Relationship between Aflatoxin Contamination and Physiological Responses of Corn Plants under Drought and Heat Stress
Abstract
:1. Introduction
2. Results
2.1. Environmental Moisture and Temperature Conditions
2.2. Aflatoxin Contamination
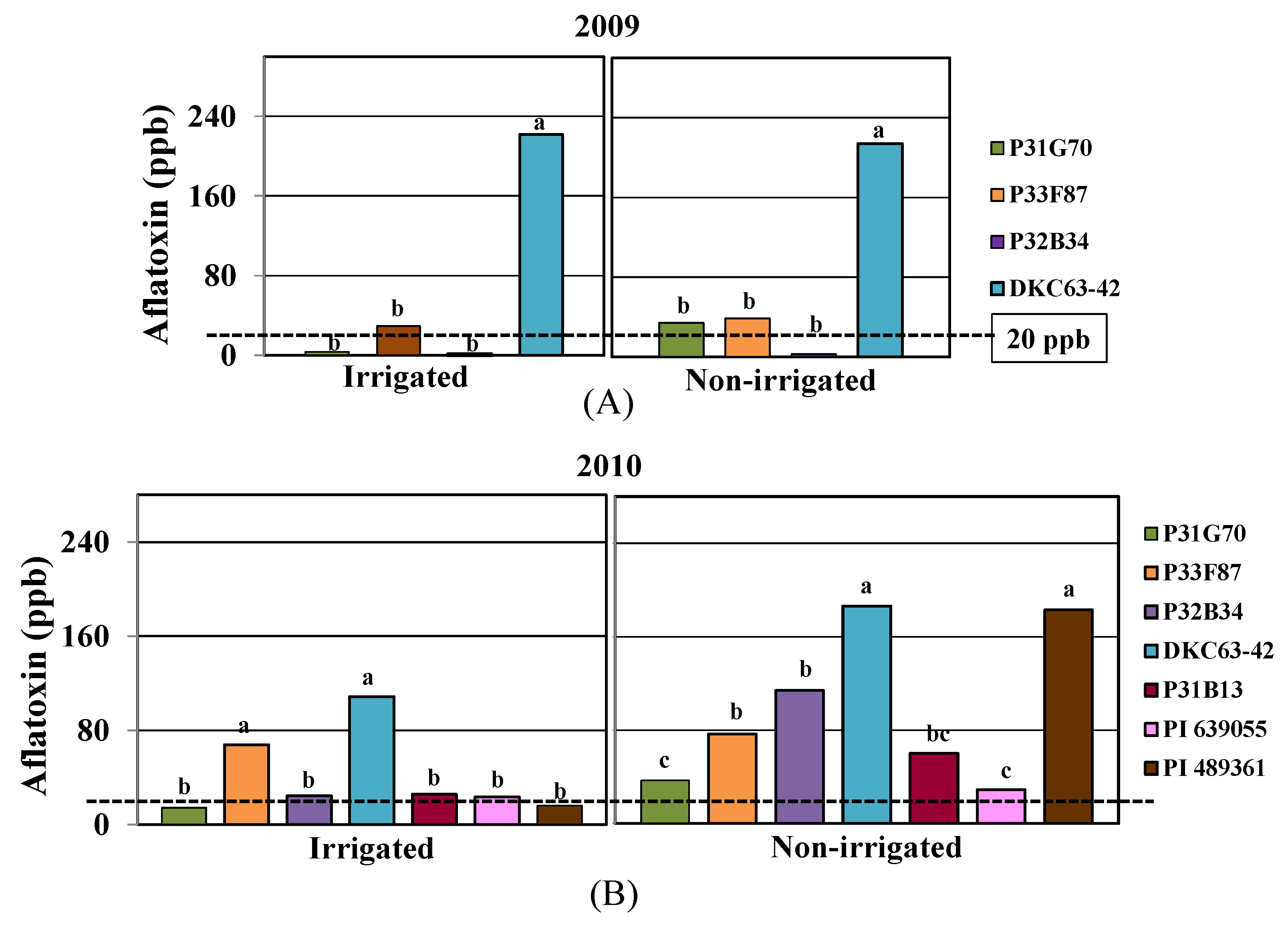
2.3. Physiological Responses to Drought and Heat Stress
2.3.1. Leaf Water Potential and Canopy Temperature
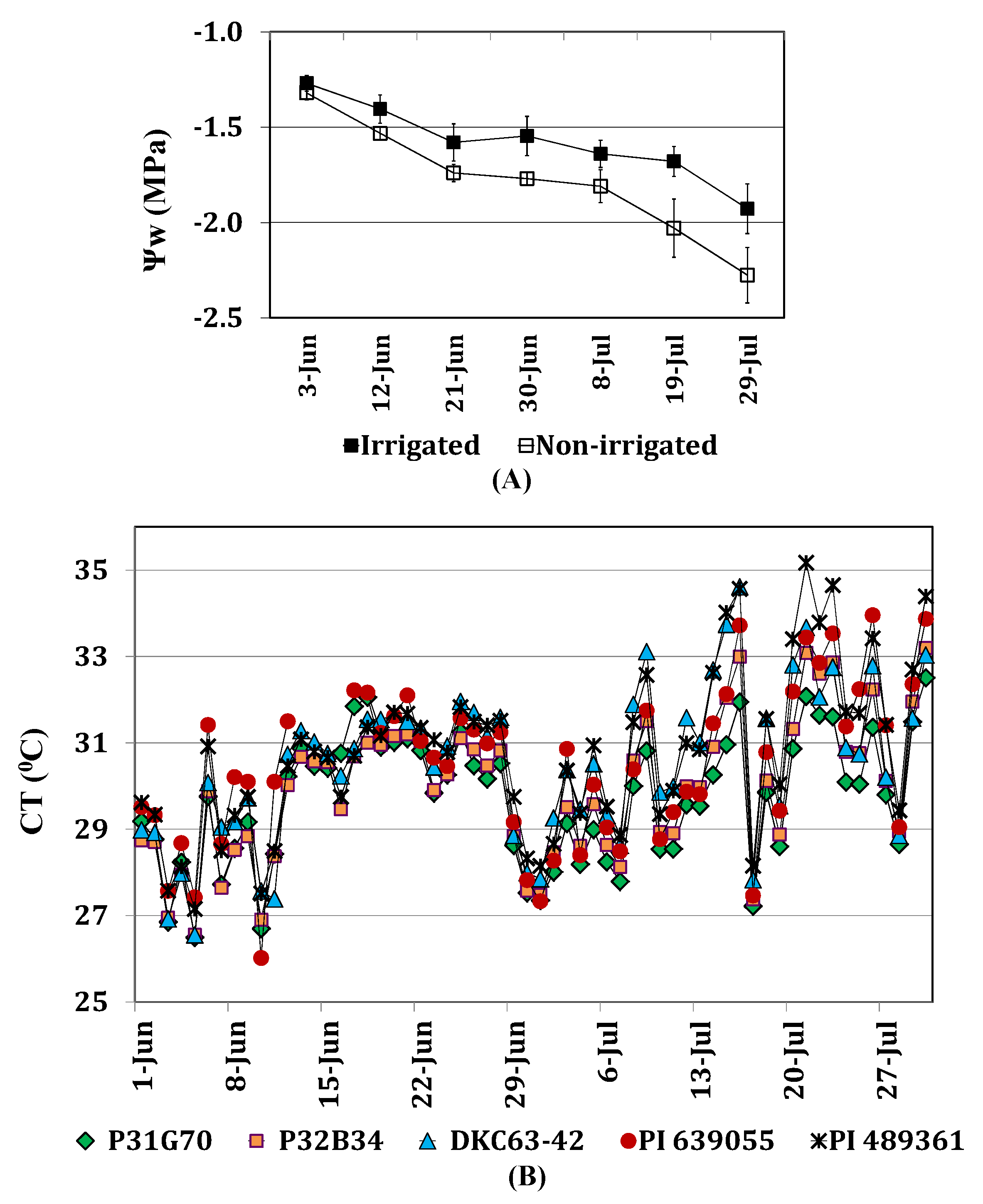
2.3.2. Photosynthetic Pigments
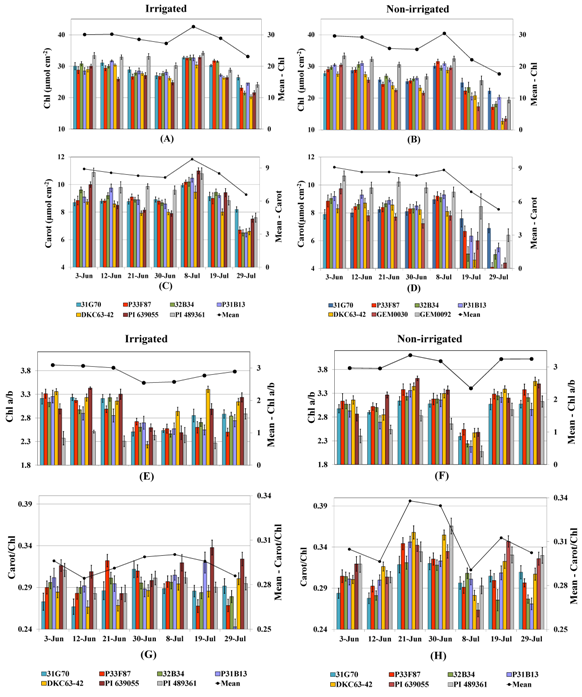
2.3.3. Chlorophyll Fluorescence and Cell Membrane Thermostability
2.3.4. Seed Composition

| Genotype | Protein (%) | Starch (%) | 100 kernel wt (g) | |||
|---|---|---|---|---|---|---|
| Irrigated | Non-irrigated | Irrigated | Non-irrigated | Irrigated | Non-irrigated | |
| 2009 | ||||||
| P31G70 | 7.5 c | 8.1 b | 63.3 b | 62.4 b | 28.8 b | 28.7 a |
| P33F87 | 7.4 c | 8.0 b | 64.5 a | 63.3 a | 30.2 a | 29.6 a |
| P32B34 | 8.1 b | 8.3 b | 63.4 b | 62.5 b | 29.5 a | 30.0 a |
| DKC63-42 | 9.0 a | 9.0 a | 62.7 c | 62.6 b | 30.2 a | 29.0 a |
| 2010 | ||||||
| P31G70 | 7.9 b | 8.8 a | 63.0 b | 62.6 b | 28.5 b | 27.9 b |
| P33F87 | 7.5 c | 8.2 b | 65.1 a | 62.5 b | 31.5 a | 30.9 a |
| P32B34 | 7.9 b | 7.8 b | 63.1 b | 62.7 b | 32.2 a | 29.1 a |
| P31B13 | 7.6 b | 8.1 b | 62.7 b | 62.0 b | 31.0 a | 29.1 a |
| DKC63-42 | 9.2 a | 9.0 a | 63.3 b | 62.8 a | 29.8 b | 27.5 b |
| PI 639055 | 9.3 a | 9.2 a | 61.8 b | 62.7 b | 29.6 b | 28.7 b |
| PI 489361 | 8.5 ab | 8.9 a | 62.5 b | 63.2 a | 30.0 b | 28.5 b |
3. Discussion
