1. Introduction
Rotaviruses have been identified as the major causative agents of diarrheal disease in humans and a wide variety of animals [
1]. It is estimated that rotaviruses account for more than a million human deaths annually in developing countries [
1,
2,
3], and 20–60 deaths each year in the U.S. [
4]. Rotavirus infections are also of high agricultural importance because of the impact diarrheal disease can have on neonatal and post-weaning animals, especially pigs and calves. There is a shortage of recent literature on the economic burden of rotavirus on livestock, but studies [
5,
6] have found that morbidity due to rotavirus infections in pigs and calves can be as high as 80% with mortality reaching 60%. High morbidity and mortality in livestock animals leads to economic losses from the loss of animals, treatment costs, and reduced growth rates.
Each year, it is estimated that livestock animals in the U.S. produce as much as 20 times more manure than people living in the U.S., or as much as 1.37 billion tons of waste [
7]. Sewage treatment plants are required for human waste. However, no such requirements exist for livestock facilities. While manure is a valuable co-product of the farming industry, large quantities become problematic. Since many farms, especially concentrated animal feeding operations (CAFOs), no longer produce their own feed onsite, the farms themselves do not have the necessary land area to apply the manure. This may lead to over-application of manure, which will quickly overload the soil with nutrients, chemicals, and pathogens. It is critically important that land receiving manure be designed and maintained to effectively and efficiently remove these nutrients, chemicals, and pathogens from surface and near-surface water transport. The increased use of antibiotics on livestock animals may promote antimicrobial resistance in pathogens, further necessitating an immediate need to remove pathogens before entering our surface water supplies. There is even evidence that living in close proximity of a CAFO operation can increase human health deterioration, including elevated cases of gastrointestinal illness, headaches, sore throats, sinusitis, and childhood asthma [
8,
9].
Vegetative filter strips (VFS) are one best management practice (BMP) that is an effective, sustainable, and low-maintenance solution for removing nutrients and pesticides in agricultural runoff. However, to date, direct experimental evidence to confirm the capacity of VFS-mediated removal of pathogens from discharge water is critically lacking, in large part because VFS are not currently being designed for this purpose. VFS are vegetated areas that receive manure from livestock operations and are closely aligned with NRCS Conservation Practice Standards 635 (Vegetated Treatment Area) and 393 (Filter Strip). In terms of mechanism, the vegetation in a VFS enhances infiltration and facilitates the filtering process. By design, VFS effectively removes nutrients and pesticides from agricultural runoff; however, pathogens are also present and pose significant economic and health burdens. There are a number of components in a VFS that are in contact with surface runoff. The plant stem, the decaying plant material, and the underlying soil are all potential adsorption sites. Adsorption of microorganisms to plant or soil particles can be due to many factors that include particle size, mobility, surface charge, and surface area. Currently, very little work has been done in this area to characterize virus adsorption to these particles.
Due to the small size of viruses, transport is influenced by the adsorption to soil particles. Virus adsorption is also affected by the pH of the soil-water system and surface properties of the virus [
10]. As the soil pH increases, adsorption of viruses to soil becomes more of a factor. The mobility of viruses is determined by the properties of the amphoteric protein coat. It has been accepted that the formation of virus-cation-clay bridges increases with an increase in the positive charge on the virus protein coat [
11]. Other researchers have shown that viruses have been adsorbed by clays, glass, iron oxides, silica, and aluminum [
12,
13,
14,
15,
16].
Virus transport through soil is enhanced in the presence of water or other transporting agents [
17]. Movement slows or ceases when water drains out of soil; therefore, there is increased movement and a faster flow rate of virus in saturated soils [
11]. There is a lack of agreement among published studies regarding whether viruses primarily interact with clay particles
versus sand particles. Studies showing a strong interaction between clay particles and viruses attribute the interaction to the increased surface area of clay particles and high cation exchange capacity (CEC) [
18,
19]. In contrast, however, other studies show the primary interaction being between sand particles and viruses. For instance, Blanc and Nasser (1996) [
20] attributed the lower sorption of viruses to loamy soil to the high content of organic material in the soil. Natural organic matters have a tendency to be heterogeneous and, therefore, vary in their hydrophilic and hydrophobic properties. In addition, organic matter sorbed on soil particles can provide additional negative charges that repulse viruses or cover positively charged sites, which may decrease the electrostatic interactions between viruses and soil particles. Davis
et al. (2006) [
21] reached a similar conclusion when they found that dairy manure wastewater (high organic matter content) decreased viral adsorption to sandy soil. Furthermore, they determined that dairy manure wastewater increased the release of viruses attached to soil. These findings are supported by findings from Davidson
et al. (2013) [
22] that showed rotavirus has a high affinity for sand particles and also findings from Chrysikopoulos and Aravantinou (2014) [
23] showing that the size of sand particles is an important factor.
