Natural Occurrence of Ochratoxin A in Musts, Wines and Grape Vine Fruits from Grapes Harvested in Argentina
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of Standard Solutions of OTA
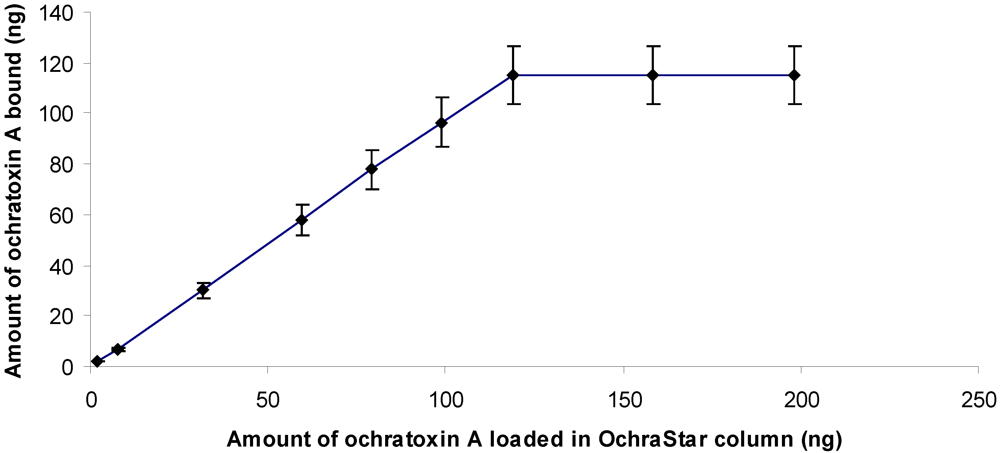
2.3. Column Capacity
2.4. Extraction and Clean-up with IAC
- (a) Extraction with a solution containing acetonitrile: water (60:40) and clean up with IAC. Briefly, 25 g of must was diluted with 100 mL of acetonitrile: water (60:40), mixed, and filtered to remove particulate matter. An 8 mL portion of this dilution was evaporated to dryness and resuspended in 56 mL of PBS (acetonitrile never exceeds 7.5% v/v); finally the pH value was adjusted to 7.4 and added to the IAC. The IAC was washed twice with 10 mL of ammonium acetate (AcNH4) 0.2 M. Ochratoxin A was eluted from the column with 1.5 mL of methanol (HPLC grade), at a flow rate of 1–2 drops per second.
- (b) Must dilution with PBS and clean up with IAC. Briefly, 8 g of must was diluted with 56 mL of PBS buffer, mixed, filtered to remove particulate matter, and added to the IAC. The column was washed twice with 10 mL of ammonium acetate (AcNH4) 0.2 M. OTA was eluted from the column with 1.5 mL of methanol: acetic acid (98:2), at a flow rate of 1–2 drops per second.
- (c) Must dilution with water solution containing polyethylene glycol 8000 (1%) and NaHCO3 (5%) and clean-up on IAC according to the methodology proposed by Visconti et al. (1999) [34] for wine samples. In brief, must was diluted with water solution containing 1% PEG and 5% NaHCO3, mixed, filtered to remove particulate matter, and a 10 mL portion was taken and added to the IAC. The IAC was washed with 5 mL of an aqueous solution of 2.5% NaCl and 0.5% NaHCO3 and then with 5 mL double distilled water. OTA was eluted from the column with 1.5 mL of methanol (HPLC grade), at a flow rate of 1–2 drops per second.
