Small Ruminant Lentiviruses (SRLVs) Break the Species Barrier to Acquire New Host Range
Abstract
:1. Introduction
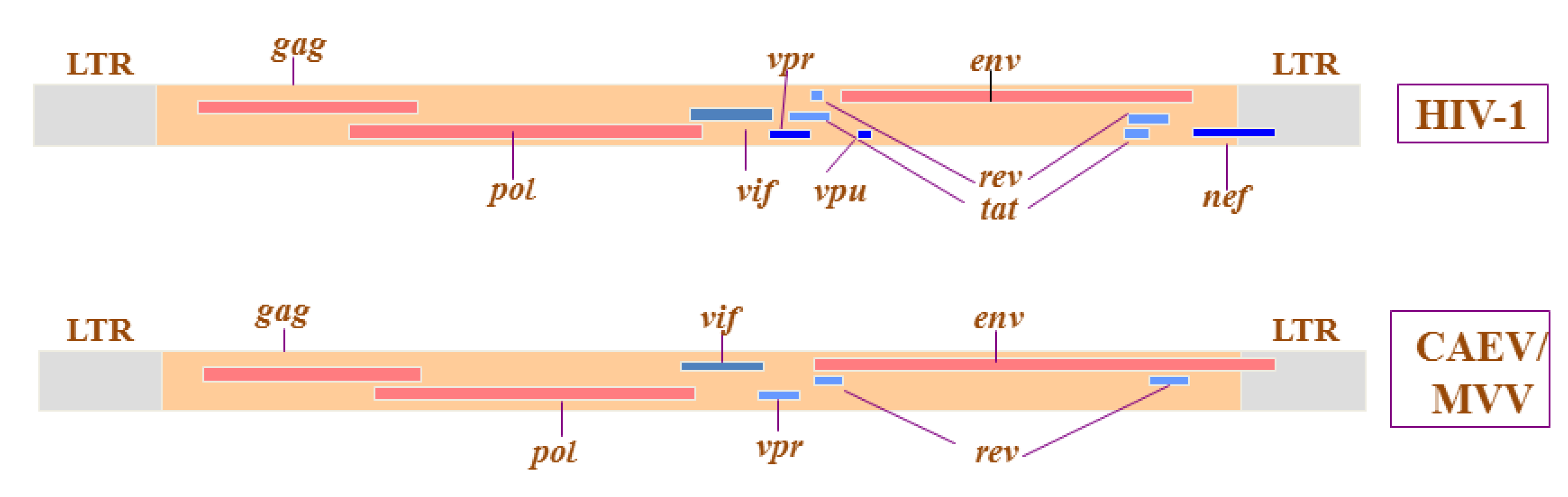
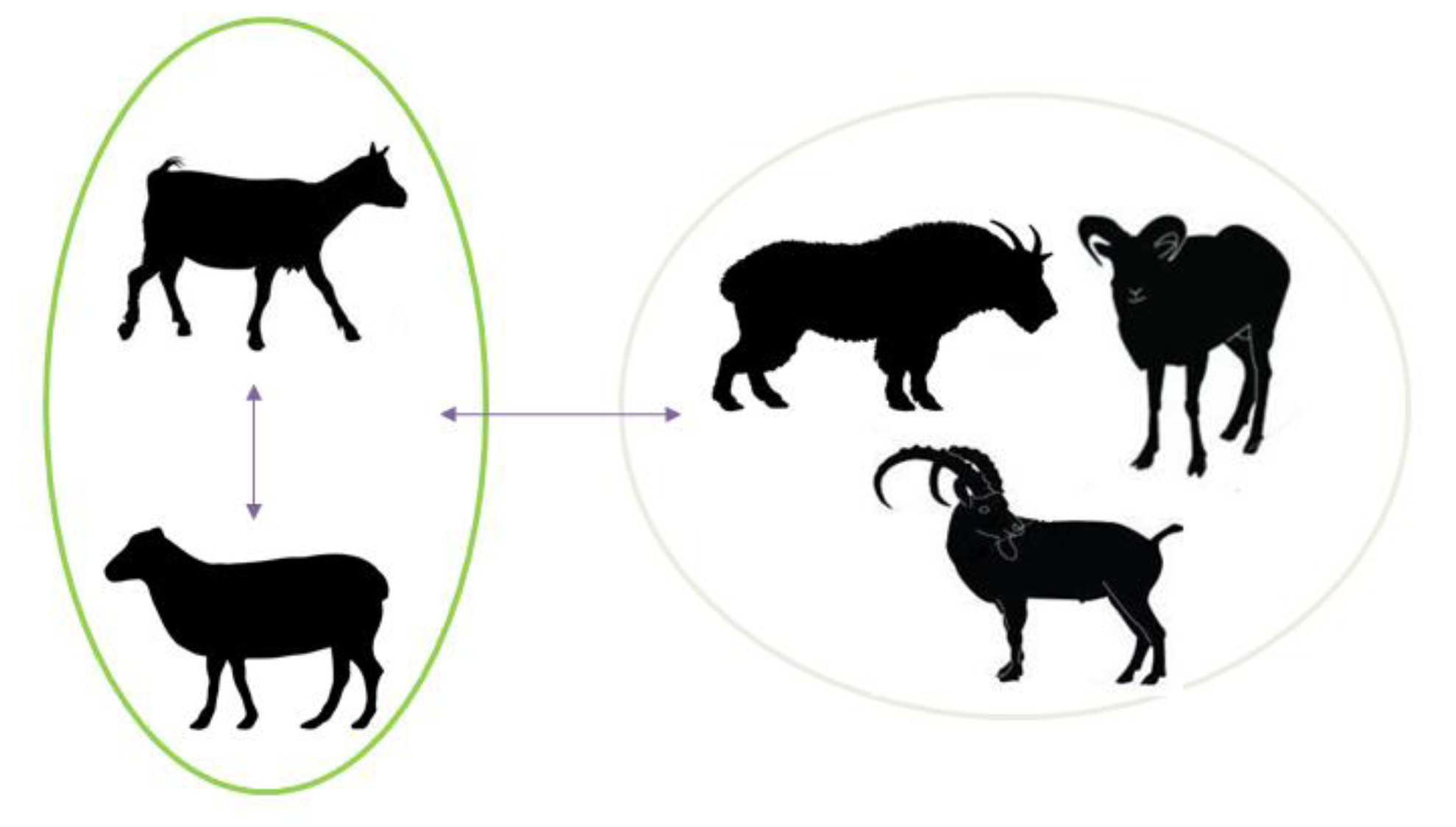

2. Implications of SRLV Genetic Diversity and Cross-Species Transmission in Control and Eradication Strategies
3. Aspects of SRLVs Emergence in Wildlife Species
4. Conclusions
Acknowledgements
Conflict of Interest
References and Notes
- Pecon-Slattery, J.; Troyer, J.L.; Johnson, W.E.; O’Brien, S.J. Evolution of feline immunodeficiency virus in Felidae: Implications for human health and wildlife ecology. Vet. Immunol. Immunopathol. 2008, 123, 32–44. [Google Scholar] [CrossRef]
- Leroux, C.; Cruz, J.C.M.; Mornex, J.F. SRLVs: A genetic continuum of lentiviral species in sheep and goats with cumulative evidence of cross species transmission. Curr. HIV Res. 2010, 8, 94–100. [Google Scholar] [CrossRef]
- Thorma, H. Maedi-visna virus and its relationship to human immunodeficiency virus. AIDS Rev. 2005, 7, 233–245. [Google Scholar]
- Smyth, R.P.; Davenport, M.P.; Mak, J. The origin of genetic diversity in HIV-1. Virus Res. 2012, 169, 415–429. [Google Scholar] [CrossRef]
- Hemelaar, J. Implications of HIV diversity for the HIV-1 pandemic. J. Infect. 2013, 66, 391–400. [Google Scholar] [CrossRef]
- Leroux, C.; Chastang, J.; Greenland, T.; Mornex, J.F. Genomic heterogeneity of small ruminant lentiviruses: Existence of heterogeneous populations in sheep and of the same lentiviral genotypes in sheep and goats. Arch. Virol. 1997, 142, 1125–1137. [Google Scholar] [CrossRef]
- Ravazzolo, A.P.; Nenci, C.; Vogt, H.R.; Waldvogel, A.; Obexer-Ruff, G.; Peterhans, E.; Bertoni, G. Viral load, organ distribution, histopathological lesions, and cytokine mRNA expression in goats infected with a molecular clone of the caprine arthritis encephalitis virus. Virology 2006, 350, 116–127. [Google Scholar] [CrossRef]
- Shah, C.; Huder, J.B.; Böni, J.; Schönmann, M.; Mühlherr, J.; Lutz, H.; Schüpbach, J. Direct evidence for natural transmission of small-ruminant lentiviruses of subtype A4 from goats to sheep and vice versa. J. Virol. 2004, 78, 7518–7522. [Google Scholar] [CrossRef]
- Pisoni, G.; Bertoni, G.; Puricelli, M.; Maccalli, M.; Moroni, P. Demonstration of coinfection with and recombination by caprine arthritis-encephalitis virus and maedi-visna virus in naturally infected goats. J. Virol. 2007, 81, 4948–4955. [Google Scholar] [CrossRef]
- Reina, R.; Bertolotti, L.; dei Giudici, S.; Puggioni, G.; Ponti, N.; Profiti, M.; Patta, C.; Rosati, S. Small ruminant lentivirus genotype E is widespread in Sarda goat. Vet. Microbiol. 2010, 144, 24–31. [Google Scholar] [CrossRef]
- Patton, K.M.; Bildfell, R.J.; Anderson, M.L.; Cebra, C.K.; Valentine, B.A. Fatal Caprine arthritis encephalitis virus-like infection in 4 Rocky Mountain goats (Oreamnos americanus). J. Vet. Diagn. Invest. 2012, 24, 392–396. [Google Scholar] [CrossRef]
- Erhouma, E.; Guiguen, F.; Chebloune, Y.; Gauthier, D.; Lakhal, L.M.; Greenland, T.; Mornex, J.F.; Leroux, C.; Alogninouwa, T. Small ruminant lentivirus proviral sequences from wild ibexes in contact with domestic goats. J. Gen. Virol. 2008, 89, 1478–1484. [Google Scholar] [CrossRef]