Previous studies [
22,
24,
25,
26] found that a small soil chamber produced results accurately reflecting the transport kinetics of
Cryptosporidium parvum as may be seen in a slightly larger soil chamber studied by Trask
et al. (2004) [
27]. The ability to achieve consistent results using a small, lab-bench soil chamber provides the opportunity to refine methodologies to more accurately detect and monitor
C. parvum (and other microbial pathogens), and then extrapolate results to larger-scale situations. This study briefly describes the design and construction of the small-scale soil box and tilting chamber used in conjunction with the rainfall simulator developed by Davidson (2007) [
28] and used by Koch
et al. (2013) [
25] and Davidson
et al. (2014) [
26]. In this study, rotavirus transport was examined in overland flow with the aim of better characterizing the interactions between the pathogen, soil particles, and vegetation.
Since the mere presence of rotavirus does not indicate infectivity, this study used a tissue-culture infectivity assay to determine what fraction of the rotavirus particles present in runoff, soil and vegetation extracts was capable of causing infection. In addition, factors affecting virus removal during land treatment of wastewater, such as adsorption to soils, have not been studied extensively, and little is known about the survival or fate of viruses applied to the soil. If viruses are not retained by the soil, they may migrate vertically, resulting in groundwater contamination. Therefore, the overall objective of this study was to characterize the overland transport of rotavirus, identifying factors that are critical for survival in a natural soil-water environment. Specifically, this study investigated the infectivity of rotavirus particles in surface runoff water, soil cores, and near-surface runoff water from three different soil types and then examined the ability of vegetation to inhibit overland transport of infective rotavirus particles.
2. Materials and Methods
The overland transport studies investigating the kinetics of rotavirus transport in the presence of three different soils and two different vegetation types are discussed in the following section. The detailed methodology is described in Davidson
et al. (2013) [
22], Koch
et al. (2013) [
25], and Davidson
et al. (2014) [
26], and is also briefly summarized here. Three soil types were chosen to span the range of soils typically found in Illinois, in terms of sand, silt, and clay contents. All samples (runoff water, soil extracts, and vegetation) were analyzed using a tissue-culture infectivity assay to determine what fraction of the rotavirus particles remained infective after transport through the respective matrix. Only infective rotavirus particles are measured in this study, as the inactivated particles were not deemed to be of concern to downstream users. Therefore, while inactivated rotavirus particles may be transported through a soil-water environment, only the active and viable particles were detected in the water, soil extracts, and vegetation samples.
2.1. Construction of a Small-Scale Rainfall Simulator
A small-scale rainfall simulator was designed and constructed to simulate low to medium rainfall intensities to relatively small areas of interest. The overall dimensions of the rainfall simulator were 1.07 m by 0.66 m. For the rainfall simulation, mister nozzles were used because of their ability to supply a very low rate of water flow. Since the nozzles produced a mist rather than droplets, a common fiberglass window screen was placed below the arrangement of nozzles. The screen contained small square openings, allowing water to easily pass through the screen without causing pooling or impeding the rainfall distribution. The mister nozzles sprayed water onto the screen and droplets were formed and allowed to fall onto the surface below, more closely mimicking natural rainfall droplets. The rainfall simulator was capable of producing intensities ranging from 23 mm/h to 90 mm/h. In this study, however, a constant 65 mm/h rainfall intensity was used.
2.2. Preparation of Soil Bed
Plexiglas soil boxes measuring 0.305 m wide, 0.610 m long, and 0.015 m deep were designed and constructed to match the rainfall footprint of the small-scale rainfall simulator. The soil boxes were sealed with epoxy and silicone caulk to prevent leaks. Holes were installed in both the downslope end of the soil box, just above the height of the soil surface, and also along the bottom of the soil box to collect surface and near-surface water runoff, respectively. The Plexiglas soil boxes were placed on a frame that allowed for the adjustment of the slope of the soil beds. Aluminum trays were installed for the collection of surface and near-surface runoff and flexible tubing was used to connect the trays to sampling containers.
