Microfluidic Platform for Enzyme-Linked and Magnetic Particle-Based Immunoassay
Abstract
:1. Introduction
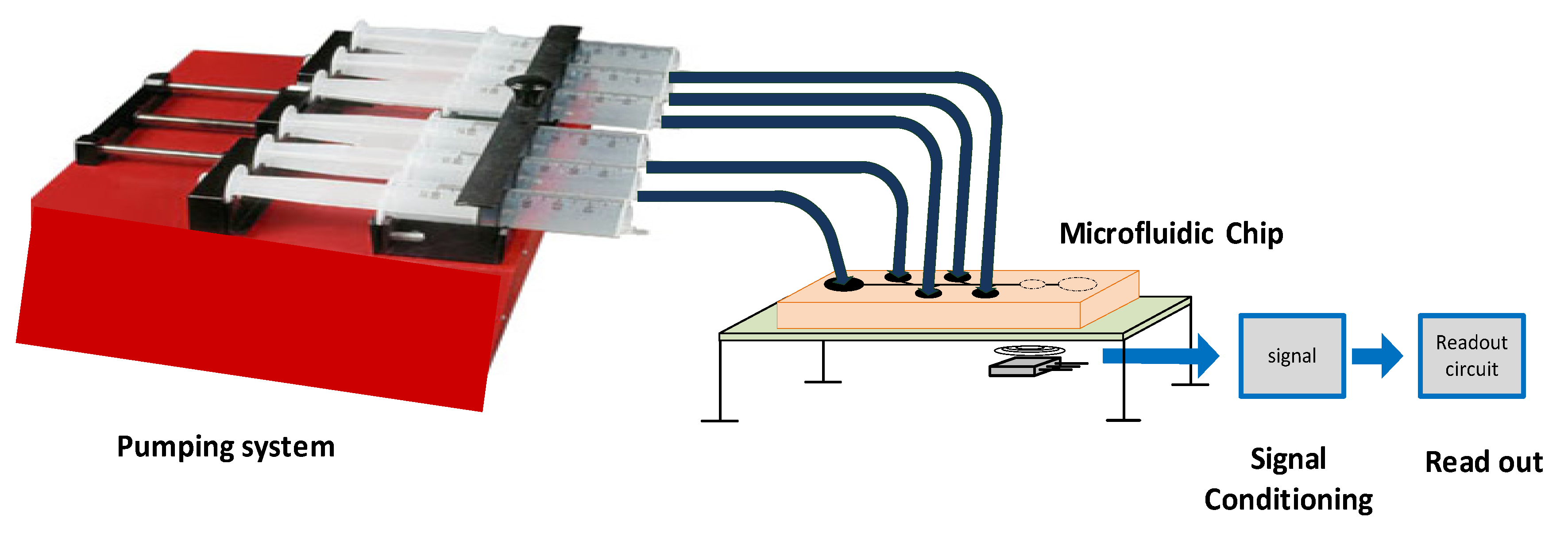
2. Experimental Section
2.1. Materials and Immunoassay Protocol
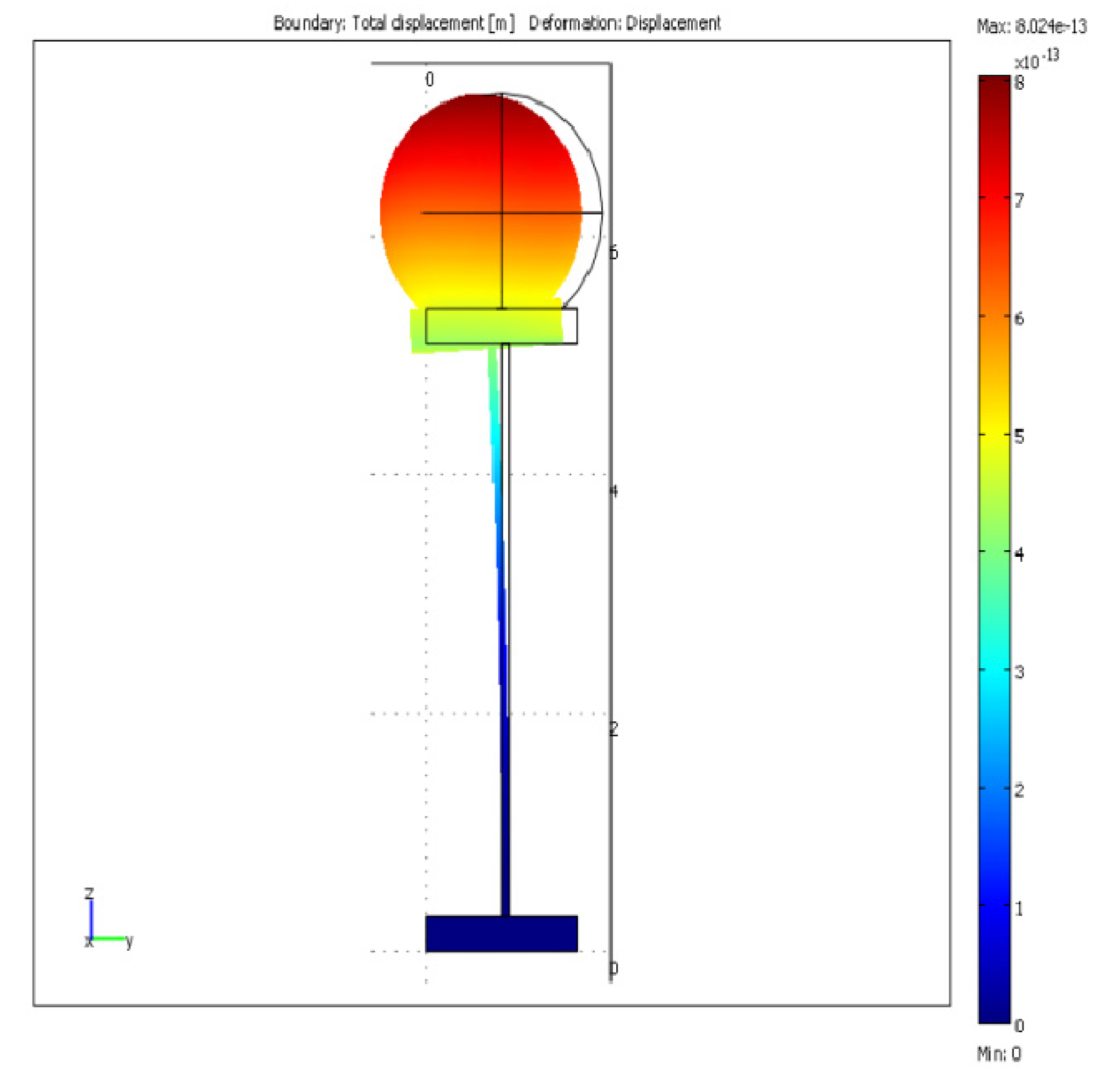
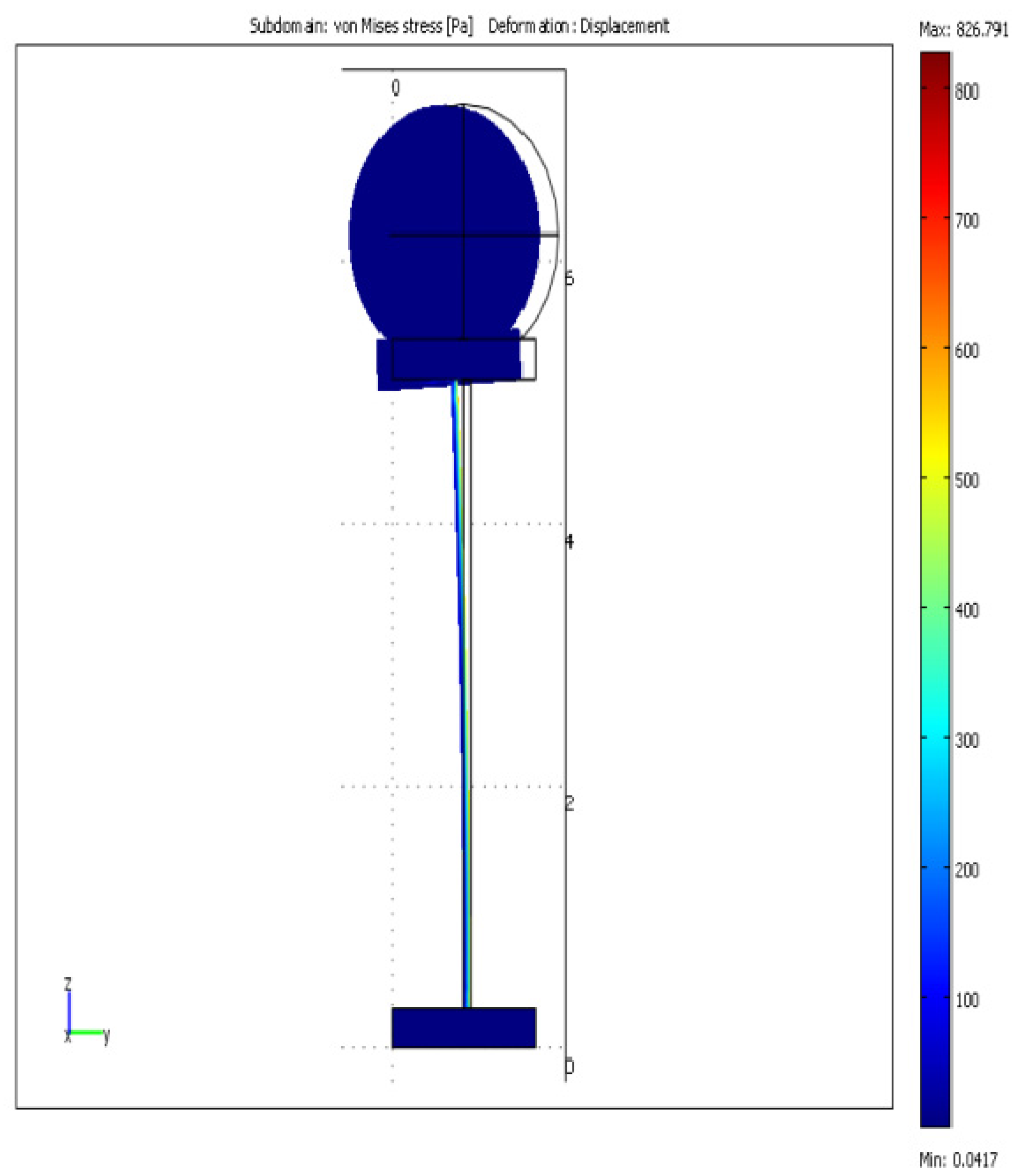
2.2. Bio-Complex Mechanical Modeling
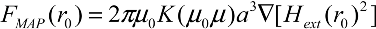
| Average Weight (Kilograms) | Length (Angstroms) | Width (Angstroms) | |
|---|---|---|---|
| Adiponectin | 1.66 × 10−27 | 500 | 8 |
| Antibody/Anti-Antibody | 6.64 × 10−23 | 30 | 150 |
| Magnetic Bead | 10 × 10−18 | Average spherical diameter of 10 nm | |
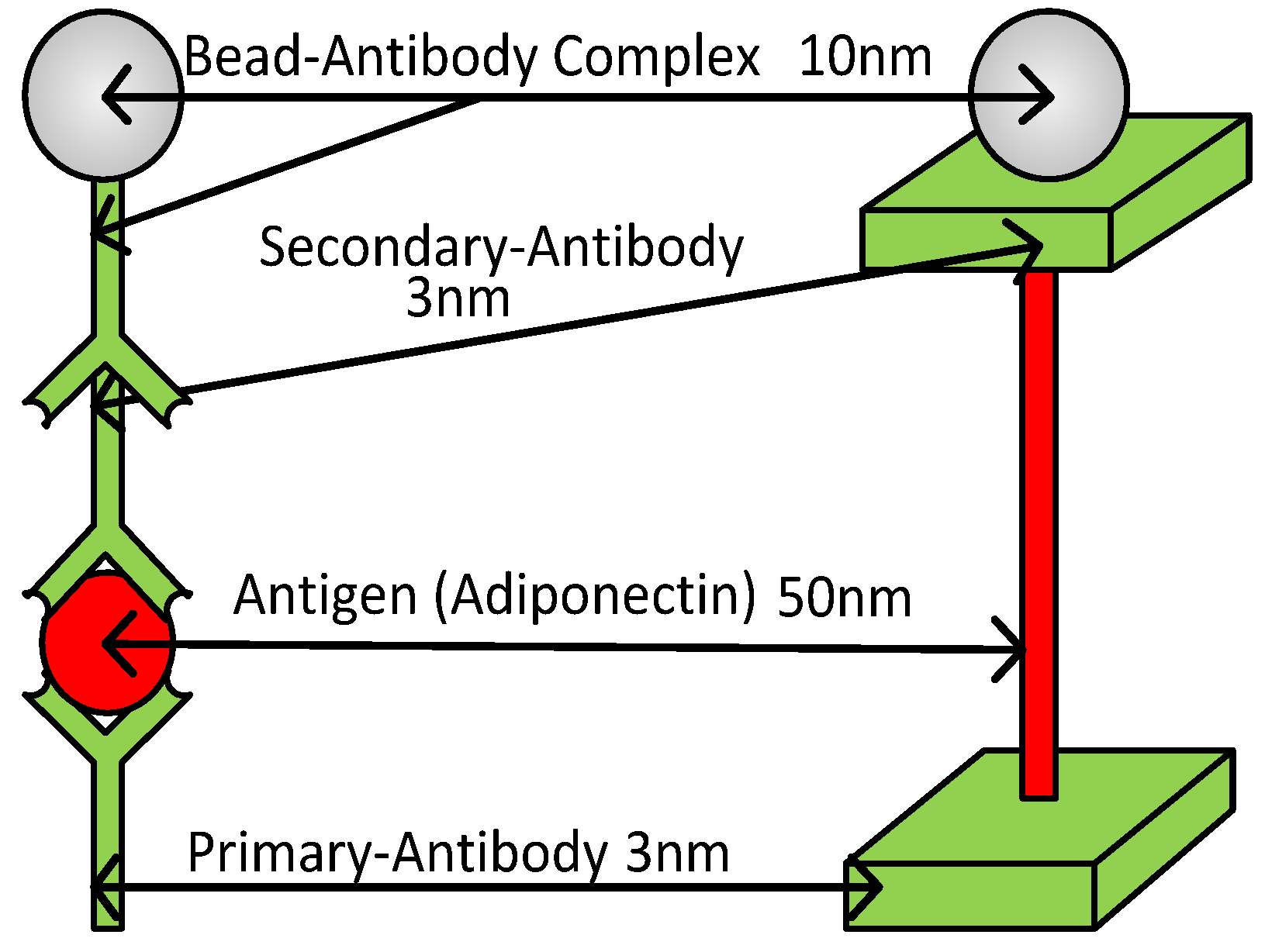
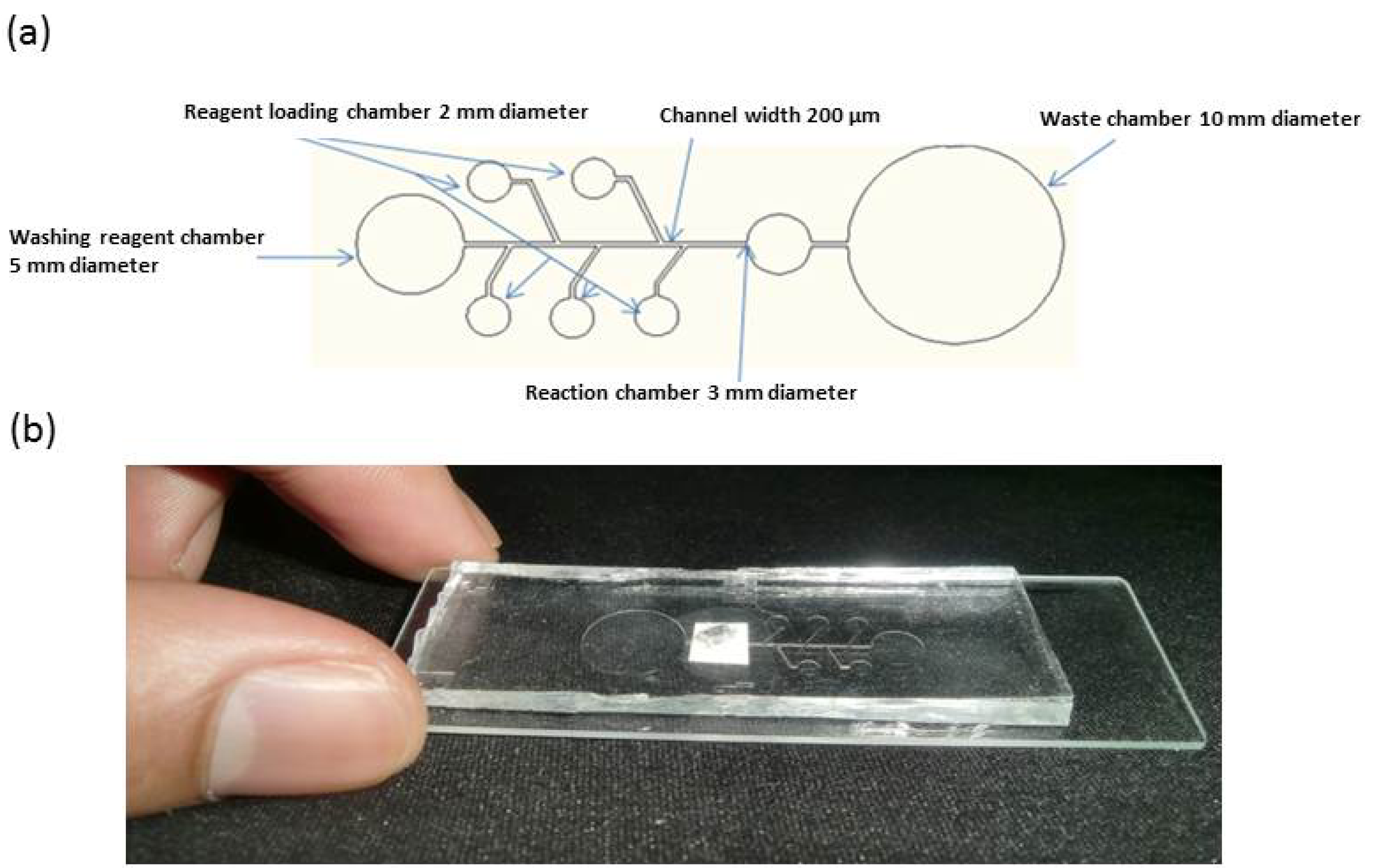
2.3. Fabrication of Microfluidic Platform
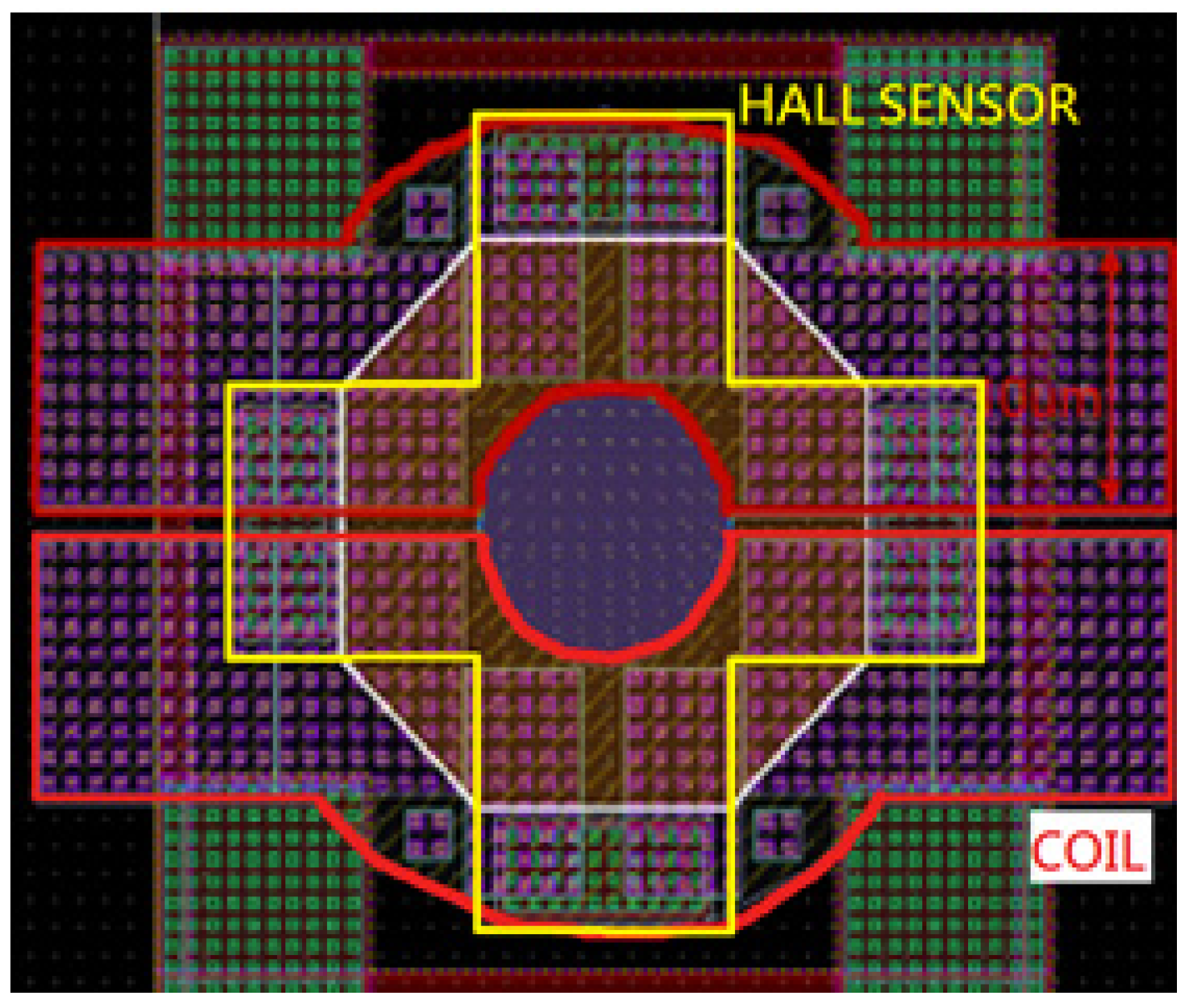
2.4. Hall Sensor Fabrication
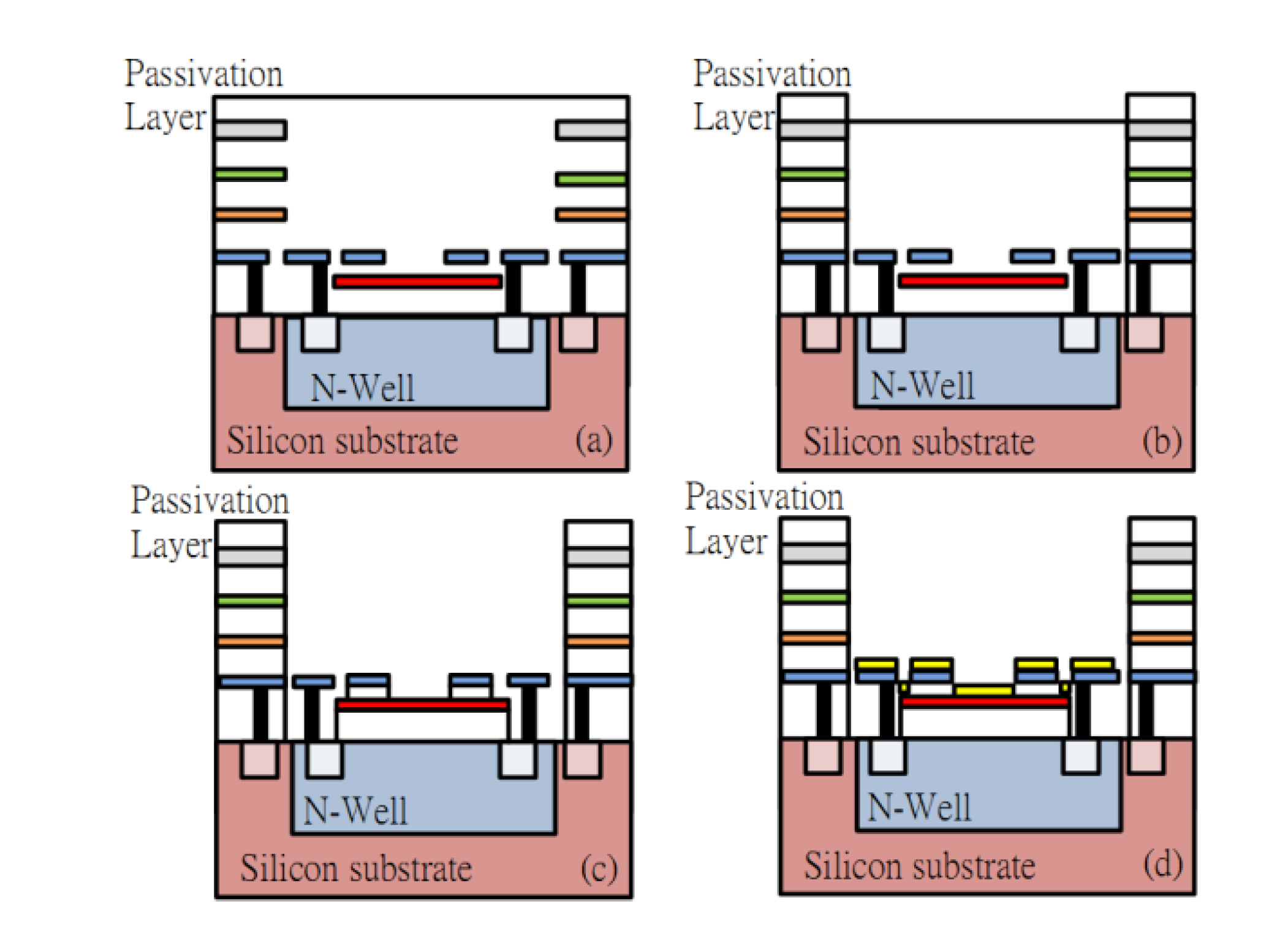
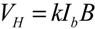
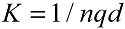
