The Impact of Fusarium Mycotoxins on Human and Animal Host Susceptibility to Infectious Diseases
Abstract
:1. Introduction
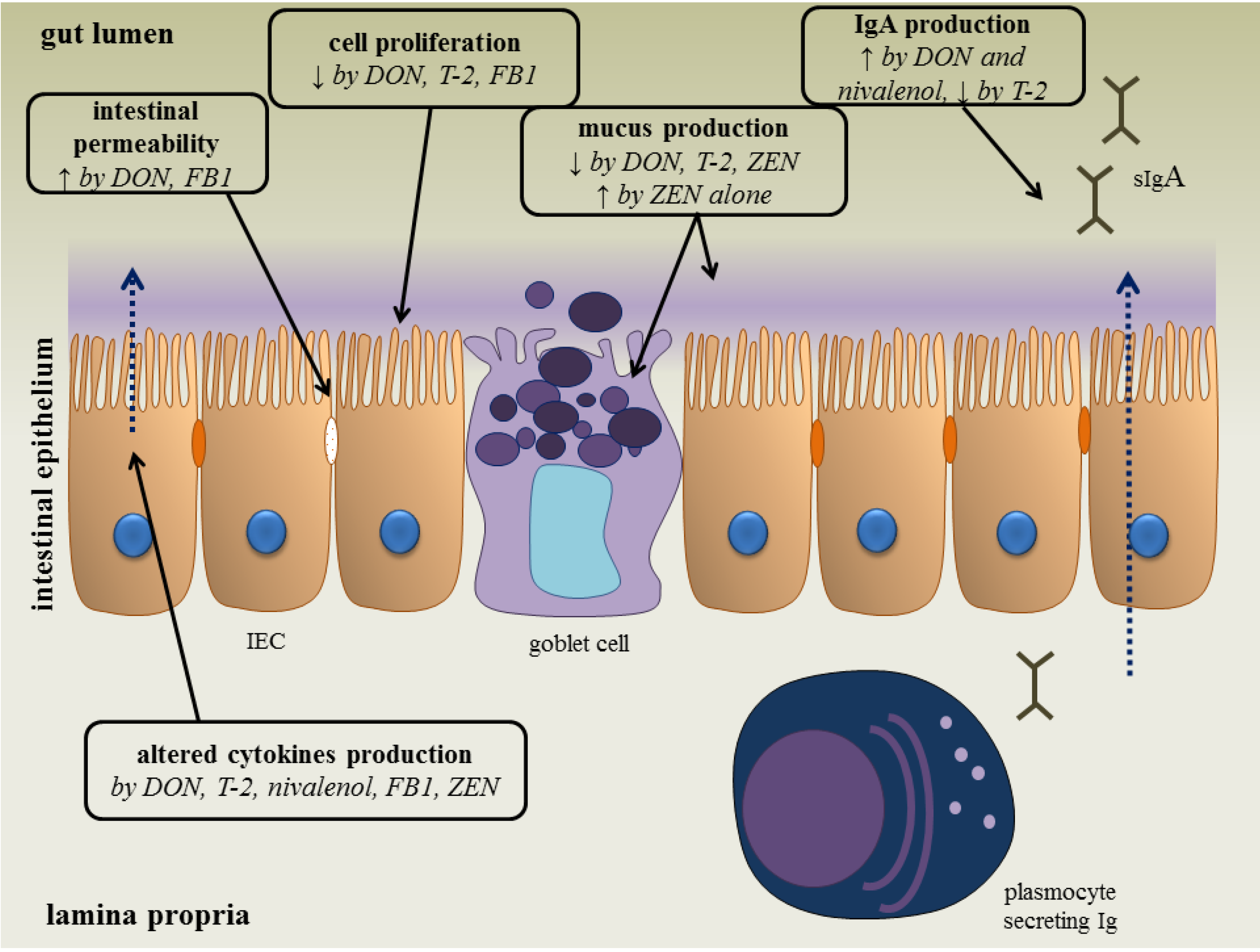
2. Effect of Fusarium Toxins on Parasitic Diseases
Coccidiosis
3. Effect of Fusarium Toxins on Bacterial Diseases
3.1. Salmonellosis

3.2. Colibacillosis
3.3. Necrotic Enteritis in Broilers
3.4. Edwardsiella ictaluri Infection in Catfish
Conflicts of Interest
Acknowledgments
Appendix
| Mycotoxin | Exposure dose | Exposure period | Cell line (host species) | Pathogen | Effect | Reference(s) |
|---|---|---|---|---|---|---|
| DON or T-2 | >25 ng DON/mL or 5 ng T-2/mL; ≥0.75 µg DON/mL or ≥2.5 ng T-2/mL | 24 h | undifferentiated IPEC1-J2; differentiated IPEC1-J2; (pig) | Salmonella Typhimurium | ↑ invasion | [27, 28] |
| DON or T-2 | 0.5 µg DON/mL or ≥1.0 ng T-2/mL | 24 h | differentiated IPEC1-J2 (pig) | Salmonella Typhimurium | ↑ translocation | [27, 28] |
| DON or T-2 | 0.025 µg DON/mL or 1 ng T-2/mL | 24 h | PAM2 (pig) | Salmonella Typhimurium | ↑ invasion | [28, 41] |
| DON | 5–50 µM (1.5–15 µg/mL) | 48 h | IPEC1-J1 (pig) | E. coli (SEPEC)3 | ↑ translocation | [14] |
| T-2 | 0.001 µM | 6 h | peritoneal macrophages (mouse) | P. aeruginosa4 | ↓ phagocytosis | [48] |
| T-2 | 0.01−0.05 µM | 20 h | alveolar macrophages (rat) | S. cerevisiae5 | ↓ phagocytosis | [106] |
| T-2 | 0.1 µM | 6 h | alveolar macrophages (rat) | S. aureus6 | ↓ phagocytosis | [106] |
| T-2 | 1–5 ng/mL; 2–5 ng/mL | 24 h | HD-11 cell line8 (chicken) | A. fumigatus7 | ↓ phagocytosis; ↑ immune response(A); ↑ germination | [80] |
| Mycotoxin | Exposure dose | Exposure period | Animal species | Age | Pathogen | Effect: compared to negative control | Reference(s) |
|---|---|---|---|---|---|---|---|
| DON, 15-acetylDON, ZEN and fumonisins | 6.5 mg DON, 0.44 mg 15-acetylDON, 0.59 mg ZEN and 0.37 mg fumonisins/kg feed | 6 weeks | chicken (broiler) | 1 day | E. maxima 1 | ↓ percentage of CD4+ and CD8+ T cells in jejunal mucosa | [35] |
| DON, 15-acetylDON and ZEN | 3.8 mg DON and 0.3 mg 15-acetylDON and 0.2 mg ZEN/kg feed | 10 weeks | chicken (broiler) | 1 day | E. acervulina 1, E. maxima 1, E. tenella 1 | ↓ level of blood monocytes at end of challenge period; percentage of CD8+ T-cells not. Restored at end of recovery period; ↑ IFN-γ gene expression | [34] |
| DON, 15-acetylDON and ZEN | 3.8 mg DON, 0.3 mg 15-acetylDON and 0.2 mg ZEN/kg feed | 10 weeks | chicken (broiler) | 1 day | E. acervulina 1, E. maxima 1, E. tenella 1 | ↓ intestinal recovery: duodenal villus height and apparent villus surface area | [36] |
| DON | 1 µg/mL | 6 h | pig | 5 weeks | Salmonella Typhimurium | synergistic ↑ gene expression IL-12, TNF-α, IL-1β, IL-8, MCP-1 and IL-6 | [27] |
| T-2 | 15 and 83 µg/kg feed | 23 days | pig | 3 weeks | Salmonella Typhimurium | ↓ colonization of the cecum | [28] |
| FB1 and FB2 | 8.6 mg FB1 and 3.2 mg FB2/kg feed | 9 weeks | pig | 4 weeks | Salmonella Typhimurium | synergistic transient effect digestive microbiota balance | [42] |
| T-2 | 2 mg/kg BW | 2 days | chicken (broiler) | 1 day | Salmonella Typhimurium | ↑ mortality | [45] |
| T-2 | 1 mg/kg BW | 3 weeks | mouse | 5–6 weeks | Salmonella Typhimurium | ↑ mortality | [46] |
| T-2 | 1 mg/kg BW | 10 days | mouse | 5–6 weeks | Salmonella Typhimurium | ↑ bacteria-related organ lesions | [47] |
| T-2 | 2 mg/kg BW | s.a. | mouse | - | Salmonella Typhimurium | ↑ mortality | [48] |
| DON | 1 mg/L drinking water | 3 weeks | mouse | 7 weeks | Salmonella Enteritidis | ↑ translocation to mesenteric lymph node, liver and spleen | [51] |
| FB1 | 150 mg/kg feed | 6 weeks | Japanese quail | 1 day | Salmonella Gallinarum | ↑ clinical signs and mortality; ↓ blood lymhocyte number | [107] |
| FB1 | 0.5 mg/kg BW | 6 days | pig | 3 weeks | E. coli (SEPEC) 2 | ↑ intestinal colonization; ↑ translocation to the mesenteric lymph node, lung, liver and spleen | [56] |
| FB1 | 1 mg/kg BW | 10 days | pig | 3–4 weeks | E. coli (ETEC) 3 | intestinal infection prolonged; impaired function of intestinal antigen presenting cells | [58] |
| fumonisins and aflatoxin | a 50–350 ng fumonisins /mL and 1–3 ng aflatoxin/mL | – | calf | <1 month | E. coli (STEC) 4 | ↑ susceptibility to hemorrhagic enteritis | [57] |
