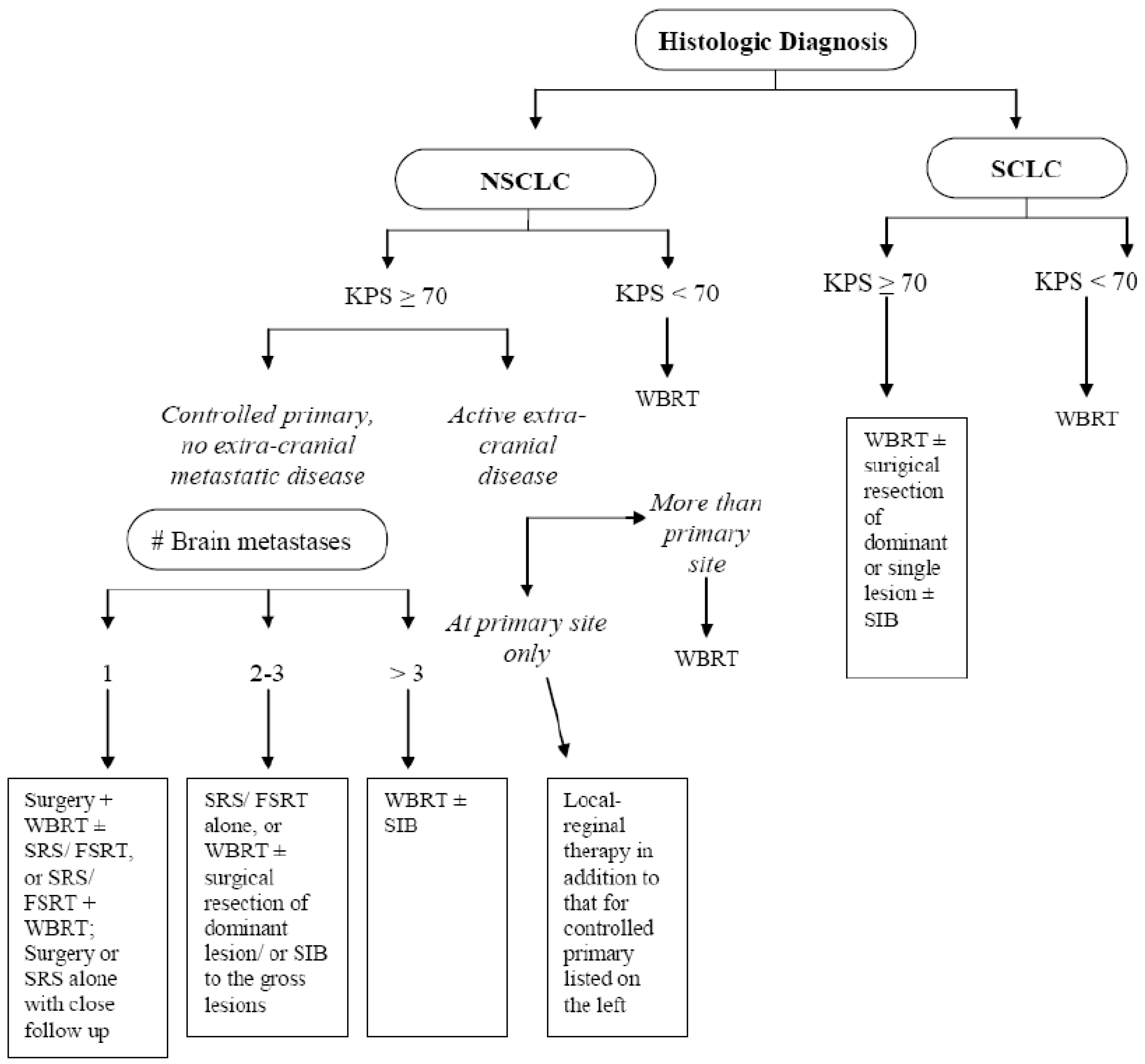
1. Introduction
Brain metastases are the most common intracranial neoplasm, occurring in 8–10% of cancer patients, and are a significant cause of cancer-related morbidity and mortality worldwide [
1,
2] The incidence of brain metastases is rising with an annual incidence of approximately 170,000 to 200,000 in the United States [
3]. This is caused by a combination of factors including the improved therapeutic efficacy of current cancer treatments, which leads to longer survival, such as the addition of bevacizumab to chemotherapy as the first-line treatment of metastatic non-small cell lung cancer; and failure in a potential systemic therapy sanctuary site, or more frequent brain surveillance for specific cancers that have a predilection for brain metastases; and improvements in modern imaging technology, which lead to the diagnosis of brain metastases at an earlier stage [
4,
5]. However, such an increase in the incidence of brain metastases in recent years has not been observed in all studies, and may possibly be attributed to under-diagnosis in earlier years [
1,
6]. The most common origins of brain metastasis include primary cancers of the lung, breast, skin (melanoma), and the GI tract. Among these, primary tumors in the lung are the most common cause of brain metastases, as up to 65% of patients with lung cancer will ultimately develop brain metastases [
7].
