Virus-Induced Aggregates in Infected Cells
Abstract
:1. Introduction
2. Virus Factories Are the Sites of Accumulation and Assembly of DNA and RNA Viruses in Mammalian Cells
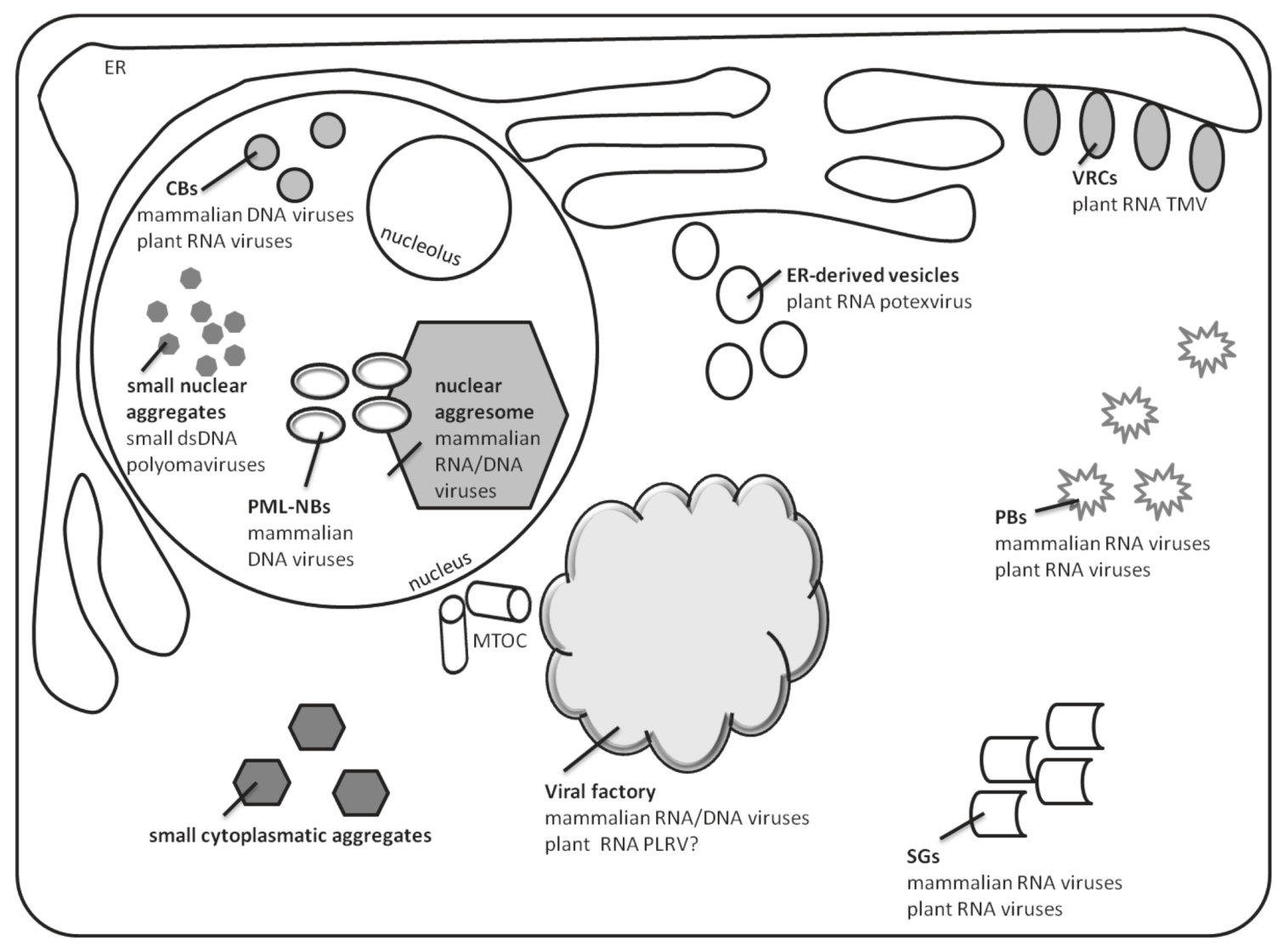
3. Sites of Accumulation and Assembly of RNA Viruses in Plant Cells
4. Viral Components Localized in Nuclear Aggregates
5. Presence of Viral Components in Cytoplasmic Small and Intermediate Aggregates
5.1. Processing Bodies
5.2. Cytoplasmic Stress Granules
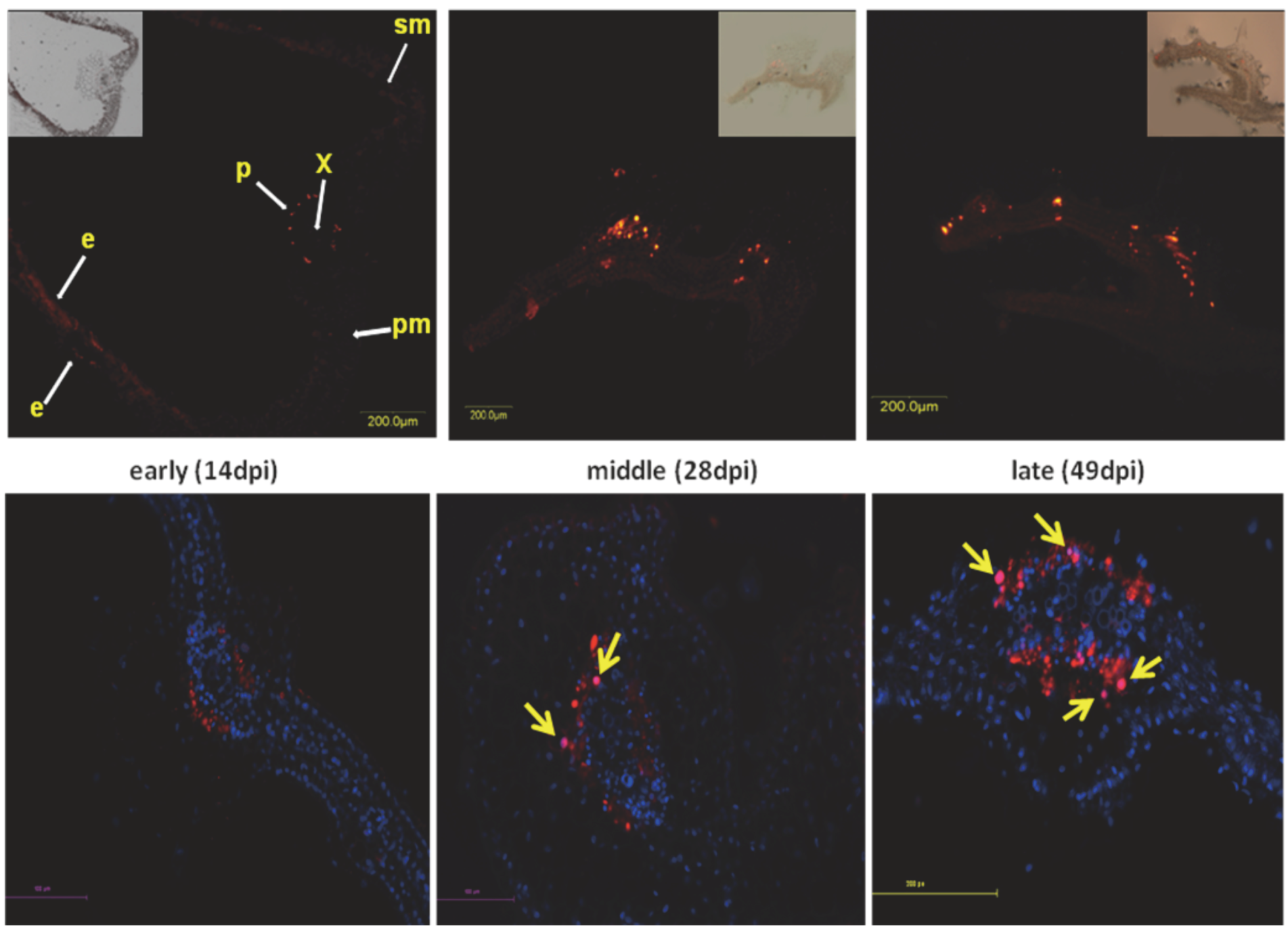
6. Aggregates Induced by Plant DNA Viruses
7. Discussion
Conflict of Interest
Acknowledgments
References and Notes
- Novoa, R.; Calderita, G; Arranz, R.; Fontana, J.; Granzow, H.; Risco, C. Virus factories: Associations of cell organelles for viral replication and morphogenesis. Biol. Cell 2005, 97, 147–172. [Google Scholar]
- Netherton, C.; Moffat, K.; Brooks, E.; Wileman, T. A guide to viral inclusions, membrane rearrangements, factories, and viroplasm produced during virus replication. Adv. Virus Res. 2007, 70, 101–182. [Google Scholar] [CrossRef]
- Netherton, C.; Wileman, T. Virus factories, double membrane vesicles and viroplasm generated in animal cells. Curr. Opin. Virol. 2011, 1, 381–387. [Google Scholar] [CrossRef]
- Wileman, T. Aggresomes and pericentriolar sites of virus assembly: Cellular defense or viral design? Annu. Rev. Microbiol. 2007, 61, 149–167. [Google Scholar] [CrossRef]
