Vitamin D Levels Are Associated with Cardiac Autonomic Activity in Healthy Humans
Abstract
:1. Introduction
2. Methods
2.1. Cardiac Autonomic Activity
2.2. Biochemical Analysis
2.3. Statistical Analysis
3. Results
3.1. Subject Characteristics
| Stratification by 25-hydroxy vitamin D status | Stratification by 1,25-dihydroxy vitamin D status | ||||||
|---|---|---|---|---|---|---|---|
| All Subjects (n = 34) | Deficient <50 nmol/L (n = 7) | Non-Deficient >50 nmol/L (n = 27) | p-value | Below 25th percentile <76 pmol/L (n = 8) | Above 25th percentile >76 pmol/L (n = 26) | p-value | |
| Age (years) | 38 ± 2 | 37 ± 5 | 38 ± 3 | 0.96 | 37 ± 4 | 38 ± 3 | 0.92 |
| Caucasian (%) | 25 (74%) | 4 (57%) | 21 (77%) | 0.08 | 7 (88%) | 18 (69%) | 0.56 |
| Female (%) | 22 (65%) | 5 (71%) | 17 (63%) | 0.69 | 6 (75%) | 16 (62%) | 0.22 |
| BMI (kg/m2) | 26 ± 1 | 27 ± 1 | 25 ± 1 | 0.43 | 27 ± 2 | 25 ± 1 | 0.21 |
| SBP (mmHg) | 115 ± 2 | 112 ± 4 | 115 ± 3 | 0.63 | 115 ± 2 | 115 ± 3 | 0.37 |
| DBP (mmHg) | 68 ± 1 | 64 ± 5 | 69 ± 2 | 0.28 | 70 ± 3 | 68 ± 2 | 0.44 |
| MAP (mmHg) | 84 ± 2 | 80 ± 4 | 84 ± 2 | 0.37 | 85 ± 2 | 83 ± 2 | 0.41 |
| 25-hydroxy vitamin D (nmol/L) | 71 ± 4 | 38 ± 2 | 81 ± 4 | <0.001 | 61 ± 7 | 74 ± 5 | 0.16 |
| 1,25-dihydroxy vitamin D (pmol/L) | 105 ± 6 | 86 ± 9 | 107 ± 7 | 0.15 | 64 ± 3 | 117 ± 6 | <0.001 |
| PTH (ng/L) | 37 ± 2 | 45 ± 9 | 34 ± 2 | 0.046 | 33 ± 6 | 38 ± 3 | 0.29 |
| Serum calcium (mmol/L) | 2.2 ± 0.01 | 2.3 ± 0.04 | 2.2 ± 0.01 | 0.47 | 2.2 ± 0.03 | 2.2 ± 0.02 | 0.49 |
| Serum phosphate (mmol/L) | 1.0 ± 0.03 | 0.9 ± 0.08 | 1.01 ± 0.03 | 0.35 | 1.03 ± 0.04 | 0.97 ± 0.04 | 0.29 |
| HDL (mmol/L) | 1.4 ± 0.05 | 1.3 ± 0.1 | 1.5 ± 0.05 | 0.21 | 1.45 ± 0.07 | 1.4 ± 0.06 | 0.62 |
| LDL (mmol/L) | 2.2 ± 0.1 | 1.8 ± 0.2 | 2.3 ± 0.2 | 0.17 | 2.1 ± 0.4 | 2.2 ± 0.2 | 0.65 |
| NE (nmol/L) | 1.3 ± 0.2 | 1.2 ± 0.06 | 1.3 ± 0.2 | 0.79 | 1.5 ± 0.3 | 1.3 ± 0.2 | 0.37 |
| Epi (pmol/L) | 97 ± 13 | 69 ± 11 | 117 ± 17 | 0.019 | 76 ± 13 | 103 ± 15 | 0.64 |
| Urinary sodium (mmol/day) | 373 ± 20 | 355 ± 48 | 379 ± 25 | 0.65 | 348 ± 25 | 380 ± 27 | 0.40 |