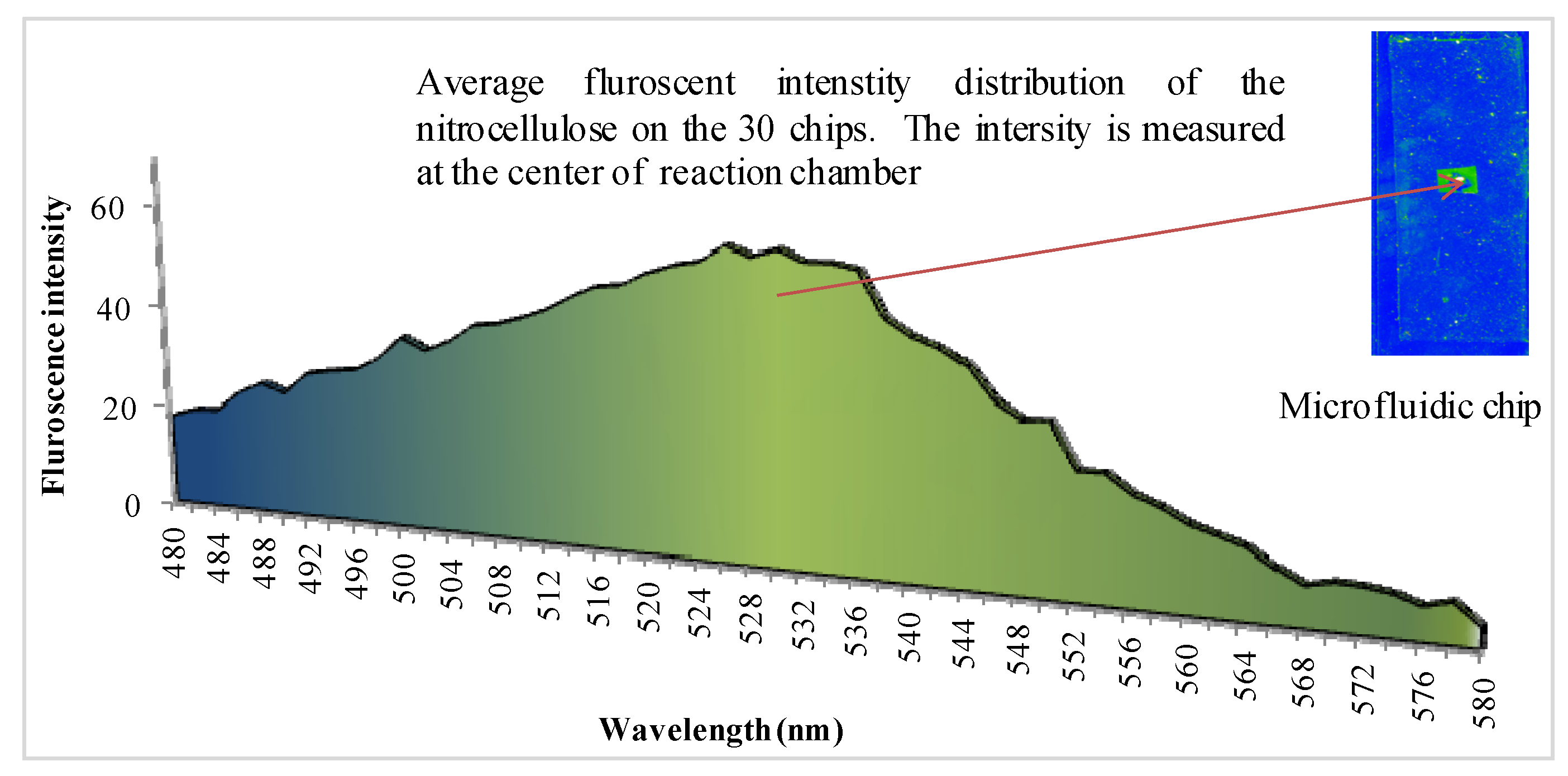
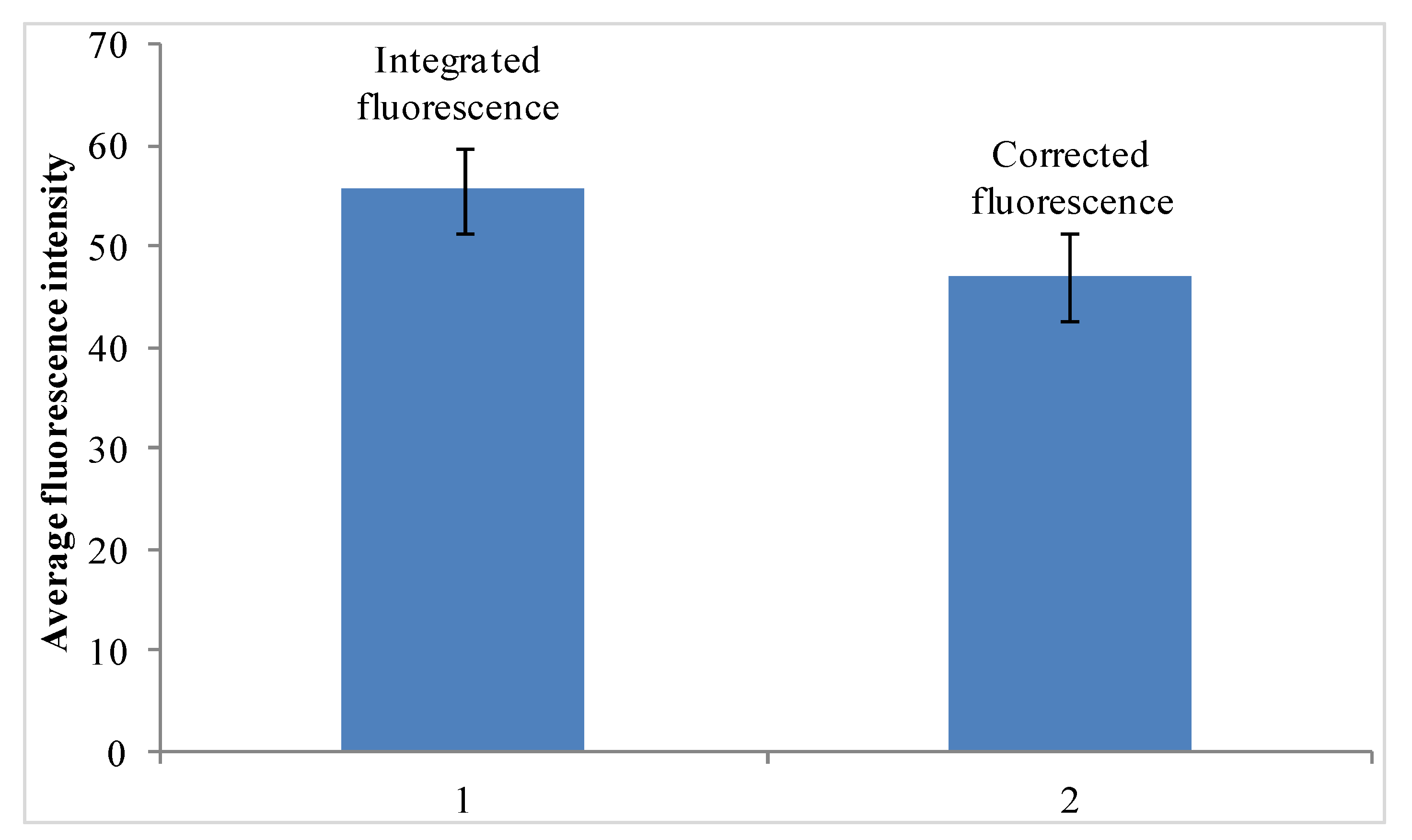
3. Results and Discussion
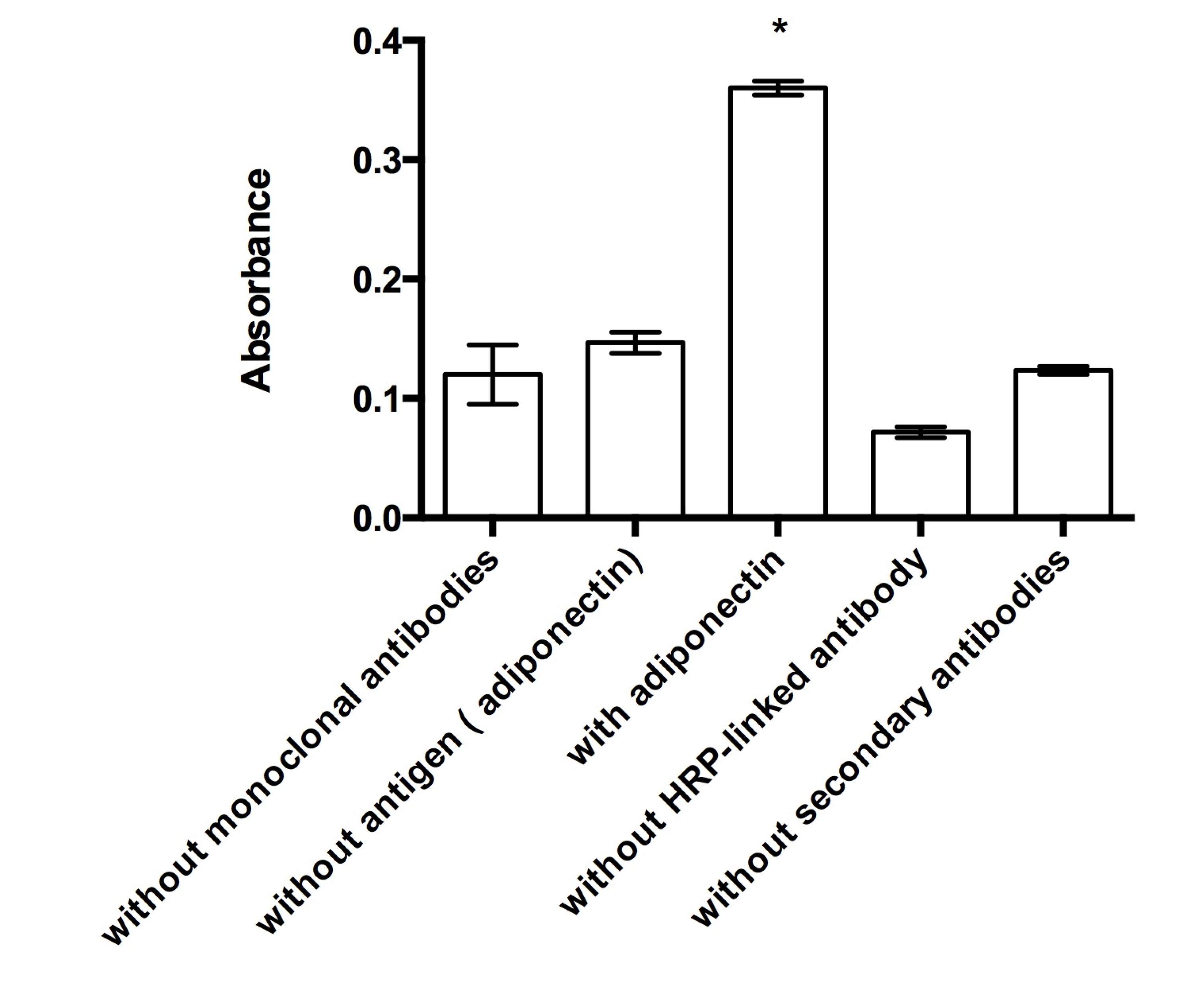
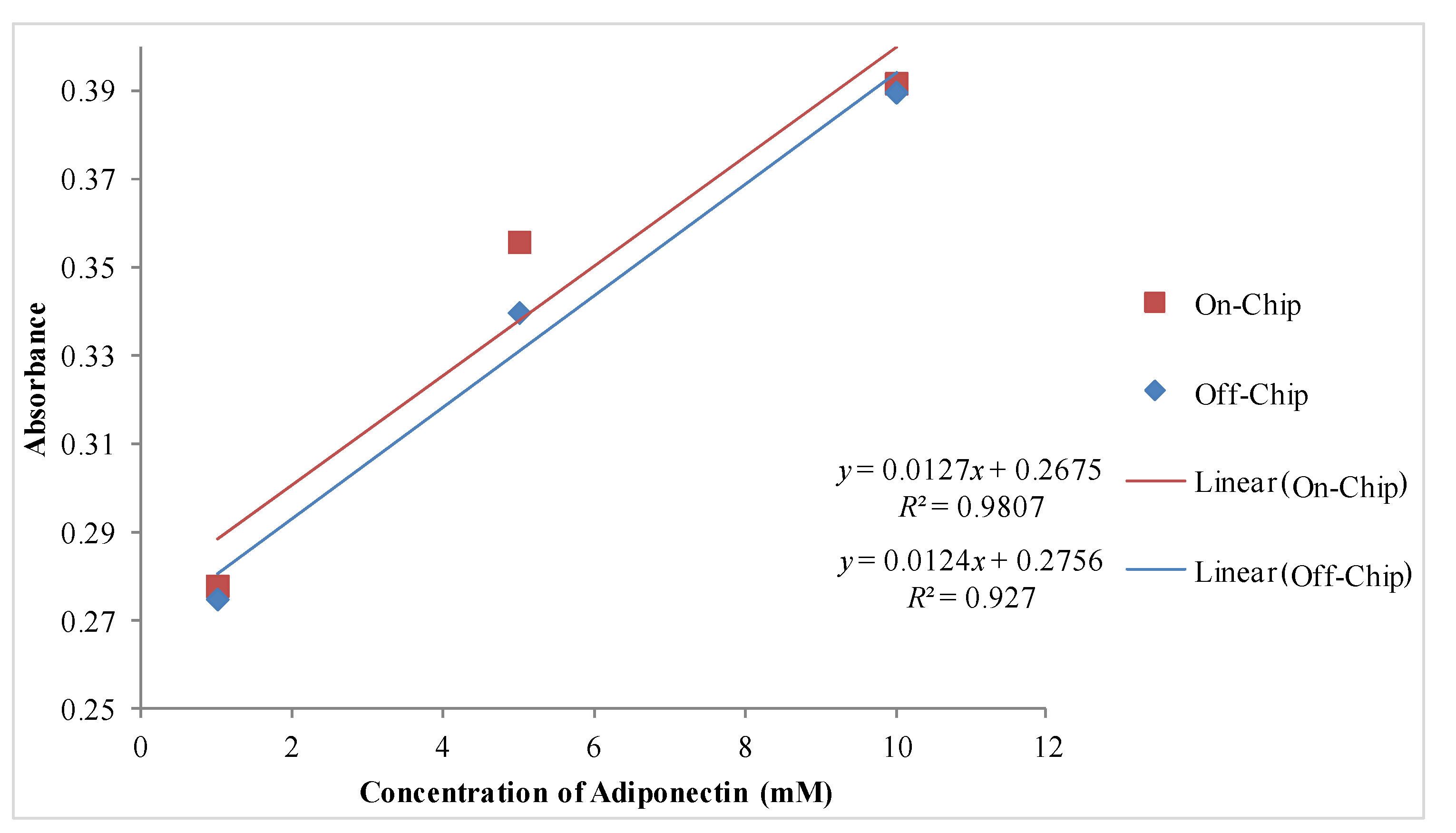

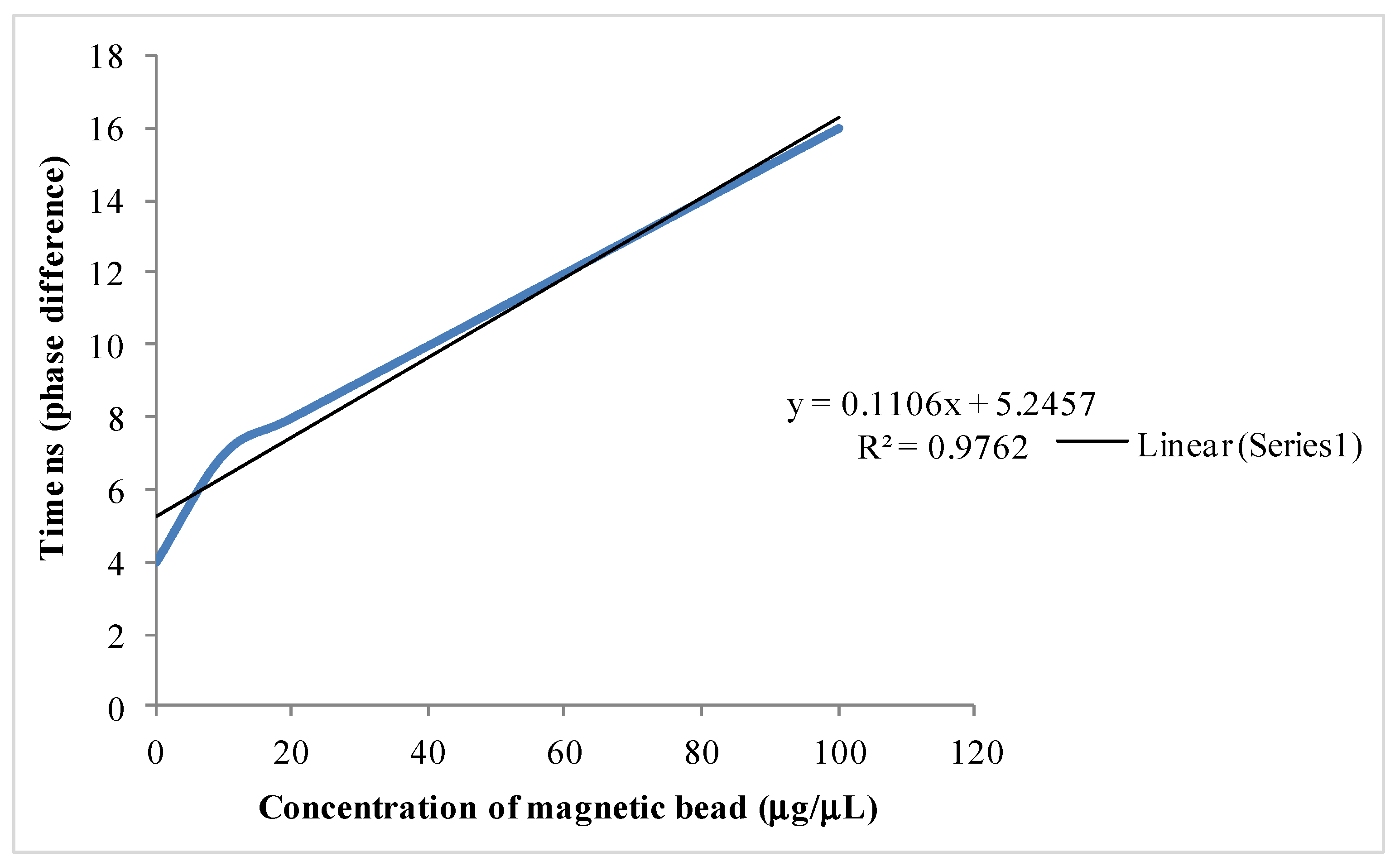

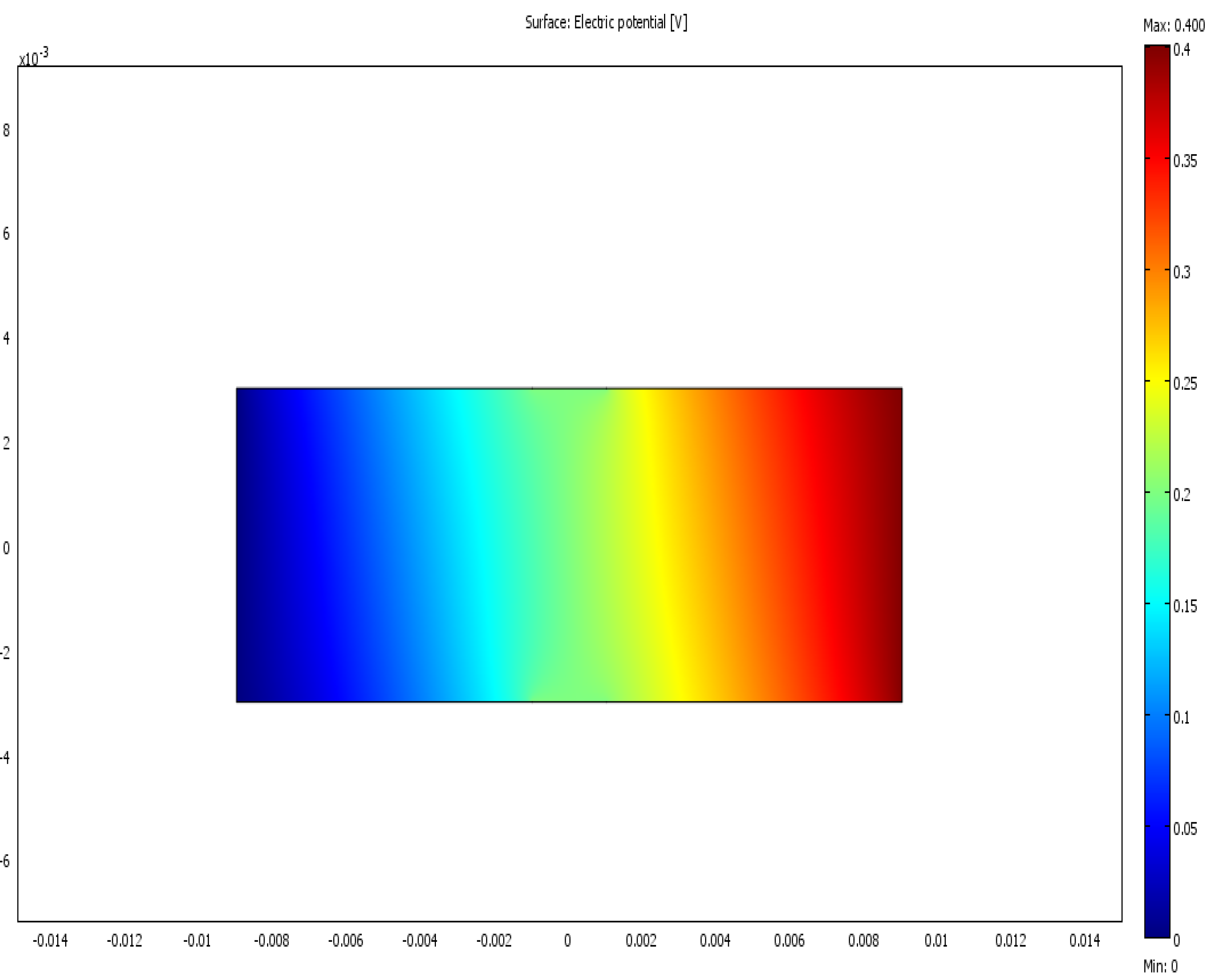
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Chin, C.D.; Laksanasopin, T.; Cheung, Y.K.; Steinmiller, D.; Linder, V.; Parsa, H.; Wang, J.; Moore, H.; Rouse, R.; Umviligihozo, G.; et al. Microfluidics-based diagnostics of infectious diseases in the developing world. Nat. Med. 2011, 17, 1015–1019. [Google Scholar] [CrossRef]
- Srinivasan, V.; Pamula, V.K.; Fair, R.B. Droplet-based microfluidic lab-on-a-chip for glucose detection. Anal. Chim. Acta 2004, 507, 145–150. [Google Scholar] [CrossRef]
- Kergaravat, S.V; Beltramino, L.; Garnero, N.; Trotta, L.; Wagener, M.; Isabel Pividori, M.; Hernandez, S.R. Electrochemical magneto immunosensor for the detection of anti-TG2 antibody in celiac disease. Biosens. Bioelectron. 2013, 48C, 203–209. [Google Scholar]