- Guiguen, F.; Mselli-Lakhal, L.; Durand, J.; Du, J.; Favier, C.; Fornazero, C.; Grezel, D.; Balleydier, S.; Hausmann, E.; Chebloune, Y. Experimental infection of Mouflon-domestic sheep hybrids with caprine arthritis-encephalitis virus. Am. J. Vet. Res. 2000, 61, 456–461. [Google Scholar] [CrossRef]
- Morin, T.; Guiguen, F.; Bouzar, B.A.; Villet, S.; Greenland, T.; Grezel, D.; Gounel, F.; Gallay, K.; Garnier, C.; Durand, J.; et al. Clearance of a productive lentivirus infection in calves experimentally inoculated with caprine arthritis-encephalitis virus. J. Virol. 2003, 77, 6430–6437. [Google Scholar] [CrossRef]
- Valas, S.; Benoit, C.; Guionaud, C.; Perrin, G.; Mamoun, R.Z. North American and French caprine arthritis-encephalitis viruses emerge from ovine maedi-visna viruses. Virology 1997, 237, 307–318. [Google Scholar] [CrossRef]
- Haase, A.T. Pathogenesis of lentivirus infections. Nature. 1986, 322, 130–136. [Google Scholar] [CrossRef]
- Angelopoulou, K.; Karanikolaou, K.; Papanastasopoulou, M.; Koumpati-Artopiou, M.; Vlemmas, I.; Papadopoulos, O.; Koptopoulos, G. First partial characterisation of small ruminant lentiviruses from Greece. Vet. Microbiol. 2005, 109, 1–9. [Google Scholar] [CrossRef]
- Ravazzolo, A.P.; Reischak, D.; Peterhans, E.; Zanoni, R. Phylogenetic analysis of small ruminant lentiviruses from Southern Brazil. Virus Res. 2001, 79, 117–123. [Google Scholar] [CrossRef]
- Pisoni, G.; Bertoni, G.; Boettcher, P.; Ponti, W.; Moroni, P. Phylogenetic analysis of the gag region encoding the matrix protein of small ruminant lentiviruses: Comparative analysis and molecular epidemiological applications. Virus Res. 2006, 116, 159–167. [Google Scholar] [CrossRef]
- Saltarelli, M.; Querat, G.; Konings, D.A.; Vigne, R.; Clements, J.E. Nucleotide sequence and transcriptional analysis of molecular clones of CAEV which generate infectious virus. Virology 1990, 179, 347–364. [Google Scholar] [CrossRef]
- Sonigo, P.; Alizon, M.; Staskus, K.; Klatzmann, D.; Cole, S.; Danos, O.; Retzel, E.; Tiollais, P.; Haase, A.; Wain-Hobson, S. Nucleotide sequence of the visna lentivirus, relationship to the AIDS virus. Cell 1985, 42, 369–382. [Google Scholar] [CrossRef]
- Shah, C.; Böni, J.; Huder, J.B.; Vogt, H.R.; Mühlherr, J.; Zanoni, R.; Miserez, R.; Lutz, H.; Schüpbach, J. Phylogenetic analysis and reclassification of caprine and ovine lentiviruses based on 104 new isolates: Evidence for regular sheep-to-goat transmission and worldwide propagation through livestock trade. Virology 2004, 319, 12–26. [Google Scholar] [CrossRef]
- Querat, G.; Audoly, G.; Sonigo, P.; Vigne, R. Nucleotide sequence analysis of SA-OMVV, a visna-related ovine lentivirus: Phylogenetic history of lentiviruses. Virology 1990, 175, 434–447. [Google Scholar] [CrossRef]
- Sargan, D.R.; Bennet, I.D.; Cousens, C.; Roy, D.J.; Blacklaws, B.A.; Dalziel, R.G.; Watt, N.J.; McConnell, I. Nucleotide sequence of EV1, a British isolate of maedi-visna virus. J. Gen. Virol. 1991, 72, 1893–1903. [Google Scholar] [CrossRef]
- Grego, E.; Bertolotti, L.; Quasso, A.; Profiti, M.; Lacerenza, D.; Muz, D.; Rosati, S. Genetic characterization of small ruminant lentivirus in Italian mixed flocks: Evidence for a novel genotype circulating in a local goat population. J. Gen. Virol. 2007, 88, 3423–3427. [Google Scholar] [CrossRef]
