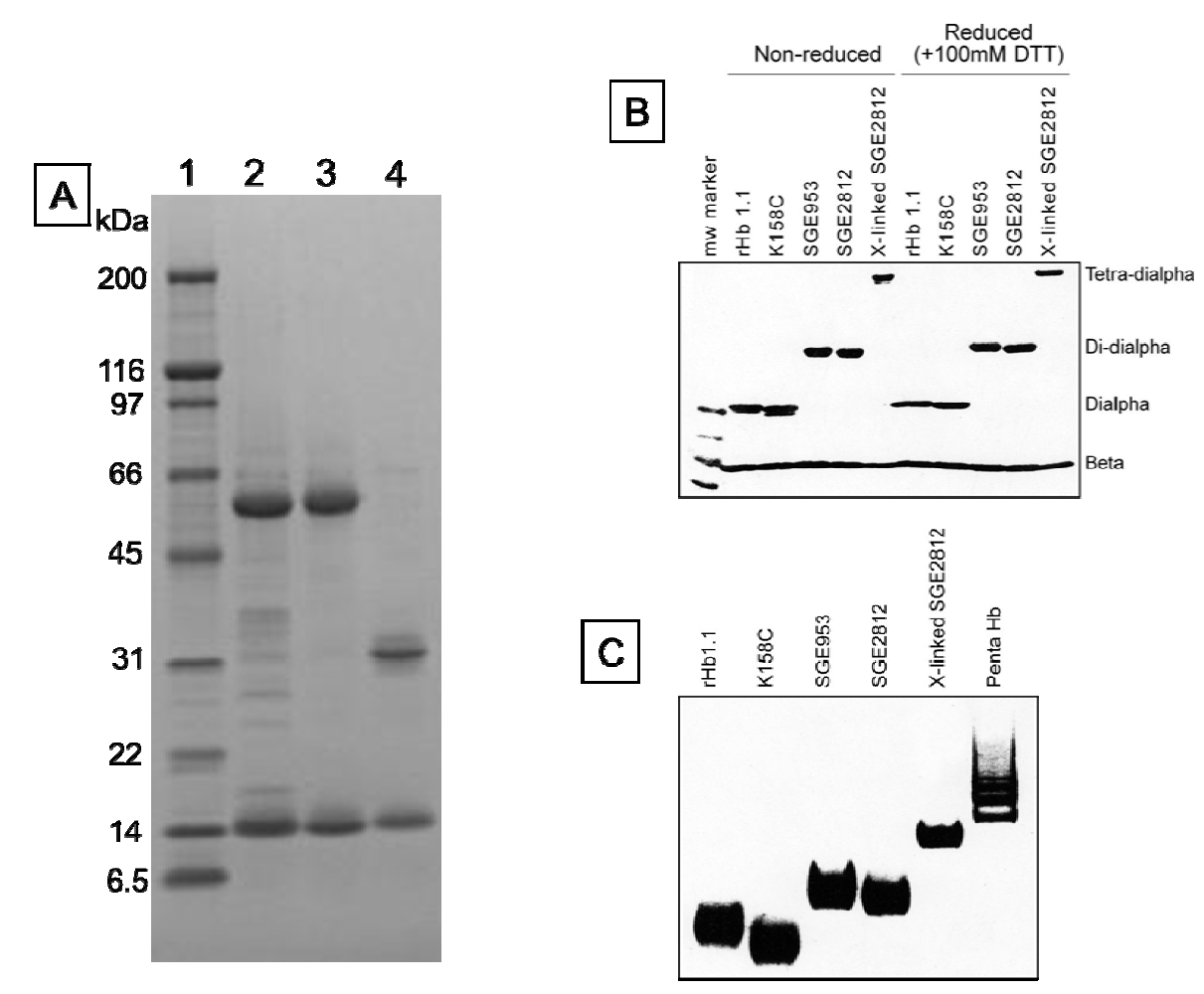
Figure 3.
