A Blood Meal Enhances Innexin mRNA Expression in the Midgut, Malpighian Tubules, and Ovaries of the Yellow Fever Mosquito Aedes aegypti
Abstract
:1. Introduction
2. Methods
2.1. Mosquito Rearing
2.2. Blood Feeding
2.3. Dissection, RNA Extraction, and cDNA Synthesis
2.4. qPCR
2.5. dsRNA Synthesis and Injections
2.6. Fecundity and Viability Assays
2.7. Data Analysis
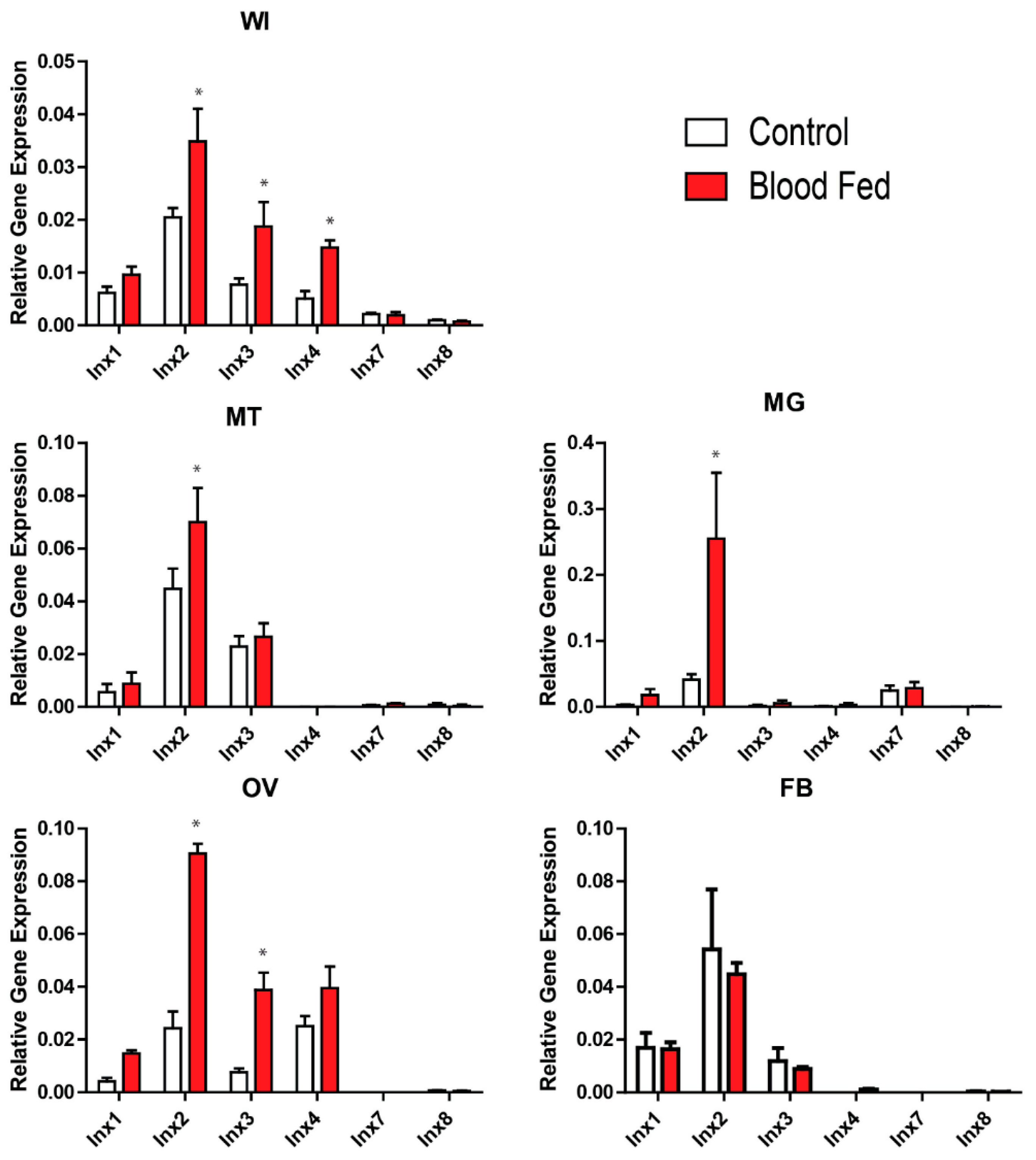
3. Results
3.1. mRNA Expression
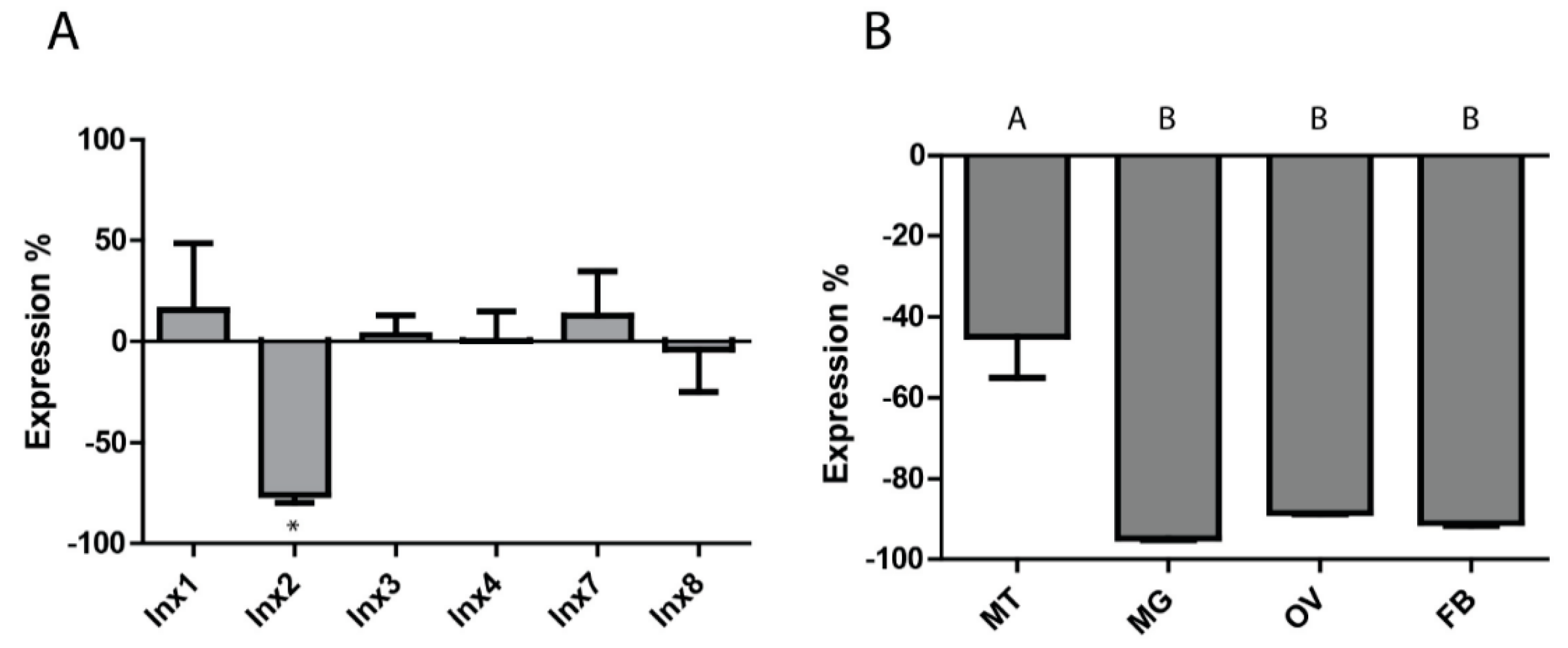
3.2. RNAi
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hansen, I.A.; Attardo, G.M.; Rodriguez, S.D.; Drake, L.L. Four-way regulation of mosquito yolk protein precursor genes by juvenile hormone-, ecdysone-, nutrient-, and insulin-like peptide signaling pathways. Front. Physiol. 2014, 5, 103. [Google Scholar] [CrossRef] [PubMed]
- Phelan, P.; Starich, T.A. Innexins get into the gap. Bioessays 2001, 23, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Phelan, P.; Stebbings, L.A.; Baines, R.A.; Bacon, J.P.; Davies, J.A.; Ford, C. Shaking-B protein forms gap junctions in paired Xenopus oocytes. Nature 1998, 391, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Phelan, P.; Nakagawa, M.; Wilkin, M.B.; Moffat, K.G.; O’Kane, C.J.; Davies, J.A.; Bacon, J.P. Mutations in shaking-B prevent electrical synapse formation in the Drosophila giant fiber system. J. Neurosci. 1996, 16, 1101–1113. [Google Scholar] [PubMed]
- Bosco, D.; Haefliger, J.-A.; Meda, P. Connexins: Key mediators of endocrine function. Physiol. Rev. 2011, 91, 1393–1445. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, F.; Giuliani, G.; Bauer, R.; Rabouille, C. Innexin 3, a new gene required for dorsal closure in Drosophila embryo. PLoS ONE 2013, 8, e69212. [Google Scholar] [CrossRef] [PubMed]