| moniliformin | 75–100 mg/kg feed | 3 weeks | chicken (broiler) | 0 day | E. coli (APEC) 5 | ↓ bacterial clearance | [60] |
| moniliformin and FB1 | 100 mg moniliformin and 200 mg FB1/kg feed | 3 weeks | turkey | 0 day | E. coli3 (APEC) 5 | ↓ bacterial clearance | [61] |
| DON | 4–5 mg/kg feed | 3 weeks | chicken (broiler) | 1 day | C. perfringens 6 | ↑ number of chickens with necrotic enteritis | [66] |
| DON | 5–10 mg/kg feed | 10 weeks | channel catfish | juvenile | E. ictaluri 7 | ↓ mortality | [71] |
| T-2 | 1–2 mg/kg | 6 weeks | channel catfish | juvenile | E. ictaluri 7 | ↑ mortality | [70] |
| FB1, FB2 and FB3 | 20 mg FB1, 3.5 mg FB2 and 1.9 mg FB3/kg feed | 42 days | pig | 3 days | M. hyopneumoniae 8 | ↑ severity of the pathological changes | [76] |
| FB1 | 10 mg/kg feed | 24 days | pig | 3 days | B. bronchiseptica 9 and P. multocida 10 (type D) | ↑ extent and severity of the pathological changes | [73] |
| FB1 | 0.5 mg/kg BW | 7 days | pig | piglets | P. multocida 10 (type A) | ↓ growth rate and ↑ coughing; ↑ total number of cells, number of macrophages and lymphocytes in BALF; ↑ gross pathological lesions and histopathological lesion of lungs | [74] |
| T-2 | mg/mouse ≈ 3.3 mg/kg BW | 20 days | mouse | adult | M. tuberculosis 11 (H37RvR-KM) | ↑ bacterial count in spleen | [108] |
| T-2 | 0.1 mg/mouse ≈ 3.3 mg/kg BW | 20 days | mouse | adult | M. bovis 12 | ↓ mouse survival time | [108] |
| T-2 | 0.5 mg/kg BW | 21 days | rabbit | – | A. fumigatus 13 | ↓ phagocytosis by alveolar macrophages | [79] |
| T-2 | 2 mg/kg BW | s.a. | mouse | – | P. aeruginosa 14 | ↓ phagocytosis by peritoneal macrophages | [48] |
| DON | 25 mg/kg BW | s.a. | mouse | 7–10 weeks | reovirus (serotype 1) | ↓ viral clearance and ↑ fecal shedding↓ Th1 response by ↓ IFN-γ gene expression↑ intestinal IgA and ↑ Th 2 response: by ↑ IL-4, IL-6 and IL-10 gene expression | [82] |
| T-2 | 1.75 mg/kg BW | s.a. | mouse | 7–10 weeks | reovirus (serotype 1) | ↓ viral clearance and ↑ fecal shedding; ↓ Th1 response by ↓ IFN-γ gene expression | [86] |
| FB1 | 12 mg/kg BW | 18 days | pig | 1 month | PRRSV15 | ↑ histopathological lesions of lungs | [89] |
| Mycotoxin | Foodstuffs for human consumption/finished animal feed | Maximum levels (µg/kg) |
|---|---|---|
| DON | unprocessed cereals other than durum wheat, oats and maize | 1250 |
| unprocessed durum wheat and oats | 1750 | |
| unprocessed maize, with the exception of unprocessed maize intended to be processed by wet milling | 1750 | |
| cereals intended for direct human consumption, cereal flour, bran and germ as end product marketed for direct human consumption, with the exception of foodstuffs listed in (1). | 750 | |
| pasta (dry) | 750 | |
| bread (including small bakery wares), pastries, biscuits, cereal snacks and breakfast cereals | 500 | |
| (1) processed cereal-based foods and baby foods for infants and young children | 200 | |
| feed materials: | ||
| cereals and cereal products with the exception of maize by-products | 8000 | |
| maize by-products | 12,000 | |
| complementary and complete feedingstuffs: | ||
| all animal species with the exception of (2) | 5000 | |
| (2) complementary and complete feedingstuffs for pigs | 900 | |
| (2) complementary and complete feedingstuffs for calves (<4 months), lambs and kids | 2000 | |
| ZEN | unprocessed cereals other than maize | 100 |
| unprocessed maize with the exception of unprocessed maize intended to be processed by wet milling | 350 | |
| cereals intended for direct human consumption, cereal flour, bran and germ as end product marketed for direct human consumption, with the exception of foodstuffs listed in (2) | 75 | |
| refined maize oil | 400 | |
| bread (including small bakery wares), pastries, biscuits, cereal snacks and breakfast cereals, excluding maize snacks and maize-based breakfast cereals | 50 | |
| (2) maize intended for direct human consumption, maize-based snacks and maize-bases breakfast cereals | 100 | |
| (2) processed cereal-based foods (excluding processed maize-based foods) and baby foods for infants and young children | 20 | |
| (2) processed maize-based foods for infants and young children | 20 | |
| feed materials: | ||
| cereals and cereal products with the exception of maize by-products | 2000 | |