Tris-glycine gels (4%–20%) stained with coomassie brilliant blue. (a) SDS-PAGE of SGE 953 and rHb1.1. Lane 1, MW markers. MWs in kDa are indicated. Lane 2, partially purified dihemoglobin from SGE 953. This material is the collected fraction from the first chromatography step. Lane 3, fully purified dihemoglobin from SGE 953. Lane 4, purified rHb1.1; (b) SDS-PAGE of rHb1.1, rHb1.1 K158C, SGE 953 and SGE 2812 before and after chemical crosslinking; (c) Native PAGE of rHb1.1, rHb1.1 K158C, SGE 953, SGE 2812 before and after chemical crosslinking, and a glutaraldehyde-crosslinked “penta-Hb”.
Figure 3.
Tris-glycine gels (4%–20%) stained with coomassie brilliant blue. (a) SDS-PAGE of SGE 953 and rHb1.1. Lane 1, MW markers. MWs in kDa are indicated. Lane 2, partially purified dihemoglobin from SGE 953. This material is the collected fraction from the first chromatography step. Lane 3, fully purified dihemoglobin from SGE 953. Lane 4, purified rHb1.1; (b) SDS-PAGE of rHb1.1, rHb1.1 K158C, SGE 953 and SGE 2812 before and after chemical crosslinking; (c) Native PAGE of rHb1.1, rHb1.1 K158C, SGE 953, SGE 2812 before and after chemical crosslinking, and a glutaraldehyde-crosslinked “penta-Hb”.
Genes for three different dihemoglobins have been constructed and cloned into low and high copy expression vectors (
Figure 1 and
Figure 2). The design of the expression vector allows for facile insertion of a linker of any desired amino acid sequence between the unique
SpeI and
PstI restriction sites flanking the fusion region. At the end of fermentation there is no evidence from restriction mapping of other plasmid species which might arise from the deletion of α globin sequences (data not shown), thus the plasmid appears to be stable over many generations.
We were concerned that the approach to the generation of larger hemoglobins described herein would yield unstable and/or misfolded proteins. Dihemoglobin is expressed at high levels in
E. coli and purification of dihemoglobins from bacterial lysate to greater than 95% homogeneity (
Figure 3) is readily achieved using the three column process described in
Experimental Procedures. After the final purification step the major contaminants are monohemoglobin species (
Figure 4B).
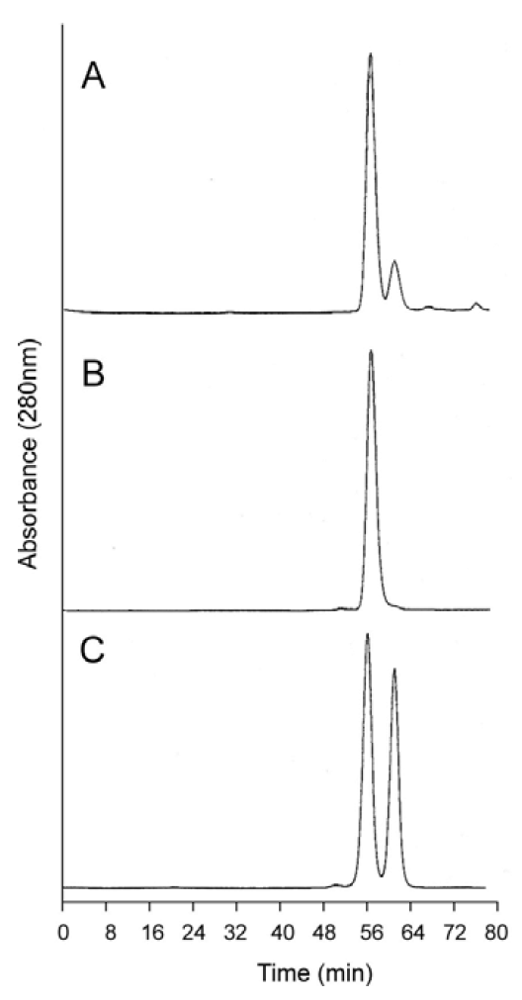
Figure 4A shows an analytical SEC chromatogram of the dihemoglobin protein recovered from a fermentation of expression strain SGE 946 after the first column purification. At this stage the protein mixture is 82% dihemoglobin and 18% monohemoglobin. For all the dihemoglobin preparations described in this report, 10%–20% of the globins recovered after the first column purification appear to be monohemoglobin species. Mass spectral and sequencing analyses of the monohemoglobin fraction indicate that it is heterogeneous and suggest a non-specific degradation of the dihemoglobin molecule. Misfolded proteins are likely to be degraded, however, correctly folded dihemoglobins may also be substrates for degradative processes. It is possible that the presence of the peptide linking the diα globins causes local destabilization of the hemoglobin in the fusion region, thereby increasing the susceptibility of this region to proteolysis.