- Pisoni, G.; Bertoni, G.; Manarolla, G.; Vogt, H.R.; Scaccabarozzi, L.; Locatelli, C.; Moroni, P. Genetic analysis of small ruminant lentiviruses following lactogenic transmission. Virology 2010, 407, 91–99. [Google Scholar] [CrossRef]
- Cork, L.C.; Hadlow, W.J.; Crawford, T.B.; Gorham, J.R.; Piper, R.C. Infectious leuko-encephalomyelitis of young goats. J. Infect. Dis. 1974, 129, 134–141. [Google Scholar] [CrossRef]
- Bertolotti, L.; Mazzei, M.; Puggioni, G.; Carrozza, M.L.; dei Giudici, S.; Muz, D.; Juganaru, M.; Patta, C.; Tolari, F.; Rosati, S. Characterization of new small ruminant lentivirus subtype B3 suggests animal trade within the Mediterranean Basin. J. Gen. Virol. 2011, 92, 1923–1929. [Google Scholar] [CrossRef]
- Gjerset, B.; Storset, A.K.; Rimstad, E. Genetic diversity of small-ruminant lentiviruses: Characterization of Norwegian isolates of Caprine arthritis encephalitis virus. J. Gen. Virol. 2006, 87, 573–580. [Google Scholar] [CrossRef]
- Glaria, I.; Reina, R.; Crespo, H.; de Andrés, X.; Ramírez, H.; Biescas, E.; Pérez, M.M.; Badiola, J.; Luján, L.; Amorena, B.; de Andrés, D. Phylogenetic analysis of SRLV sequences from an arthritic sheep outbreak demonstrates the introduction of CAEV-like viruses among Spanish sheep. Vet. Microbiol. 2009, 138, 156–162. [Google Scholar] [CrossRef]
- Pisoni, G.; Quasso, A.; Moroni, P. Phylogenetic analysis of small-ruminant lentivirus subtype B1 in mixed flocks: Evidence for natural transmission from goats to sheep. Virology 2005, 339, 147–152. [Google Scholar] [CrossRef]
- Grego, E.; Lacerenza, D.; Arias, R.R.; Profiti, M.; Rosati, S. Serological characterization of the new genotype E of small ruminant lentivirus in Roccaverano goat flocks. Vet. Res. Commun. 2009, 33, 137–140. [Google Scholar] [CrossRef]
- Villet, S.; Faure, C.; Bouzar, B.A.; Morin, T.; Verdier, G.; Chebloune, Y.; Legras, C. Lack of trans-activation function for Maedi Visna virus and Caprine arthritis encephalitis virus Tat proteins. Virology 2003, 307, 317–327. [Google Scholar] [CrossRef]
- Villet, S.; Bouzar, B.A.; Morin, T.; Verdier, G.; Legras, C.; Chebloune, Y. Maedi visna virus and caprine arthritis encephalitis virus genomes encode a Vpr-like but no Tat protein. J. Virol. 2003, 77, 9632–9638. [Google Scholar] [CrossRef]
- Clements, J.E.; Wall, R.J.; Narayan, O.; Hauer, D.; Schoborg, R.; Sheffer, D.; Powell, A.; Carruth, L.M.; Zink, M.C.; Rexroad, C.E. Development of transgenic sheep that express the visna virus envelope gene. Virology 1994, 200, 370–380. [Google Scholar] [CrossRef]
- Butler, I.F.; Pandrea, I.; Marx, P.A.; Apetrei, C. HIV genetic diversity: Biological and public health consequences. Curr. HIV Res. 2007, 5, 23–45. [Google Scholar] [CrossRef]
- Pisoni, G.; Moroni, P.; Turin, L.; Bertoni, G. Compartmentalization of small ruminant lentivirus between blood and colostrum in infected goats. Virology 2007, 369, 119–130. [Google Scholar] [CrossRef]
- Kuzmak, J.; Rola, M.; Gallay, K.; Chebloune, Y. Molecular characterization of lentiviruses from goats from Poland based on gag gene sequence analysis. Comp. Immunol. Microbiol. Infect. Dis. 2007, 30, 211–223. [Google Scholar] [CrossRef]