4. Materials and Methods
4.1. Plant Material, and Moisture and Temperature Measurements
| Name | Type | Trait |
|---|---|---|
| P31G70 | commercial hybrid | drought stress tolerant + stay-green |
| P33F87 | commercial hybrid | drought stress tolerant |
| P32B34 | commercial hybrid | needs irrigation for good performance |
| P31B13 | commercial hybrid | corn borer resistant (Bt) |
| DKC63-42 | commercial hybrid | aflatoxin susceptible |
| PI 639055 (GEMS-0030) | Inbred germplasm (tropical background) | aflatoxin resistant |
| PI 489361 (GEMS-0092) | Inbred germplasm (tropical background) | drought stress tolerant |
4.2. Fungal Inoculation and Aflatoxin Analysis
4.3. Physiological Measurements
4.3.1. Plant Water Status
4.3.2. Photosynthetic Pigments
4.3.3. Heat Stress Measurements
4.3.4. Seed Composition Analysis
4.4. Statistical Analysis
5. Conclusion
Disclaimer
Conflict of Interest
Acknowledgements
References
- Payne, G.A. Process of Contamination by Aflatoxin-Producing Fungi and Their Impact on Crops. In Mycotoxins in Agriculture and Food Safety; Sinha, K.K., Bhatnagar, D., Eds.; Marcel Dekker: New York, NY, USA, 1998; pp. 279–306. [Google Scholar]
- Abbas, H.K.; Williams, W.P.; Windham, G.L.; Pringle III, H.C.; Xie, W.; Shier, W.T. Aflatoxin and fumonisin contamination of commercial corn (Zea mays) hybrids in Mississippi. J. Agric. Food Chem. 2002, 50, 5246–5254. [Google Scholar] [CrossRef]
- Cole, R.J.; Sanders, T.H.; Hill, R.A.; Blankenship, P.D. Mean geocarposphere temperatures that induce preharvest aflatoxin contamination of peanuts under drought stress. Mycopathologia 1985, 91, 41–46. [Google Scholar]
