The Migration of Cancer Cells in Gradually Varying Chemical Gradients and Mechanical Constraints
Abstract
:1. Introduction
2. Experiments
2.1. Device Design and Fabrication
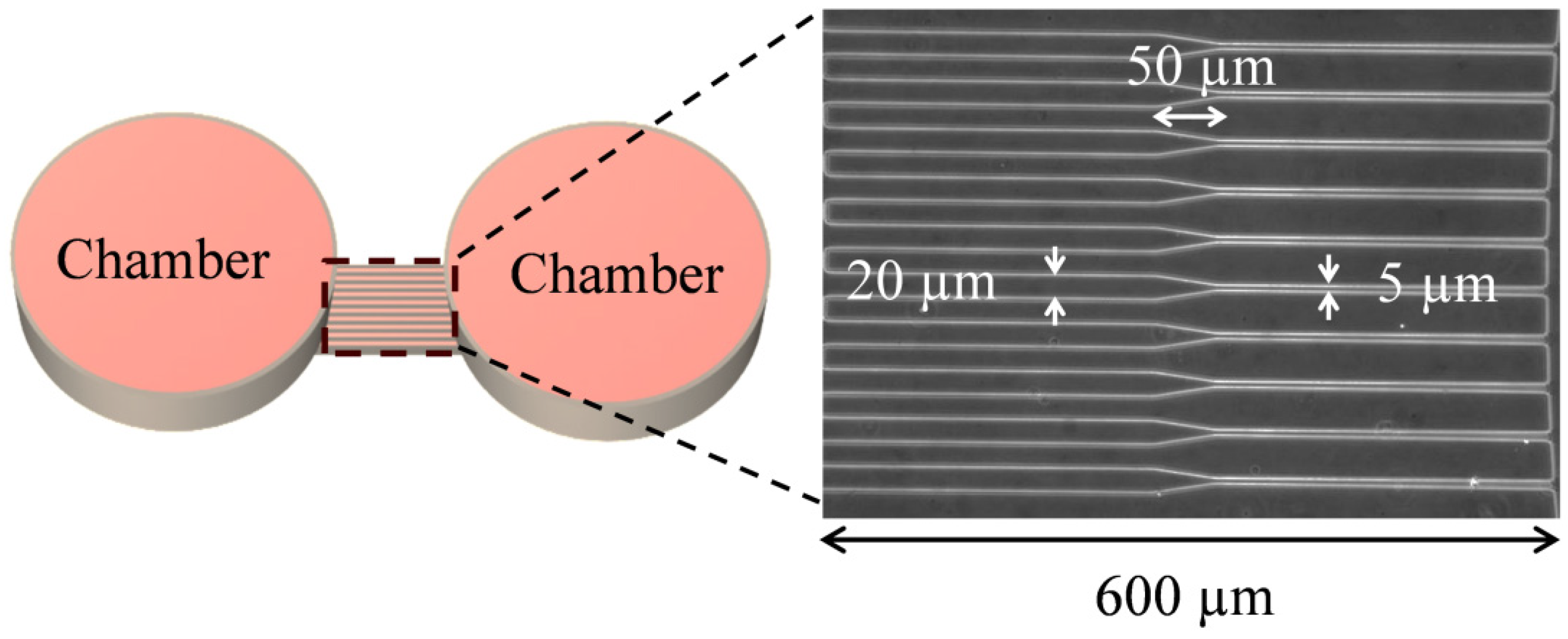
2.2. Cell Migration



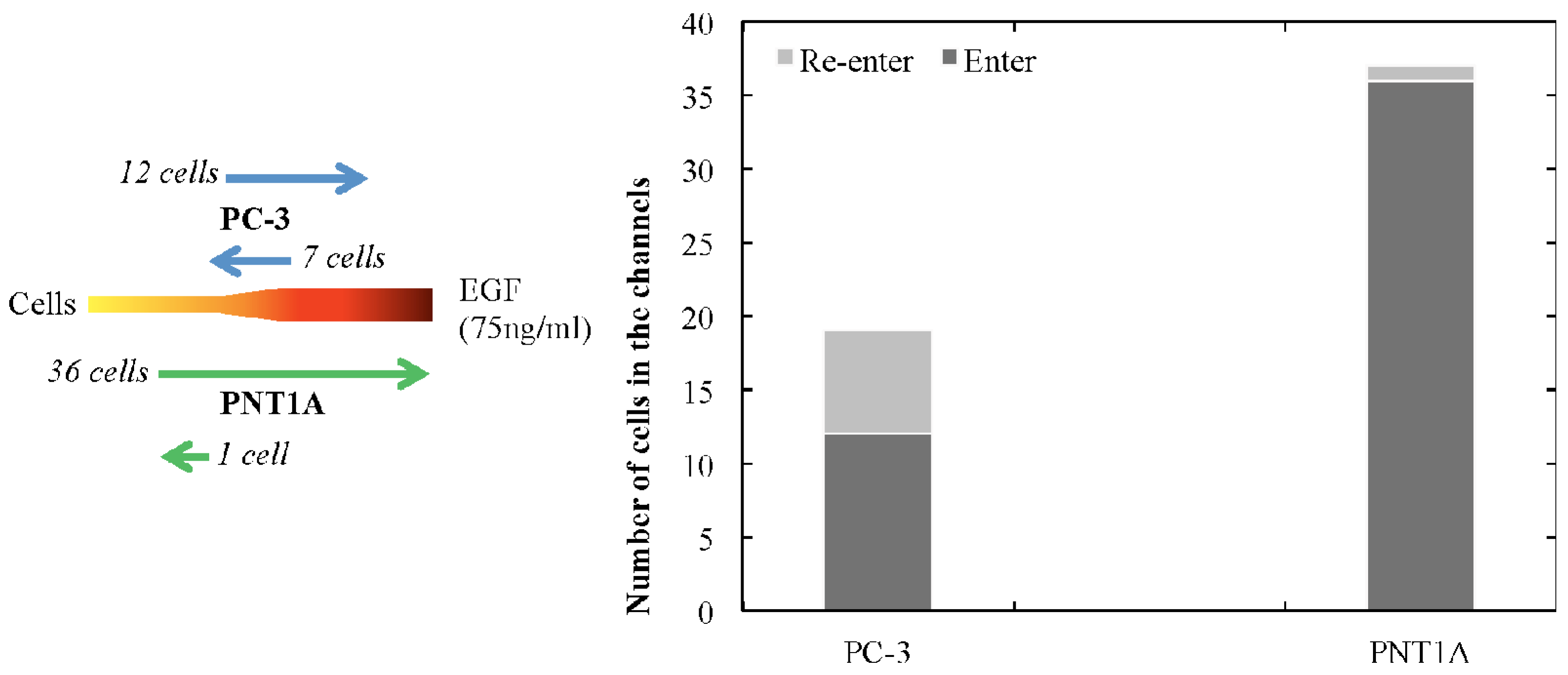
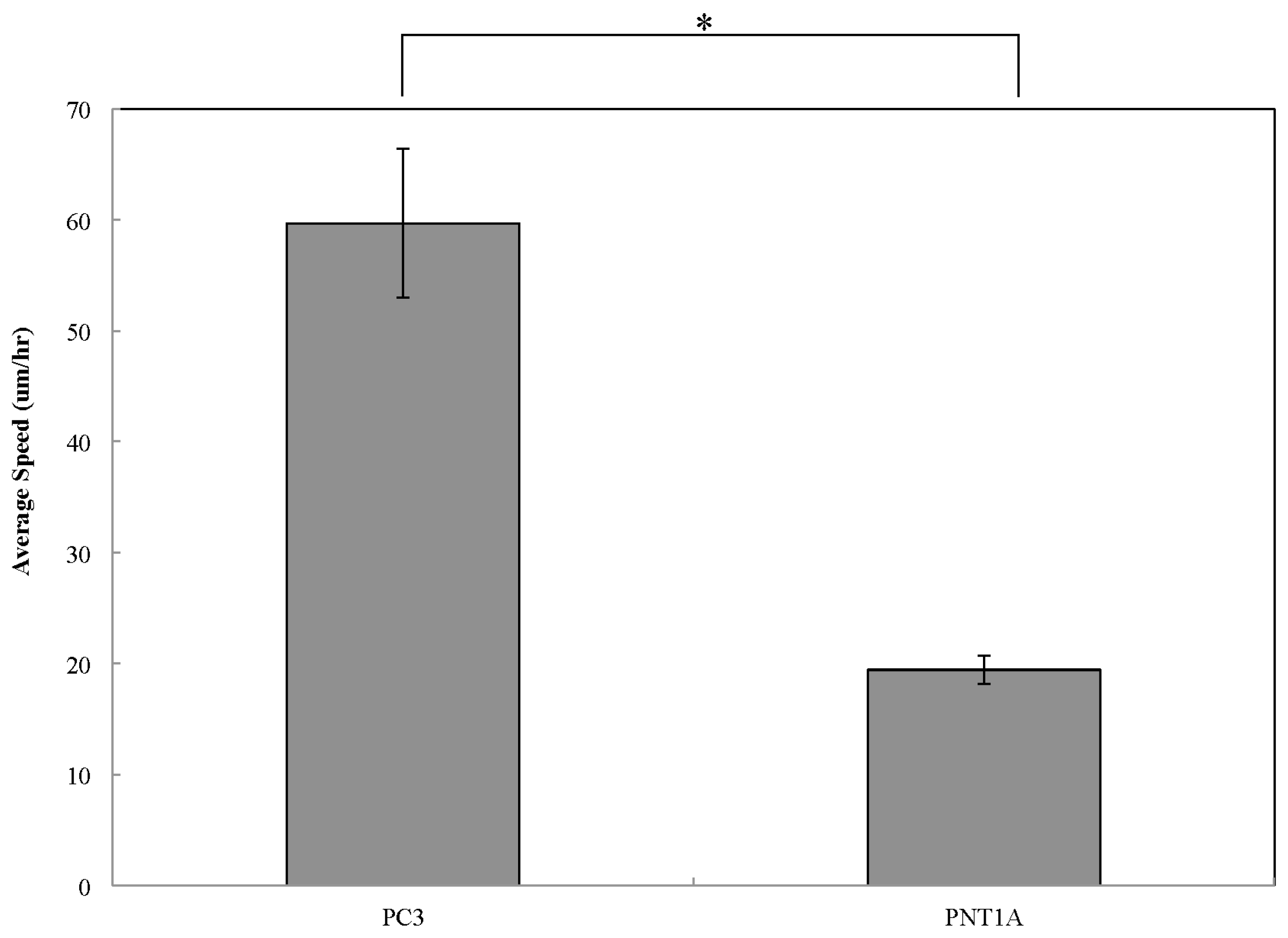
3. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Transforming Growth Factor-β in Cancer Therapy. In Cancer Treatment and Therapy; Jakowlew, S.B. (Ed.) Humana Press: Totowa, NJ, USA, 2008; Volume II.