A moderately drained silt-loam (Catlin series: 24% sand, 50% silt, 26% clay) soil was collected from a site in Champaign, IL, and a well-drained fine sandy loam (Alvin series: 60% sand, 25% silt, 15% clay) soil and poorly-drained silty-clay (Darwin series: 5% sand, 50% silt, 45% clay) soil were collected from a farm near Newton, IL. The three soil types span the range of soil textures found in Illinois and collectively represent typical soils found in central Illinois.
The soil was sieved to remove gravel and plant residue and then oven-dried for 24 h to reduce existing microbial contamination, to produce uniform antecedent moisture conditions, and to allow for easier separation of soil particles. The soil was added to each soil box in three one-inch (25.4 mm) layers, with each layer moistened and lightly tamped. Rainfall was then simulated periodically to simulate natural climatic conditions and to approach a natural compaction level after several cycles of wetting and drying. Next, grass was seeded accordingly. Smooth Brome and Tall Fescue (purchased at Illini FS, Urbana, IL, USA) grass varieties were used in this study. The chambers were seeded by hand until a uniform distribution of seed was achieved. The recommended seeding rate of 15 lbs/acre was used for each grass. Approximately 3–6 months was necessary to achieve the desired maturity level of vegetation before use in the transport studies.
2.3. Source of Rotavirus
Rotavirus was acquired for this study in the same manner as Davidson
et al. (2013) [
22]. For convenience, the details are briefly summarized here. Group A porcine rotavirus OSU strain ([
7], p. 9, G5) was obtained from the American Type Culture Collection catalog #VR-892 and passaged two additional times in cultured Ma104 cells [
29]. Infectious units of activity were determined in a focus forming unit assay (FFU) as described by Rolsma
et al. (1998) [
30]. Three colostrum-deprived newborn piglets were inoculated with 1 mL (3 × 10
6 FFU) each and the feces were pooled and collected over a two day period. Rotavirus was also prepared by cultivation in Ma104 cells and purified as described by Rolsma
et al. (1998) [
30], excluding the last CsCl gradient step.
2.4. Experiments
The experimental setup is depicted in
Figure 1 below. All experiments were completed in a laboratory and all runoff was contained, collected, and disposed of accordingly. A series of experiments were completed to investigate the overland transport kinetics of rotavirus through three different surface conditions (bare soil and two vegetation types) and three different soil types, as described earlier. All experiments were conducted with a constant rainfall intensity of 65 mm/h, as this intensity is common in the Midwestern U.S. A bed slope of 2.5% was chosen to mimic the typical slope of existing vegetative filter strips. A rainfall duration of 20 min was used in order to observe the entire transport curve.
Experiments began after the vegetated beds reached a canopy cover of approximately 50% or greater, or after a somewhat natural soil compaction was achieved through repeated rainfall events for the bare soil conditions. To ensure the initial surface moisture conditions were similar for each experiment, rainfall was simulated until flow began. The fecal slurry was then applied after both surface and near-surface flow ceased. The moisture content of the soil was then near field-capacity. Approximately 15 mL of slurry containing 108 rotavirus particles/mL was applied in a band approximately 5 cm (2 in) from the up-slope end of the soil bed with an additional slurry aliquot taken for initial concentration analysis. Surface and near-surface runoff samples were collected routinely throughout the entire flow event. In addition, soil cores were taken with a 2.1 cm diameter cork borer to a depth of 7.6 cm (3 in) after the rainfall event and when all runoff had ceased to determine the transport of rotavirus particles in the soil. A total of eight soil cores were collected; four cores evenly spaced along a transect 5 cm (2 inches) from the up-slope edge of the soil bed, corresponding to the location of the slurry application, and four additional cores evenly spaced along a transect 30.5 cm (12 inches) from the upslope edge of the soil bed. Each of the eight soil cores was then divided into three 2.54 cm (1 inch) depths. These cores were analyzed to determine the number of infective rotavirus particles at each depth increment. All vegetation was clipped at the soil surface and collected from a 2.54 cm (1 inch) transect across the entire width of the soil bed prior to soil sampling, coinciding with the transects for soil core collection.