As the leading cause of cancer mortality, and the most prevalent cancer in men, lung cancer accounted for an estimated 161,840 deaths in the United States with an incidence of 215,020 in 2008. Furthermore, approximately 1.35 million cases were diagnosed worldwide with 1.18 million deaths in 2002 [
8]. Therefore, brain metastasis is a very important problem in the overall management of lung cancer. Among the various histologies, small cell lung cancer (SCLC) is the most likely to metastasize to the brain with an 80% probability of brain metastasis after two years from diagnosis [
6]. Brain metastases develop in approximately 30% of patients with non-small cell lung cancer (NSCLC) [
9]. Among the various histologies of NSCLC, the relative frequency of brain metastases in patients with adenocarcinoma and large cell carcinoma was much higher than that in patients with squamous cell carcinoma [
10,
11].
Most patients present with significant neurological signs and symptoms that are related to the location and extent of brain involvement. They include both focal neurological changes and general symptoms secondary to increased intracranial pressure [
12]. Major clinical presentations are listed in
Table 1 [
13]. Contrast-enhanced MRI is the preferred imaging study for the diagnosis of brain metastases over non-enhanced MRI or computed tomography (CT) scans in detecting cerebral metastases and in differentiating metastases from other central nervous system (CNS) lesions [
14,
15]. The recommended pregadolinium studies include T2-weighted and T1-weighted sequences, and the recommended postgadolinium studies include the T1-weighted and fluid-attenuated inversion-recovery (FLAIR) sequences [
5]. Thinner axial slices without skips may be necessary to detect the smallest lesions. If the diagnosis is still in doubt, biopsy should be considered. Brain metastases are usually found at the junction of the grey and white matters, with circumscribed margins and large amounts of vasogenic edema relative to the size of the lesion. Furthermore, they usually present as multiple lesions as a result of a lung primary [
16].
Without treatment, the median survival of patients is 4–7 weeks [
17,
18,
19]. The treatment can usually be divided into symptomatic and therapeutic strategies. Symptomatic relief is most commonly achieved with corticosteroids to reduce peritumoral edema and anticonvulsants to prevent recurrent seizures. Systemic steroids alone improve neurological function and prolong survival to approximately two months [
20]. Whole brain radiotherapy (WBRT), as the primary treatment approach for brain metastases, improves neurological function and prolongs median survival to three to five months [
12]. Due to the poor survival outcomes associated with brain metastases, more aggressive treatments for patients have been sought and investigated. In general, the therapeutic approach largely depends on the number and location of metastases, as well as the extent of extra-cranial tumor involvement. In the following sections, prognostic factors that may influence treatment selection and the various treatment approaches will be reviewed.
Table 1.
Clinical presentation of brain metastasis.
Table 1.