- Hung, L.-Y.; Chuang, Y.-H.; Kuo, H.-T.; Wang, C.-H.; Hsu, K.-F.; Chou, C.-Y.; Lee, G.-B. An integrated microfluidic platform for rapid tumor cell isolation, counting and molecular diagnosis. Biomed. Microdevices 2013, 15, 339–352. [Google Scholar] [CrossRef]
- Srinivasan, V.; Pamula, V.K.; Fair, R.B. An integrated digital microfluidic lab-on-a-chip for clinical diagnostics on human physiological fluids. Lab Chip 2004, 4, 310–315. [Google Scholar] [CrossRef]
- Yeo, L.Y.; Chang, H.-C.; Chan, P.P.Y.; Friend, J.R. Microfluidic devices for bioapplications. Small 2011, 7, 12–48. [Google Scholar] [CrossRef]
- Fair, R.B. Digital microfluidics: Is a true lab-on-a-chip possible? Microfluid. Nanofluid. 2007, 3, 245–281. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Waki, H.; Terauchi, Y.; Kubota, N.; Hara, K.; Mori, Y.; Ide, T.; Murakami, K.; Tsuboyama-Kasaoka, N.; et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 2001, 7, 941–946. [Google Scholar] [CrossRef]
- Bruls, D.M.; Evers, T.H.; Kahlman, J.A.H.; van Lankvelt, P.J.W.; Ovsyanko, M.; Pelssers, E.G.M.; Schleipen, J.J.H.B.; de Theije, F.K.; Verschuren, C.A.; van der Wijk, T.; et al. Rapid integrated biosensor for multiplexed immunoassays based on actuated magnetic nanoparticles. Lab Chip 2009, 9, 3504–3510. [Google Scholar] [CrossRef]
- Owen, D.; Mao, W.; Alexeev, A.; Cannon, J.; Hesketh, P. Multiplexed electrochemical immunoassay of biomarkers using metal sulfide quantum dot nanolabels and trifunctionalized magnetic beads. Micromachines 2013, 4, 103–115. [Google Scholar] [CrossRef]
- Owen, D.; Mao, W.; Alexeev, A.; Cannon, J.; Hesketh, P. Electrochemical magneto immunosensor for the detection of anti-TG2 antibody in celiac disease. Micromachines 2013, 4, 103–115. [Google Scholar] [CrossRef]