- Holcroft, C.E.; Jackson, W.D.; Lin, W.-H.; Bassiri, K.; Baines, R.A.; Phelan, P. Innexins Ogre and Inx2 are required in glial cells for normal postembryonic development of the Drosophila central nervous system. J. Cell Sci. 2013, 126, 3823–3834. [Google Scholar] [CrossRef] [PubMed]
- Li, M.W.M.; Wang, J.; Zhao, Y.O.; Fikrig, E. Innexin AGAP001476 is critical for mediating anti-Plasmodium responses in Anopheles mosquitoes. J. Biol. Chem. 2014, 1–21. [Google Scholar] [CrossRef]
- Tazuke, S.I.; Schulz, C.; Gilboa, L.; Fogarty, M.; Mahowald, A.P.; Guichet, A.; Ephrussi, A.; Wood, C.G.; Lehmann, R.; Fuller, M.T. A germline-specific gap junction protein required for survival of differentiating early germ cells. Development 2002, 129, 2529–2539. [Google Scholar] [PubMed]
- Hasegawa, D.K.; Turnbull, M.W. Recent findings in evolution and function of insect innexins. FEBS Lett. 2014, 588, 1403–1410. [Google Scholar] [CrossRef] [PubMed]
- Calkins, T.L.; Woods-Acevedo, M.A.; Hildebrandt, O.; Piermarini, P.M. The molecular and immunochemical expression of innexins in the yellow fever mosquito, Aedes aegypti: Insights into putative life stage- and tissue-specific functions of gap junctions. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2015, 183, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Calkins, T.L.; Piermarini, P.M. Pharmacological and genetic evidence for gap junctions as potential new insecticide targets in the yellow fever mosquito, Aedes aegypti. PLoS ONE 2015, 10, e0137084. [Google Scholar] [CrossRef] [PubMed]
- Weng, X.-H.; Piermarini, P.M.; Yamahiro, A.; Yu, M.-J.; Aneshansley, D.J.; Beyenbach, K.W. Gap junctions in Malpighian tubules of Aedes aegypti. J. Exp. Biol. 2008, 211, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Piermarini, P.M.; Calkins, T.L. Evidence for intercellular communication in mosquito renal tubules: A putative role of gap junctions in coordinating and regulating the rapid diuretic effects of neuropeptides. Gen. Comp. Endocrinol. 