Figure 4.
Analytical size exclusion chromatography of dihemoglobin SGE 953. (
a) Partially purified SGE 953. This material is the collected fraction from the first chromatography step (see lane 2 of
Figure 3A); (
b) Purified SGE 953; (
c) Coinjection of purified SGE 953 and purified rHb1.1.
Figure 4.
Analytical size exclusion chromatography of dihemoglobin SGE 953. (
a) Partially purified SGE 953. This material is the collected fraction from the first chromatography step (see lane 2 of
Figure 3A); (
b) Purified SGE 953; (
c) Coinjection of purified SGE 953 and purified rHb1.1.
The proper assembly of the desired tetra-α globin and four β-globin chains was confirmed by SDS-PAGE (
Figure 3) and mass spectral analysis of the isolated globin chains. Integration of the area under the peaks of analytical reversed phase HPLC chromatograms indicates that the tetra-α and β globins are present in the expected stoichiometry of 1:4 (data not shown).
Analytical SEC shows good separation between rHb1.1 and the faster migrating dihemoglobin when the two proteins are co-injected (
Figure 4C). The apparent molecular weights of the purified dihemoglobins were estimated from their retention times on analytical SEC columns that had been calibrated with protein MW standards (
Table 1). These analyses yielded MWs for the dihemoglobins that are twice that for rHb1.1. Dynamic light scattering (
Table 1) also shows that particle sizes of the hemoglobins increase going from rHb1.1 (64 kDa) to SGE 946 or 953 (130 kDa) to SGE 2812 (260 kDa).
Table 1.
Apparent molecular weights and particle sizes of dihemoglobins.
Table 1.
Apparent molecular weights and particle sizes of dihemoglobins.
| Hemoglobin | Calculated MW (kDa) | Estimated MW from SEC (kDa) | Observed MW from sedimentation (kDa) | Observed diameter from light scattering (nm) |
|---|
| rHb1.1 | 64.6 | 52.5 | 63–64 | 6.0 |
| SGE 946 | 130 | 102 | 126–128 | 10.4 |
| SGE 953 | 130 | 102 | 127–129 | 9.7 |
| SGE 2812 | 260 | 173 | n.d. | 12.4 |
A quantitative determination of MW was made by analytical ultracentrifugation. Observed values of MW obtained from curve fitting are in excellent agreement with expected MWs for the three dihemoglobins and rHb1.1 (
Table 1). By varying the concentration of the protein and analyzing the signal at various speeds, the individual curves could be combined and fit globally, thus reducing the influence of either concentration or rotor speed. The curve fitting equation included a term for non-ideality that arises due to the finite size of the protein and its associated charge; both can lead to an apparent decrease in MW with increasing concentration [
29]. The samples displayed homogeneity, as no increase in the molecular weight from the meniscus position to the bottom of the cell was detected and plots of ln(conc)
vs. r
2 did not exhibit upward curvature.