Clinical presentation of brain metastasis.
| Symptom | Percentage (%) | Sign | Percentage (%) |
|---|
| Headache | 49 | Hemiparesis | 59 |
| Mental problems | 32 | Cognitive deficits | 58 |
| Focal weakness | 30 | Sensory deficits | 21 |
| Ataxia | 21 | Papilledema | 20 |
| Seizures | 18 | Ataxia | 19 |
| Speech problems | 12 | Apraxia | 18 |
2. Prognostic Factors
A retrospective recursive partitioning analysis (RPA) was performed based on three consecutive Radiation Therapy Oncology Group (RTOG) trials, which included approximately 1200 patients with brain metastases [
21]. Three prognostic classes (RPA class I, II and III) were found to be associated with the overall survival of patients with brain metastases. This classification scheme is based on age at diagnosis, presence of extracranial disease, Karnofsky performance status (KPS), and the status of the primary cancer. RPA class I includes patients who are younger than 65 years of age, have a KPS score of ≥70, tumor controlled at the primary site, and no extracranial disease. RPA class III patients have a KPS score of less than 70. All other patients are in RPA class II. The median survival times for the RPA classes I-III were 7.1, 4.2, and 2.3 months, respectively. This RPA classification is the most commonly used prognostic system for brain metastases, with further validation in Phase III and major institutional studies for both NSCLC and SCLC [
22,
23,
24,
25]. Despite the common adaptation of RPA classification, clinicians are still faced with the dilemma of tailoring treatments to individual patients because factors such as the number or volume of brain metastases were not included in the RPA initially, estimation of systemic disease was not consistently reliable,
etc. As newer data came out, a new prognostic index, the graded prognostic assessment (GPA), was generated based on data from five randomized RTOG studies involving brain metastases [
26]. Please refer to
Table 2 for details of the GPA scoring system. The median survival times according to GPA score were: GPA 0–1, 2.6 months; GPA 1.5–2.5, 3.8 months; GPA 3, 6.9 months; and GPA 3.5–4.0, 11.0 months (
p < 0.05). The GPA prognostic index was further validated based on specific diagnosis at the primary site due to the heterogeneous response of brain metastases to various treatment approaches based on histology and the various patterns of systemic disease and response to systemic therapy for different types of primary tumor [
27]. For both NSCLC and SCLC, all four prognostic factors remained significant, confirming the prognostic value of the original GPA for lung cancer.
Table 2.
The graded prognostic assessment (GPA).
Table 2.
The graded prognostic assessment (GPA).
| Score | 0 | 0.5 | 1.0 |
|---|
| Age | >60 | 50–59 | <50 |
| KPS | <70 | 70–80 | 90–100 |
| No. of CNS metastases | >3 | 2–3 | 1 |
| Extracranial metastases | Present | - | None |
3. Symptomatic Management
The management of symptoms from brain metastases has primarily consisted of the usage of corticosteroids (e.g., dexamethasone or methylprednisolone) and anticonvulsants. Corticosteroids are given upon initial diagnosis to relieve the symptoms associated with peritumoral edema in approximately two-thirds of patients because of their ability to reduce the permeability of tumor capillaries [
28,
29]. In a study by Vecht
et al., doses of 8
versus 16 mg/day with tapering schedules over four weeks and doses of 4
versus 16 mg/day with continuation of these doses for 28 days prior to tapering demonstrated similar KPS improvements at seven days (54% to 70%) and 28 days (50% to 81%) in patients treated with WBRT and concurrent ranitidine [
30]. However, patients in the 4 mg/day arm experienced a higher rate of drug reinstitution than in patients treated with 8 or 16 mg/day. Furthermore, the greatest KPS improvement was observed in patients in the 16 mg/day arm when this dose was tapered over four weeks. These findings suggest that greater KPS improvement arose from the maximal anti-inflammatory effects of the initial higher doses, while the late toxicity associated with corticosteroids was minimized with gradual tapering. A commonly used dexamethasone regimen in patients with brain metastases is a 10-mg intravenous (IV) bolus, followed by 4 to 6 mg PO every six to eight hours before gradual tapering with caution. However, initial corticosteroid use may be reserved for symptomatic patients owing to the common side effects of dexamethasone, including hyperglycemia, peripheral edema, psychiatric disorder, oropharyngeal candidiasis, Cushing’s syndrome, muscular weakness, and pulmonary embolism [
31].