| Serum creatinine (umol/L) | 69 ± 4 | 69 ± 7 | 70 ± 3 | 0.92 | 70 ± 5 | 67 ± 4 | 0.98 |
| PRA (ng/L/s) | 0.21 ± 0.03 | 0.23 ± 0.04 | 0.20 ± 0.03 | 0.48 | 0.15 ± 0.03 | 0.23 ± 0.03 | 0.19 |
| Ang II (ng/L) | 18 ± 1 | 18 ± 3 | 16 ± 1 | 0.38 | 18 ± 2 | 18 ± 1 | 0.81 |
| Aldo (pmol/L) | 106 ± 10 | 90 ± 8 | 110 ± 12 | 0.61 | 99 ± 14 | 109 ± 12 | 0.91 |
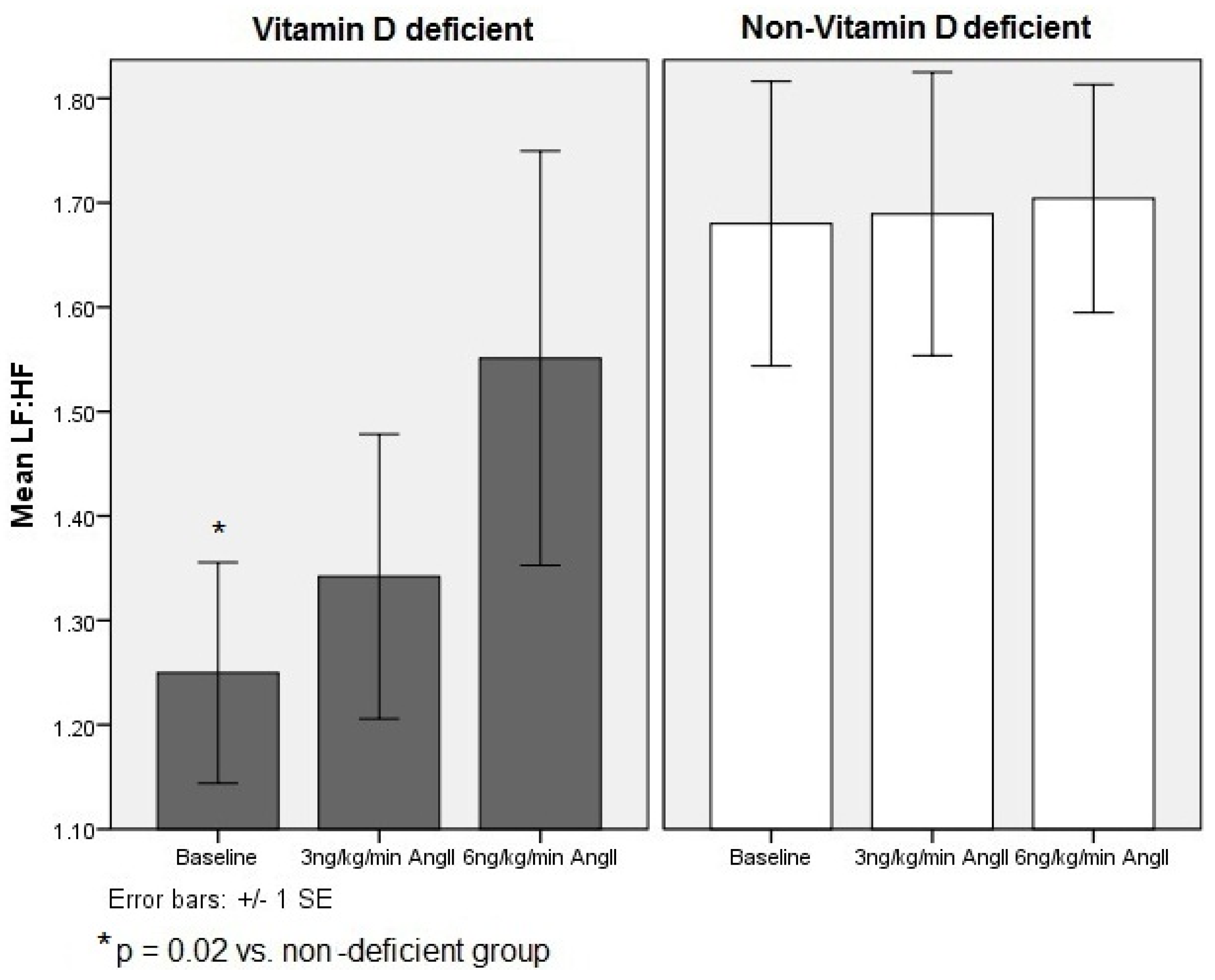
3.2. Cardiac Autonomic Responses in 25-Hydroxy Vitamin D Deficient vs. Non-Vitamin D Deficient Subjects
| Baseline | 3 ng/kg/min AngII | 6 ng/kg/min AngII | |
|---|---|---|---|
| Heart rate (bpm) | |||
| Vitamin D deficient Non-vitamin D deficient | 62 ± 3 57 ± 2 | 56 ± 3 56 ± 3 | 65 ± 4 58 ± 2 |
| TP (ms) | |||
| Vitamin D deficient | 56 ± 7 | 66 ± 7 | 63 ± 8 |
| Non-vitamin D deficient | 69 ± 6 | 74 ± 7 | 73 ± 7 |
| VLF (ms) | |||
| Vitamin D deficient | 39 ± 6 | 48 ± 6 | 47 ± 6 |
| Non-vitamin D deficient | 51 ± 5 | 55 ± 7 | 56 ± 5 |
| LF (ms) | |||
| Vitamin D deficient | 31 ± 4 | 34 ± 4 | 33 ± 5 |
| Non-vitamin D deficient | 37 ± 4 | 37 ± 3 | 37 ± 3 |
| LF (ln ms2) | |||
| Vitamin D deficient | 6.76 ± 0.3 | 6.93 ± 0.3 | 6.84 ± 0.3 |
| Non-vitamin D deficient | 6.99 ± 0.2 | 6.97 ± 0.2 | 7.00 ± 0.2 |
| LF (nu) | |||
| Vitamin D deficient | 61 ± 5 | 58 ± 3 | 62 ± 4 |
| Non-vitamin D deficient | 67 ± 3 | 66 ± 3 | 68 ± 2 |
| HF (ms) | |||
| Vitamin D deficient | 25 ± 3 | 26 ± 3 | 24 ± 4 |
| Non-vitamin D deficient | 25 ± 3 | 24 ± 3 | 23 ± 2 |
| HF (ln ms2) | |||
| Vitamin D deficient | 6.36 ± 0.3 | 6.40 ± 0.3 | 6.06 ± 0.6 |
| Non-vitamin D deficient | 6.12 ± 0.2 | 6.02 ± 0.2 | 5.98 ± 0.2 |
| HF (nu) | |||
| Vitamin D deficient | 41 ± 7 | 37 ± 4 | 32 ± 5 |
| Non-vitamin D deficient | 33 ± 3 | 28 ± 3 | 26 ± 2 |
| LF:HF | |||
| Vitamin D deficient | 1.25 ± 0.1 | 1.34 ± 0.2 | 1.55 ± 0.2 |
| Non-vitamin D deficient | 1.66 ± 0.1 † | 1.72 ± 0.1 | 1.74 ± 0.1 |
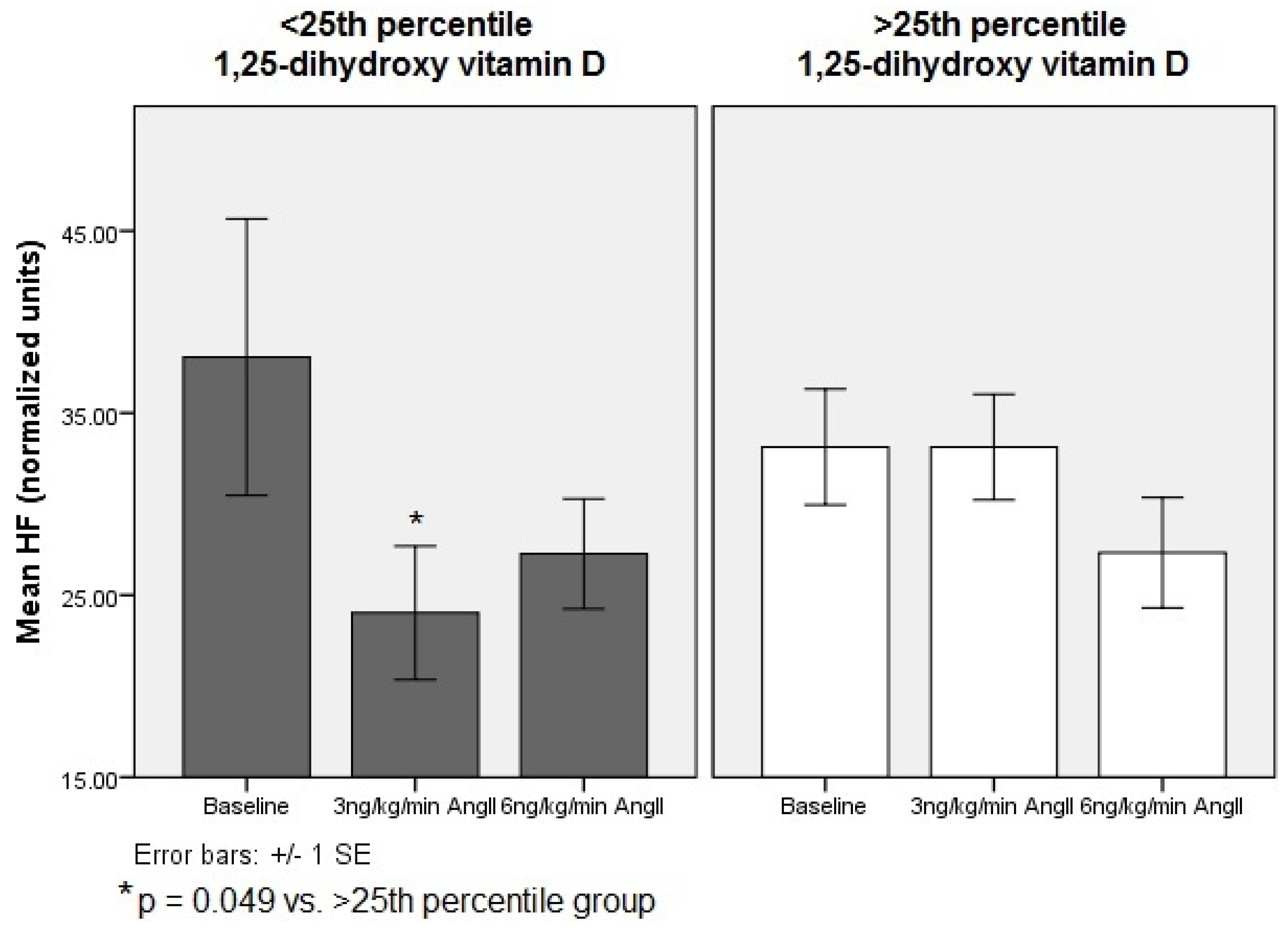
3.3. Cardiac Autonomic Responses below vs. above 25th Percentile 1,25-Dihydroxy Vitamin D Subjects
| Baseline | 3 ng/kg/min AngII | 6 ng/kg/min AngII | |
