Durum Wheat (Triticum Durum Desf.) Lines Show Different Abilities to Form Masked Mycotoxins under Greenhouse Conditions
Abstract
:1. Introduction
2. Results and Discussion
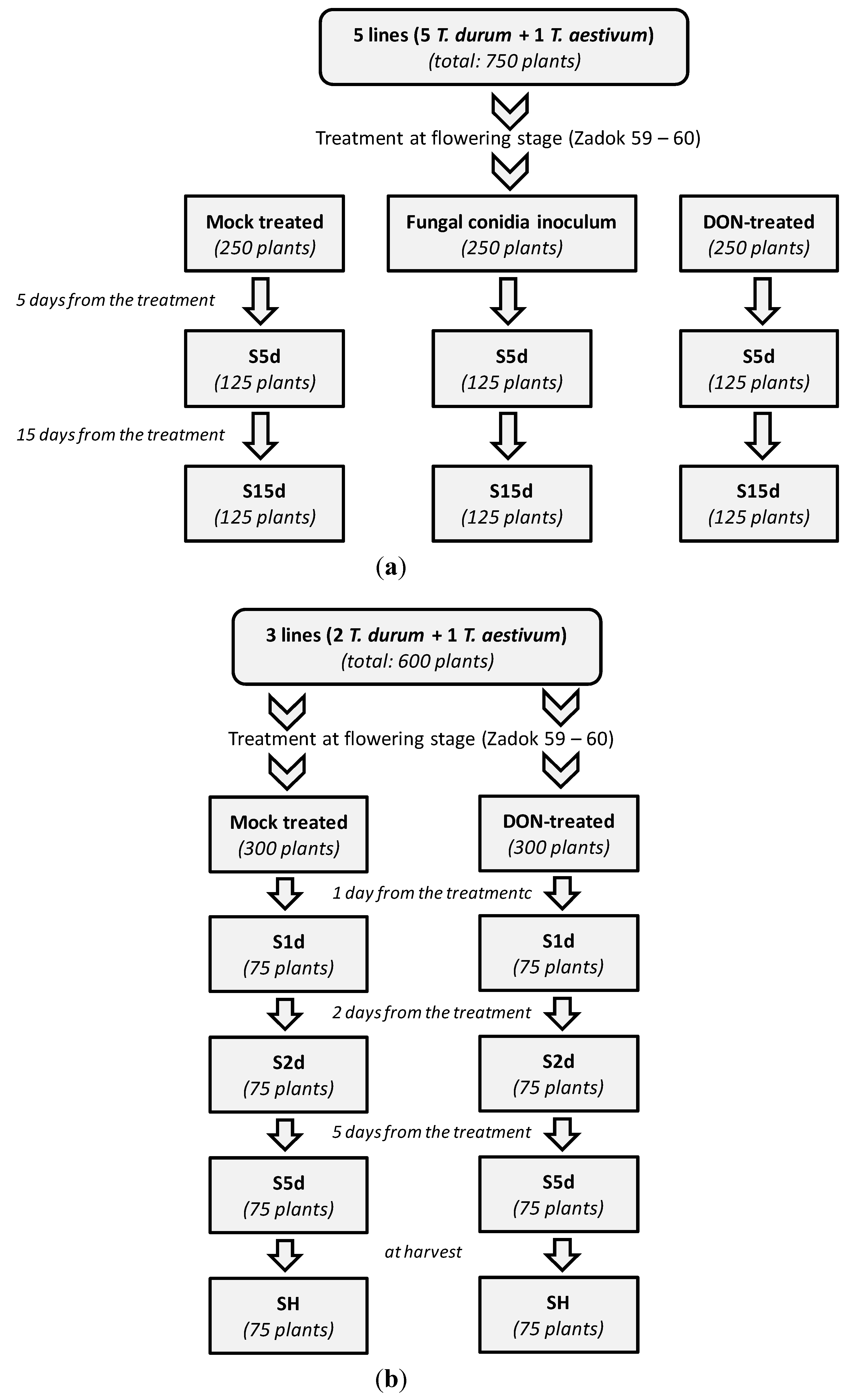
2.1. Set up of the First Trial
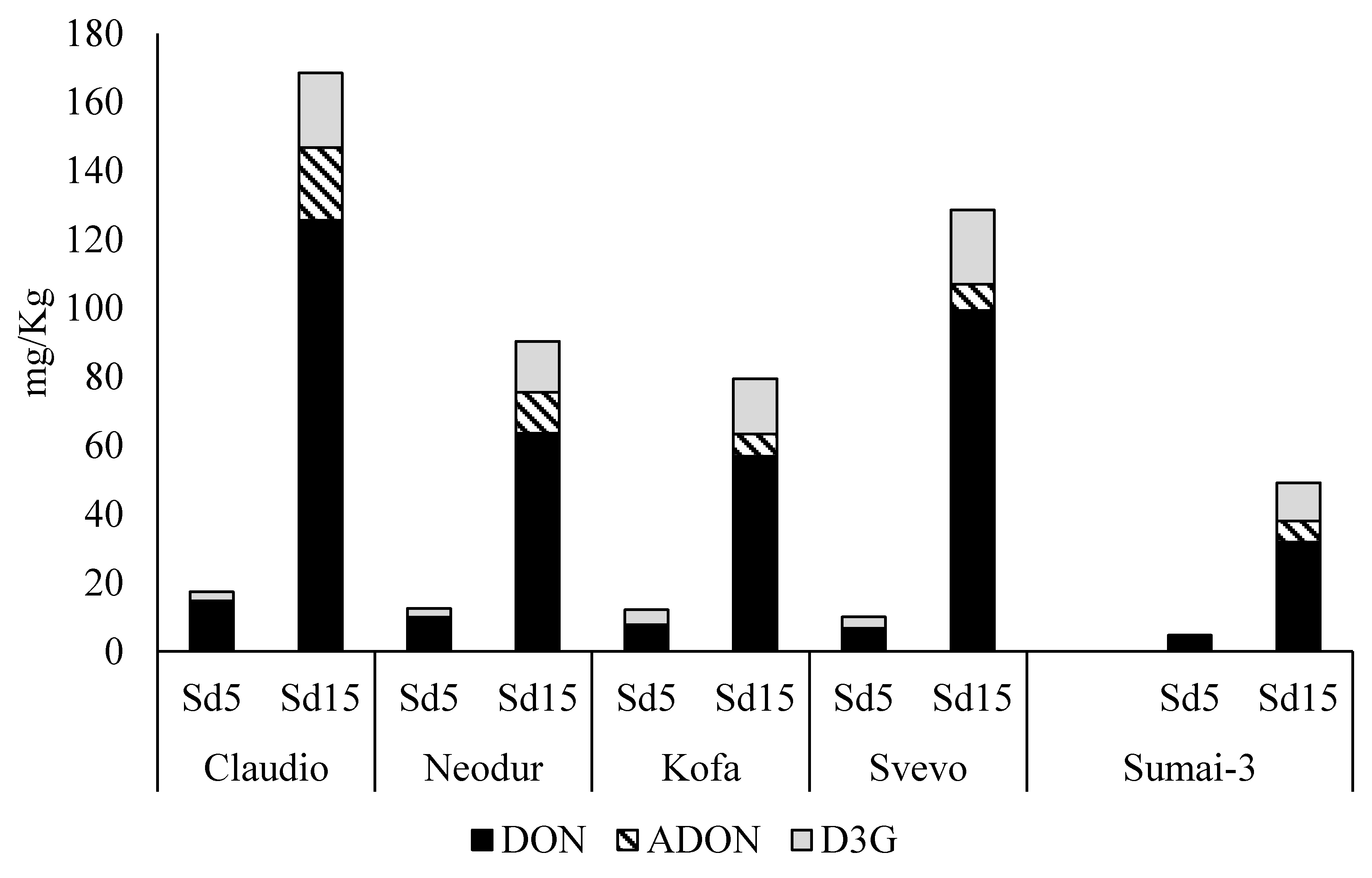
2.1.1. Fusarium graminearum Treated Samples
| Cultivars | TD (mg/Kg) | Ergosterol (mg/Kg) |
|---|---|---|
| Claudio | 168 ± 34 | 22.0 ± 7.7 |
| Neodur | 90 ± 20 | 5.2 ± 1.8 |
| Kofa | 79 ± 5 | 4.0 ± 1.3 |
| Svevo | 129 ± 6 | 10.1 ± 0.7 |
| Sumai-3 | 49 ± 6 | 2.5 ± 0.2 |
| Cultivars | [D3G]/[TDON] | |
|---|---|---|
| DON-Treated Samples | F. graminearum Inoculated Samples | |
| Claudio | 0.292 b | 0.144 b |
| Neodur | 0.371 a | 0.182 b |
| Kofa | 0.407 a | 0.252 a |
| Svevo | 0.273 b | 0.282 a |
2.1.2. DON-Treated Samples
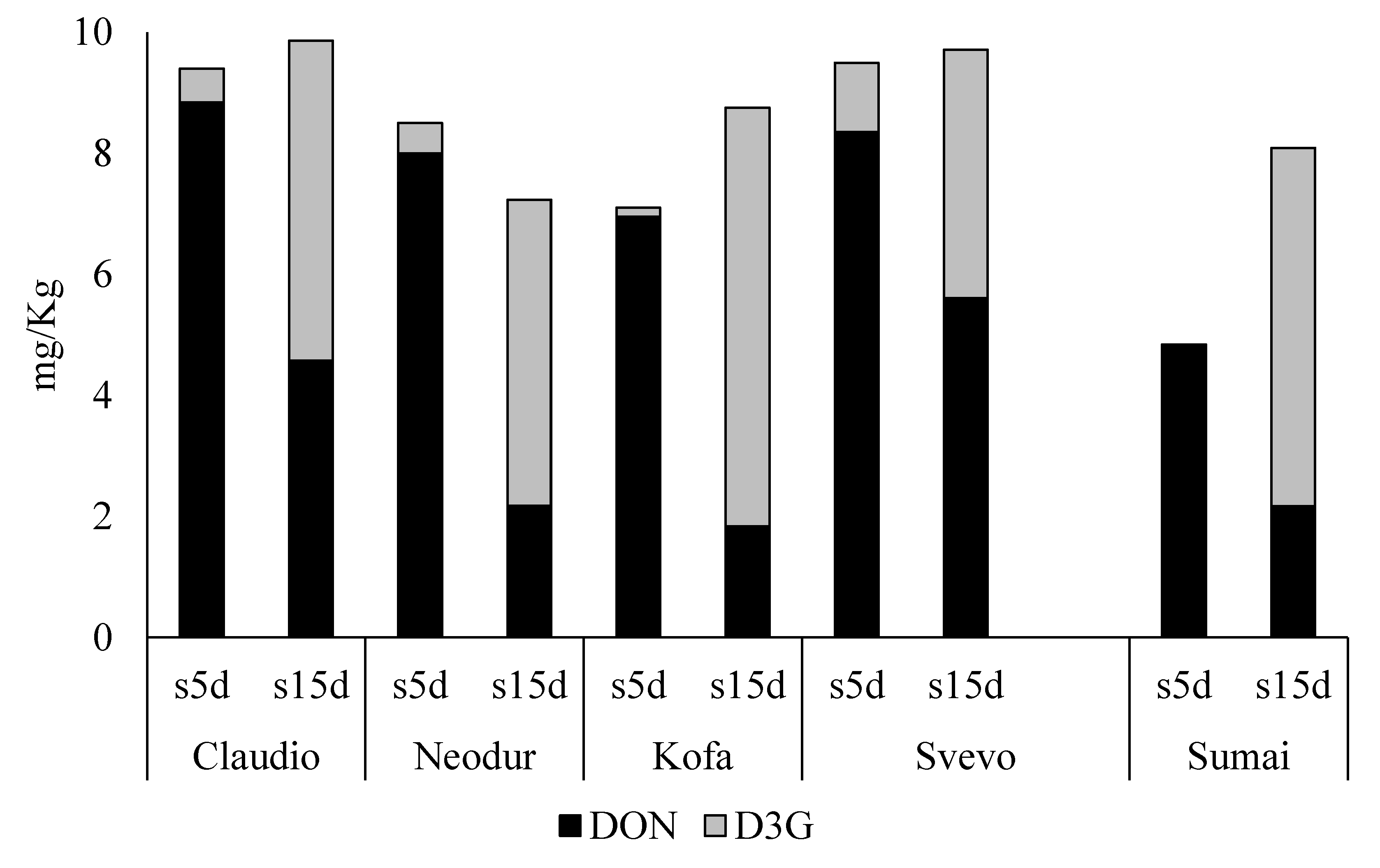
2.2. Set up of the Second Trial
DON-Treated Samples
| Sampling dates | Kofa | Svevo | Sumai-3 |
|---|---|---|---|
| Sd1 | 0.046 | 0.056 | 0.246 |
| Sd2 | 0.391 | 0.154 | 0.655 |
| Sd5 | 0.421 | 0.115 | 0.619 |
| SH | 0.530 | 0.312 | 0.843 |
3. Experimental Section
3.1. Chemicals
3.2. Wheat Lines Background Information
3.3. Fusarium graminearum Strain Background Information
3.4. Design of the Greenhouse Experiment
3.5. Free and Masked Mycotoxins Analysis
3.6. Ergosterol Extraction and Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- McMullen, M.; Jones, R.; Gallenberg, D. Scab of wheat and barley: A re-emerging disease of devastating impact. Plant Dis. 1997, 81, 1340–1348. [Google Scholar] [CrossRef]
- Champeil, A.; Doré, T.; Fourbet, J.F. Fusarium head blight: Epidemiological origin of the effects of cultural practices on head blight attacks and the production of mycotoxins by Fusarium in wheat grains. Plant Sci. 2004, 166, 1389–1415. [Google Scholar]