The suitability of dihemoglobin to function as an O
2 delivery platform was assessed by measuring equilibrium oxygen binding, oxygen content, and the stoichiometry of ligand binding. Dihemoglobin displays equilibrium oxygen binding behavior similar to rHb1.1 (
Table 2), with observed values of P
50 and n
max that are suitable for an oxygen-delivering therapeutic. The dihemoglobins showed a modest reduction in the values of P
50 and n
max relative to rHb1.1. The observed values of these parameters indicate that dihemoglobin could function as an efficacious oxygen delivery molecule. Theoretical fractional saturation of hemoglobin is calculated using the following equation:
where
![Jfb 03 00061 i002]()
is fractional saturation,
![Jfb 03 00061 i003]()
is the partial pressure of oxygen,
P50 is the
![Jfb 03 00061 i003]()
required to achieve 50% saturation of the ligand binding sites, and n is the Hill coefficient. Given the observed values of these constants for the dihemoglobins (
Table 2) and assuming an oxygen partial pressure of 90 torr in the lungs and 40 torr in the capillary beds, these molecules could theoretically release 21% of bound oxygen compared to a theoretical 30% release of oxygen from whole blood.
Table 2.
Oxygen binding and NO scavenging rates for dihemoglobins.
Table 2.
Oxygen binding and NO scavenging rates for dihemoglobins.
| | rHb1.1 | SGE 946 | SGE 953 |
|---|
| P50 (torr) | 31 | 24 | 25 |
| nmax | 2.2 | 2.0 | 2.1 |
| k’NO (μM−1s−1) | 71 | 71 | 66 |
The observed reductions in P
50 and n
max may reflect stabilization of the R state conformation that results from the linking of the two hemoglobins. Marked reductions of P
50 and n
max have been reported for natural [
30] and directed mutants [
31] of the penultimate Tyr in α globin (Tyr140-α). This residue is thought to stabilize the T-state structure and promote tetramer assembly [