|---|---|---|---|
| Heart rate (bpm) | 59 ± 3 58 ± 2 | 60 ± 2 56 ± 2 | 62 ± 3 58 ± 2 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| TP (ms) | 70 ± 15 66 ± 5 | 83 ± 16 69 ± 6 | 85 ± 18 67 ± 5 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| VLF (ms) | 50 ± 12 48 ± 4 | 66 ± 13 50 ± 5 | 58 ± 11 53 ± 4 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| LF (ms) | 40 ± 8 35 ± 3 | 43 ± 8 34 ± 3 | 40 ± 10 35 ± 2 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| LF (ln ms2) | 7.04 ± 0.4 6.91 ± 0.2 | 7.28 ± 0.4 6.87 ± 0.2 | 6.89 ± 0.5 6.99 ± 0.2 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| LF (nu) | 67 ± 4 65 ± 3 | 72 ± 4 62 ± 3 | 69 ± 4 66 ± 2 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| HF (ms) | 25 ± 5 25 ± 3 | 23 ± 4 25 ± 2 | 21 ± 5 24 ± 2 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| HF (ln ms2) | 6.15 ± 0.4 6.17 ± 0.2 | 6.04 ± 0.3 6.12 ± 0.2 | 5.79 ± 0.4 6.06 ± 0.2 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| HF (nu) | 37 ± 7 34 ± 3 | 23 ± 3 32 ± 3 † | 25 ± 3 28 ± 3 |
| Below 25th percentile | |||
| Above 25th percentile | |||
| LF:HF | 1.6 ± 0.1 1.6 ± 0.1 | 1.9 ± 0.2 1.6 ± 0.1 † | 1.8 ± 0.2 0.7 ± 0.1 |
| Below 25th percentile | |||
| Above 25th percentile |
3.4. Systemic Responses to Angiotensin II Challenge
4. Discussion
4.1. Vitamin D and Cardiovascular Risk
4.2. Vitamin D and Cardiac Autonomic Activity
4.3. Challenges and Limitations
4.4. Implications
Funding Sources
Conflict of Interest
References
- Holick, M.F. Vitamin D. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Dobnig, H.; Pilz, S.; Scharnagl, H.; Renner, W.; Seelhorst, U.; Wellnitz, B.; Kinkeldi, J.; Boehm, B.O.; Weihrauch, G.; Maerz, W. Independent association of low serum 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D levels with all-cause and cardiovascular mortality. Arch. Intern. Med. 2008, 168, 1340–1349. [Google Scholar] [CrossRef]