Surface runoff samples were collected every minute after runoff began. Near-surface runoff samples were collected on longer time intervals (as needed) since near-surface runoff volume was much lower than surface runoff volume. The runoff volume was measured for each collected sample. The grass clippings were placed in 15 mL conical tubes, one tube for each of the two transects of clippings. Each tube of grass clippings was then mixed with approximately 8 mL PBS and allowed to sit for approximately four hours. The samples were then mixed thoroughly by shaking and vortexing, centrifuged to pellet the clippings, and the supernatant was collected.
2.5. Extraction of Rotavirus from Soil Cores
At the time of sampling, a volume of TNC approximately equal to the porosity of the soil sample was added to the tube and the contents mixed thoroughly. The tube was centrifuged at 1260 g for 10 min at 4 °C. The aqueous solution was then removed from the tube and saved. This process was repeated for a total of three extractions. All three extractions were combined.
Three extractions were chosen because, theoretically, 87.5% of the virus should be extracted with three extractions, assuming 100% availability of the virus and no loss of infectivity after each addition of TNC and mixing. With uniform mixing of 1 mL virus suspension, 1 mL TNC, and soil, 50% of the virus would be removed during the first and subsequent extractions. A combination of the three extracts should, therefore, contain 87.5% of the original virus. Further extractions would yield more rotavirus but would substantially dilute the sample, making it more difficult to assay such a low concentration of rotavirus and introducing more error into the analysis. The data was normalized for the remaining 12.5% that was not extracted.
2.6. Preparation of Samples for Rotavirus Analysis
The surface and near-surface runoff samples were collected and analyzed for infective rotavirus particles. The collected samples contained a relatively high concentration of rotavirus particles, requiring a dilution of the samples for analysis. The procedure for diluting the samples consisted of measuring the total volume, shaking the samples and allowing soil particles to settle, and then taking a given amount from each sample. A 1/5 dilution was then prepared by combining 200 µL of each sample with 800 µL of serum-free MEM and applying 500 µL directly to the plate wells. Soil core samples were prepared by combining 250 µL of sample with 750 µL of serum-free MEM. Again, 500 µL was applied directly to the plate wells.
2.7. Focus Forming Unit (FFU) Assay
The focus forming unit (FFU) assay was performed following previously published methods [
30]. A summary of this method, as modified for analysis of rotavirus recovery for soil samples, is briefly described below.
Confluent monolayers of Ma104 cells in 24-well plates were rinsed twice with phosphate-buffered saline (PBS). Each plate well was then inoculated with the 500 µL of sample and allowed to incubate for 30 min at 37 °C in a 5% CO2 incubator. After the short incubation, the sample was aspirated from the cells, rinsed one time with 1 mL PBS, and allowed to incubate overnight (37 °C in a 5% CO2) with 1 mL MEM + antibiotics.
After overnight incubation, the plate wells were rinsed twice with 1 mL of PBS and fixed using 1 mL MeOH-glacial acetic acid (9:1) per well. The monolayers were rehydrated using a 70% ethanol solution followed by the addition of a 50% ethanol solution. The rehydration was followed by rinsing with a wash buffer (WB; 125 mM Tris, 350 mM NaCl, 0.25% Triton X-100, pH 7.6). Endogenous peroxidase activity was inhibited by addition of 3% H2O2 diluted in WB. Nonspecific primary antibody binding was inhibited by addition of a 5% normal goat serum (Vector Laboratories, Burlingame, CA, USA) diluted in wash buffer. After a short incubation of 20 min, the normal goat serum was removed and the primary antibody (rabbit anti-human rotavirus; Dako; catalog No. B218) was added to the monolayers. After rinsing, a secondary antibody of biotinylated goat anti-rabbit immunoglobulin G (Vector Elite Kit; Vector Laboratories, #BA1000) diluted in WB with normal goat serum was added. The monolayers were rinsed and incubated 20 min with an ABC reagent (Vector Standard Elite Kit, #PK6100). The ABC reagent is a horse radish peroxidase-conjugated biotin-streptavidin complex that binds to biotinylated 2° antibody. After washing out unbound conjugate, peroxidase substrates DAB (#54-10-00) and H2O2 were added and incubated 10 min at room temperature (Kirkegaard and Perry, Gaithersburg, MD, USA). The DAB is a precipitation substrate for the peroxidase that turns brown during the reaction (oxidation) to reveal the cytochemical location of the rotavirus antigen. Excess substrates were washed out and brown foci quantified.