| maize by-products | 3000 | |
| complementary and complete feedingstuffs: | ||
| complementary and complete feedingstuffs for piglets and gilts (young sows) | 100 | |
| complementary and complete feedingstuffs for sows and fattening pigs complementary and complete feedingstuffs for calves, dairy cattle, sheep (including lamb) and goats (including kids) | 250 | |
| complementary and complete feedingstuffs for calves, dairy cattle, sheep (including lamb) and goats (including kids) | 500 | |
| Fumonisins (sum FB1 + FB2) | unprocessed maize with the exception of unprocessed maize intended to be processed by wet milling | 4000 |
| maize intended for direct human consumption, maize-based foods for direct human consumption, with the exception of foodstuffs listed in (3) | 1000 | |
| (3) maize-based breakfast cereals and maize-based snacks | 800 | |
| (3) processed maize-based foods and baby foods for infants and young children | 200 | |
| feed materials: | ||
| maize and maize products | 60,000 | |
| complementary and complete feedingstuffs: | ||
| complementary and complete feedingstuffs for pigs, horses ( Equidae), rabbits and pet animals | 5000 | |
| complementary and complete feedingstuffs for fish | 10,000 | |
| complementary and complete feedingstuffs for poultry, calves (<4 months), lambs and kids | 20,000 | |
| complementary and complete feedingstuffs for adult ruminants (>4 months) and mink | 50,000 | |
| Sum T-2 and HT-2 | unprocessed cereals: | |
| barley (including malting barley) and maize | 200 | |
| oats (with husk) | 1000 | |
| wheat, rye and other cereals | 100 | |
| cereal grains for direct human consumption: | ||
| oats | 200 | |
| maize | 100 | |
| other cereals | 50 | |
| cereal products for human consumption: | ||
| oat bran and flaked oats | 200 | |
| cereal bran except oat bran, oat milling products other than oat bran and flaked oats, and maize milling products | 100 | |
| other cereal milling products | 50 | |
| breakfast cereals including formed cereal flakes | 75 | |
| bread (including small bakery wares), pastries, biscuits, cereal snacks, pasta | 25 | |
| cereal-based foods for infants and young children | 15 | |
| cereal products for feed: | ||
| oat milling products (husks) | 2000 | |
| other cereal products | 500 | |
| compound feed: | ||
| compound feed, with the exception of feed for cats | 250 |
References
- Binder, E.M. Managing the risk of mycotoxins in modern feed production. Anim. Feed Sci. Tech. 2007, 133, 149–166. [Google Scholar] [CrossRef]
- Filtenborg, O.; Frisvad, J.C.; Thrane, U. Moulds in food spoilage. Int. J. Food Microbiol. 1996, 33, 85–102. [Google Scholar] [CrossRef]
- Placinta, C.; D’mello, J.; Macdonald, A. A review of worldwide contamination of cereal grains and animal feed with Fusarium mycotoxins. Anim. Feed Sci. Tech. 1999, 78, 21–37. [Google Scholar] [CrossRef]
- D’mello, J.; Placinta, C.; Macdonald, A. Fusarium mycotoxins: A review of global implications for animal health, welfare and productivity. Anim. Feed Sci. Tech. 1999, 80, 183–205. [Google Scholar] [CrossRef]
- Smith, T.K.; Diaz, G.; Swamy, H. Current Concepts in Mycotoxicoses in Swine. In The Mycotoxin Blue Book; Diaz, D.E., Ed.; Nottingham University Press: Nottingham, UK, 2005; pp. 235–248. [Google Scholar]
- Devegowda, G.; Murthy, T. Mycotoxins: Their Effects in Poultry and Some Practical Solutions. In The Mycotoxin Blue Book; Diaz, D.E., Ed.; Nottingham University Press: Nottingham, UK, 2005; pp. 25–56. [Google Scholar]
- Devreese, M.; de Backer, P.; Croubels, S. Overview of the most important mycotoxins for the pig and poultry husbandry. Vlaams Diergeneeskundig Tijdschrift 2013, 82, 171–180. [Google Scholar]
- Streit, E.; Naehrer, K.; Rodrigues, I.; Schatzmayr, G. Mycotoxin occurrence in feed and feed raw materials worldwide-long term analysis with special focus on Europe and Asia. J. Sci. Food Agric. 2013, 93, 2892–2899. [Google Scholar] [CrossRef]
- Bouhet, S.; Oswald, I.P. The effects of mycotoxins, fungal food contaminants, on the intestinal epithelial cell-derived innate immune response. Vet. Immunol. Immun. 2005, 108, 199–209. [Google Scholar] [CrossRef]
- Oswald, I.P. Role of intestinal epithelial cells in the innate immune defence of the pig intestine. Vet. Res. 2006, 37, 359–368. [Google Scholar] [CrossRef]