2.5. Ochratoxin A Detection and Quantification
2.6. Assay: Spiking and Recovery of OTA in Musts, Wines and Dried Vine Fruits
2.7. Origin of Samples for OTA Natural Occurrence Determination
3. Results and Discussion
| Grapes by Product | Extraction/Clean Up Procedure | Mean Recovery (%) ± SD | RSDr (%) | LOD | LOQ |
|---|---|---|---|---|---|
| Must | (a) Extraction with Acetonitrile:Water (60:40) + OchraStarTM clean-up. | 98.9 ± 4.84 | 4.80 | 0.05 a | 0.1 a |
| (b) Must diluted with PBS + OchraStarTM clean-up. | 40.35 ± 12.42 | 22.3 | 0.05 a | 0.1 a | |
| (c) Dilution with PEG-NaHCO3 + OchraStarTM clean-up | 100.3 ± 2.4 | 2.35 | 0.05 a | 0.1 a | |
| Wine | Official method proposed by Visconti et al. 2001 [29] | 90 ± 2 | 2.50 | 0.01 a | 0.1 a |
| Dried vine fruits | Method proposed by Möller et al. 2003 [35] | 95 ± 2.5 | 2.60 | 0.1 b | 1 b |
| Region of Origin | Number of Positive Samples/Total samples | Range of OTA (ng/mL) | Mean OTA Levels (ng/mL) |
|---|---|---|---|
| Mendoza province | 3/30 | ≤0.1–0.16 | 0.12 ± 0.04 |
| Chubut province | 0/2 | ND | ND |
| Region of Origin | Positive Sample/Total Samples | Concentration Range of OTA (ng/mL) |
|---|---|---|
| Famatina Valley (La Rioja province) | 1/6 | 2 |
| Tulum Valley (San Juan province) | 0/6 | ND |
| ZARM (Mendoza Province) | 0/8 | ND |
| Uco Valley (Mendoza province) | 1/11 | 0.02 |
| North of Mendoza | 1/6 | 3.14 |
| South of Mendoza | 1/7 | 4.82 |
| Neuquén – Río Negro | 0/3 | ND |
| Total | 4/47 | 0.02–4.82 |
| Number of Positive Samples/ Total Samples | Rangeof OTA (ng/g) | Mean OTA Levels (ng/g) |
|---|---|---|
| 2/16 | 8.66–20.28 | 14.47 ± 8.21 |
| 8/16 | 0.26–3.1 | 1.07 ± 1.01 |
| 6/16 | ND | ND |
4. Conclusions
Acknowledgements
References
- IARC, Monographs on the Evaluation of Carcinogenic Risks to Humans. Some Naturally Occurring Substance, Food Items and Constituents, Heteroxyclic Aromatic Amines and Mycotoxins. IARC International Agency for Research on Cancer: Lyon, France, 1993; 56, pp. 489–521.
- Joint FAO/WHO Experts Committee on Food Additives. Available online: http://www.who.int/ipcs/food/jecfa/en/ (accessed on 28 July 2010).
- Park, J.W.; Chung, S.H.; Kim, Y.B. Ochratoxin A in Korean food commodities: Occurrence and safety evaluation. J. Agric. Food Chem. 2005, 53, 4637–4642. [Google Scholar]
- Overy, D.P.; Seifert, K.A.; Savard, M.E.; Frisvad, J.C. Spoilage fungi and their mycotoxins in commercially marketed chestnuts. Int. J. Food Microbiol. 2003, 88, 69–77. [Google Scholar]
- Drusch, S.; Ragab, W. Mycotoxins in fruits, fruit juices, and dried fruits. J. Food Prot. 2003, 66, 1514–1527. [Google Scholar]
- Magnoli, C.E.; Ponsone, M.L.; Combina, M.; Palacio, G.; da Rocha Rosa, C.A.; Dalcero, A. Survey of mycoflora and ochratoxin A in dried vine fruits from Argentina markets. Lett. Appl. Microbiol. 2004, 39, 326–331. [Google Scholar]
- Meyvaci, K.B.; Altindisli, A.; Aksoy, U.; Eltem, R.; Turgut, H.; Arasiler, Z.; Kartal, N. Ochratoxin A in sultanas from Turkey I: Survey of unprocessed sultanas from vineyards and packing-houses. Part A: Chemistry, Analysis, Control, Exposure and Risk Assessment. Food Addit. Contam. 2005, 22, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Aksoy, U.; Eltem, R.; Meyvaci, K.B.; Altindisli, A.; Karabat, S. Five-year survey of ochratoxin A in processed sultanas from Turkey. Food Addit. Contam. 2007, 24, 292–296. [Google Scholar]
- Meletis, K.; Meniades-Meimaroglou, S.; Markaki, P. Determination of ochratoxin A in grapes of Greek origin by immunoaffinity and high-performance liquid chromatography. Food Addit. Contam. 2007, 24, 1275–1282. [Google Scholar]