31]. The linker may alter the conformation of this residue in dihemoglobin and thereby alter the equilibrium oxygen binding properties of the dihemoglobin relative to rHb1.1. The covalent linkage of the tetramers may also result in inter-tetramer interactions that perturb the T-R equilibrium in favor of the R-state.
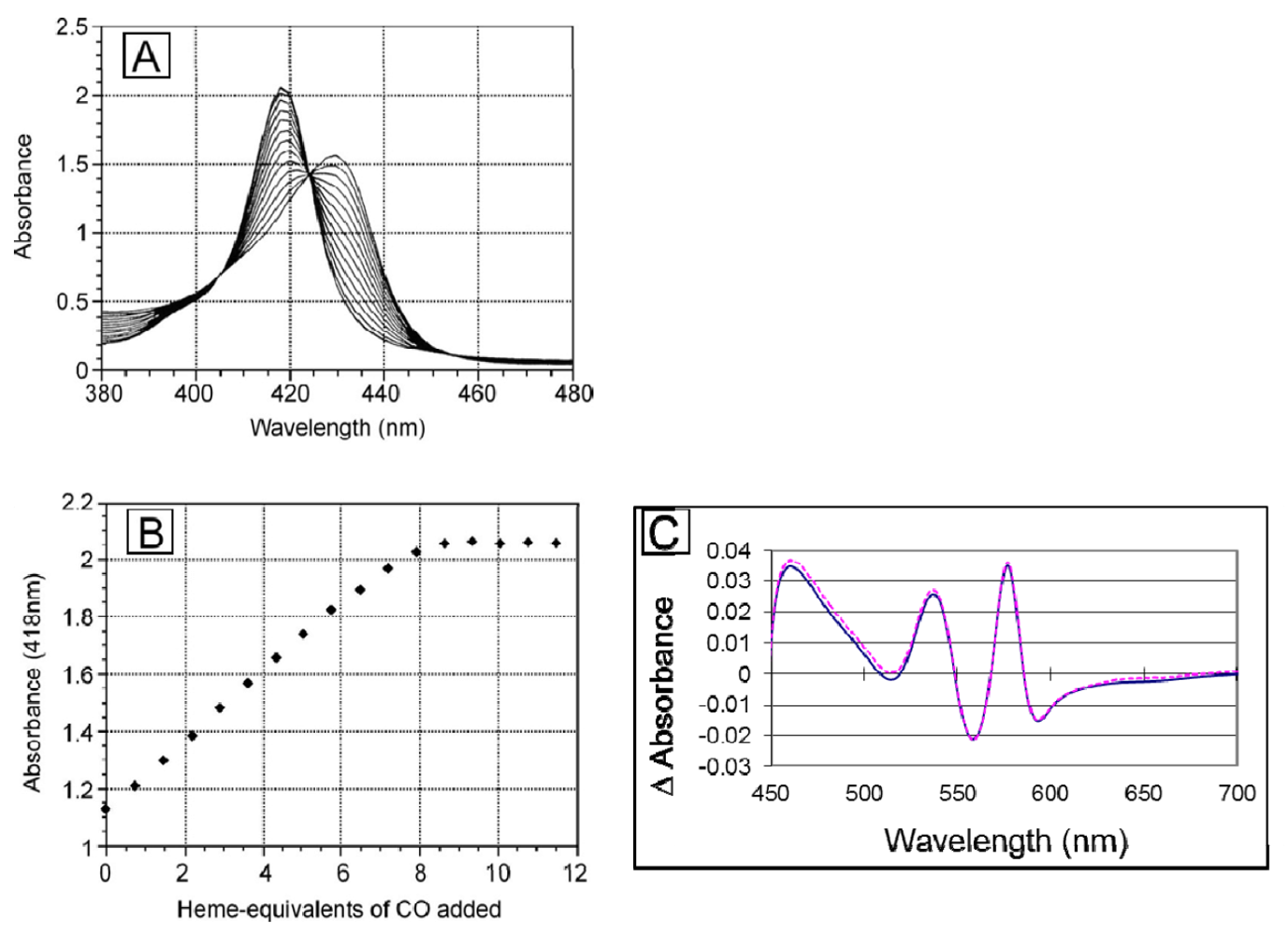
To ascertain that the linker does not render any of the ligand binding sites non-functional, we titrated the deoxy form of the dihemoglobin with CO-saturated buffer (
Figure 5). Dihemoglobin has 7.9 (of 8 expected) CO binding sites (
Figure 5), and rHb1.1 has 3.7 (of 4 expected) CO binding sites (data not shown). When the spectra acquired during the titration are overlaid (
Figure 5A), three isosbestic points emerge, indicating that there are not multiple absorbing species in solution and that the heme environment is similar at all ligand binding sites in the dihemoglobin molecule. In addition, the O
2 content of dihemoglobin and tetrahemoglobin prepared for preclinical studies was measured as described in
Experimental Procedures and shown to be 95% of the expected value (data not shown). Finally, the oxy-deoxy difference spectra for rHb1.1 and dihemoglobin (
Figure 5C) are essentially identical, indicating no significant perturbation of the heme binding pocket in dihemoglobin compared to rHb1.1. These data taken together support the conclusion that covalent linkage of the hemoglobins has not hampered ligand binding at any of the eight heme centers in the dihemoglobin molecule.
Figure 5.
Ligand binding to SGE 953. (a) The stoichiometry of ligand binding was determined by titrating deoxy dihemoglobin with CO-saturated buffer at 20 °C. An overlay of the spectra recorded after each CO addition during the titration is shown; (b) The data from panel (a) are plotted at 418 nm; (c) The (oxy di-Hb)-(deoxy di-Hb) difference spectra for SGE 953 (magenta, dashed line) and rHb1.1 (blue, solid line).
Figure 5.