- Wileman, T. Aggresomes and autophagy generate sites for viral infection. Science 2006, 312, 875–878. [Google Scholar] [CrossRef]
- Kopito, R. Aggresomes, inclusion bodies and protein aggregation. Trends Cell Biol. 2000, 10, 524–530. [Google Scholar]
- Kaganivich, D.; Kopito, R.; Frydman, J. Misfolded proteins partition between two distinct quality control compartments. Nature 2008, 454, 1088–1095. [Google Scholar] [CrossRef]
- Dohner, K.; Nagel, C.-H.; Sodeik, B. Viral stop-and-go along microtubules: Taking a ride with dynein and kinesins. Trends Microbiol. 2005, 13, 320–327. [Google Scholar] [CrossRef]
- Vogel, F.; Hofius, D.; Sonnewald, U. Intracellular trafficking of potato leafroll virus movement protein in transgenic arabidopsis. Traffic 2007, 8, 1205–1214. [Google Scholar] [CrossRef]
- Reichel, C.; Beachy, R.N. Degradation of tobacco mosaic virus movement protein by the 26S proteasome. J. Virol. 2000, 74, 3330–3337. [Google Scholar] [CrossRef]
- Kawakami, S.; Watanabe, Y.; Beachy, R.N. Tobacco mosaic virus infection spreads cell to cell as intact replication complexes. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 6291–6296. [Google Scholar] [CrossRef]
- den Boon, J.A.; Diaz, A.; Ahlquist, P. Cytoplasmic viral replication complexes. Cell Host Micro. 2010, 8, 77–85. [Google Scholar] [CrossRef]