- Zinedine, A.; Soriano, J.M.; Juan, C.; Mojemmi, B.; Molto, J.C.; Bouklouze, A.; Cherrah, Y.; Idrissi, L.; El Aouad, R.; Manes, J. Incidence of ochratoxin A in rice and dried fruits from Rabat and Sale area, Morocco. Food Addit. Contam. 2007, 24, 285–291. [Google Scholar]
- Solfrizzo, M.; Panzarini, G.; Visconti, A. Determination of Ochratoxin A in Grapes, Dried Vine Fruits, and Winery Byproducts by High-Performance Liquid Chromatography with Fluorometric Detection (HPLC−FLD) and Immunoaffinity Cleanup. J. Agric. Food Chem. 2008, 56, 11081–11086. [Google Scholar]
- Bircan, C. Incidence of ochratoxin A in dried fruits and co-occurrence with aflatoxins in dried figs. Food Chem. Toxicol. 2009, 47, 1996–2001. [Google Scholar]
- Napolitano, A.; Fogliano, V.; Tafuri, A.; Ritieni, A. Natural occurrence of ochratoxin A and antioxidant activities of green and roasted coffees and corresponding by-products. J. Agric. Food Chem. 2007, 55, 10499–10504. [Google Scholar]
- Skaug, M.A. Analysis of Norwegian milk and infant formulas for ochratoxin A. Food Addit. Contam. 1999, 16, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Tangni, E.K.; Ponchaut, S.; Maudoux, M.; Rozenberg, R.; Larondelle, Y. Ochratoxin A in domestic and imported beers in Belgium: Occurrence and exposure assessment. Food Addit. Contam. 2002, 12, 1169–1179. [Google Scholar]
- Pietri, A.; Bertuzzi, T.; Pallaroni, L.; Piva, G. Occurrence of ochratoxin A in Italian wines. Food Addit. Contam. 2001, 18, 647–654. [Google Scholar] [PubMed]
- Rosa, C.A.R.; Magnoli, C.E.; Fraga, M.E.; Dalcero, A.M.; Santana, D.N.M. Ocurrence of ochratoxin A in wine and grape juice marketed in Rio de Janeiro, Brazil. Food Addit. Contam. 2004, 21, 358–364. [Google Scholar]
- Zimmerli, B.; Dick, R. Ochratoxin A in table wine and grape-juice: Occurrence and risk assessment. Food Addit. Contam. 1996, 13, 655–668. [Google Scholar]
- Visconti, A.; Perrone, G.; Cozzi, G.; Solfrizzo, M. Managing ochratoxin A risk in the grape-wine food chain. Food Addit. Contam. 2008, 25, 193–202. [Google Scholar]
- Kozakiewicz, Z.; Battilani, P. Making wine safer: The case of ochratoxin A. The Second World Mycotoxin Forum, Amsterdam, The Netherlands; 17–18 2 2003. [Google Scholar]
- Ponsone, M.L.; Combina, M.; Dalcero, A.; Chulze, S. Occurrence of Ochratoxin A and Aspergillus species ochratoxin A producers in Argentinian wine grapes. Int. J. Food Microbiol. 2007, 114, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Cozzi, G.; Pascale, M.; Perrone, G.; Visconti, A.; Logrieco, A. Effect of Lobesia botrana damages on black aspergilli rot and ochratoxin A content in grapes. Int. J. Food Microbiol. 2006, 111, 88–92. [Google Scholar]
- Leong, S.L.; Hocking, A.D.; Pitt, J.I.; Kazi, B.A.; Emmett, R.W.; Scott, E.S. Australian research on ochratoxigenic fungi and ochratoxin A. Int. J. Food Microbiol. 2006, 111, 10–17. [Google Scholar]
- Ponsone, M.L.; Chiotta, M.L.; Combina, M.; Dalcero, A.M.; Chulze, S.N. Fate of ochratoxin A content in Argentinean red wine during a pilot scale vinification. Revista Argentina de Microbiología 2009, 41, 245–250. [Google Scholar] [PubMed]
- Jordan, K.J. Ready-to-eat dry fruit products and process. United States Patent 7569244, 8 April 2009. [Google Scholar]
- INV. Estadísticas de cosecha 2009. Producción de uvas según provincia de origen. Available online: http://www.inv.gov.ar/PDF/Cosecha2009/ProduccionPorProvincia_pasas.PDF (accessed on 27 July 2010).