Approximately 15% of patients with brain metastases present with seizures, and seizure is frequently associated with supratentorial lesions. Seizures can be managed with antiseizure medications, but anticonvulsants are generally not given prophylactically. In a prospectively randomized study by Forsyth
et al. [
32], one hundred patients with newly diagnosed brain tumors were randomized to prophylactic anticonvulsants or no anticonvulsants. After a median follow up of 5.44 months, no difference in the rates of seizures at three months or seizure-free survival were observed, suggesting that antiseizure prophylaxis in brain tumor patients is not necessary.
5. Neurocognitive Functioning after Brain Irradiation
WBRT is associated with many acute, subacute, and late side effects. The acute toxicities such as fatigue, hair loss, and skin reaction are mild and self-limiting. The late toxicities are usually observed in patients with limited brain metastases and well controlled extracranial disease because these patients tend to survive longer. Late toxicities include diffuse white matter injury or cerebral atrophy and neurocognitive deficits. Neurocognitive function after cranial irradiation is being evaluated more closely as the efficacy of systemic therapy improves over time. This is of special importance in advanced stage lung cancer owing to the high frequency and short onset of brain metastases from lung cancer.
Neurocognitive impairment has been found frequently in long-term survivors of SCLC after prophylactic WBRT [
81,
82] and has been seen in patients with existing brain metastases as well. In a cohort of 98 patients with single brain metastasis, four of 38 patients (11%) who survived ≥ 1 year after postoperative WBRT developed severe dementia associated with ataxia and urinary incontinence [
83]. All four patients were among a group of 23 patients (17%) who were treated with hypofractionated WBRT with fractions larger than 3 Gy/day. These toxicities were not observed in the patients who were treated with fractions ≤3 Gy/day. However, similar toxicities were seen in one patient who was treated with 3 Gy/day combined with intra-arterial chemotherapy. These findings suggest that large fractions and radiosensitizers, such as chemotherapy, may contribute to severe neurocognitive deficits in long-term survivors from brain metastases. However, such effects may not surface if the patients survive for less than one year. On the other hand, neurocognitive impairment was observed shortly after starting WBRT when patients underwent serial neurocognitive testing as shown by Welzel
et al. [
84]. But those authors recommended not avoiding WBRT since the neurocognitive dysfunction was restricted mainly to verbal memory. In addition, the risk of disease progression will always outweigh the risk of neurocognitive deficits secondary to brain irradiation since most recurrences can be associated with a neurologic deficit [
85].
Some investigators believe that the neurocognitive outcome is directly related to intracranial tumor response after cranial irradiation, as neurocognitive deficits can be partially explained by intracranial tumor progression [
86]. Furthermore, improvement in neurocognitive function in responding patients with multiple brain metastases also depends on the initial and posttreatment tumor volume [
87,
88]. In a study by Li
et al., a battery of standardized neurocognitive tests was administered monthly for six months and then every three months until death by trained and certified nurses or clinical research associates to patients with unresectable brain metastases who were treated with WBRT [
88]. At two months, patients with greater tumor shrinkage were found to have longer median survival, higher survival rate at one year, and longer time to neurocognitive deterioration. The cognitive gain was especially prominent in executive function and fine motor coordination. Nine patients were alive at 15 months, and the correlation between tumor shrinkage and executive function as well as fine motor coordination persisted. Furthermore, neurocogntive function was found to be influenced mostly by disease progression early on after WBRT. The patients who became long-term survivors also experienced larger tumor volume reductions after WBRT, and they had the best neurocognitive outcome.