- Schenk, M.; Mueller, C. The mucosal immune system at the gastrointestinal barrier. Best Pract. Res. CL GA 2008, 22, 391–409. [Google Scholar] [CrossRef]
- Maresca, M.; Mahfoud, R.; Garmy, N.; Fantini, J. The mycotoxin deoxynivalenol affects nutrient absorption in human intestinal epithelial cells. J. Nutr. 2002, 132, 2723–2731. [Google Scholar]
- Sergent, T.; Parys, M.; Garsou, S.; Pussemier, L.; Schneider, Y.-J.; Larondelle, Y. Deoxynivalenol transport across human intestinal Caco-2 cells and its effects on cellular metabolism at realistic intestinal concentrations. Toxicol. Lett. 2006, 164, 167–176. [Google Scholar] [CrossRef]
- Pinton, P.; Nougayrède, J.-P.; Del Rio, J.-C.; Moreno, C.; Marin, D.E.; Ferrier, L.; Bracarense, A.-P.; Kolf-Clauw, M.; Oswald, I.P. The food contaminant deoxynivalenol, decreases intestinal barrier permeability and reduces claudin expression. Toxicol. Appl. Pharm. 2009, 237, 41–48. [Google Scholar] [CrossRef]
- Bouhet, S.; Hourcade, E.; Loiseau, N.; Fikry, A.; Martinez, S.; Roselli, M.; Galtier, P.; Mengheri, E.; Oswald, I.P. The mycotoxin fumonisin B1 alters the proliferation and the barrier function of porcine intestinal epithelial cells. Toxicol. Sci. 2004, 77, 165–171. [Google Scholar]
- Awad, W.A.; Bohm, J.; Razzazi-Fazeli, E.; Zentek, J. Effects of feeding deoxynivalenol contaminated wheat on growth performance, organ weights and histological parameters of the intestine of broiler chickens. J. Anim. Physiol. Anim. Nutr. 2006, 90, 32–37. [Google Scholar] [CrossRef]
- Yunus, A.W.; Blajet-Kosicka, A.; Kosicki, R.; Khan, M.Z.; Rehman, H.; Bohm, J. Deoxynivalenol as a contaminant of broiler feed: Intestinal development, absorptive functionality and metabolism of the mycotoxin. Poult. Sci. 2012, 91, 852–861. [Google Scholar] [CrossRef]
- Hoerr, F.; Carlton, W.; Yagen, B. Mycotoxicosis caused by a single dose of T-2 toxin or diacetoxyscirpenol in broiler chickens. Vet. Pathol. 1981, 18, 652–664. [Google Scholar]
- Awad, W.A.; Hess, M.; Twarużek, M.; Grajewski, J.; Kosicki, R.; Böhm, J.; Zentek, J. The impact of the fusarium mycotoxin deoxynivalenol on the health and performance of broiler chickens. Int. J. Mol. Sci. 2011, 12, 7996–8012. [Google Scholar] [CrossRef]
- Maresca, M. From the gut to the brain: Journey and pathophysiological effects of the food-associated trichothecene mycotoxin deoxynivalenol. Toxins 2013, 5, 784–820. [Google Scholar] [CrossRef]
- Obremski, K.; Zielonka, Ł.; Gajecka, M.; Jakimiuk, E.; Bakuła, T.; Baranowski, M.; Gajecki, M. Histological estimation of the small intestine wall after administration of feed containing deoxynivalenol, T-2 toxin and zearalenone in the pig. Pol. J. Vet. Sci. 2008, 11, 339–345. [Google Scholar]
- Obremski, K.; Gajecka, M.; Zielonka, L.; Jakimiuk, E.; Gajecki, M. Morphology and ultrastructure of small intestine mucosa in gilts with zearalenone mycotoxicosis. Pol. J. Vet. Sci. 2004, 8, 301–307. [Google Scholar]
- Bondy, G.S.; Pestka, J.J. Immunomodulation by fungal toxins. J. Toxicol. Env. Heal. B 2000, 3, 109–143. [Google Scholar] [CrossRef]
- Osselaere, A.; Devreese, M.; Goossens, J.; Vandenbroucke, V.; de Baere, S.; de Backer, P.; Croubels, S. Toxicokinetic study and absolute oral bioavailability of deoxynivalenol, T-2 toxin and zearalenone in broiler chickens. Food Chem. Toxicol. 2012, 51, 350–355. [Google Scholar]
- Corrier, D. Mycotoxicosis: Mechanisms of immunosuppression. Vet. Immunol. Immun. 1991, 30, 73–87. [Google Scholar] [CrossRef]
- Oswald, I.; Marin, D.; Bouhet, S.; Pinton, P.; Taranu, I.; Accensi, F. Immunotoxicological risk of mycotoxins for domestic animals. Food Addit. Contam. 2005, 22, 354–360. [Google Scholar] [CrossRef]
- Vandenbroucke, V.; Croubels, S.; Martel, A.; Verbrugghe, E.; Goossens, J.; van Deun, K.; Boyen, F.; Thompson, A.; Shearer, N.; de Backer, P. The mycotoxin deoxynivalenol potentiates intestinal inflammation by Salmonella Typhimurium in porcine ileal loops. PLoS One 2011, 6. [Google Scholar] [CrossRef]