- Kang, Y.; He, W.; Tulley, S.; Gupta, G.P.; Serganova, I.; Chen, C.R.; Manova-Todorova, K.; Blasberg, R.; Gerald, W.L.; Massague, J. Breast cancer bone metastasis mediated by the smad tumor suppressor pathway. Proc. Natl. Acad. Sci. USA 2005, 102, 13909–13914. [Google Scholar] [CrossRef]
- Polacheck, W.J.; Li, R.; Uzel, S.G.M.; Kamm, R.D. Microfluidic platforms for mechanobiology. Lab Chip 2013, 13, 2252–2267. [Google Scholar] [CrossRef]
- Seton-Rogers, E.S.; Lu, Y.; Hines, M.L.; Koundinya, M.; LaBaer, J.; Muthuswamy, K.S.; Brugge, S.J. Cooperation of the ErbB2 receptor and transforming growth factor beta in induction of migration and invasion in mammary epithelial cells. Proc. Natl. Acad. Sci. USA 2004, 101, 1257–1262. [Google Scholar] [CrossRef]
- Xie, L.; Law, K.B.; Chytil, M.A.; Brown, A.K.; Aakre, E.M.; Moses, L.H. Activation of the Erk pathway is required for TGF-β1-induced EMT in vitro. Neoplasia 2004, 6, 603–610. [Google Scholar] [CrossRef]
- Serganova, I.; Moroz, E.; Vider, J.; Gogiberidze, G.; Moroz, M.; Pillarsetty, N.; Doubrovin, M.; Minn, A.; Thaler, H.T.; Massague, J.; et al. Multimodality imaging of TGFβ signaling in breast cancer metastases. FASEB J. 2009, 23, 2662–2672. [Google Scholar] [CrossRef]
- Kaminska, B.; Wesolowska, A.; Danilkiewicz, M. TGF Beta signalling and its role in tumour pathogenesis. Acta Biochim. Pol. 2005, 52, 329–337. [Google Scholar]
- Kanzaki, T.; Shiina, R.; Saito, Y.; Morisaki, H.O.A.N. Role of latent TGF-β1 binding protein in vascular remodeling. Biochem. Biophys. Res. Commun. 1998, 246, 26–30. [Google Scholar] [CrossRef]
- Wikström, P.; Stattin, P.; Franck-Lissbrant, I.; Anders Bergh, J.-E.D. Transforming growth factor β1 is associated with angiogenesis, metastasis, and poor clinical outcome in prostate cancer. Prostate 1998, 37, 19–29. [Google Scholar] [CrossRef]
- Rajan, R.; Vanderslice, R.; Kapur, S.; Lynch, J.; Thompson, R.; Djakiew, D. Epidermal growth factor (EGF) promotes chemomigration of a human prostate tumor cell line, and EGF immunoreactive proteins are present at sites of metastasis in the stroma of lymph nodes and medullary bone. Prostate 1996, 28, 1–9. [Google Scholar] [CrossRef]
- Wang, J.H.; Li, B. Mechanics rules cell biology. Sports Med. Arthrosc. Rehabil. Ther. Technol. 2010, 2. [Google Scholar] [CrossRef]
- Ingber, D.E. Cellular mechanotransduction: Putting all the pieces together again. FASEB J. 2006, 20, 811–827. [Google Scholar] [CrossRef]
- Chambers, A.F.; Groom, A.C.; MacDonald, I.C. Dissemination and growth of cancer cells in metastatic sites. Nat. Rev. Cancer 2002, 2, 563–572. [Google Scholar] [CrossRef]
- Dieterich, P.; Klages, R.; Preuss, R.; Schwab, A. Anomalous dynamics of cell migration. Proc. Natl. Acad. Sci. USA 2008, 105, 459–463. [Google Scholar] [CrossRef]
- Liu, B.; Kim, T.J.; Wang, Y. Live cell imaging of mechanotransduction. J. R. Soc. Interf. 2010, 7, S365–S375. [Google Scholar] [CrossRef]
- Orr, A.W.; Helmke, B.P.; Blackman, B.R.; Schwartz, M.A. Mechanisms of mechanotransduction. Dev. Cell 2006, 10, 11–20. [Google Scholar] [CrossRef]
- Bio-MEMS Technologies and Applications, 1st ed.; Wang, W.; Soper, S.A. (Eds.) CRC Press: Boca Raton, FL, USA, 2006.
- Microfluidics for Biological Applications; Tian, W.; Finehout, E. (Eds.) Springer: New York, NY, USA, 2008.