The combination of SRS and WBRT over WBRT alone has been supported by the results of RTOG 9508 for patients with single brain metastasis, good functional performance status, and no active extracranial disease [
62]. No difference in neurological deaths or mental status at six months between the two arms of this study was found. In addition, the rate of neurological deaths in the SRS boost arm was within the 25–50% range reported in other surgery or SRS series [
62]. Because of the known toxicity associated with WBRT and the lack of any difference in survival between SRS alone and SRS plus WBRT as described in previous sections, the use of SRS alone as initial treatment for patients with a limited number of lesions has been advocated by many. The difference in neurocognitive function between patients undergoing SRS alone and those undergoing SRS and WBRT has been investigated in two prospective randomized controlled trials [
66,
89]. In the study by Aoyama
et al. [
66], neurocognitive function was assessed by serial MMSE after SRS + WBRT or SRS alone. No statistical difference in MMSE scores was found between the two arms, nor was any statistically significant difference found in the rate of MMSE score deterioration after a median follow up of 5.3 months. However, the time to neurological deterioration was significantly longer in patients who received SRS + WBRT than in those who received SRS alone (16.5 months
versus 7.6 months,
p = 0.05). This was thought to reflect the higher number of intracranial recurrences in the SRS alone group (11
versus 3 patients,
p < 0.0001). Five patients who underwent SRS + WBRT, but none in the SRS alone arm, suffered a radiation toxic event. Although not statistically significant, a trend of continuous neurocognitive deterioration became prominent after 24 months in long-term survivors after SRS and WBRT. These findings from Aoyama
et al. corroborate those from Regine
et al. and Li
et al. in that WBRT may help to improve neurocognitive function in patients with brain metastasis through its therapeutic effects on a short-term basis [
66,
86,
88]. Moreover, significant numbers of patients treated with SRS alone may experience recurrence with neurological symptoms, leading to the recommendation that WBRT be used whenever indicated [
85]. However, the late toxicity in terms of neurocognitive function from WBRT in long-term survivors cannot be ignored and warrants further investigation, as such effects may be masked owing to the short survival time of many patients on these studies of mostly far less than two years. Recently, the effects of initial treatment with SRS alone or SRS combined with WBRT on learning and memory function were investigated in a prospective randomized study by Chang
et al. [
89]. Most patients in this study had NSCLC, 1-2 lesions, and RPA class I or II. The GPA indices between the two arms were also well balanced. This study was designed to detect a 5-point decline in the Hopkins Verbal Learning Test–Revised (HVLT-R). The study was stopped when a significant decline in the HVLT-R score at four months was observed in the SRS plus WBRT arm compared with the SRS alone arm after accrual of 58 patients. Overall, the total recall difference persisted at six months. The patients who received SRS + WBRT demonstrated greater declines in executive function as well. Increased intracranial failure was observed in the SRS alone arm, with approximately 87% of the patients requiring salvage therapy. However, the one year survival rate was higher in the SRS alone arm (63%
versus 21%,
p = 0.003), possibly because of earlier systemic therapy in the SRS group and greater systemic disease burden in the SRS + WBRT arm. The authors argued for the initial treatment to be SRS alone with close follow up since intracranial recurrences are likely to be asymptomatic if discovered in their early stages by imaging studies.
Given the evidence described above, WBRT does seem to have a toxic effect on neurocognitive function over time. However, neurocognitive deterioration can be observed only in long-term survivors. Therefore, WBRT may be omitted in patients with good functional performance status and limited numbers of metastases if those patients have limited extracranial disease and are aware of the risk of intracranial failure associated with SRS alone and the risk of potential neurological deficits as a result of such failures. Thus, we recommend offering SRS alone to patients who can be monitored closely (e.g., every two months) with MRI. SRS plus WBRT should still be given serious consideration for patients with good functional performance status with controlled extracranial disease and single brain metastasis given the observed survival benefit observed in RTOG 9508 [
62]. In contrast, WBRT can actually improve neurocognitive function of patients with radiosensitive tumors, such as lung cancer, poor prognosis, and a short lifespan. Thus WBRT should be recommended for such patients. In recent years, donepezil, a drug used to treat Alzheimer’s disease, was shown to have a positive effect on the cognitive function of patients who underwent irradiation for brain tumors [
90]. The potential role of memantine, an agent that blocks the pathologic stimulation of the
N-methyl-
d-aspartate (NMDA) receptor (a receptor involved in learning and memory), in alleviating neurocognitive deficits from WBRT is being investigated in a randomized Phase III study, RTOG 0614, with results pending [
91].