- Leonard, K.J.; Bushnell, W.R. Fusarium Head Blight of Wheat and Barley; APS Press: Saint Paul, MN, USA, 2003. [Google Scholar]
- Desjardins, A.E.; Hohn, T.M.; McCormick, S.P. Trichothecene biosynthesis in Fusarium species: Chemistry, genetics, and significance. Microbiol. Rev. 1993, 57, 595–604. [Google Scholar]
- Buerstmayr, H.; Ban, T.; Anderson, J.A. QTL mapping and marker-assisted selection for Fusarium head blight resistance in wheat: A review. Plant Breed. 2009, 128, 1–26. [Google Scholar] [CrossRef]
- Anderson, J.A.; Stack, R.W.; Liu, S.; Waldron, B.L.; Fjeld, A.D.; Coyne, C.; Moreno-Sevilla, B.; Fetch, J.M.; Song, Q.J.; Cregan, P.B.; et al. DNA markers for Fusarium head blight resistance QTLs in two wheat populations. Theor. Appl. Genet. 2001, 102, 1164–1168. [Google Scholar] [CrossRef]
- Buerstmayr, H.; Lemmens, M.; Hartl, L.; Doldi, L.; Steiner, B.; Stierschneider, M.; Ruckenbauer, P. Molecular mapping of QTLs for Fusarium head blight resistance in spring wheat. I. Resistance to fungal spread (type II resistance). Theor. Appl. Genet. 2002, 104, 84–91. [Google Scholar] [CrossRef]
- Buerstmayr, H.; Stierschneider, M.; Steiner, B.; Lemmens, M.; Griesser, M.; Nevo, E.; Fahima, T. Variation for resistance to head blight caused by Fusarium graminearum in wild emmer (Triticum dicoccoides) originating from Israel. Euphytica 2003, 130, 17–23. [Google Scholar] [CrossRef]
- Stack, R.W.; Elias, E.M.; Mitchell Fetch, J.; Miller, J.D.; Joppa, L.R. Fusarium head blight reaction of Langdon durum-Triticum dicoccoides chromosome substitution lines. Crop Sci. 2002, 42, 637–642. [Google Scholar] [CrossRef]