2.8. Quantification of Rotavirus Infectivity
The rotavirus FFUs were enumerated by counting the number of stained viral-antigen-positive cells, or FFUs, present in each sample. The FFUs were observed at a magnification of 100× using a Nikon TS 100 inverted microscope equipped with a computer-controlled electronic stage and Spot RT-slider CCD camera (Nikon Instruments Inc., New York, NY, USA). Twenty-five digital images were automatically collected from each plate well using Metamorph software (Molecular Devices, Inc., Sunnyvale, CA, USA) to develop and capture images within a scan grid which covered >80% of the well surface area. The number of FFUs was either counted manually or automatically using integrated morphometric parameters within the software to recognize FFU. The ability to automatically count FFUs was dependent on the degree of background staining, which varied between different antibody lots. The number of FFUs present in each sample was divided by the number of FFUs present in the initial fecal slurry applied to the respective soil bed, providing a percent recovery of rotavirus relative to the amount applied.
2.9. Statistical Analysis
In this study, the laboratory experiments were conducted using three soil types (Catlin, Darwin, and Alvin) and three surface cover conditions (bare soil, Smooth Brome, and Fescue). A rainfall intensity of 65 mm/h was applied to each soil bed. Statistical analyses were conducted for rotavirus recovery in surface runoff for all cover conditions and soil types using R. The parameters were evaluated using a 10% (p = 0.10) significance level. As there were a large number of variables involved with this study, it was not possible to consider all of the possible main effects and interactions between the variables (ground cover, soil, slope, intensity, and runoff volume). Since true repetitions were not possible in this study because of the length and intensity of the analysis methods, mean square error (MSE) could not be computed. A series of analysis of variance (ANOVA) was performed to evaluate the interaction between variables to determine which interactions were negligible (small sums of squares). Another ANOVA was also conducted to compare the main effects of soil property (% sand and % clay), % vegetation cover, and runoff volume, and the interactions between these variables that seemed to be of most importance.
3. Results and Discussion
3.1. Effect of Soil Type
The three soil types are first compared in this section according to the surface condition in order to examine the effect of soil type on the recovery of infective rotavirus particles during overland transport.
Figure 2 shows the results from the three soil types separated by surface condition.
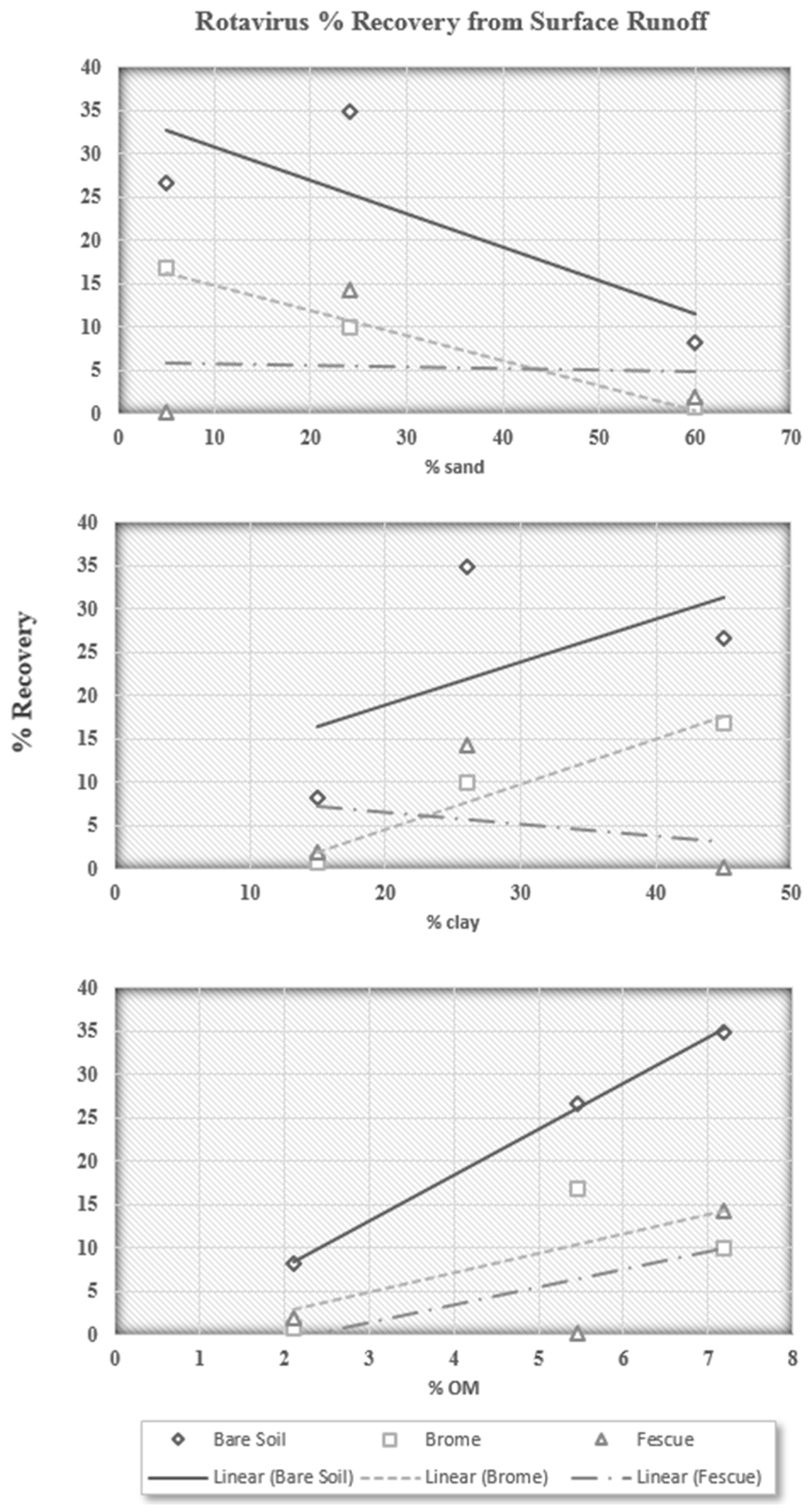
From these graphics, it appears that soils with considerable amounts of clay (Catlin and Darwin) increase the amount of infective rotavirus particles recovered in surface runoff, which is in agreement with our previous findings that rotavirus particles preferentially interact with and are removed by sand particles. However, this percent recovery trend with surface runoff in