- Reina, R.; Mora, M.I.; Glaria, I.; García, I.; Solano, C.; Luján, L.; Badiola, J.J.; Contreras, A.; Berriatua, E.; Juste, R.; et al. Molecular characterization and phylogenetic study of Maedi Visna and Caprine Arthritis Encephalitis viral sequences in sheep and goats from Spain. Virus Res. 2006, 121, 189–198. [Google Scholar] [CrossRef]
- Gjerset, B.; Jonassen, C.M.; Rimstad, E. Natural transmission and comparative analysis of small ruminant lentiviruses in the Norwegian sheep and goat populations. Virus Res. 2007, 125, 153–161. [Google Scholar] [CrossRef]
- Benavides, J.; Fuertes, M.; García-Pariente, C.; Ferreras, M.C.; García Marín, J.F.; Pérez, V. Natural cases of visna in sheep with myelitis as the sole lesion in the central nervous system. J. Comp. Pathol. 2006, 134, 219–230. [Google Scholar] [CrossRef]
- Benavides, J.; Gómez, N.; Gelmetti, D.; Ferreras, M.C.; García-Pariente, C.; Fuertes, M.; García-Marín, J.F.; Pérez, V. Diagnosis of the nervous form of Maedi-Visna infection with a high frequency in sheep in Castilla y Leon, Spain. Vet. Rec. 2006, 158, 230–235. [Google Scholar] [CrossRef]
- Benavides, J.; García-Pariente, C.; Fuertes, M.; Ferreras, M.C.; García-Marín, J.F.; Juste, R.A.; Pérez, V. Maedi-visna: The meningoencephalitis in naturally occurring cases. J. Comp. Pathol. 2009, 140, 1–11. [Google Scholar] [CrossRef]
- Herrmann-Hoesing, L.M. Diagnostic assays used to control small ruminant lentiviruses. J. Vet. Diagn. Invest. 2010, 22, 843–855. [Google Scholar] [CrossRef]
- Rimstad, E.; East, N.E.; Torten, M.; Higgins, J.; DeRock, E.; Pedersen, N.C. Delayed seroconversion following naturally acquired caprine arthritis-encephalitis virus infection in goats. Am. J. Vet. Res. 1993, 54, 1858–1862. [Google Scholar]
- Blacklaws, B.A.; Berriatua, E.; Torsteinsdottir, S.; Watt, N.J.; de Andres, D.; Klein, D.; Harkiss, G.D. Transmission of small ruminant lentiviruses. Vet. Microbiol. 2004, 101, 199–208. [Google Scholar] [CrossRef]
- Chebloune, Y.; Karr, B.; Sheffer, D.; Leung, K.; Narayan, O. Variations in lentiviral gene expression in monocyte-derived macrophages from naturally infected sheep. J. Gen. Virol. 1996, 77, 2037–2051. [Google Scholar] [CrossRef]
- Karr, B.M.; Chebloune, Y.; Leung, K.; Narayan, O. Genetic characterization of two phenotypically distinct North American ovine lentiviruses and their possible origin from caprine arthritis-encephalitis virus. Virology 1996, 225, 1–10. [Google Scholar] [CrossRef]
- Singh, D.K.; Chebloune, Y.; Mselli-Lakhal, L.; Karr, B.M.; Narayan, O. Ovine lentivirus-infected macrophages mediate productive infection in cell types that are not susceptible to infection with cell-free virus. J. Gen. Virol. 1999, 80, 1437–1444. [Google Scholar]
- De Andres, D.; Klein, D.; Watt, N.J.; Berriatua, E.; Torsteinsdottir, S.; Blacklaws, B.A.; Harkiss, G.D. Diagnostic tests for small ruminant lentiviruses. Vet. Microbiol. 2005, 107, 49–62. [Google Scholar] [CrossRef]
- Reina, R.; Grego, E.; Profiti, M.; Glaria, I.; Robino, P.; Quasso, A.; Amorena, B.; Rosati, S. Development of specific diagnostic test for small ruminant lentivirus genotype E. Vet. Microbiol. 2009, 138, 251–257. [Google Scholar] [CrossRef]