- Tubajika, K.M.; Damann, K.E. Sources of resistance to aflatoxin production in maize. J. Agric. Food Chem. 2001, 49, 2652–2656. [Google Scholar] [CrossRef]
- Guo, B.; Chen, Z.-Y.; Lee, R.D.; Scully, B.T. Drought stress and preharvest aflatoxin contamination in agricultural commodity: Genetics, genomics and proteomics. J. Integr. Plant Biol. 2008, 50, 1281–1291. [Google Scholar] [CrossRef]
- Hill, R.A.; Blankenship, P.D.; Cole, R.J.; Sanders, T.H. Effects of soil moisture and temperature on preharvest invasion of peanuts by the Aspergillus flavus group and subsequent aflatoxin development. Appl. Environ. Microbiol. 1983, 45, 628–633. [Google Scholar]
- Bruns, H.A. Controlling aflatoxin and fumonisin in maize by crop management. Toxin Rev. 2003, 22, 153–173. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Brown, R.L.; Cleveland, T.E. Evidence for an association in corn between stress tolerance and resistance to Aspergillus flavus infection and aflatoxin contamination. Afr. J. Biotechnol. 2004, 3, 693–699. [Google Scholar]
- Jayashree, T.; Subramanyam, C. Oxidative stress as a prerequisite for aflatoxin production by Aspergillus parasiticus. Free Radic. Biol. Med. 2000, 29, 981–985. [Google Scholar]
- Narasaiah, K.V.; Sashidhar, R.B.; Subramanyam, C. Biochemical analysis of oxidative stress in the production of aflatoxin and its precursor intermediates. Mycopathologia 2006, 162, 179–189. [Google Scholar] [CrossRef]