- Verbrugghe, E.; Vandenbroucke, V.; Dhaenens, M.; Shearer, N.; Goossens, J.; de Saeger, S.; Eeckhout, M.; D'herde, K.; Thompson, A.; Deforce, D. T-2 toxin induced Salmonella Typhimurium intoxication results in decreased Salmonella numbers in the cecum contents of pigs, despite marked effects on Salmonella-host cell interactions. Vet. Res. 2012, 43, 1–18. [Google Scholar] [CrossRef]
- Lillehoj, H.S.; Lillehoj, E.P. Avian coccidiosis. A review of acquired intestinal immunity and vaccination strategies. Avian Dis. 2000, 44, 408–425. [Google Scholar] [CrossRef]
- Chapman, H.D.; Barta, J.R.; Blake, D.; Gruber, A.; Jenkins, M.; Smith, N.C.; Suo, X.; Tomley, F.M. A selective review of advances in coccidiosis research. Adv. Parasitol. 2013, 83, 93–171. [Google Scholar]
- Lillehoj, H. Role of T lymphocytes and cytokines in coccidiosis. Int. J. Parasitol. 1998, 28, 1071–1081. [Google Scholar] [CrossRef]
- Lillehoj, H.; Min, W.; Dalloul, R. Recent progress on the cytokine regulation of intestinal immune responses to Eimeria. Poult. Sci. 2004, 83, 611–623. [Google Scholar]
- Lillehoj, H.; Kim, C.; Keeler, C.; Zhang, S. Immunogenomic approaches to study host immunity to enteric pathogens. Poult. Sci. 2007, 86, 1491–1500. [Google Scholar]
- Girgis, G.N.; Sharif, S.; Barta, J.R.; Boermans, H.J.; Smith, T.K. Immunomodulatory effects of feed-borne fusarium mycotoxins in chickens infected with coccidia. Exp. Biol. Med. 2008, 233, 1411–1420. [Google Scholar] [CrossRef]
- Girgis, G.N.; Barta, J.R.; Girish, C.K.; Karrow, N.A.; Boermans, H.J.; Smith, T.K. Effects of feed-borne fusarium mycotoxins and an organic mycotoxin adsorbent on immune cell dynamics in the jejunum of chickens infected with Eimeria maxima. Vet. Immunol. Immun. 2010, 138, 218–223. [Google Scholar] [CrossRef]
- Girgis, G.; Barta, J.; Brash, M.; Smith, T. Morphologic changes in the intestine of broiler breeder pullets fed diets naturally contaminated with fusarium mycotoxins with or without coccidial challenge. Avian Dis. 2010, 54, 67–73. [Google Scholar] [CrossRef]
- Békési, L.; Hornok, S.; Szigeti, G.; Dobos-Kovács, M.; Széll, Z.; Varga, I. Effect of F-2 and T-2 fusariotoxins on experimental Cryptosporidium baileyi infection in chickens. Int. J. Parasitol. 1997, 27, 1531–1536. [Google Scholar] [CrossRef]
- Varga, I.; Ványi, A. Interaction of T-2 fusariotoxin with anticoccidial efficacy of lasalocid in chickens. Int. J. Parasitol. 1992, 22, 523–525. [Google Scholar] [CrossRef]
- Andrews-Polymenis, H.L.; Bäumler, A.J.; McCormick, B.A.; Fang, F.C. Taming the elephant: Salmonella biology, pathogenesis, and prevention. Infect. Immun. 2010, 78, 2356–2369. [Google Scholar] [CrossRef]
- Ohl, M.E.; Miller, S.I. Salmonella: A model for bacterial pathogenesis. Annu. Rev. Med. 2001, 52, 259–274. [Google Scholar] [CrossRef]
- Vandenbroucke, V.; Croubels, S.; Verbrugghe, E.; Boyen, F.; De Backer, P.; Ducatelle, R.; Rychlik, I.; Haesebrouck, F.; Pasmans, F. The mycotoxin deoxynivalenol promotes uptake of Salmonella Typhimurium in porcine macrophages, associated with ERK1/2 induced cytoskeleton reorganization. Vet. Res. 2009, 40. [Google Scholar] [CrossRef]
- Burel, C.; Tanguy, M.; Guerre, P.; Boilletot, E.; Cariolet, R.; Queguiner, M.; Postollec, G.; Pinton, P.; Salvat, G.; Oswald, I.P. Effect of low dose of fumonisins on pig health: Immune status, intestinal microbiota and sensitivity to Salmonella. Toxins 2013, 5, 841–864. [Google Scholar]
- Skjolaas, K.; Burkey, T.; Dritz, S.; Minton, J. Effects of Salmonella enterica serovars Typhimurium (st) and Choleraesuis (sc) on chemokine and cytokine expression in swine ileum and jejunal epithelial cells. Vet. Immunol. Immun. 2006, 111, 199–209. [Google Scholar] [CrossRef]
- Boyen, F.; Haesebrouck, F.; Maes, D.; van Immerseel, F.; Ducatelle, R.; Pasmans, F. Non-typhoidal Salmonella infections in pigs: A closer look at epidemiology, pathogenesis and control. Vet. Microbiol. 2008, 130, 1–19. [Google Scholar] [CrossRef]
- Ziprin, R.; Elissalde, M. Effect of T-2 toxin on resistance to systemic Salmonella Typhimurium infection of newly hatched chickens. Am. J. Vet. Res. 1990, 51, 1869–1872. [Google Scholar]
- Tai, J.; Pestka, J. Impaired murine resistance to Salmonella Typhimurium following oral exposure to the trichothecene t-2 toxin. Food Cem. Toxicol. 1988, 26, 691–698. [Google Scholar] [CrossRef]