- Travassos, C.; Benoit, C.; Valas, S.; da Silva, A.; Perrin, G. Detection of caprine arthritis encephalitis virus in sperm of experimentally infected bucks. Vet. Res. 1998, 29, 579–584. [Google Scholar]
- Ali Al Ahmad, M.Z.; Fieni, F.; Pellerin, J.L.; Guiguen, F.; Cherel, Y.; Chatagnon, G.; Bouzar, A.B.; Chebloune, Y. Detection of viral genomes of caprine arthritis-encephalitis virus (CAEV) in semen and in genital tract tissues of male goat. Theriogenology 2008, 69, 473–480. [Google Scholar] [CrossRef]
- Peterson, K.; Brinkhof, J.; Houwers, D.J.; Colenbrander, B.; Gadella, B.M. Presence of pro-lentiviral DNA in male sexual organs and ejaculates of small ruminants. Theriogenology 2008, 69, 433–442. [Google Scholar] [CrossRef]
- Ramirez, H.; Román, B.S.; Glaria, I.; Reina, R.; Hernández, M.M.; de Andrés, X.; Crespo, H.; Hichou, B.; Cianca, S.; Goñi, C.; et al. Antibody-based diagnosis of small ruminant lentivirus infection in seminal fluid. Theriogenology 2009, 72, 1085–1096. [Google Scholar] [CrossRef]
- Fieni, F.; Pellerin, J.L.; Roux, C.; Poulin, N.; Baril, G.; Fatet, A.; Valas, S.; Chatagnon, G.; Mermillod, P.; Guignot, F. Can caprine arthritis encephalitis virus (CAEV) be transmitted by in vitro fertilization with experimentally infected sperm? Theriogenology 2011, 77, 644–651. [Google Scholar]
- Reina, R.; Glaria, I.; Cianca, S.; Crespo, H.; de Andrés, X.; Goñi, C.; Lasarte, J.M.; Luján, L.; Amorena, B.; de Andrés, D.F. Use of small ruminant lentivirus-infected rams for artificial insemination. Vet. J. 2011, 189, 106–117. [Google Scholar] [CrossRef]
- Reina, R.; Berriatua, E.; Luján, L.; Juste, R.; Sánchez, A.; de Andrés, D.; Amorena, B. Prevention strategies against small ruminant lentiviruses: An update. Vet. J. 2009, 182, 31–37. [Google Scholar] [CrossRef]
- Beyer, J.C.; Chebloune, Y.; Mselli-Lakhal, L.; Hötzel, I.; Kumpula-McWhirter, N.; Cheevers, W.P. Immunization with plasmid DNA expressing the caprine arthritis-encephalitis virus envelope gene: Quantitative and qualitative aspects of antibody response to viral surface glycoprotein. Vaccine 2001, 19, 1643–1651. [Google Scholar] [CrossRef]
- Cheevers, W.P.; Beyer, J.C.; Hotzel, I. Plasmid DNA encoding caprine interferon gamma inhibits antibody response to caprine arthritis-encephalitis virus (CAEV) surface protein encoded by a co-administered plasmid expressing CAEV env and tat genes. Vaccine 2001, 19, 3209–3215. [Google Scholar] [CrossRef]
- Cheevers, W.P.; Snekvik, K.R.; Trujillo, J.D.; Kumpula-McWhirter, N.M.; Pretty On Top, K.J.; Knowles, D.P. Prime-boost vaccination with plasmid DNA encoding caprine-arthritis encephalitis lentivirus env and viral SU suppresses challenge virus and development of arthritis. Virology 2003, 306, 116–125. [Google Scholar] [CrossRef]
- Fluri, A.; Nenci, C.; Zahno, M.L.; Vogt, H.R.; Charan, S.; Busato, A.; Pancino, G.; Peterhans, E.; Obexer-Ruff, G.; Bertoni, G. The MHC-haplotype influences primary, but not memory, immune responses to an immunodominant peptide containing T- and B-cell epitopes of the caprine arthritis encephalitis virus Gag protein. Vaccine 2006, 24, 597–606. [Google Scholar] [CrossRef]