Also worth mentioning is the potential contribution of anticonvulsants to the development of late neurological symptoms from WBRT [
92]. Therefore, any systemic agents (e.g., anticonvulsants, steroids) that can possibly influence the symptomatic outcome from brain irradiation should be carefully assessed and controlled for in future prospective studies to reach firm conclusions regarding the incidence of late radiation toxicity from brain irradiation.
8. Future Investigations
Given the lack of any survival benefit demonstrated for adjuvant WBRT after surgical resection or SRS and its potential neurotoxicity, the utility of SRS to provide a boost dose to the tumor bed after craniotomy for patients with limited numbers of brain metastases has been investigated in recent years. In a study of 72 patients (43% NSCLC) with 1-4 brain metastases and 76 cavities after surgical resection, a median dose of 18.0 Gy was delivered to the median 79% isodose line at the periphery of the tumor bed [
138]. The actuarial local control rate in this study was 79% at two years, and the distant control rate was 47% at 12 months. Three patients underwent surgical resection of a region of necrosis. Use of less conformal plans translated into a local control rate of 100%, thus the authors recommended a planning target volume margin of 2 mm around the resection cavity. In a similar study of 52 patients (46% NSCLC) with up to four lesions, a local failure rate of 7.7% was observed after a median follow up of 13 months [
139]. The distant failure rate was 44% after a median of 16 months after resection, and the median survival was 15 months. Similar results have been reported in other single-institution studies [
141,
142]. Equivalent local control between surgery followed by adjuvant WBRT or SRS is suggested by these small studies, but these findings remain to be validated in a prospective randomized study. However, the risk of distant recurrence remains high with adjuvant SRS alone, making this approach inappropriate for patients with solitary brain metastasis, good functional performance status, and primary disease controlled locally because of the potential for aggressive treatments to improve survival in these patients, especially for patients with lung cancer [
142,
143]. Therefore, WBRT may still be warranted in patients with good prognosis in addition to surgery and adjuvant SRS, if the potential toxicity is tolerable. The feasibility of this approach was investigated in a small study of 27 patients (70% NSCLC); the actuarial two-year local control was 94%, and the two year actuarial incidence of new brain metastasis was 30% [
144]. Only one patient required reoperation for symptomatic radiation necrosis at 16 months after treatment. The median survival was 17.6 months. Whether this approach will lead to a survival benefit still requires further investigation in a randomized trial.
As mentioned previously, SRS can spare adjacent normal tissue by achieving a sharp dose gradient at the periphery of the tumor target volume. However, this advantage is diminished with large lesions. To spare normal brain tissue, the dose delivered needs to be decreased to avoid potential neurotoxicity [
51,
145]. The tumor response is usually impaired as a result of this [
146]. Therefore, fractionated stereotactic radiotherapy has been proposed owing to the advantages of reoxygenation of hypoxic cells within large lesions and significant increases in late-responding tissue sparing if the radiation dose is fractionated [
147]. Thus, the therapeutic ratio can be significantly increased when large brain metastases are treated with a high dose delivered in several fractions with a stereotactic set-up. This concept has been validated in a small study, in which patients with large brain metastases (average volume 21.2 cm
3) were treated safely with fractionated stereotactic radiotherapy, and a local control rate of 83% has been achieved [
148]. These findings were confirmed in a larger retrospective study of patients with large brain metastases treated with this technique [
149]. Similarly, many single-institution studies have shown excellent clinical outcome and toxicity profile from fractionated stereotactic radiotherapy with or without use of a frame (
Table 10). These studies suggest that local control seems to be related to tumor size [
155] and that intracranial control outside of the treated area seems to be poor when fractionated stereotactic radiotherapy is used alone. However, its combination with WBRT was shown to be feasible with good intracranial control and toxicity profile [
156,
157], which may be a better option for patients who present with neurological deficits from large, but few, brain metastases. However, the patient selection criteria for this technique, alone or as a boost, needs further investigation in prospective studies. Also, it is important to be aware that a bigger margin than that used for SRS may be needed when patients are not precisely immobilized.