Ligand binding to SGE 953. (a) The stoichiometry of ligand binding was determined by titrating deoxy dihemoglobin with CO-saturated buffer at 20 °C. An overlay of the spectra recorded after each CO addition during the titration is shown; (b) The data from panel (a) are plotted at 418 nm; (c) The (oxy di-Hb)-(deoxy di-Hb) difference spectra for SGE 953 (magenta, dashed line) and rHb1.1 (blue, solid line).
Given the encouraging
in vitro characteristics of dihemoglobin we undertook further studies to compare the
in vivo properties of these novel hemoglobin polymers to the well-characterized recombinant hemoglobin rHb1.1. Administration of hemoglobin solutions has resulted in undesired vasoactivity in humans [
2,
3,
4]. Two different mechanisms have been proposed to account for this vasoactivity. One attributes the effects to increased O
2 concentrations in non-capillary vasculature, via facilitated diffusion of O
2 by cell-free hemoglobin (see [
32] and references therein). The other proposes that oxy-Hb extravasation and the subsequent scavenging of NO by oxy-Hb is responsible for the observed effects [
10]. Polymeric hemoglobins have been reported to possess increased retention in circulation [
13,
14], and we hypothesized that a larger molecule, such as dihemoglobin, would extravasate to a lesser extent than rHb1.1. Attenuated extravasation would presumably reduce or eliminate NO scavenging by oxy-Hb and thereby lead to a reduced pressor response. The rates of NO scavenging of oxydihemoglobin and oxy-rHb1.1 are identical (
Table 2), thus the effect of apparent MW on the reduction of the pressor response
in vivo can be directly compared for rHb1.1 and dihemoglobin.
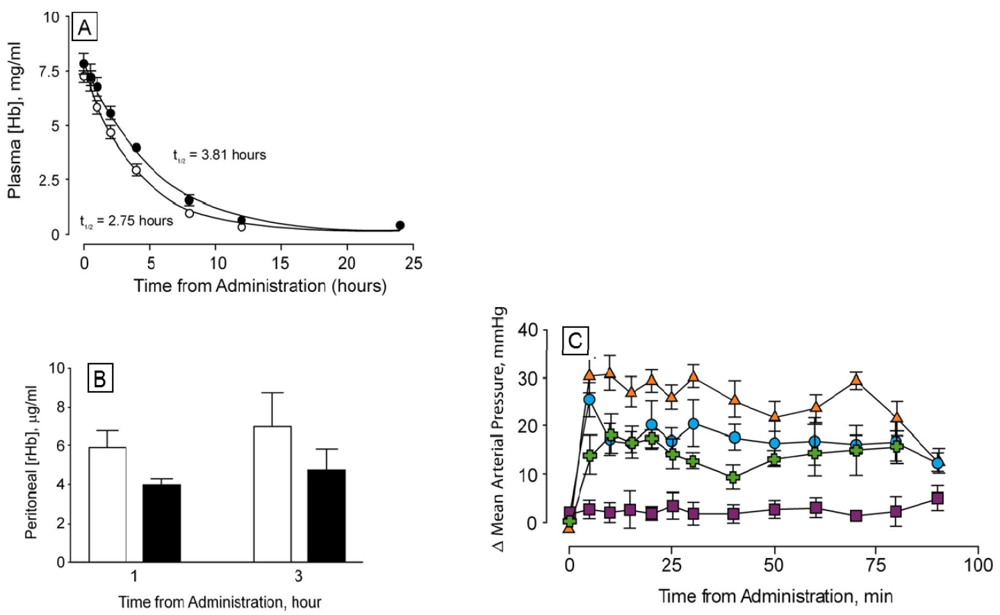
Pharmacokinetic measurements show that the circulating halflife of dihemoglobin is greater than that of rHb1.1 by a factor of 1.4. When administered at a dose of 350 mg/kg, rHb1.1 has a halflife of 2.75 h whereas the halflife of dihemoglobin from SGE 953 is 3.81 h (
Figure 6A). Dihemoglobin also extravasates to a lesser degree than rHb1.1 (
Figure 6B). Peritoneal lavage after hemoglobin administration in mice showed approximately 30% less dihemoglobin had entered the peritoneal cavity compared to rHb1.1. The difference between rHb1.1 and dihemoglobin was statistically significant at the 1 h time point (p < 0.05). In addition to increased retention in the vasculature, dihemoglobin does not raise mean arterial pressure (MAP) to the same extent as rHb1.1 when administered to conscious rats (
Figure 6C). The difference between rHb1.1 and dihemoglobin was statistically significant (p < 0.05, repeated measures ANOVA). Data obtained in experiments with a related dihemoglobin show that most of the MAP elevation can be attributed to an increase in cardiac output rather than peripheral vasoconstriction (M. Doyle, unpublished). Taken together, these data support the hypothesis that increased vascular retention of hemoglobin can minimize the observed pressor response, and that even a relatively modest two-fold increase in MW can increase vascular retention.