- Leitner, G.; Krifucks, O.; Weisblit, L.; Lavi, Y.; Bernstein, S.; Merin, U. The effect of caprine arthritis encephalitis virus infection on production in goats. Vet. J. 2009, 183, 328–331. [Google Scholar]
- Peterhans, E.; Greenland, T.; Badiola, J.; Harkiss, G.; Bertoni, G.; Amorena, B.; Eliaszewicz, M.; Juste, R.A.; Krassnig, R.; Lafont, J.P.; et al. Routes of transmission and consequences of small ruminant lentiviruses (SRLVs) infection and eradication schemes. Vet. Res. 2004, 35, 257–274. [Google Scholar] [CrossRef]
- Valas, S.; le Ven, A.; Croise, B.; Maquigneau, M.; Perrin, C. Interference of vaccination against bluetongue virus serotypes 1 and 8 with serological diagnosis of small-ruminant lentivirus infection. Clin. Vaccine Immunol. 2011, 18, 513–517. [Google Scholar] [CrossRef]
- Herrmann-Hoesing, L.M.; Broughton-Neiswanger, L.E.; Gouine, K.C.; White, S.N.; Mousel, M.R.; Lewis, G.S.; Marshall, K.L.; Knowles, D.P. Evaluation of a caprine arthritis-encephalitis virus/maedi-visna virus indirect enzyme-linked immunosorbent assay in the serological diagnosis of ovine progressive pneumonia virus in U.S. sheep. Clin. Vaccine Immunol. 2010, 17, 307–310. [Google Scholar] [CrossRef]
- Pecon-Slattery, J.; McCracken, C.L.; Troyer, J.L.; VandeWoude, S.; Roelke, M.; Sondgeroth, K.; Winterbach, C.; Winterbach, H.; O’Brien, S.J. Genomic organization, sequence divergence, and recombination of feline immunodeficiency virus from lions in the wild. BMC Genomics 2008, 9, 1–13. [Google Scholar] [CrossRef]
- Hemelaar, J. The origin and diversity of the HIV-1 pandemic. Trends Mol. Med. 2012, 18, 182–192. [Google Scholar] [CrossRef]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Emerging infectious diseases of wildlife—Threats to biodiversity and human health. Science 2000, 287, 443–449. [Google Scholar] [CrossRef]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Anthropogenic environmental change and the emergence of infectious diseases in wildlife. Acta. Trop. 2001, 78, 103–116. [Google Scholar] [CrossRef]
- Fox, J.; Fidler, S. Sexual transmission of HIV-1. Antivir. Res. 2010, 85, 276–285. [Google Scholar] [CrossRef]
- Morse, S.S.; Mazet, J.A.; Woolhouse, M.; Parrish, C.R.; Carroll, D.; Karesh, W.B.; Zambrana-Torrelio, C.; Lipkin, W.I.; Daszak, P. Prediction and prevention of the next pandemic zoonosis. Lancet 2012, 380, 1956–1965. [Google Scholar] [CrossRef]
- Konishi, M.; Nagura, Y.; Fujita, N.M.; Hayashi, K.; Tsukioka, M.; Yamamoto, T.; Kameyama, K.; Sentsui, H.; Murakami, K. Combined eradication strategy for CAE in a dairy goat farm in Japan. Small Rumin. Res. 2011, 99, 65–71. [Google Scholar] [CrossRef]
- Brinkhof, J.; van Maanen, C. Evaluation of five enzyme-linked immunosorbent assays and an agar gel immunodiffusion test for detection of antibodies to small ruminant lentiviruses. Clin. Vaccine Immunol. 2007, 14, 1210–1214. [Google Scholar] [CrossRef]
- Brinkhof, J.M.; Moll, L.; van Maanen, C.; Houwers, D.J. Use of serology and polymerase chain reaction for the rapid eradication of small ruminant lentivirus infections from a sheep flock: A case report. Res. Vet. Sci. 2010, 88, 41–53. [Google Scholar] [CrossRef]