- Tai, J.-H.; Pestka, J. T-2 toxin impairment of murine response to Salmonella Typhimurium: A histopathologic assessment. Mycopathologia 1990, 109, 149–155. [Google Scholar] [CrossRef]
- Vidal, D.; Mavet, S. In vitro and in vivo toxicity of t-2 toxin, a fusarium mycotoxin, to mouse peritoneal macrophages. Infect. Immun. 1989, 57, 2260–2264. [Google Scholar]
- Mittrücker, H.-W.; Kaufmann, S. Immune response to infection with Salmonella Typhimurium in mice. J. Leukocyte Biol. 2000, 67, 457–463. [Google Scholar]
- Tai, J.; Pestka, J. Synergistic interaction between the trichothecene T-2 toxin and Salmonella Typhimurium lipopolysaccharide in C3H/HeN and C3H/HeJ mice. Toxicol. Lett. 1988, 44, 191–200. [Google Scholar] [CrossRef]
- Hara-Kudo, Y. Effects of deoxynivalenol on Salmonella Enteritidis infection. Mycotoxins 1996, 1996, 42, 51–56. [Google Scholar]
- Rothkötter, H.; Sowa, E.; Pabst, R. The pig as a model of developmental immunology. Hum. Exp. Toxicol. 2002, 21, 533–536. [Google Scholar] [CrossRef]
- Meurens, F.; Summerfield, A.; Nauwynck, H.; Saif, L.; Gerdts, V. The pig: A model for human infectious diseases. Trends Microbiol. 2012, 20, 50–57. [Google Scholar] [CrossRef]
- Kaper, J.B.; Nataro, J.P.; Mobley, H.L. Pathogenic Escherichia coli. Nat. Rev. Microbiol. 2004, 2, 123–140. [Google Scholar] [CrossRef]
- Nataro, J.P.; Kaper, J.B. Diarrheagenic Escherichia coli. Clin. Microbiol. Rev. 1998, 11, 142–201. [Google Scholar]
- Oswald, I.P.; Desautels, C.; Laffitte, J.; Fournout, S.; Peres, S.Y.; Odin, M.; le Bars, P.; le Bars, J.; Fairbrother, J.M. Mycotoxin fumonisin B1 increases intestinal colonization by pathogenic escherichia coli in pigs. Appl. Environ. Microb. 2003, 69, 5870–5874. [Google Scholar] [CrossRef]
- Baines, D.; Sumarah, M.; Kuldau, G.; Juba, J.; Mazza, A.; Masson, L. Aflatoxin, fumonisin and shiga toxin-producing Escherichia coli infections in calves and the effectiveness of celmanax/dairyman’s choice applications to eliminate morbidity and mortality losses. Toxins 2013, 5, 1872–1895. [Google Scholar] [CrossRef]
- Devriendt, B.; Verdonck, F.; Wache, Y.; Bimczok, D.; Oswald, I.P.; Goddeeris, B.M.; Cox, E. The food contaminant fumonisin B1 reduces the maturation of porcine CD11r1+ intestinal antigen presenting cells and antigen-specific immune responses, leading to a prolonged intestinal etec infection. Vet. Res. 2009, 40, 1–14. [Google Scholar]
- Grenier, B.; Applegate, T.J. Modulation of intestinal functions following mycotoxin ingestion: Meta-analysis of published experiments in animals. Toxins 2013, 5, 396–430. [Google Scholar]
- Li, Y.; Ledoux, D.; Bermudez, A.; Fritsche, K.; Rottinghaust, G. Effects of moniliformin on performance and immune function of broiler chicks. Poult. Sci. 2000, 79, 26–32. [Google Scholar]
- Li, Y.; Ledoux, D.; Bermudez, A.; Fritsche, K.; Rottinghaus, G. The individual and combined effects of fumonisin B1 and moniliformin on performance and selected immune parameters in turkey poults. Poult. Sci. 2000, 79, 871–878. [Google Scholar]
- Barbara, A.J.; Trinh, H.T.; Glock, R.D.; Glenn Songer, J. Necrotic enteritis-producing strains of Clostridium perfringens displace non-necrotic enteritis strains from the gut of chicks. Vet. Microbiol. 2008, 126, 377–382. [Google Scholar]
- Timbermont, L.; Haesebrouck, F.; Ducatelle, R.; van Immerseel, F. Necrotic enteritis in broilers: An updated review on the pathogenesis. Avian Pathol. 2011, 40, 341–347. [Google Scholar] [CrossRef]
- Williams, R. Intercurrent coccidiosis and necrotic enteritis of chickens: Rational, integrated disease management by maintenance of gut integrity. Avian Pathol. 2005, 34, 159–180. [Google Scholar] [CrossRef]
- Keyburn, A.L.; Boyce, J.D.; Vaz, P.; Bannam, T.L.; Ford, M.E.; Parker, D.; di Rubbo, A.; Rood, J.I.; Moore, R.J. NetB, a new toxin that is associated with avian necrotic enteritis caused by Clostridium perfringens. PLoS Pathog. 2008, 4. [Google Scholar] [CrossRef]
- Antonissen, G.; Van Immerseel, F.; Pasmans, F.; Ducatelle, R.; Haesebrouck, F.; Timbermont, L.; Verlinden, M.; Janssens, G.P.J.; Eeckhout, M.; de Saeger, S.; et al. Deoxynivalenol predisposes for necrotic enteritis by affecting the intestinal barrier in broilers. In Proceedings of the International Poultry Scientific Forum, Atlanta, Georgia, USA, 28–29 January 2013; pp. 9–10.