Table 10.
Selected studies of fractionated stereotactic radiotherapy.
Table 10.
Selected studies of fractionated stereotactic radiotherapy.
| | N (patients/ metastases) | Dose (Gy) | Response | 1-year Overall Survival (%) | Distant Failure in the Brain | Toxicity |
|---|
| De Salles et al. [148] | 26/41 | 6 Gy × 2-3 | LC 83% | NR | NR | NR |
| FU 2-18 mo |
| Nishizaki et al. [149] | 71/148 | 7.8-30.1 Gy/1-3 fractions | Median FU: 11 mo | 47 | 35.2 % at a median FU of 6.6 mo | No permanent symptoms from radionecrosis |
| LC: 83% |
| Manning et al. [150] | 32/57 | 6-12 Gy × 3; | 31% ≤ 25% | 44 | 13% after ≥ 6 mo | Seizure: 12% |
| WBRT for all patients | 31% > 25% | Radionecrosis: 6% |
| 16% PD | |
| Aoyama et al. [151] | 87/159 | 8.75 Gy × 4 | 1 yr LC: 81% | 39 | 60% at 1 yr | Symptomatic radionecrosis: 2.7% |
| 2 yr LC: 69% |
| Aoki et al. [152] | 44/65 | 18-30 Gy/ 3-5 fractions | 1 yr LC: 71.9% | 50.8 | 31% at 1 yr | No severe complications |
| Ernst-Stecken et al. [153] | 51/72 | 6 Gy × 5 | 1 yr LC: 76% | Median survival: 11 mo | NR | Increased radionecrosis if V4Gy > 23 cm3 (70% vs. 14%, p = 0.001) |
| 7 Gy × 5 |
| SRT alone or with WBRT |
| Fahrig et al. [154] | 150/228 | 6-7 Gy × 5 | CR: 46% | 66 | NR | 10% toxicity; 1 symptomatic hemorrhage from melanoma; 2 had to be operated on for radionecrosis; |
| 4 Gy × 10 | 31% |
| 5 Gy × 7 | 47% |
| | Median FU: 28 mo | No grade 5 toxicity; |
| No toxicity with 4 Gy × 10. |
| Kwon et al. [155] | 36/66 | 20-36 Gy/ 4-6 | 1 yr LC: 68.2%; | 43.9 | 25.9% at a median FU of 6.47 mo | Radionecrosis: 5.8% |
| Tumors < 1 cm3 had better LC |
| Lindvall et al. [156] | 61/77 | 8 Gy × 5 | LC: 84% (no WBRT); 100% (WBRT); mean 3.7 mo after SRT | Mean survival from time of SRT: 6.1 mo | 25% after a median of 3.7 mo after SRT alone, but none in WBRT + SRT patients who were followed radiologically | 4.7% radionecrosis after SRT + WBRT |
| ± WBRT |
| Giubilei et al. [157] | 30/44 | 6 Gy × 3 | 1 yr LC: 86.1% | 36.6 | 12.1% at 1 yr | No acute or late complications |
| 8 Gy × 4 |
| WBRT for all patients |
In patients with good prognostic factors, such as 1–3 brain metastases, RPA class 1–2, controlled extracranial disease, GPA of ≥ 2.5, young age, and high KPS scores, aggressive treatment of brain metastasis with WBRT followed by a regular external beam boost to the gross tumor or the surgical bed has been investigated. This approach has been consistently shown to be associated with increased local control comparable to that reported for WBRT + SRS, as well as a median survival time of over 12 months, significantly better than WBRT alone with or without surgical resection [
158,
159]. Radiation delivered as a simultaneous integrated boost with intensity-modulated radiotherapy has been shown to produce a sharper dose gradient than that from WBRT followed by SRS for the treatment of brain metastasis [
160,
161]. This leads to improved normal tissue sparing owing to the ability to optimize the dose to the normal brain and to account for dose spillage from the boost dose to the adjacent brain tissue in the planning of WBRT. This not only spares more normal brain tissue but also shortens treatment time for patients with multiple lesions. The feasibility of this approach has been demonstrated in a single-institution Phase I study through the use of helical tomotherapy, which combines delivery of intensity-modulated radiotherapy with megavoltage CT imaging for precise radiation delivery through image guidance [