- Somers, D.J.; Fedak, G.; Clarke, J.; Cao, W. Mapping of FHB resistance QTLs in tetraploid wheat. Genome 2006, 49, 1586–1593. [Google Scholar] [CrossRef]
- Oliver, R.E.; Cai, X.; Friesen, T.L.; Halley, S.; Stack, R.W.; Xu, S.S. Evaluation of Fusarium head blight resistance in tetraploid wheat (Triticum turgidum L.). Crop Sci. 2008, 48, 213–222. [Google Scholar] [CrossRef]
- Gilbert, J.; Procunier, J.D.; Aung, T. Influence of the D genome in conferring resistance to fusarium head blight in spring wheat. Euphytica 2000, 114, 181–186. [Google Scholar] [CrossRef]
- Oliver, R.E.; Stack, R.W.; Miller, J.D.; Cai, X. Reaction of wild emmer wheat accessions to fusarium head blight. Crop Sci. 2007, 47, 893–899. [Google Scholar] [CrossRef]
- Lu, Q.; Lillemo, M.; Skinnes, H.; He, X.; Shi, J.; Ji, F.; Dong, Y.; Bjørnstad, A. Anther extrusion and plant height are associated with Type I resistance to Fusarium head blight in bread wheat line “Shanghai-3/Catbird”. Theor. Appl. Genet. 2013, 126, 317–334. [Google Scholar] [CrossRef]
- Lin, F.; Xue, S.L.; Zhang, Z.Z.; Zhang, C.Q.; Kong, Z.X.; Yao, G.Q.; Tian, D.G.; Zhu, H.L.; Li, C.J.; Cao, Y.; et al. Mapping QTL associated with resistance to Fusarium head blight in the Nanda2419 × Wangshuibai population. II: Type I resistance. Theor. Appl. Genet. 2006, 112, 528–535. [Google Scholar] [CrossRef]
- Buerstmayr, M.; Alimari, A.; Steiner, B.; Buerstmayr, H. Genetic mapping of QTL for resistance to Fusarium head blight spread (type 2 resistance) in a Triticum dicoccoides × Triticum durum backcross-derived population. Theor. Appl. Genet. 2013, 126, 2825–2834. [Google Scholar] [CrossRef]