- Kim, J.H.; Campbell, B.C.; Yu, J.; Mahoney, N.; Chan, K.L.; Molyneux, R.J.; Bhatnagar, D.; Cleveland, T.E. Examination of fungal stress response genes using Saccharomyces cerevisiae as a model system: Targeting genes affecting aflatoxin biosynthesis by Aspergillus flavus. Appl. Microbiol. Biotechnol. 2005, 67, 807–815. [Google Scholar] [CrossRef]
- Kim, J.H.; Yu, J.J.; Mahoney, N.; Chan, K.L.; Molyneux, R.J.; Varga, J.; Bhatnagar, D.; Cleveland, T.E.; Nierman, W.C.; Campbell, B.C. Elucidation of the functional genomics of antioxidant-based inhibition of aflatoxin biosynthesis. Int. J. Food Microbiol. 2008, 122, 49–60. [Google Scholar] [CrossRef]
- Huang, J.Q.; Jiang, H.F.; Zhou, Y.Q.; Lei, Y.; Wang, S.Y.; Liao, B.S. Ethylene inhibited aflatoxin biosynthesis is due to oxidative stress alleviation and related to glutathione redox state changes in Aspergillus flavus. Int. J. Food Microbiol. 2009, 130, 17–21. [Google Scholar] [CrossRef]
- Mahoney, N.; Molyneux, R.J.; Kim, J.H.; Campbell, B.C.; Waiss, A.C.; Hagerman, A.E. Aflatoxigenesis induced in Aspergillus flavus by oxidative stress and reduction by phenolic antioxidants from tree nuts. World Mycotoxin J. 2010, 3, 49–57. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Botany 2012. [Google Scholar]
- Cui, L.; Li, J.; Fan, Y.; Xu, S.; Zhang, Z. High temperature effects on photosynthesis, PS II functionality and antioxidant activity of two Festuca arundinacea cultivars with different heat susceptibility. Bot. Stud. 2006, 47, 61–69. [Google Scholar]