- Owen, D.; Mao, W.; Alexeev, A.; Cannon, J.; Hesketh, P. Microbeads for sampling and mixing in a complex sample. Micromachines 2013, 4, 103–115. [Google Scholar] [CrossRef]
- Aytur, T.; Foley, J.; Anwar, M.; Boser, B.; Harris, E.; Beatty, P.R. A novel magnetic bead bioassay platform using a microchip-based sensor for infectious disease diagnosis. J. Immunol. Methods 2006, 314, 21–29. [Google Scholar] [CrossRef]
- Lu, M.; Zhai, H.; Magnusson, R. Focusing light with curved guided-mode resonance reflectors. Micromachines 2011, 2, 150–156. [Google Scholar] [CrossRef]
- Chang, A.-Y.; Lu, M.S.-C. A CMOS magnetic microbead-based capacitive biosensor array with on-chip electromagnetic manipulation. Biosens. Bioelectron. 2013, 45, 6–12. [Google Scholar] [CrossRef]
- Florescu, O.; Wang, K.; Au, P.; Tang, J.; Harris, E.; Beatty, P.R.; Boser, B.E. On-chip magnetic separation of superparamagnetic beads for integrated molecular analysis. J. Appl. Phys. 2010, 107, 54702. [Google Scholar] [CrossRef]