2014, 203, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Magnusson, K.; Mendes, A.M.; Windbichler, N.; Papathanos, P.-A.; Nolan, T.; Dottorini, T.; Rizzi, E.; Christophides, G.K.; Crisanti, A. Transcription regulation of sex-biased genes during ontogeny in the malaria vector Anopheles gambiae. PLoS ONE 2011, 6, e21572. [Google Scholar] [CrossRef] [PubMed]
- Piermarini, P.M.; Hine, R.M.; Schepel, M.; Miyauchi, J.; Beyenbach, K.W. Role of an apical K, Cl cotransporter in urine formation by renal tubules of the yellow fever mosquito (Aedes aegypti). Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 301, R1318–R1337. [Google Scholar] [CrossRef] [PubMed]
- Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium extraction. Anal. Biochem. 1987, 159, 156–159. [Google Scholar] [CrossRef]
- Song, J.; Bai, Z.; Han, W.; Zhang, J.; Meng, H.; Bi, J.; Ma, X.; Han, S.; Zhang, Z. Identification of suitable reference genes for qPCR analysis of serum microRNA in gastric cancer patients. Dig. Dis. Sci. 2012, 57, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Silver, N.; Best, S.; Jiang, J.; Thein, S.L. Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time PCR. BMC Mol. Biol. 2006, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Holt, R.; Subramanian, G.; Halpern, A.; Sutton, G.; Charlab, R.; Nusskern, D.; Wincker, P.; Clark, A.; Ribeiro, J.; Wides, R.; et al. The genome sequence of the malaria mosquito Anopheles gambiae. Science 2002, 298, 129–149. [Google Scholar] [CrossRef] [PubMed]
- Sanders, H.R.; Evans, A.M.; Ross, L.S.; Gill, S.S. Blood meal induces global changes in midgut gene expression in the disease vector, Aedes aegypti. Insect Biochem. Mol. Biol. 2013, 33, 1105–1122. [Google Scholar] [CrossRef]