- Maruta, T.; Noshi, M.; Tanouchi, A.; Tamoi, M.; Yabuta, Y.; Yoshimura, K.; Ishikawa, T.; Shigeoka, S. H2O2-triggered retrograde signaling from chloroplasts to nucleus plays a specific role in the response to stress. J. Biol. Chem. 2012, 287, 11717–11729. [Google Scholar]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Wahid, A.; Gelani, S.; Ashraf, M.; Foola, M.R. Heat tolerance in plants: An overview. Environ. Exp. Bot. 2007, 61, 199–223. [Google Scholar] [CrossRef]
- Kebede, H.; Fisher, D.K.; Young, L.D. Determination of moisture deficit and heat stress tolerance in corn using physiological measurements and a low-cost microcontroller-based monitoring system. J. Agron. Crop Sci. 2011, 198, 118–129. [Google Scholar]
- US Food and Drug Administration. Action levels for poisonous or deleterious substances in human food and animal feed. Available online: http://www.cfsan.fda.gov/_lrd/fdaact.html (accessed on 20 September 2002).
- Jones, R.K.; Duncan, H.E.; Hamilton, P.B. Planting date, harvest date, and irrigation effects on infection and aflatoxin production by Aspergillus flavus in field corn. Phytopathology 1981, 71, 810–816. [Google Scholar] [CrossRef]
- Gomathi, R.; Rakkiyapan, P. Comparative lipid peroxidation, leaf membrane thermostability, and antioxidant system in four sugarcane genotypes differing in salt tolerance. Int. J. Plant Physiol. Biochem. 2011, 3, 67–74. [Google Scholar]
- Pioneer corn product catalog. Available online: https://www.pioneer.com/home/site/us/products/corn/ (accessed on 6 January 2009).