- Deo, R.; Katz, R.; Shlipk, M.G.; Sotoodehnia, N.; Psaty, B.M.; Sarnak, M.J.; Fried, L.F.; Chonchol, M.; de Boer, I.H.; Enquobahrie, D.; et al. Vitamin D, paraythyroid hormone and sudden cardiac death: Results from the Cardiovascular Health Study. Hypertension 2011, 58, 1021–1028. [Google Scholar] [CrossRef]
- De Novellis, V.; Loffreda, A.; Vitagliano, S. Effects of dietary vitamin D deficiency on the cardiovascular system. Res. Commun. Chem. Pathol. Pharmacol. 1994, 83, 125–144. [Google Scholar]
- Chan, C.T.; Levin, N.W.; Chertow, G.M.; Larive, B.; Schulman, G.; Kotanko, P.; The Frequent Hemodialysis Network Daily Trial Group. Determinants of cardiac autonomic dysfunction in ESRD. J. Am. Soc. Nephrol. 2010, 5, 1821–1827. [Google Scholar] [CrossRef]
- Kneip, C.F.; Mallet, R.T.; Williams, A.G.; Hamdan, M.H.; Smith, M.L. Vagal modulation of heart rate variability during atrial fibrillation in pigs. Exp. Bil. Med. (Maywood) 2010, 235, 1007–1014. [Google Scholar]
- Tsuji, H.; Venditti, F.J.; Manders, E.S.; Evans, J.C.; Feldman, C.L.; Levy, D. Impact of reduced heart rate variability on risk for cardiac events: The framingham heart study. Circulation 1996, 94, 2850–2855. [Google Scholar] [CrossRef]
- La Rovere, M.T.; Pinna, G.D.; Maestri, R.; Mortara, A.; Capomolla, S.; Febo, O.; Ferrari, R.; Franchini, M.; Gnemmi, M.; Opasich, C.; et al. Short-term heart rate variability strongly predicts sudden cardiac death in chronic heart failure patients. Circulation 2003, 107, 565. [Google Scholar] [CrossRef]
- Ranpuria, R.; Hall, M.; Chan, C.; Unruh, M. Heart rate variability (HRV) in kidney failure: Measurement and consequences of HRV. Neph. Dial. Transplant. 2007, 23, 444–449. [Google Scholar] [CrossRef]
- Goldberger, J.J.; Cain, M.E.; Hohnloser, S.H.; Kadish, A.H.; Knight, B.P.; Lauer, M.S.; Maron, B.J.; Page, R.L.; Passman, R.S.; Siscovick, D.; et al. American Heart Association/American College of Cardiology Foundation/Heart Rhythm Society Scientific Statement on noninvasive risk stratification techniques for identifying patients at risk for sudden cardiac death. Circulation 2008, 118, 1497–1518. [Google Scholar] [CrossRef]