Figure 2 is likely due to the reduced infiltration rate of soils with high clay contents. The rotavirus particles may never be coming into contact with the clay soil particles: suspended in surface water and being directly transported in surface runoff. An inability of water to initially infiltrate the soil would cause this initial flush of rotavirus particles. Over time, the recovery from Catlin soil decreases and follows that of Darwin soil very closely. The recovery of infective rotavirus particles in surface runoff from Alvin soil is much lower than that from both the Catlin and Darwin soils. The total recovery in surface runoff for Alvin soil is only 8.28%, compared to 34.92% for Catlin soil and 26.56% for Darwin soil (
Table 1). Again, this is likely due to characteristics of the soil and the ability of water to better infiltrate a sandy soil such as Alvin. With more water infiltrating, there will also be more rotavirus particles entering the soil. Even with the increased infiltration rate, no infective rotavirus particles were detected in the near-surface runoff and only trace amounts in the soil core extractions from the Alvin soil.
With the addition of Brome vegetation, recovery of infective rotavirus particles is highest for Darwin soil. With the increased ability of water to infiltrate the soil due to macropores created from the vegetation root system and impeded surface flow due to surface obstructions from the vegetation, there is now a greater opportunity for interaction between clay particles and rotavirus particles. The Alvin soil has a much lower recovery than Catlin or Darwin soil. This reduced recovery from Alvin soil is probably mostly controlled by flow characteristics, with water more easily penetrating a sandy soil. However, the effect of interactions between sand particles and rotavirus particles cannot be eliminated here.
With Fescue vegetative cover, no significant amount of infective rotavirus particles is recovered from Darwin or Alvin soils. The Catlin soil had the lowest percentage of vegetative cover and, therefore, did not have as much obstruction to surface flow. This would account for the initial pulse of rotavirus particles, followed by a quick decrease to less than two percent recovery. The total surface runoff volume from Catlin soil was substantially higher than that from either Darwin or Alvin soils. A summary of infective rotavirus particles recovered from surface runoff tabulated in conjunction with the seven experimental parameters collected for each combination of soil and vegetation type is presented in
Table 2.
The data in
Table 2 were used to analyze trends between individual characteristics of each condition and the actual percent recovery of infective rotavirus particles detected in the surface runoff. Since it is reasonable that rotavirus particles may never interact with the soil in surface runoff (Darwin bare soil condition) due to very low initial soil infiltration rates, trends were plotted for individual characteristics of each condition
versus the percent recovery of infective rotavirus particles detected in both the surface runoff (
Figure 3) and soil cores (
Figure 4).