- Esquivel, C.J.; Cassone, B.J.; Piermarini, P.M. Transcriptomic evidence for a dramatic functional transition of the malpighian tubules after a blood meal in the Asian tiger mosquito Aedes albopictus. PLoS Negl. Trop. Dis. 2014, 8, e2929. [Google Scholar] [CrossRef] [PubMed]
- Esquivel, C.J.; Cassone, B.J.; Piermarini, P.M. A de novo transcriptome of the Malpighian tubules in non-blood-fed and blood-fed Asian tiger mosquitoes Aedes albopictus: Insights into diuresis, detoxification, and blood meal processing. PeerJ 2016, 4, e1784. [Google Scholar] [CrossRef] [PubMed]
- Koller, C.N.; Raikhel, A.S. Initiation of vitellogenin uptake and protein-synthesis in the mosquito (Aedes aegypti) ovary in response to a blood meal. J. Insect Physiol. 1991, 37, 703–711. [Google Scholar] [CrossRef]
- Raikhel, A.S. Vitellogenesis in mosquitoes. Adv. Dis. Vector Res. 1992, 9, 1–39. [Google Scholar]
- Nunes, F.M.F.; Aleixo, A.C.; Barchuk, A.R.; Bomtorin, A.D.; Grozinger, C.M.; Simões, Z.L.P. Non-target effects of green fluorescent protein (GFP)-derived double-stranded RNA (dsRNA-GFP) used in honey bee RNA interference (RNAi) assays. Insects 2013, 4, 90–103. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calkins, T.L.; Piermarini, P.M. A Blood Meal Enhances Innexin mRNA Expression in the Midgut, Malpighian Tubules, and Ovaries of the Yellow Fever Mosquito Aedes aegypti. Insects 2017, 8, 122. https://doi.org/10.3390/insects8040122
Calkins TL, Piermarini PM. A Blood Meal Enhances Innexin mRNA Expression in the Midgut, Malpighian Tubules, and Ovaries of the Yellow Fever Mosquito Aedes aegypti. Insects. 2017; 8(4):122. https://doi.org/10.3390/insects8040122
Chicago/Turabian StyleCalkins, Travis L., and Peter M. Piermarini. 2017. "A Blood Meal Enhances Innexin mRNA Expression in the Midgut, Malpighian Tubules, and Ovaries of the Yellow Fever Mosquito Aedes aegypti" Insects 8, no. 4: 122. https://doi.org/10.3390/insects8040122