To explore further the effect of increased MW on vasoactivity, we produced a 240 kDa tetrahemoglobin by chemical crosslinking of a dihemoglobin containing a single reactive Cys residue in the C-terminal α-globin of the tetra-α-globin chain (
Figure 1). Lysine 16 (wild-type Hb numbering) is known to react with chemical modifying agents such as glutaraldehyde (12) and is solvent accessible in the rHb1.1 structure (
Figure 2B). We mutated this site (amino acid residue 457 in SGE 953) to produce a di-Hb with a uniquely reactive site for chemical crosslinking under deoxy conditions (under oxy conditions β-Cys93 is also reactive toward maleimide reagents). This combined site-directed mutagenesis and chemical crosslinking approach allowed us to produce a monodisperse tetra-Hb (
Figure 7B). A preparation of glutaraldehyde linked “penta-Hb” contains significant concentrations of tetra-, hexa- and septa-Hb polymers in addition to the predominant penta-Hb (see
Figure 3C). In contrast, SGE 2812 is composed of a single polymeric species (
Figure 3B,C).
The tetrahemoglobin produced in this fashion shows reduced elevation in MAP compared to rHb1.1, but the observed change is not significantly different from that seen for dihemoglobin (
Figure 6C). This observation is consistent with a recent report that glutaraldehyde crosslinked poly-Hb solutions containing oligomers of <500 kDa exhibited significant vasoactivity, whereas those containing oligomers of >500 kDa showed minimal vasoactivity in preclinical testing [
33].
Figure 6.
Determination of circulating halflife and hemodynamic response of di-Hb in vivo (a) Circulating halflife was determined in male Sprague-Dawley rats. Top-load doses of protein were administered via intravenous infusion to six rats each in experimental (SGE 953, filled circles) and control (rHb1.1, open circles) groups. Time-dependent curves of plasma hemoglobin concentrations are shown; (b) Hemoglobin concentrations in peritoneal lavage fluid at two time points following administration of rHb1.1 (open bars) or dihemoglobin from SGE 953 (filled bars) to separate groups of Balb/C mice; (c) Changes in mean arterial pressure following administration of hemoglobins to conscious, unrestrained rats. Top-load doses of rHb1.1 (filled triangles, orange), SGE 953 di-Hb (filled circles, blue), crosslinked SGE 2812 tetra-Hb (filled crosses, green), or HSA (50 mg/mL, filled squares, purple) were administered to separate groups of rats (n = 6 in all groups).
Figure 6.
Determination of circulating halflife and hemodynamic response of di-Hb in vivo (a) Circulating halflife was determined in male Sprague-Dawley rats. Top-load doses of protein were administered via intravenous infusion to six rats each in experimental (SGE 953, filled circles) and control (rHb1.1, open circles) groups. Time-dependent curves of plasma hemoglobin concentrations are shown; (b) Hemoglobin concentrations in peritoneal lavage fluid at two time points following administration of rHb1.1 (open bars) or dihemoglobin from SGE 953 (filled bars) to separate groups of Balb/C mice; (c) Changes in mean arterial pressure following administration of hemoglobins to conscious, unrestrained rats. Top-load doses of rHb1.1 (filled triangles, orange), SGE 953 di-Hb (filled circles, blue), crosslinked SGE 2812 tetra-Hb (filled crosses, green), or HSA (50 mg/mL, filled squares, purple) were administered to separate groups of rats (n = 6 in all groups).