162]. In this study, 48 patients (50% of whom had lung cancer) were treated with WBRT 30 Gy in 10 fractions and a simultaneous integrated boost to the brain metastases safely escalated from 5 to 30 Gy in 10 fractions. No grade 3–5 dose limiting toxicity was encountered. However, this study had a median follow up of only 7.72 months and a median overall survival time of only 5.29 months. Given the small number of patients in this study, no firm conclusions can be made regarding tumor response and survival outcome. Worth mentioning is the application of helical tomotherapy in the treatment of recurrent brain metastases from lung and breast cancer with a simultaneous integrated boost. The safe delivery of 30 Gy to the gross disease and 15 Gy to the whole brain in 10 fractions for up to 11 lesions was reported by Sterzing
et al. [
163]. In this report, an excellent dose conformality index was achieved, and no severe toxicity was observed; the patients remained recurrence-free at six and 12 months of follow up. Based on this limited evidence, the simultaneous integrated boost approach may be an excellent treatment approach with intensity modulation of doses to the target and the adjacent brain tissue. Depending on the degree of immobilization, no standard has been established regarding the planning target volume margin for gross disease, and margins from 0 to 10 mm have been reported [
158,
161,
162,
163]. Further conclusions regarding this matter can be made as the existing data mature. Recently, excellent dose sparing of radiosensitive structures, such as the hippocampus, was reported when brain metastases were treated with the simultaneous integrated boost approach, which further supports the use of this approach for normal tissue sparing [
164,
165].
Other future work involves targeting therapy to the epidermal growth factor receptor (EGFR) family of four homologous receptors, EGFR (ERBB1), HER-2/neu (ERBB2), HER-3 (ERBB3), and HER-4 (ERBB4). EGFR activation leads to receptor tyrosine-kinase activation and activation of a series of downstream signaling activities that mediate tumor cell proliferation, migration, invasion, and the suppression of apoptosis [
166]. As a result, inhibiting EGFR by binding its intracellular adenosine triphosphate-binding site with small-molecule tyrosine kinase inhibitors has been investigated as a treatment strategy for NSCLC [
167]. Among these inhibitors, erlotinib has shown a survival benefit when combined with chemotherapy for advanced stage NSCLC [
168]. However, tumor response is mainly limited to patients who possess somatic mutations in the kinase domain of the EGFR gene [
169]. These patients are usually of East Asian descent, female, nonsmokers, with adenocarcinoma [
170]. In a small series of 41 patients with brain metastasis from lung adenocarcinoma, gefitinib was shown to have antitumor activity (10% major response). Intracranial control was associated with previous WBRT [
171]. In another study by Kim
et al. [
172], median progression-free survival and overall survival times of 7.1 and 18.8 months were observed in East Asian, nonsmoking patients with lung adenocarcinoma and asymptomatic synchronous brain metastasis after treatment with either gefitinib 250 mg or erlotinib 150 mg once daily. Both studies suggest a potential role for EGFR inhibitors in the treatment of brain metastases. Additive effects may be produced when both EGFR inhibitors and WBRT were delivered, as patients who received both treatments have better disease control and longer overall survival [
171,
172]. Several studies have demonstrated increased response to EGFR inhibitors, as well as prolonged time to intracranial progression and improved overall survival, in patients with mutations in the EGFR gene [
173,
174]. The presence of EGFR mutations was shown to enhance radiation response; for patients with EGFR mutation and brain metastases from lung adenocarcinoma, WBRT delivered concurrently with EGFR inhibitors produced a response rate of 84% [
175]. However, severe toxicities, including grade 5 interstitial lung disease, have been reported in patients treated with concurrent erlotinib and WBRT [
176,
177]. The unexpected lung toxicity from EGFR inhibitors needs further investigation for the safe administration of these drugs.