- Lauer, M.S. Autonomic function and prognosis. Cleve Clin. J. Med. 2009, 76, S18–S22. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Forman, J.P.; Williams, J.S.; Fisher, N.D. Plasma 25-hydroxyvitamin D and regulation of the renin-angiotensin system. Hypertension 2010, 55, 1283–1288. [Google Scholar] [CrossRef]
- Vaidya, A.; Forman, J.P.; Williams, J.S. Vitamin D and the vascular sensitivity to angiotensin II in obese Caucasians with hypertension. J. Hum. Hypertens. 2011, 25, 672–678. [Google Scholar] [CrossRef]
- Shoback, D.M.; Williams, G.H.; Swartz, S.L.; Davies, R.O.; Hollenberg, N.K. Time course and effect of sodium intake on vascular and hormonal responses to enalapril (MK 421) in normal subjects. J. Cardiovasc. Pharmacol. 1983, 5, 1010–1018. [Google Scholar] [CrossRef]
- Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. Circulation 1996, 93, 1043–1065.
- Zhong, Y.; Jan, K.M.; Ju, K.H.; Chon, K.H. Quantifying cardiac sympathetic and parasympathetic nervous activities using principal dynamic modes analysis of heart rate variability. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H1475–H1483. [Google Scholar] [CrossRef]
- Taylor, J.A.; Carr, D.L.; Myers, C.W.; Eckberg, D.L. Mechanisms underlying very-low frequency RR-interval oscillations in humans. Circulation 1998, 98, 547–555. [Google Scholar] [CrossRef]
- Tripathi, K.K. Very low frequency oscillations in the power spectra of heart rate variability during dry supine immersion and exposure to non-hypoxic hypobaria. Physiol. Meas. 2011, 32, 717–729. [Google Scholar] [CrossRef]
- Grasso, R.; Schena, F.; Gulli, G.; Cevese, A. Does low-frequency variability of heart period reflect a specific parasympathetic mechanism? J. Auton. Nerv. Syst. 1997, 63, 30–38. [Google Scholar] [CrossRef]
- Mann, M.C.; Exner, D.V.; Hemmelgarn, B.R.; Sola, D.Y.; Turin, T.C.; Ahmed, S.B. Impact of gender on the cardiac autonomic response to angiotensin II in healthy humans. J. Appl. Physiol. 2012, 112, 1001–1007. [Google Scholar] [CrossRef]