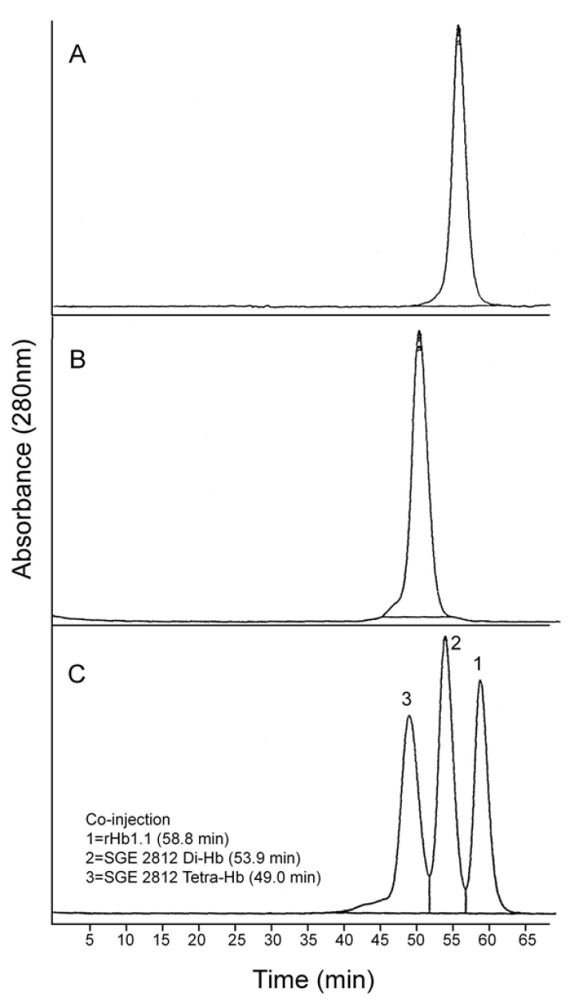
Figure 7.
Analytical size exclusion chromatography of dihemoglobin SGE 2812. (A) Purified SGE 2812 prior to crosslinking; (B) Purified SGE 2812 after crosslinking with bismaleimidohexane; (C) Coinjection of rHb1.1 (tr = 58.8 min) and purified SGE 2812 before (tr = 53.9 min) and after (tr = 49.0 min) BMH crosslinking.
Figure 7.
Analytical size exclusion chromatography of dihemoglobin SGE 2812. (A) Purified SGE 2812 prior to crosslinking; (B) Purified SGE 2812 after crosslinking with bismaleimidohexane; (C) Coinjection of rHb1.1 (tr = 58.8 min) and purified SGE 2812 before (tr = 53.9 min) and after (tr = 49.0 min) BMH crosslinking.
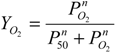
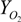 is fractional saturation,
is fractional saturation, 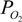 is the partial pressure of oxygen, P50 is the
is the partial pressure of oxygen, P50 is the 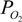 required to achieve 50% saturation of the ligand binding sites, and n is the Hill coefficient. Given the observed values of these constants for the dihemoglobins (Table 2) and assuming an oxygen partial pressure of 90 torr in the lungs and 40 torr in the capillary beds, these molecules could theoretically release 21% of bound oxygen compared to a theoretical 30% release of oxygen from whole blood.
required to achieve 50% saturation of the ligand binding sites, and n is the Hill coefficient. Given the observed values of these constants for the dihemoglobins (Table 2) and assuming an oxygen partial pressure of 90 torr in the lungs and 40 torr in the capillary beds, these molecules could theoretically release 21% of bound oxygen compared to a theoretical 30% release of oxygen from whole blood.