- Florescu, O.; Mattmann, M.; Boser, B. Fully integrated detection of single magnetic beads in complementary metal-oxide-semiconductor. J. Appl. Phys. 2008, 103, 046101. [Google Scholar] [CrossRef]
- Bettazzi, F.; Hamid-Asl, E.; Esposito, C.L.; Quintavalle, C.; Formisano, N.; Laschi, S.; Catuogno, S.; Iaboni, M.; Marrazza, G.; Mascini, M.; et al. Electrochemical detection of miRNA-222 by use of a magnetic bead-based bioassay. Anal. Bioanal.Chem. 2013, 405, 1025–1034. [Google Scholar] [CrossRef]
- Zhou, C.-H.; Long, Y.-M.; Qi, B.-P.; Pang, D.-W.; Zhang, Z.-L. A magnetic bead-based bienzymatic electrochemical immunosensor for determination of H9N2 avian influenza virus. Electrochem. Commun. 2013, 31, 129–132. [Google Scholar]
- Choi, J.-W.; Oh, K.W.; Thomas, J.H.; Heineman, W.R.; Halsall, H.B.; Nevin, J.H.; Helmicki, A.J.; Henderson, H.T.; Ahn, C.H. An integrated microfluidic biochemical detection system for protein analysis with magnetic bead-based sampling capabilities. Lab Chip 2002, 2, 27–30. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Wang, S.-H.; Wu, M.-H.; Pan, T.-M.; Lai, C.-S.; Luo, J.-D.; Chiou, C.-C. Integrating solid-state sensor and microfluidic devices for glucose, urea and creatinine detection based on enzyme-carrying alginate microbeads. Biosens. Bioelectron. 2013, 43, 328–335. [Google Scholar] [CrossRef]
- Zhuang, J.; Fu, L.; Xu, M.; Zhou, Q.; Chen, G.; Tang, D. DNAzyme-based magneto-controlled electronic switch for picomolar detection of lead (II) coupling with DNA-based hybridization chain reaction. Biosens. Bioelectron. 2013, 45, 52–57. [Google Scholar] [CrossRef]
- Lu, A.-H.; Salabas, E.L.; Schüth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bhalla, N.; Chung, D.W.Y.; Chang, Y.-J.; Uy, K.J.S.; Ye, Y.Y.; Chin, T.-Y.; Yang, H.C.; Pijanowska, D.G. Microfluidic Platform for Enzyme-Linked and Magnetic Particle-Based Immunoassay. Micromachines 2013, 4, 257-271. https://doi.org/10.3390/mi4020257
Bhalla N, Chung DWY, Chang Y-J, Uy KJS, Ye YY, Chin T-Y, Yang HC, Pijanowska DG. Microfluidic Platform for Enzyme-Linked and Magnetic Particle-Based Immunoassay. Micromachines. 2013; 4(2):257-271. https://doi.org/10.3390/mi4020257
Chicago/Turabian StyleBhalla, Nikhil, Danny Wen Yaw Chung, Yaw-Jen Chang, Kimberly Jane S. Uy, Yi Ying Ye, Ting-Yu Chin, Hao Chun Yang, and Dorota G. Pijanowska. 2013. "Microfluidic Platform for Enzyme-Linked and Magnetic Particle-Based Immunoassay" Micromachines 4, no. 2: 257-271. https://doi.org/10.3390/mi4020257
APA StyleBhalla, N., Chung, D. W. Y., Chang, Y.-J., Uy, K. J. S., Ye, Y. Y., Chin, T.-Y., Yang, H. C., & Pijanowska, D. G. (2013). Microfluidic Platform for Enzyme-Linked and Magnetic Particle-Based Immunoassay. Micromachines, 4(2), 257-271. https://doi.org/10.3390/mi4020257