- Lin, F.; Kong, Z.X.; Zhu, H.L.; Xue, S.L.; Wu, J.Z.; Tian, D.G.; Wei, J.B.; Zhang, C.Q.; Ma, Z.Q. Mapping QTL associated with resistance to Fusarium head blight in the Nanda2419 × Wangshuibai population. I. Type II resistance. Theor. Appl. Genet. 2004, 109, 1504–1511. [Google Scholar] [CrossRef]
- Arunachalam, C.; Doohan, F.M. Trichothecene toxicity in eukaryotes: Cellular and molecular mechanisms in plants and animals. Toxicol. Lett. 2013, 217, 149–158. [Google Scholar] [CrossRef]
- Lemmens, M.; Scholz, U.; Berthiller, F.; Dall’Asta, C.; Koutnik, A.; Schumacher, R.; Adam, G.; Buerstmayr, H.; Mesterhazy, A.; Krska, R.; et al. The ability to detoxify the mycotoxin deoxynivalenol colocalizes with a major quantitative trait locus for Fusarium Head Blight resistance in wheat. Mol. Plant Microbe. Interact. 2005, 18, 1318–1324. [Google Scholar] [CrossRef]
- Schweiger, W.; Boddu, J.; Shin, S.; Poppenberger, B.; Berthiller, F.; Lemmens, M.; Muehlbauer, G.J.; Adam, G. Validation of a candidate deoxynivalenol-inactivating UDP-glucosyltransferase from barley by heterologous expression in yeast. Mol. Plant Microbe Interact. 2010, 23, 977–986. [Google Scholar] [CrossRef]
- Poppenberger, B.; Berthiller, F.; Lucyshyn, D.; Sieberer, T.; Schuhmacher, R.; Krska, R.; Kuchler, K.; Glössl, J.; Luschnig, C.; Adam, G. Detoxification of the Fusarium mycotoxin deoxynivalenol by a UDP-glucosyltransferase from Arabidopsis thaliana. J. Biol. Chem. 2003, 278, 47905–47914. [Google Scholar] [CrossRef]
- Gunnaiah, R.; Kushalappa, A.C.; Duggavathi, R.; Fox, S.; Somers, D.J. Integrated metabolo-proteomic approach to decipher the mechanisms by which wheat QTL (Fhb1) contributes to resistance against Fusarium graminearum. PLoS One 2012, 7. [Google Scholar] [CrossRef]