- Brinkhof, J.M.; Houwers, D.J.; Moll, L.; Dercksen, D.; van Maanen, C. Diagnostic performance of ELISA and PCR in identifying SRLV-infected sheep and goats using serum, plasma and milk samples and in early detection of infection in dairy flocks through bulk milk testing. Vet. Microbiol. 2010, 142, 193–198. [Google Scholar] [CrossRef]
- Martin, C.; Pastoret, P.P.; Brochier, B.; Humblet, M.F.; Saegerman, C. A survey of the transmission of infectious diseases/infections between wild and domestic ungulates in Europe. Vet. Res. 2011, 42, 1–16. [Google Scholar]
- Boyazoglu, J.; Morand-Fehr, P. Mediterranean dairy sheep and goat products and their quality: A critical review. Small Rumin. Res. 2001, 40, 1–11. [Google Scholar] [CrossRef]
- Klinger, I.; Rosenthal, I. Public health and the safety of milk and milk products from sheep and goats. Rev. Sci. Tech. 1997, 16, 482–488. [Google Scholar]
- Corsetti, A.; Caldini, G.; Mastrangelo, M.; Trotta, F.; Valmorri, S.; Cenci, G. Raw milk traditional Italian ewe cheeses as a source of Lactobacillus casei strains with acid-bile resistance and antigenotoxic properties. Int. J. Food Microbiol. 2008, 125, 330–335. [Google Scholar] [CrossRef]
- Tesoro-Cruz, E.; Hernandez-Gonzalez, R.; Kretschmer-Schmid, R.; Aguilar-Setien, A. Cross-reactivity between caprine arthritis-encephalitis virus and type 1 human immunodeficiency virus. Arch. Med. Res. 2003, 34, 362–366. [Google Scholar] [CrossRef]
- Tesoro-Cruz, E.; Feria-Romero, I.A.; Orozco-Suárez, S.; Hernández-González, R.; Silva-García, R.; Valladares-Salgado, A.; Bekker-Méndez, V.C.; Blanco-Favela, F.; Aguilar-Setién, A. Frequency of the serological reactivity against the caprine arthritis encephalitis lentivirus gp135 in children who consume goat milk. Arch. Med. Res. 2009, 40, 204–207. [Google Scholar] [CrossRef]
- Mselli-Lakhal, L.; Favier, C.; Leung, K.; Guiguen, F.; Grezel, D.; Miossec, P.; Mornex, J.F.; Narayan, O.; Querat, G.; Chebloune, Y. Lack of functional receptors is the only barrier that prevents caprine arthritis-encephalitis virus from infecting human cells. J. Virol. 2000, 74, 8343–8348. [Google Scholar] [CrossRef]
- Gjerset, B.; Rimstad, E.; Teige, J.; Soetaert, K.; Jonassen, C.M. Impact of natural sheep-goat transmission on detection and control of small ruminant lentivirus group C infections. Vet. Microbiol. 2009, 135, 231–238. [Google Scholar] [CrossRef]
- Mariante Ada, S.; Egito, A.A. Animal genetic resources in Brazil: Result of five centuries of natural selection. Theriogenology 2002, 57, 223–235. [Google Scholar] [CrossRef]
- Mselli-Lakhal, L.; Guiguen, F.; Greenland, T.; Mornex, J.F.; Chebloune, Y. In vitro cross-species infections using a caprine arthritis encephalitis lentivirus carrying the GFP marker gene. J. Virol. Methods 2007, 143, 11–15. [Google Scholar] [CrossRef]
- VandeWoude, S.; Troyer, J.; Poss, M. Restrictions to cross-species transmission of lentiviral infection gleaned from studies of FIV. Vet. Immunol. Immunopathol. 2010, 134, 25–32. [Google Scholar] [CrossRef]
- Falconi, C.; Lopez-Olvera, J.R.; Gortazar, C. BTV infection in wild ruminants, with emphasis on red deer: A review. Vet. Microbiol. 2011, 151, 209–219. [Google Scholar] [CrossRef]
- Gufler, H.; Moroni, P.; Casu, S.; Pisoni, G. Seroprevalence, clinical incidence, and molecular and epidemiological characterisation of small ruminant lentivirus in the indigenous Passirian goat in northern Italy. Arch. Virol. 2008, 153, 1581–1585. [Google Scholar] [CrossRef]