- Abhyankar, V.V.; Toepke, M.W.; Cortesio, C.L.; Lokuta, M.A.; Huttenlocher, A.; Beebe, D.J. A platform for assessing chemotactic migration within a spatiotemporally defined 3D microenvironment. Lab Chip 2008, 8, 1507–1515. [Google Scholar] [CrossRef]
- Campbell, C.J.; Grzybowski, B.A. Microfluidic mixers: From microfabricated to self-assembling devices. Philos. Transact. A Math. Phys. Eng. Sci. 2004, 362, 1069–1086. [Google Scholar] [CrossRef]
- Cha, J.; Kim, J.; Ryu, S.-Y.; Park, J.; Jeong, Y.; Park, S.; Park, S.; Kim, H.C.; Chun, K. A highly efficient 3D micromixer using soft PDMS bonding. J. Micromech. Microeng. 2006, 16, 1778–1782. [Google Scholar] [CrossRef]
- Chang, J.K.; Bang, H.; Park, S.J.; Chung, S.; Han, C.; Chung, D.C. Fabrication of the PDMS Microchip for Serially Diluting Sample with Buffer. In Microsystem Technologies; Springer-Verlag: Berlin, Germany, 2003; Volume 9, pp. 555–558. [Google Scholar]
- Diao, J.; Young, L.; Kim, S.; Fogarty, E.A.; Heilman, S.M.; Zhou, P.; Shuler, M.L.; Wu, M.; DeLisa, M.P. A Three-channel microfluidic device for generating static linear gradients and its application to the quantitative analysis of bacterial chemotaxis. Lab Chip 2006, 6, 381–388. [Google Scholar] [CrossRef]
- Duffy, D.C.; McDonald, J.C.; Schueller, O.J.A.; Whitesides, G.M. Rapid prototyping of microfluidic systems in poly(dimethylsiloxane). Anal. Chem. 1998, 70, 4974–4984. [Google Scholar] [CrossRef]
- Ghitun, M.; Bonneil, E.; Fortier, M.; Yin, H.; Killeen, K.; Thibault, P. Integrated microfluidic devices with enhanced separation performance: Application to phosphoproteome analyses of differentiated cell model systems. J. Seper. Sci. 2006, 29, 1539–1549. [Google Scholar] [CrossRef]
- Irimia, D.; Geba, D.A.; Toner, M. Universal microfluidic gradient generator. Anal. Chem. 2006, 78, 3472–3477. [Google Scholar] [CrossRef]
- Jeon, N.L.; Dertinger, S.K.W.; Chiu, D.T.; Choi, I.S.; Stroock, A.D.; Whitesides, G.M. Generation of solution and surface gradients using microfluidic systems. Langmuir 2000, 16, 8311–8316. [Google Scholar] [CrossRef]
- Jiang, X.; Bruzewicz, D.A.; Wong, A.P.; Piel, M.; Whitesides, G.M. Directing cell migration with asymmetric micropatterns. Proc. Natl. Acad. Sci. USA 2005, 102, 975–978. [Google Scholar]
- Leclerc, E.; Sakai, Y.; Fujii, T. A Multi-Layer PDMS Microfluidic Device for Tissue Engineering Applications. In Proceedings of the IEEE the 16th Annual International Conference on Micro Electro Mechanical Systems, Kyoto, Japan, 23 January 2003; pp. 415–418.
- Li, J.; LeRiche, T.; Tremblay, T.-L.; Wang, C.; Bonneil, E.; Harrison, D.J.; Thibault, P. Application of microfluidic devices to proteomics research: Identification of trace-level protein digests and affinity capture of target peptides. Mol. Cell. Proteomics 2002, 1, 157–168. [Google Scholar] [CrossRef]
- Martin, R.S.; Root, P.D.; Spence, D.M. Microfluidic technologies as platforms for performing quantitative cellular analyses in an in vitro environment. Analyst 2006, 131, 1197–1206. [Google Scholar] [CrossRef]
- McDonald, J.C.; Duffy, D.C.; Anderson, J.R.; Chiu, D.T.; Wu, H.; Schueller, O.J.; Whitesides, G.M. Fabrication of microfluidic systems in poly(Dimethylsiloxane). Electrophoresis 2000, 21, 27–40. [Google Scholar] [CrossRef]
- McDonald, J.C.; Whitesides, G.M. Poly(Dimethylsiloxane) as a Material for fabricating microfluidic devices. Acc. Chem. Res. 2002, 35, 491–499. [Google Scholar] [CrossRef]
- Nie, F.; Kobayashi, J.; Yamada, M.; Yamato, M.; Kikuchi, A.; Okano, T. Cell Migration Assay Using Multiple Laminar Flows in PDMS Microchannel. In Proceedings of the International Symposium on Micro-NanoMechatronics and Human Science MHS ’07, Nagoya, Japan, 11–14 November 2007; pp. 406–409.