- Crumlish, M.; Dung, T.; Turnbull, J.; Ngoc, N.; Ferguson, H. Identification of Edwardsiella ictaluri from diseased freshwater catfish, Pangasius hypophthalmus (sauvage), cultured in the mekong delta, Vietnam. J. Fish Dis. 2002, 25, 733–736. [Google Scholar] [CrossRef]
- Ferguson, H.; Turnbull, J.; Shinn, A.; Thompson, K.; Dung, T.T.; Crumlish, M. Bacillary necrosis in farmed Pangasius hypophthalmus (sauvage) from the Mekong delta, Vietnam. J. Fish Dis. 2001, 24, 509–513. [Google Scholar] [CrossRef]
- Newton, J.; Wolfe, L.; Grizzle, J.; Plumb, J. Pathology of experimental enteric septicaemia in channel catfish, Ictalurus punctatus (rafinesque), following immersion-exposure to Edwardsiella ictaluri. J. Fish Dis. 1989, 12, 335–347. [Google Scholar] [CrossRef]
- Manning, B.B.; Terhune, J.S.; Li, M.H.; Robinson, E.H.; Wise, D.J.; Rottinghaus, G.E. Exposure to feedborne mycotoxins T-2 toxin or ochratoxin a causes increased mortality of channel catfish challenged with Edwardsiella ictaluri. J. Aquat. Anim. Health 2005, 17, 147–152. [Google Scholar] [CrossRef]
- Manning, B.B.; Abbas, H.K.; Wise, D.J.; Greenway, T. The effect of feeding diets containing deoxynivalenol contaminated corn on channel catfish (Ictalurus punctatus) challenged with edwardsiella ictaluri. Aquac. Res. 2013. [Google Scholar] [CrossRef]
- Hooft, J.M.; Elmor, A.E.H.I.; Encarnação, P.; Bureau, D.P. Rainbow trout (Oncorhynchus mykiss) is extremely sensitive to the feed-borne Fusarium mycotoxin deoxynivalenol. Aquaculture 2011, 311, 224–232. [Google Scholar] [CrossRef]
- Pósa, R.; Donkó, T.; Bogner, P.; Kovács, M.; Repa, I.; Magyar, T. Interaction of Bordetella bronchiseptica, Pasteurella multocida, and fumonisin B1 in the porcine respiratory tract as studied by computed tomography. Can. J. Vet. Res. 2011, 75, 176–182. [Google Scholar]
- Halloy, D.J.; Gustin, P.G.; Bouhet, S.; Oswald, I.P. Oral exposure to culture material extract containing fumonisins predisposes swine to the development of pneumonitis caused by Pasteurella multocida. Toxicology 2005, 213, 34–44. [Google Scholar] [CrossRef]
- Maes, D.; Segales, J.; Meyns, T.; Sibila, M.; Pieters, M.; Haesebrouck, F. Control of Mycoplasma hyopneumoniae infections in pigs. Vet. Microbiol. 2008, 126, 297–309. [Google Scholar] [CrossRef]
- Pósa, R.; Magyar, T.; Stoev, S.; Glávits, R.; Donkó, T.; Repa, I.; Kovács, M. Use of computed tomography and histopathologic review for lung lesions produced by the interaction between Mycoplasma hyopneumoniae and fumonisin mycotoxins in pigs. Vet. Pathol. 2013, 50. [Google Scholar] [CrossRef]
- Chanter, N.; Magyar, T.; Rutter, J.M. Interactions between Bordetella bronchiseptica and toxigenic Pasteurella multocida in atrophic rhinitis of pigs. Res. Vet. Sci. 1989, 47, 48–53. [Google Scholar]
- Davies, R.L.; MacCorquodale, R.; Baillie, S.; Caffrey, B. Characterization and comparison of Pasteurella multocida strains associated with porcine pneumonia and atrophic rhinitis. J. Med. Microbiol. 2003, 52, 59–67. [Google Scholar] [CrossRef]
- Niyo, K.; Richard, J.; Niyo, Y.; Tiffany, L. Effects of T-2 mycotoxin ingestion on phagocytosis of Aspergillus fumigatus conidia by rabbit alveolar macrophages and on hematologic, serum biochemical, and pathologic changes in rabbits. Am. J. Vet. Res. 1988, 49, 1766–1773. [Google Scholar]
- Li, S.-J.; Pasmans, F.; Croubels, S.; Verbrugghe, E.; Van Waeyenberghe, L.; Yang, Z.; Haesebrouck, F.; Martel, A. T-2 toxin impairs antifungal activities of chicken macrophages against Aspergillus fumigatus conidia but promotes the pro-inflammatory responses. Avian Pathol. 2013, 42, 457–463. [Google Scholar] [CrossRef]
- Nibert, M.; Furlong, D.; Fields, B. Mechanisms of viral pathogenesis. Distinct forms of reoviruses and their roles during replication in cells and host. J. Clin. Invest. 1991, 88, 727–934. [Google Scholar] [CrossRef]
- Li, M.; Cuff, C.F.; Pestka, J. Modulation of murine host. Response to enteric reovirus infection by the trichothecene deoxynivalenol. Toxicol. Sci. 2005, 87, 134–145. [Google Scholar] [CrossRef]
- Jones, R. Avian reovirus infections. Rev. Sci. Tech. OIE 2000, 19, 614–625. [Google Scholar]
- Jones, R.; Kibenge, F. Reovirus-induced tenosynovitis in chickens: The effect of breed. Avian Pathol. 1984, 13, 511–528. [Google Scholar] [CrossRef]
- Benavente, J.; Martínez-Costas, J. Avian reovirus: Structure and biology. Virus Res. 2007, 123, 105–119. [Google Scholar] [CrossRef]
- Li, M.; Cuff, C.F.; Pestka, J.J. T-2 toxin impairment of enteric reovirus clearance in the mouse associated with suppressed immunoglobulin and IFN-γ responses. Toxicol. Appl. Pharm. 2006, 214, 318–325. [Google Scholar] [CrossRef]
- Chand, R.J.; Trible, B.R.; Rowland, R.R. Pathogenesis of porcine reproductive and respiratory syndrome virus. Curr. Opin. Virol. 2012, 2, 256–263. [Google Scholar] [CrossRef]
- Rowland, R.; Morrison, R. Challenges and opportunities for the control and elimination of porcine reproductive and respiratory syndrome virus. Transbound. Emerg. Dis. 2012, 59, 55–59. [Google Scholar] [CrossRef]
- Ramos, C.M.; Martinez, E.M.; Carrasco, A.C.; Puente, J.H.L.; Quezada, F.; Perez, J.T.; Oswald, I.P.; Elvira, S.M. Experimental trial of the effect of fumonisin B and the PRRS virus in swine. J. Anim. Vet. Adv. 2010, 9, 1301–1310. [Google Scholar] [CrossRef]
- Bane, D.P.; Neumann, E.J.; Hall, W.F.; Harlin, K.S.; Slife, R.L.N. Relationship between fumonisin contamination of feed and mystery swine disease-a case-control study. Mycopathologia 1992, 117, 121–124. [Google Scholar]
- Wu, F. Measuring the economic impacts of Fusarium toxins in animal feeds. Anim. Feed Sci. Tech. 2007, 137, 363–374. [Google Scholar] [CrossRef]