- Gortazar, C.; Ferroglio, E.; Höfe, U.; Frölich, K.; Vicente, J. Disease shared between wildlife and livestock: A european perspective. Eur. J. Wildl. Res. 2007, 53, 241–256. [Google Scholar] [CrossRef]
- Troyer, J.L.; Vandewoude, S.; Pecon-Slattery, J.; McIntosh, C.; Franklin, S.; Antunes, A.; Johnson, W.; O’Brien, S.J. FIV cross-species transmission: An evolutionary prospective. Vet. Immunol. Immunopathol. 2008, 123, 159–166. [Google Scholar] [CrossRef]
- Cruz, J.C.M.; Gouveia, A.M.G.; Souza, K.C.; Braz, G.F.; Teixeira, B.M.; Heinemann, M.B.; Leite, R.C.; Reis, J.K.P.; Pinheiro, R.R.; Andrioli, A. Caprine arthritis-encephalitis virus (CAEV) detection in semen of endangered goat breeds by nested polymerase chain reaction. Small Rumin. Res. 2009, 85, 149–152. [Google Scholar] [CrossRef]
- Paula, N.R.O.; Andriolib, A.; Cardosoc, J.F.S.; Pinheirob, R.R.; Sousad, F.M.L.; Souzad, K.C.; Alvesb, F.S.F.; Campelloa, C.C.; Ricartea, A.R.F.; Teixeiraa, M.F.S. Profile of the Caprine arthritis-encephalitis virus (CAEV) in blood, semen from bucks naturally and experimentally infected in the semi-arid region of Brazil. Small Rumin. Res. 2009, 85, 27–33. [Google Scholar] [CrossRef]
- Pedersen, A.B.; Jones, K.E.; Nunn, C.L.; Altizer, S. Infectious diseases and extinction risk in wild mammals. Conserv. Biol. 2007, 21, 1269–1279. [Google Scholar] [CrossRef]
- Acevedo-Whitehouse, K.; Duffus, A.L. Effects of environmental change on wildlife health. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 3429–3438. [Google Scholar] [CrossRef]
- Maillard, J.C.; Gonzalez, J.P. Biodiversity and emerging diseases. Ann. NY Acad. Sci. 2006, 1081, 1–16. [Google Scholar] [CrossRef]
- Vistnes, I.; Nelleman, C. The matter of spatial and temporal scales: A review of reindeer and caribou response to human activity. Polar Biol. 2008, 31, 399–407. [Google Scholar] [CrossRef]
- Cardinaux, L.; Zahno, M.L.; Deubelbeiss, M.; Zanoni, R.; Vogt, H.R.; Bertoni, G. Virological and phylogenetic characterization of attenuated small ruminant lentivirus isolates eluding efficient serological detection. Vet. Microbiol. 2013, 162, 572–581. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Minardi da Cruz, J.C.; Singh, D.K.; Lamara, A.; Chebloune, Y. Small Ruminant Lentiviruses (SRLVs) Break the Species Barrier to Acquire New Host Range. Viruses 2013, 5, 1867-1884. https://doi.org/10.3390/v5071867
Minardi da Cruz JC, Singh DK, Lamara A, Chebloune Y. Small Ruminant Lentiviruses (SRLVs) Break the Species Barrier to Acquire New Host Range. Viruses. 2013; 5(7):1867-1884. https://doi.org/10.3390/v5071867
Chicago/Turabian StyleMinardi da Cruz, Juliano Cezar, Dinesh Kumar Singh, Ali Lamara, and Yahia Chebloune. 2013. "Small Ruminant Lentiviruses (SRLVs) Break the Species Barrier to Acquire New Host Range" Viruses 5, no. 7: 1867-1884. https://doi.org/10.3390/v5071867
APA StyleMinardi da Cruz, J. C., Singh, D. K., Lamara, A., & Chebloune, Y. (2013). Small Ruminant Lentiviruses (SRLVs) Break the Species Barrier to Acquire New Host Range. Viruses, 5(7), 1867-1884. https://doi.org/10.3390/v5071867