- O’Neill, A.T.; Monteiro-Riviere, N.; Walker, G.M. A serial dilution microfluidic device for cytotoxicity assays. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2006, 1, 2836–2839. [Google Scholar]
- Park, J.W.; Vahidi, B.; Taylor, A.M.; Rhee, S.W.; Jeon, N.L. Microfluidic culture platform for neuroscience research. Nat. Protoc. 2006, 1, 2128–2136. [Google Scholar] [CrossRef]
- Saadi, W.; Wang, S.; Lin, F.; Jeon, N.L. A parallel-gradient microfluidic chamber for quantitative analysis of breast cancer cell chemotaxis. Biomed. Microdevices 2006, 8, 109–118. [Google Scholar] [CrossRef]
- Saadi, W.; Rhee, S.W.; Lin, F.; Vahidi, B.; Chung, B.G.; Jeon, N.L. Generation of stable concentration gradients in 2D and 3D environments using a microfluidic ladder chamber. Biomed. Microdevices 2007, 9, 627–635. [Google Scholar] [CrossRef]
- Sia, S.K.; Whitesides, G.M. Microfluidic devices fabricated in poly(Dimethylsiloxane) for biological studies. Electrophoresis 2003, 24, 3563–3576. [Google Scholar] [CrossRef]
- Situma, C.; Hashimoto, M.; Soper, S.A. Merging microfluidics with microarray-based bioassays. Biomol. Eng. 2006, 23, 213–231. [Google Scholar] [CrossRef]
- Snyder, J.D.; Desai, T.A. Microscale three-dimensional polymeric platforms for in vitro cell culture systems. J. Biomater. Sci. Polym. Ed. 2001, 12, 921–932. [Google Scholar] [CrossRef]
- Taylor, A.M.; Rhee, S.W.; Jeon, N.L. Microfluidic chambers for cell migration and neuroscience research. Methods Mol. Biol. 2006, 321, 167–177. [Google Scholar]
- Walker, G.M.; Monteiro-Riviere, N.; Rouse, J.; O’Neill, A.T. A linear dilution microfluidic device for cytotoxicity assays. Lab Chip 2007, 7, 226–232. [Google Scholar] [CrossRef]
- Li Jeon, N.; Baskaran, H.; Dertinger, S.K.; Whitesides, G.M.; van de Water, L.; Toner, M. Neutrophil chemotaxis in linear and complex gradients of interleukin-8 formed in a microfabricated device. Nat. Biotechnol. 2002, 20, 826–830. [Google Scholar] [CrossRef]
- Lin, F.; Nguyen, C.M.; Wang, S.; Saadi, W.; Gross, S.P.; Jeon, N.L. Neutrophil migration in opposing chemoattractant gradients using microfluidic chemotaxis devices. Ann. Biomed. Eng. 2005, 33, 475–482. [Google Scholar] [CrossRef]
- Irimia, D.; Toner, M. Spontaneous migration of cancer cells under conditions of mechanical confinement. Integr. Biol. 2009, 1, 506–512. [Google Scholar] [CrossRef]
- Rao, S.M.N.; Lin, V.K.; Tata, U.; Raj, G.V.; Hsieh, J.-T.; Nguyen, K.; Chiao, J.-C. Demonstration of cancer cell migration using a novel microfluidic device. J. Nanotechnol. Eng. Med. 2010, 1. [Google Scholar] [CrossRef]
- Tata, U.; Rao, S.M.N.; Sharma, A.; Pabba, K.; Pokhrel, K.; Adhikari, B.; Lin, V.K.; Chiao, J.C. Study of lung-metastasized prostate cancer cell line chemotaxis to epidermal growth factor with a BIOMEMS device. Adv. Nat. Sci. Nanosci. Nanotechnol. 2012, 3. [Google Scholar] [CrossRef]
- Bretscher, M.S. Getting membrane flow and the cytoskeleton to cooperate in moving cells. Cell 1996, 87, 601–606. [Google Scholar] [CrossRef]
- Iijima, M.; Huang, Y.E.; Devreotes, P. Temporal and spatial regulation of chemotaxis. Dev. Cell 2002, 3, 469–478. [Google Scholar] [CrossRef]