- Dall’Asta, C.; Dall’Erta, A.; Mantovani, P.; Massi, A.; Galaverna, G. Occurrence of deoxynivalenol and deoxynivalenol-3-glucoside in durum wheat. World Mycotoxin J. 2013, 6, 83–91. [Google Scholar] [CrossRef]
- Zhuang, Y.; Gala, A.; Yen, Y. Identification of functional genic components of major fusarium head blight resistance quantitative trait loci in wheat cultivar Sumai 3. Mol. Plant Microbe Interact. 2013, 26, 442–450. [Google Scholar] [CrossRef]
- Zhou, M.P.; Hayden, M.J.; Zhang, Z.Y.; Lu, W.Z.; Ma, H.X. Saturation and mapping of a major Fusarium head blight resistance QTL on chromosome 3BS of Sumai 3 wheat. J. Appl. Genet. 2010, 51, 19–25. [Google Scholar] [CrossRef]
- Golkari, S.; Gilbert, J.; Ban, T.; Procunier, J.D. QTL-specific microarray gene expression analysis of wheat resistance to Fusarium head blight in Sumai-3 and two susceptible NILs. Genome 2009, 52, 409–418. [Google Scholar] [CrossRef]
- Karlovsky, P. Biological detoxification of the mycotoxin deoxynivalenol and its use in genetically engineered crops and feed additives. Appl. Microbiol. Biotechnol. 2011, 91, 491–504. [Google Scholar] [CrossRef]
- Zadoks, J.C.; Chang, T.T.; Konzak, C.F. A decimal code for the growth stages of cereals. Weed Res. 1974, 14, 415–421. [Google Scholar] [CrossRef]
- Gottwald, S.; Samans, B.; Lück, S.; Friedt, W. Jasmonate and ethylene dependent defence gene expression and suppression of fungal virulence factors: Two essential mechanisms of Fusarium head blight resistance in wheat? BMC Genomics 2012, 13, 369. [Google Scholar] [CrossRef]
- Prodi, A.; Tonti, S.; Nipoti, P.; Pancaldi, D.; Pisi, A. Identification of deoxynivalenol and nivalenol producing chemotypes of Fusarium graminearum isolates from durum wheat in a restricted area of northern Italy. J. Plant Pathol. 2009, 91, 727–731. [Google Scholar]