- Dreschler, C.; Pilz, S.; Obermayer-Pietsch, B.; Verduijn, M.; Tomaschitz, A.; Krane, V.; Espe, K.; Dekker, F.; Brandenburg, V.; Marz, W.; et al. Vitamin D deficiency is associated with sudden cardiac death, combined cardiovascular events, and mortality in hemodialysis patients. Eur. Heart J. 2010, 31, 2253–2261. [Google Scholar] [CrossRef]
- Pilz, S.; Iodice, S.; Zittermann, A.; Grant, W.B.; Gandini, S. Vitamin D status and mortality risk in CKD: A meta-analysis of prospective studies. Am. J. Kidney Dis. 2011, 58, 374–382. [Google Scholar]
- Wang, T.; Pencina, M.; Booth, S.; Jacques, P.; Ingelsson, E.; Lanier, K.; Benjamin, E.; D’Agostino, R.; Wolf, M.; Vasan, R. Vitamin D deficiency and risk of cardiovascular disease. Circulation 2008, 117, 503–511. [Google Scholar] [CrossRef]
- Scorza, F.A.; Albuquerque, M.; Arida, R.M.; Tera, V.C.; Machado, H.R.; Cavalheiro, E.A. Benefits of sunlight: Vitamin D deficiency might increase the risk of sudden unexpected death in epilepsy. Med. Hypotheses 2010, 74, 158–161. [Google Scholar] [CrossRef]
- Sternberg, Z. Autonomic dysfunction: A unifying multiple sclerosis theory, linking chronic cerebrospinal venous insufficiency, vitamin D3 and Epstein-Barr virus. Autoimmun. Rev. 2012, 12, 250–259. [Google Scholar] [CrossRef]
- Tishkoff, D.X.; Nibbelink, K.A.; Holmberg, K.H.; Dandu, L.; Simpson, R.U. Functional vitamin D receptor (VITAMIN DR) in the t-tubules of cardiac myocytes: VITAMIN DR knockout cardiomyocyte contractility. Endocrinology 2008, 149, 558–564. [Google Scholar]
- Brewster, U.C.; Setaro, J.F.; Perazella, M.A. The renin-angiotensin-aldosterone system: Cardiorenal effects and implications for renal and cardiovascular disease states. Am. J. Med Sci. 2003, 326, 15–24. [Google Scholar] [CrossRef]
- Perazella, M.A.; Setaro, J.F. Renin-angiotensin-aldosterone system: Fundamental aspects and clinical implications in renal and cardiovascular disorders. J. Nucl. Cardiol. 2003, 10, 184–196. [Google Scholar] [CrossRef]
- Wolf, M.; Shah, A.; Gutierrez, O.; Ankers, E.; Monroy, M.; Tamez, H.; Steele, D.; Chang, Y.; Camargo, C.A., Jr.; Tonelli, M.; et al. Vitamin D levels and early mortality among incident hemodialysis patients. Kidney Int. 2007, 72, 1004–1013. [Google Scholar] [CrossRef]