- Pateraki, M.; Dekanea, A.; Mitchell, D.; Lydakis, D.; Magan, N. Influence of sulphur dioxide, controlled atmospheres and water availability on in vitro germination, growth and ochratoxin A production by strains of Aspergillus carbonarius isolated from grapes. Postharvest Biol. Technol. 2007, 44, 141–149. [Google Scholar]
- Magan, N.; Aldred, D. Conditions of formation of ochratoxin in drying, transport and in different commodities. Food Addit. Contam. 2005, 1S, 10–16. [Google Scholar]
- Visconti, A.; Pascale, M.; Centonze, G. Determination of ochratoxin A in wine and beer by immunoaffinity column cleanup and liquid chromatographic analysis with fluorometric detection: Collaborative study. J. AOAC Int. 2001, 84, 1818–1827. [Google Scholar]
- MacDonald, S.J.; Anderson, S.; Brereton, P.; Wood, R. Determination of ochratoxin A in currants, raisins, sultanas, mixed dried fruit, and dried figs by immunoaffinity column cleanup with liquid chromatography: Interlaboratory study. J. AOAC Int. 2003, 86, 1164–1171. [Google Scholar]
- CEN EN 14133 Foodstuffs—Determination of Ochratoxin A in Wine and Beer—HPLC Method with Immunoaffinity Column Clean-Up. BSI: Brussels, Belgium, 2009.
- OIV-Organisation Internationale de la Vigne et du Vin. Available online: http://news.reseau-concept.net/images/oiv_es/Client/OENO_16-2001es.pdf (accessed on 12 December 2004).
- Serra, R.; Mendonça, C.; Abrunhosa, L.; Pietri, A.; Venâncio, A. Determination of ochratoxin A in wine grapes: Comparison of extraction procedures and method validation. Anal. Chim. Acta 2004, 513, 41–47. [Google Scholar]
- Visconti, A.; Pascale, M.; Centonze, G. Determination of Ochratoxin A in wine by means of immunafinity column clean-up and high-performance liquid chromatography. J. Chromatogr. A 1999, 864, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Möller, T.E.; Nyberg, M. Ochratoxin A in raisins and currants: Basic extraction procedure used in two small marketing surveys of the occurrence and control of the heterogeneity of the toxins in samples. Food Addit. Contam. 2003, 20, 1072–1076. [Google Scholar]
- Catania, C.D.; Avagnina de del Monte, S.; Uliarte, E.M.; del Monte, R.F.; Tonietto, J. El clima vitícola de las regiones productoras de uvas para vinos de Argentina. In Caracterização climática de regiões vitivinícolas ibero-americanas; Embrapa Uva e Vinho: Bento Gonçalves, Brazil, 2007; pp. 9–47. [Google Scholar]
- Gilbert, J.; Anklam, E. Validation of analytical methods for determining mycotoxins in foodstuffs. Trends Anal. Chem. 2002, 21, 468–486. [Google Scholar]
- Chiotta, M.L.; Ponsone, M.L.; Combina, M.; Torres, A.; Chulze, S. Aspergillus section Nigri species isolated from different wine-grape growing regions in Argentina. Int. J. Food Microbiol. 2009, 136, 137–141. [Google Scholar]
- El Khoury, A.; Rizk, T.; Lteif, R.; Azouri, H.; Delia, M.L.; Lebrihi, A. Occurrence of ochratoxin A- and aflatoxin B1-producing fungi in Lebanese grapes and ochratoxin a content in musts and finished wines during 2004. J. Agric. Food Chem. 2006, 54, 8977–8982. [Google Scholar]
- El Khoury, A.; Rizk, T.; Lteif, R.; Azouri, H.; Delia, M.L.; Lebrihi, A. Fungal contamination and Aflatoxin B1 and Ochratoxin A in Lebanese wine–grapes and musts. Food Chem. Toxicol. 2008, 46, 2244–2250. [Google Scholar]
- Fernández, A.; Ratola, N.; Cerdeira, A.; Alves, A.; Venâncio, A. Changes in Ochratoxin A Concentration during Winemaking. Am. J. Enol. Vitic. 2007, 58, 92–96. [Google Scholar]
- Ratola, N.; Abade, E.; Simões, T.; Venâncio, A.; Alves, A. Evolution of ochratoxin A content from must to wine in Port Wine microvinification. Anal. Bioanal. Chem. 2005, 382, 405–411. [Google Scholar] [Green Version]
- Battilani, P.; Giorni, P.; Bertuzzi, T.; Formenti, S.; Pietri, A. Black aspergilli and ochratoxin A in grapes in Italy. Int. J. Food Microbiol. 2006, 111, 53–60. [Google Scholar]