- Duvick, D.N. Genetic progress in yield of United States maize (Zea mays L.). Maydica 2005, 50, 193–202. [Google Scholar]
- Van Oosterom, E.J.; Jayachandran, R.; Bidinger, F.R. Diallel analysis of the stay-green trait and its components in sorghum. Crop Sci. 1996, 36, 549–555. [Google Scholar]
- Thomas, H.; Smar, C.M. Crops that stay green. Ann. Appl. Biol. 2008, 123, 193–219. [Google Scholar] [CrossRef]
- Bekavac, G.; Purar, B.; Stojakovih, M.; Jockovih, D.J.; Ivanovih, M.; Nastasih, A. Genetic analysis of stay-green trait in broad-based maize populations. Cereal Res. Commun. 2007, 35, 31–41. [Google Scholar]
- Dorner, J.W.; Cole, R.J.; Sanders, T.H.; Blankenship, P.D. Interrelationship of kernel water activity, soil temperature, maturity, and phytoalexin production in preharvest aflatoxin contamination of drought-stressed peanuts. Mycopathologia 1989, 105, 117–128. [Google Scholar] [CrossRef]
- Brown, R.L.; Bhatnagar, D.; Cleveland, T.E.; Cary, J.W. Recent Advances in Preventing Mycotoxin Vontamination. In Mycotoxins in Agriculture and Food Safety; Sinha, K.K., Bhatnagar, D., Eds.; Marcel Dekker: New York, NY, USA, 1998; pp. 351–379. [Google Scholar]
- Chen, Z.Y.; Brown, R.L.; Lax, A.R.; Guo, B.Z.; Cleveland, T.E.; Russin, J.S. Resistance to Aspergillus flavus in corn kernels is associated with a 14-kDa protein. Phytopathology 1998, 88, 276–281. [Google Scholar] [CrossRef]
- Moore, K.G.; Price, M.S.; Boston, R.S.; Weissinger, A.K.; Payne, G.A. Chitinase from Tex6 maize kernels inhibits growth of Aspergillus flavus. Phytopathology 2004, 94, 82–87. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Brown, R.L.; Damann, K.E.; Cleveland, T.E. PR10 expression in maize and its effect on host resistance against Aspergillus flavus infection and aflatoxin production. Mol. Plant Pathol. 2010, 11, 69–81. [Google Scholar] [CrossRef]
- Baker, R.L.; Brown, R.L.; Chen, Z.Y.; Cleveland, T.E.; Fakhoury, A.M. A maize lectin-like protein with antifungal activity against Aspergillus flavus. J. Food Prot. 2009, 72, 120–127. [Google Scholar]
- Russin, J.S.; Guo, B.Z.; Tubajika, K.M.; Brown, R.L.; Cleveland, T.E.; Widstrom, N.W. Comparison of kernel wax from corn genotypes resistant or susceptible to Aspergillus flavus. Phytopathology 1997, 87, 529–533. [Google Scholar] [CrossRef]
- Gembeh, S.V.; Brown, R.L.; Grimm, C.; Cleveland, T.E. Identification of chemical components of corn kernel pericarp wax associated with resistance to Aspergillus flavus infection and aflatoxin production. J. Agric. Food Chem. 2001, 49, 4635–4641. [Google Scholar] [CrossRef]
- Cary, J.W.; Rajasekaran, K.; Brown, R.L.; Luo, M.; Chen, Z.Y.; Bhatnagar, D. Developing resistance to aflatoxin in maize and cottonseed. Toxins 2011, 3, 678–696. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Brown, R.L.; Damann, K.E.; Cleveland, T.E. Identification of maize kernel endosperm proteins associated with resistance to aflatoxin contamination by Aspergillus flavus. Phytopathology 2007, 97, 1094–1103. [Google Scholar] [CrossRef]
- Brown, R.L.; Chen, Z.Y.; Warburton, M.; Luo, M.; Menkir, A.; Fakhoury, A.; Bhatnagar, D. Discovery and characterization of proteins associated with aflatoxin-resistance: Evaluating their potential as breeding markers. Toxins 2010, 2, 919–933. [Google Scholar] [CrossRef]