- Purahong, W.; Nipoti, P.; Pisi, A.; Lemmens, M.; Prodi, A. Aggressiveness of different Fusarium graminearum chemotypes within a population from northern-central Italy. Mycoscience 2013. [Google Scholar] [CrossRef]
- Berthiller, F.; Schuhmacher, R.; Buttinger, G.; Krska, R. Rapid simultaneous determination of major type A- and B-trichothecenes as well as zearalenone in maize by high performance liquid chromatography–tandem mass spectrometry. J. Chrom. A 2005, 1062, 209–216. [Google Scholar]
- Annaratone, C.; Caligiani, A.; Cirlini, M.; Toffanin, L.; Palla, G. Sterols, sterol oxides and CLA in typical meat products from pigs fed with different diets. Czech J. Food Sci. 2009, 27, 219–222. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cirlini, M.; Generotti, S.; Dall'Erta, A.; Lancioni, P.; Ferrazzano, G.; Massi, A.; Galaverna, G.; Dall'Asta, C. Durum Wheat (Triticum Durum Desf.) Lines Show Different Abilities to Form Masked Mycotoxins under Greenhouse Conditions. Toxins 2014, 6, 81-95. https://doi.org/10.3390/toxins6010081
Cirlini M, Generotti S, Dall'Erta A, Lancioni P, Ferrazzano G, Massi A, Galaverna G, Dall'Asta C. Durum Wheat (Triticum Durum Desf.) Lines Show Different Abilities to Form Masked Mycotoxins under Greenhouse Conditions. Toxins. 2014; 6(1):81-95. https://doi.org/10.3390/toxins6010081
Chicago/Turabian StyleCirlini, Martina, Silvia Generotti, Andrea Dall'Erta, Pietro Lancioni, Gianluca Ferrazzano, Andrea Massi, Gianni Galaverna, and Chiara Dall'Asta. 2014. "Durum Wheat (Triticum Durum Desf.) Lines Show Different Abilities to Form Masked Mycotoxins under Greenhouse Conditions" Toxins 6, no. 1: 81-95. https://doi.org/10.3390/toxins6010081