- Kendrick, J.; Cheung, A.K.; Kaufman, J.S.; Green, T.; Roberta, W.L.; Smits, G.; Chonchol, M.; HOST Study Inventigators. Associations of plasma 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D concentrations with death and progression to maintenance dialysis in patients with advanced kidney disease. Am. J. Kidney Dis. 2012, 60, 567–575. [Google Scholar]
- Palmer, S.C.; Hayen, A.; Macaskill, P.; Pellegrini, F.; Craig, J.C.; Elder, G.J.; Strippoli, G.F. Serum levels of phosphorus, parathyroid hormone, and calcium and risks of death and cardiovascular disease in individuals with chronic kidney disease: A systematic review and meta-analysis. JAMA 2011, 305, 1119–1127. [Google Scholar] [CrossRef]
- McNeely, J.D.; Windham, B.G.; Anderson, D.E. Dietary sodium effects on heart rate variability in salt-sensitivity of blood pressure. Psychophysiology 2007, 45, 405–411. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mann, M.C.; Exner, D.V.; Hemmelgarn, B.R.; Sola, D.Y.; Turin, T.C.; Ellis, L.; Ahmed, S.B. Vitamin D Levels Are Associated with Cardiac Autonomic Activity in Healthy Humans. Nutrients 2013, 5, 2114-2127. https://doi.org/10.3390/nu5062114
Mann MC, Exner DV, Hemmelgarn BR, Sola DY, Turin TC, Ellis L, Ahmed SB. Vitamin D Levels Are Associated with Cardiac Autonomic Activity in Healthy Humans. Nutrients. 2013; 5(6):2114-2127. https://doi.org/10.3390/nu5062114
Chicago/Turabian StyleMann, Michelle C., Derek V. Exner, Brenda R. Hemmelgarn, Darlene Y. Sola, Tanvir C. Turin, Linda Ellis, and Sofia B. Ahmed. 2013. "Vitamin D Levels Are Associated with Cardiac Autonomic Activity in Healthy Humans" Nutrients 5, no. 6: 2114-2127. https://doi.org/10.3390/nu5062114
APA StyleMann, M. C., Exner, D. V., Hemmelgarn, B. R., Sola, D. Y., Turin, T. C., Ellis, L., & Ahmed, S. B. (2013). Vitamin D Levels Are Associated with Cardiac Autonomic Activity in Healthy Humans. Nutrients, 5(6), 2114-2127. https://doi.org/10.3390/nu5062114