- Oberschall, A.; Deak, M.; Torok, K.; Sass, L.; Vass, I.; Kovacs, I.; Feher, A.; Dudits, D.; Horvath, G.V. A novel aldose/aldehyde reductase protects transgenic plants against lipid peroxidation under chemical and drought stresses. Plant J. 2000, 24, 437–446. [Google Scholar] [CrossRef]
- Jiang, Y.; Huang, B. Physiological responses to heat stress alone or in combination with drought: A comparison between tall fescue and perennial ryegrass. HortScience 2001, 36, 682–686. [Google Scholar]
- Fisher, D.K.; Kebede, H. A low-cost microcontroller-based system to monitor crop temperature and water status. Comput. Electron. Agric. 2010, 74, 168–173. [Google Scholar] [CrossRef]
- Abbas, H.K.; Zablotowicz, R.M.; Bruns, H.A.; Abel, C.A. Biocontrol of aflatoxin in corn by inoculation with non-aflatoxigenic Aspergillus flavus isolates. Biocontrol Sci. Technol. 2006, 16, 437–449. [Google Scholar] [CrossRef]
- Abbas, H.K.; Accinelli, C.; Zablotowicz, R.M.; Abel, C.A.; Bruns, H.A.; Dong, Y.; Shier, W.T. Dynamics of mycotoxin and Aspergillus flavus levels in aging Bt and non-Bt corn residues under Mississippi no-till conditions. J. Agric. Food Chem. 2008, 56, 7578–7585. [Google Scholar] [CrossRef]
- Hendry, G.A.F.; Price, A.H. Stress Indicators: Chlorophylls and Carotenoids. In Methods in Comparative Plant Ecology; Hendry, G.A.F., Grime, J.P., Eds.; Chapman & Hall: London, UK, 1993; pp. 148–152. [Google Scholar]
- Sullivan, C.Y. Mechanisms of Heat and Drought Resistance in Grain Sorghum and Methods of Measurement. In Sorghum in the Seventies; Rao, N.G.P., House, L.R., Eds.; Oxford and IPH: New Delhi, India, 1972; pp. 247–264. [Google Scholar]
- Bellaloui, N. Effect of water stress and foliar boron application on seed protein, oil fatty acids, and nitrogen metabolism in soybean. Am. J. Plant Sci. 2011, 2, 692–701. [Google Scholar] [CrossRef]
- Reddy, K.N.; Bellaloui, N; Zablotowicz, R. Glyphosate effect on shikimate, nitrate reductase activity, yield, and seed composition in corn. J. Agric. Food Chem. 2010, 58, 3646–3650. [Google Scholar] [CrossRef]
- The Association of Official Analytical Chemists, Method 988.05. In Official Methods of Analysis, 15th; Helrich, K. (Ed.) The Association of Official Analytical Chemists, Inc.: Arlington, Virginia, VA, USA, 1990.
- The Association of Official Analytical Chemists, Method 920.39. In Official Methods of Analysis, 15t; Helrich, K. (Ed.) The Association of Official Analytical Chemists, Inc.: Arlington, Virginia, VA, USA, 1990.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kebede, H.; Abbas, H.K.; Fisher, D.K.; Bellaloui, N. Relationship between Aflatoxin Contamination and Physiological Responses of Corn Plants under Drought and Heat Stress. Toxins 2012, 4, 1385-1403. https://doi.org/10.3390/toxins4111385
Kebede H, Abbas HK, Fisher DK, Bellaloui N. Relationship between Aflatoxin Contamination and Physiological Responses of Corn Plants under Drought and Heat Stress. Toxins. 2012; 4(11):1385-1403. https://doi.org/10.3390/toxins4111385
Chicago/Turabian StyleKebede, Hirut, Hamed K. Abbas, Daniel K. Fisher, and Nacer Bellaloui. 2012. "Relationship between Aflatoxin Contamination and Physiological Responses of Corn Plants under Drought and Heat Stress" Toxins 4, no. 11: 1385-1403. https://doi.org/10.3390/toxins4111385
APA StyleKebede, H., Abbas, H. K., Fisher, D. K., & Bellaloui, N. (2012). Relationship between Aflatoxin Contamination and Physiological Responses of Corn Plants under Drought and Heat Stress. Toxins, 4(11), 1385-1403. https://doi.org/10.3390/toxins4111385