- Zain, M.E. Impact of mycotoxins on humans and animals. J. Saudi Chem. Soc. 2011, 15, 129–144. [Google Scholar] [CrossRef]
- Maresca, M.; Fantini, J. Some food-associated mycotoxins as potential risk factors in humans predisposed to chronic intestinal inflammatory diseases. Toxicon 2010, 56, 282–294. [Google Scholar] [CrossRef]
- Wagacha, J.; Muthomi, J. Mycotoxin problem in africa: Current status, implications to food safety and health and possible management strategies. Int. J. Food Microbiol. 2008, 124, 1–12. [Google Scholar] [CrossRef]
- Wild, C.P.; Gong, Y.Y. Mycotoxins and human disease: A largely ignored global health issue. Carcinogenesis 2010, 31, 71–82. [Google Scholar] [CrossRef]
- Wan, L.Y.M.; Turner, P.C.; El-Nezami, H. Individual and combined cytotoxic effects of Fusarium toxins (deoxynivalenol, nivalenol, zearalenone and fumonisins B1) on swine jejunal epithelial cells. Food Chem. Toxicol. 2013, 57, 276–283. [Google Scholar] [CrossRef]
- Grenier, B.; Oswald, I. Mycotoxin co-contamination of food and feed: Meta-analysis of publications describing toxicological interactions. World Mycotoxin J. 2011, 4, 285–313. [Google Scholar] [CrossRef]
- Grenier, B.; Loureiro‐Bracarense, A.P.; Lucioli, J.; Pacheco, G.D.; Cossalter, A.M.; Moll, W.D.; Schatzmayr, G.; Oswald, I.P. Individual and combined effects of subclinical doses of deoxynivalenol and fumonisins in piglets. Mol. Nutr. Food. Res. 2011, 55, 761–771. [Google Scholar] [CrossRef]
- De Boevre, M.; di Mavungu, J.D.; Landschoot, S.; Audenaert, K.; Eeckhout, M.; Maene, P.; Haesaert, G.; De Saeger, S. Natural occurrence of mycotoxins and their masked forms in food and feed products. World Mycotoxin J. 2012, 5, 207–219. [Google Scholar] [CrossRef]
- Nagl, V.; Schwartz, H.; Krska, R.; Moll, W.-D.; Knasmüller, S.; Ritzmann, M.; Adam, G.; Berthiller, F. Metabolism of the masked mycotoxin deoxynivalenol-3-glucoside in rats. Toxicol. Lett. 2012, 213, 367–373. [Google Scholar] [CrossRef]
- Dall’Erta, A.; Cirlini, M.; Dall’Asta, M.; del Rio, D.; Galaverna, G.; Dall’Asta, C. Masked mycotoxins are efficiently hydrolysed by the human colonic microbiota, releasing their toxic aglycones. Chem. Res. Toxicol. 2013, 26, 305–312. [Google Scholar] [CrossRef]
- Broekaert, N.; Devreese, M.; de Mil, T.; Fraeyman, S.; de Baere, S.; de Saeger, S.; de Backer, P.; Croubels, S. Development and validation of an LC-MS/MS method for the toxicokinetic study of deoxynivalenol and its acetylated derivatives in animal plasma. Anal. Bioanal. Chem. Submitted.
- Paterson, R.R.M.; Lima, N. How will climate change affect mycotoxins in food? Food Res. Int. 2010, 43, 1902–1914. [Google Scholar] [CrossRef]
- Magan, N.; Medina, A.; Aldred, D. Possible climate change effects on mycotoxin contamination of food crops pre-and postharvest. Plant Pathol. 2011, 60, 150–163. [Google Scholar] [CrossRef]
- Shuman, E.K. Global climate change and infectious diseases. N. Engl. J. Med. 2010, 362, 1061–1063. [Google Scholar] [CrossRef]
- Gerberick, G.F.; Sorenson, W.; Lewis, D. The effects of T-2 toxin on alveolar macrophage function in vitro. Environ. Res. 1984, 33, 246–260. [Google Scholar] [CrossRef]
- Deshmukh, S.; Asrani, R.; Jindal, N.; Ledoux, D.; Rottinghaus, G.; Sharma, M.; Singh, S. Effects of fusarium moniliforme culture material containing known levels of fumonisin B1 on progress of Salmonella Gallinarum infection in Japanese quail: Clinical signs and hematologic studies. Avian Dis. 2005, 49, 274–280. [Google Scholar] [CrossRef]
- Kanai, K.; Kondo, E. Decreased resistance to mycobacterial infection in mice fed a trichothecene compound (T-2 toxin). Jpn. J. Med. Sci. Biol. 1984, 37, 97. [Google Scholar]
- European Commission. Commission Regulation (EC) of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs no. 1881/2006. Off. J. Eur. Union 2006, L364, 5–24. [Google Scholar]
- European Commission. Commission Recommendation of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding (2006/576/EC). Off. J. Eur. Union 2006, L229, 7–9. [Google Scholar]
- European Commission. Commission Recommendation of 27 March 2013 on the presence of T-2 and HT-2 toxin in cereals and cereal products (2013/165/EU). Off. J. Eur. Union 2013, L91, 12–15. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Antonissen, G.; Martel, A.; Pasmans, F.; Ducatelle, R.; Verbrugghe, E.; Vandenbroucke, V.; Li, S.; Haesebrouck, F.; Van Immerseel, F.; Croubels, S. The Impact of Fusarium Mycotoxins on Human and Animal Host Susceptibility to Infectious Diseases. Toxins 2014, 6, 430-452. https://doi.org/10.3390/toxins6020430
Antonissen G, Martel A, Pasmans F, Ducatelle R, Verbrugghe E, Vandenbroucke V, Li S, Haesebrouck F, Van Immerseel F, Croubels S. The Impact of Fusarium Mycotoxins on Human and Animal Host Susceptibility to Infectious Diseases. Toxins. 2014; 6(2):430-452. https://doi.org/10.3390/toxins6020430
Chicago/Turabian StyleAntonissen, Gunther, An Martel, Frank Pasmans, Richard Ducatelle, Elin Verbrugghe, Virginie Vandenbroucke, Shaoji Li, Freddy Haesebrouck, Filip Van Immerseel, and Siska Croubels. 2014. "The Impact of Fusarium Mycotoxins on Human and Animal Host Susceptibility to Infectious Diseases" Toxins 6, no. 2: 430-452. https://doi.org/10.3390/toxins6020430
APA StyleAntonissen, G., Martel, A., Pasmans, F., Ducatelle, R., Verbrugghe, E., Vandenbroucke, V., Li, S., Haesebrouck, F., Van Immerseel, F., & Croubels, S. (2014). The Impact of Fusarium Mycotoxins on Human and Animal Host Susceptibility to Infectious Diseases. Toxins, 6(2), 430-452. https://doi.org/10.3390/toxins6020430