- Battilani, P.; Barbano, C.; Marin, S.; Sanchis, V.; Kozakiewicz, Z.; Magan, N. Mapping of Aspergillus section Nigri in Southern Europe and Israel based on geostatistical analysis. Int. J. Food Microbiol. 2006, 111, 72–82. [Google Scholar]
- Bellí, N.; Bau, M.; Marin, S.; Abarca, M.L.; Ramos, A.J.; Bragulat, M.R. Mycobiota and ochratoxin A producing fungi from Spanish wine grapes. Int. J. Food Microbiol. 2006, 111, 40–45. [Google Scholar]
- Sage, L.; Garon, D.; Seigle-Murandi, F. Fungal microflora and ochratoxin risk in French vineyards. J. Agric. Food Chem. 2004, 52, 5764–5768. [Google Scholar]
- Serra, R.; Lourenço, A.; Alípio, P.; Venâncio, A. Influence of the region of origin on the mycobiota of grapes with emphasis on Aspergillus and Penicillium species. Mycol. Res. 2006, 110, 971–978. [Google Scholar]
- MacDonald, S.; Wilson, P.; Barnes, K.; Damant, A.; Massey, R.; Mortby, E.; Shepherd, M.J. Ochratoxin A in dried vine fruit: Method development and survey. Food Addit. Contam. 1999, 16, 253–260. [Google Scholar]
- Hocking, A.D.; Varelis, P.; Pitt, J.I.; Cameron, S.; Leong, S. Occurrence of ochratoxin A in Australian wine. Aust. J. Grape Wine Res. 2003, 9, 72–78. [Google Scholar]
- European Commission 2002. SCOOP EC Directorate–General Health and Consumer Protection. Assessment of dietary intake of ochratoxin A by the population of EU Member States. Reports on tasks for scientific cooperation. Brussels, Belgium, 2002. Available online: http://ec.europa.eu/food/fs/scoop/3.2.7_en.pdf (Accessed on 5 September 2007).
- Stefanaki, I.; Foufa, E.; Tsatsou-Dritsa, A.; Dais, P. Ochratoxin A concentration in Greek domestic wines and dried vine fruits. Food Addit. Contam. 2003, 20, 74–83. [Google Scholar]
- Battilani, P.; Pietri, A.; Giorni, P.; Bertuzzi, T.; Barbano, C. Growth and ochratoxin A production of Aspergillus section Nigri isolates from Italian grapes. Aspects Appl. Biol. 2003, 68, 175–180. [Google Scholar]
- Mitchell, D.; Aldred, D.; Magan, N. Impact of ecological factors on the growth and ochratoxin A production by Aspergillus carbonarius from different regions of Europe. Aspects Appl. Biol. 2003, 68, 109–116. [Google Scholar]
- Mitchell, D.; Parra, R.; Aldred, D.; Magan, N. Water and temperature relations of growth and ochratoxin A production by Aspergillus carbonarius strains from grapes in Europe and Israel. J. Appl. Microbiol. 2004, 97, 439–445. [Google Scholar]
- Bellí, N.; Marín, S.; Sanchis, V.; Ramos, A.J. Influence of water activity and temperature on growth of isolates of Aspergillus section Nigri obtained from grapes. Int. J. Food Microbiol. 2004, 96, 19–27. [Google Scholar]
- Leong, S.L.; Hocking, A.D.; Scott, E.S. Effect of temperature and water activity on growth and ochratoxin A production by Australian Aspergillus carbonarius and A. niger isolates on a simulated grape juice medium. Int. J. Food Microbiol. 2006, 110, 209–216. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ponsone, M.L.; Chiotta, M.L.; Combina, M.; Torres, A.; Knass, P.; Dalcero, A.; Chulze, S. Natural Occurrence of Ochratoxin A in Musts, Wines and Grape Vine Fruits from Grapes Harvested in Argentina. Toxins 2010, 2, 1984-1996. https://doi.org/10.3390/toxins2081984
Ponsone ML, Chiotta ML, Combina M, Torres A, Knass P, Dalcero A, Chulze S. Natural Occurrence of Ochratoxin A in Musts, Wines and Grape Vine Fruits from Grapes Harvested in Argentina. Toxins. 2010; 2(8):1984-1996. https://doi.org/10.3390/toxins2081984
Chicago/Turabian StylePonsone, María Lorena, María Laura Chiotta, Mariana Combina, Adriana Torres, Patricia Knass, Ana Dalcero, and Sofía Chulze. 2010. "Natural Occurrence of Ochratoxin A in Musts, Wines and Grape Vine Fruits from Grapes Harvested in Argentina" Toxins 2, no. 8: 1984-1996. https://doi.org/10.3390/toxins2081984