From these trends, it appears that sand decreases the recovery of rotavirus particles in surface runoff, while clay increases recovery in surface runoff, both of which are in agreement with preliminary findings that rotavirus particles primarily interact with sand particles. However, these trends also appear to be directly related to overland flow characteristics. The higher clay content would decrease soil infiltration rates and allow more runoff. Sand, on the other hand, would provide a high soil infiltration rate and allow much more water, and rotavirus particles, to infiltrate the soil profile.
The third plot shows that the recovery of rotavirus particles increases with increasing organic matter content, which confirms findings by Blanc and Nasser (1996) [
20], who found that organic matter provides negative charge attachment sites that inhibit attachment of negatively charged microbial organisms. Clay particles are often associated with a higher organic matter content, and organic matter sorbed on soil particles can provide additional negative charges that repulse viruses or cover positively charged sites, decreasing the electrostatic interactions between viruses and soil particles.
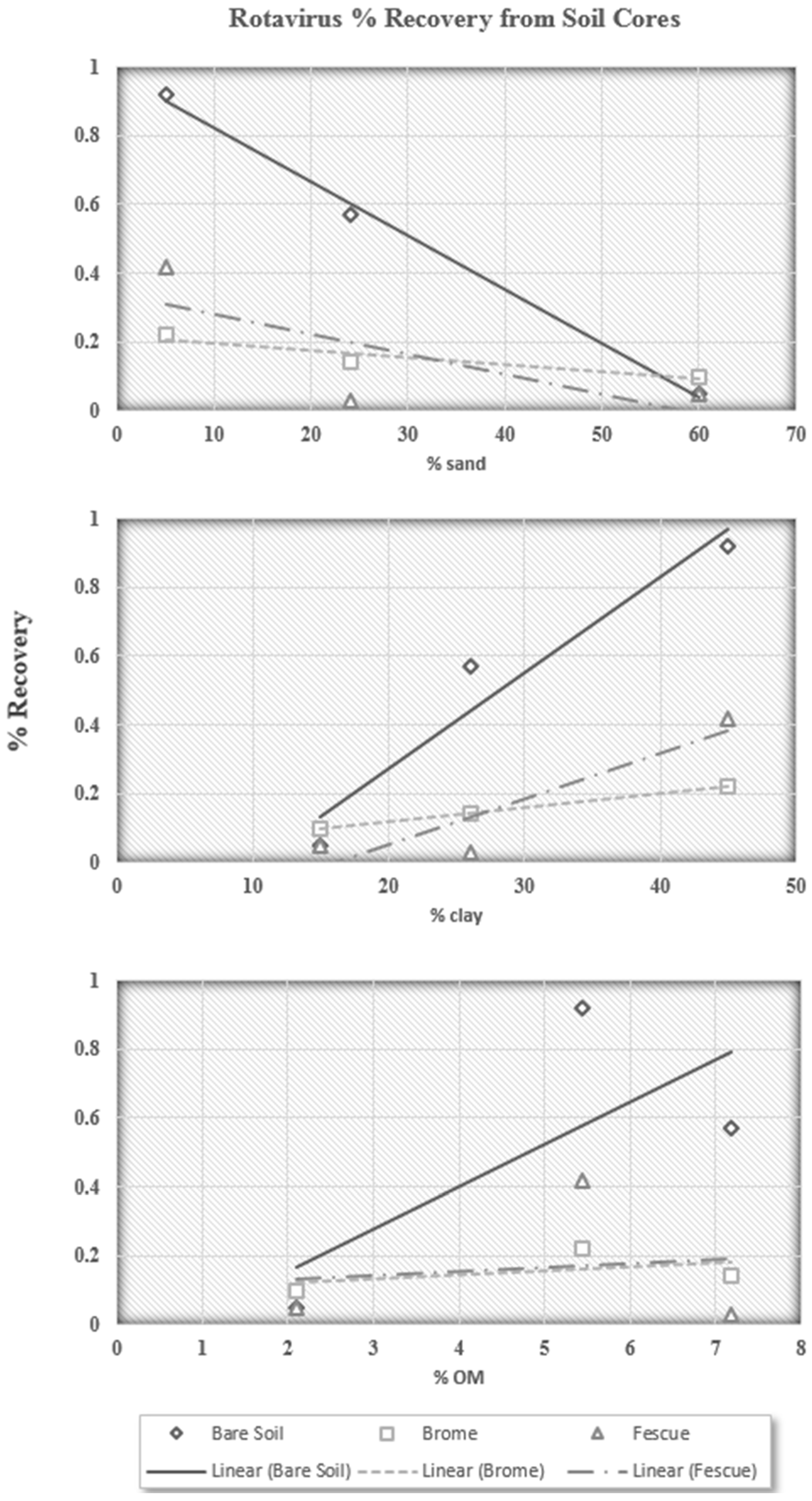
If a portion of the infective rotavirus particles are never interacting with the soil surface, it is not possible to determine if one particular fraction of soil is having a greater impact than another on rotavirus recovery. However, analysis of the soil core extractions could help explain this. The soil core extractions contain the fraction of infective rotavirus particles that successfully entered the soil and were then able to be extracted. The percent of infective rotavirus particles recovered in the soil core extractions, then, is the fraction of particles that either does not interact with soil particles, or is not affected by the interaction with the soil. The soil core extractions are plotted against percent sand, percent clay, and percent organic matter in
Figure 4.