- Charest, P.G.; Firtel, R.A. Feedback signaling controls leading-edge formation during chemotaxis. Curr. Opin. Genet. Dev. 2006, 16, 339–347. [Google Scholar] [CrossRef]
- Sasaki, A.T.; Firtel, R.A. Regulation of chemotaxis by the orchestrated activation of ras, PI3K, and TOR. Eur. J. Cell Biol. 2006, 85, 873–895. [Google Scholar] [CrossRef]
- Janetopoulos, C.; Firtel, R.A. Directional sensing during chemotaxis. FEBS Lett. 2008, 582, 2075–2085. [Google Scholar] [CrossRef]
- Steeg, P.S. Metastasis suppressors alter the signal transduction of cancer cells. Nat. Rev. Cancer 2003, 3, 55–63. [Google Scholar] [CrossRef]
- Stephens, L.; Milne, L.; Hawkins, P. Moving towards a better understanding of chemotaxis. Curr. Biol. 2008, 18, R485–R494. [Google Scholar] [CrossRef]
- Signal Transduction in Cancer Metastasis; Wu, W.; Hu, C.H. (Eds.) Springer: Dordrecht, The Netherlands, 2010.
- Mak, M.; Reinhart-King, C.A.; Erickson, D. Microfabricated physical spatial gradients for investigating cell migration and invasion dynamics. PLoS One 2011, 6. [Google Scholar] [CrossRef]
- Harris, J.; Lee, H.; Vahidi, B.; Tu, C.; Cribbs, D.; Cotman, C.; Jeon, N.L. Non-plasma bonding of PDMS for inexpensive fabrication of microfluidic devices. J. Vis. Exp. 2007, 9. [Google Scholar] [CrossRef]
- ata, U.; Rao, S.; Nguyen, K.; Lin, V.K.; Chiao, J.C. A Microfluidic Approach to Study the Effect of Growth Factors on PC3 Cell Migration. In Proceedings of the BMES Biomedical Engineering Society Annual Meeting 2011, Hartford, CT, USA, 12–15 October 2011.
- Kaighn, M.E.; Narayan, K.S.; Ohnuki, Y.; Lechner, J.F.; Jones, L.W. Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Invest. Urol. 1979, 17, 16–23. [Google Scholar]
- Cussenot, O.; Berthon, P.; Berger, R.; Mowszowicz, I.; Faille, A.; Hojman, F.; Teillac, P.; le Duc, A.; Calvo, F. Immortalization of human adult normal prostatic epithelial cells by liposomes containing large T-SV40 gene. J. Urol. 1991, 146, 881–886. [Google Scholar]
- Han, J.; Lee, J.D.; Bibbs, L.; Ulevitch, R.J. A MAP kinase targeted by endotoxin and hyperosmolarity in mammalian cells. Science 1994, 265, 808–811. [Google Scholar]
- Bohil, A.B.; Robertson, B.W.; Cheney, R.E. Myosin-X is a molecular motor that functions in filopodia formation. Proc. Natl. Acad. Sci. USA 2006, 103, 12411–12416. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rao, S.M.N.; Tata, U.; Lin, V.K.; Chiao, J.-C. The Migration of Cancer Cells in Gradually Varying Chemical Gradients and Mechanical Constraints. Micromachines 2014, 5, 13-26. https://doi.org/10.3390/mi5010013
Rao SMN, Tata U, Lin VK, Chiao J-C. The Migration of Cancer Cells in Gradually Varying Chemical Gradients and Mechanical Constraints. Micromachines. 2014; 5(1):13-26. https://doi.org/10.3390/mi5010013
Chicago/Turabian StyleRao, Smitha M. N., Uday Tata, Victor K. Lin, and Jung-Chih Chiao. 2014. "The Migration of Cancer Cells in Gradually Varying Chemical Gradients and Mechanical Constraints" Micromachines 5, no. 1: 13-26. https://doi.org/10.3390/mi5010013