The soil core extraction trends are very similar to those for surface runoff. It appears the rotavirus particles that successfully enter the soil are more affected by the sand content, as was believed from preliminary results. While the relationships for percent sand and percent clay are not conclusive, they show a strong trend confirming that soils high in sand content are better at reducing the recovery of infective rotavirus particles. In addition, rotavirus recovery increased with increasing organic matter content for all three surface conditions. This is also in agreement with previous findings by Davis
et al. (2006) [
21]. As presented in
Table 1, the number of infective rotavirus particles in the surface runoff, near-surface runoff, and soil cores was also less for Alvin soil, which is the highest in terms of sand content. Therefore, it is clear from the data in this study that infective rotavirus is less likely to be transported from a sandy soil.
3.2. Effect of Vegetation
The three surface conditions are compared in this section according to the soil type in order to examine the effect of vegetation on the recovery of rotavirus particles during overland transport.
Figure 5 shows the three surface conditions separated by soil type.
When all three surface conditions are plotted together according to soil type, the effect of surface condition is more easily understood. For Catlin soil, both vegetated conditions show a lower peak recovery than the bare soil condition. This can be explained by the reduced surface flow rate due to surface obstructions caused by the vegetation and an increased ability of water and rotavirus particles to enter the soil due to macropores created by the vegetation root system.
For Darwin soil, the bare soil condition has a much higher peak recovery than either of the vegetated conditions. As discussed previously, this is probably due to a low soil infiltration rate of the high clay soil. The roots of the vegetation create macropores that serve as small channels for water to enter the soil, reducing the volume of surface runoff as well as the number of infective rotavirus particles in the surface runoff. For the Alvin soil, the bare soil condition also has the highest peak recovery. As expected, for all three soil types, the percent recovery of infective rotavirus particles in near-surface runoff increases with the addition of vegetation, even though only extremely low quantities were detected. With a relatively shallow layer of soil, the vegetation is effective at extending through the entire soil profile and allowing transport into the near-surface runoff. It is important not to confuse near-surface runoff with groundwater in this study since the soil profile is only intended to represent a very shallow layer of soil. This study cannot make any conclusions about the transport of infective rotavirus to groundwater sources.
Again, while vegetation, in general, appears to decrease the recovery of infective rotavirus particles in surface runoff, there does not appear to be a clear advantage of any one vegetation type. Perhaps more important is the surface vegetative cover and the extent of the root system in the soil profile.
3.3. Statistics of Results
The statistical analysis indicates that % sand (
p = 0.0556) was the main sources of variation for soil and % clay did not play a considerable role in reducing the transport of rotavirus (
p = 0.3174). Vegetation cover was another factor that reduced rotavirus transport (
p = 0.0707). Runoff volume was the most significant variable for the source of variation in rotavirus transport (
p = 0.0054). The interaction between % sand and % vegetation cover (
p = 0.0369) and % vegetation cover × runoff volume (
p = 0.0234) had also played important roles in rotavirus transport. Even though true replications were not possible in this study, this general statistical analysis clearly indicates that soil properties, ground surface condition, and runoff volume are significant factors in the overland transport of rotavirus. Statistical analyses were done using a significance level of p equal to 0.10. The result of the statistical analysis is presented in
Table 3.