- Bamunusinghe, D.; Hemenway, C.L.; Nelson, R.S.; Sanderfoot, A.A.; Ye, C.M.; Silva, M.A.; Payton, M.; Verchot-Lubicz, J. Analysis of potato virus X replicase and TGBp3 subcellular locations. Virology 2009, 393, 272–285. [Google Scholar] [CrossRef]
- Tajima, H.; Iwata, Y.; Iwano, M.; Takayama, S.; Koizumi, N. Identification of an Arabidopsis transmembrane bZIP transcription factor involved in the endoplasmic reticulum stress response. Biochem. Biophys. Res. Commun. 2008, 374, 242–247. [Google Scholar] [CrossRef]
- Ye, C.; Dickman, M.B.; Whitham, S.A.; Payton, M.; Verchot, J. The unfolded protein response is triggered by a plant viral movement protein. Plant Physiol. 2011, 156, 741–755. [Google Scholar] [CrossRef]
- Verchot, J. Wrapping membranes around plant virus infection. Cur. Opin. Virol. 2011, 1, 388–395. [Google Scholar] [CrossRef]
- Maul, G.G.; Negorev, D.; Bell, P.; Ishov, A.M. Properties and assembly mechanisms of ND10, PML bodies, or PODs. J. Str. Biol. 2000, 129, 278–287. [Google Scholar] [CrossRef]
- Negorev, D.; Maul, G.G. Cellular proteins localized at and interacting within ND10/PML nuclear bodies/PODs suggest functions of a nuclear depot. Oncogene 2001, 20, 7234–7242. [Google Scholar] [CrossRef]
- Boe, S.O.; Haave, M.; Jul-Larsen, A.; Grudic, A.; Bjerkvig, R. Promyelocytic leukemia nuclear bodies are predetermined processing sites for damaged DNA. J. Cell Sci. 2006, 119, 3284–3295. [Google Scholar] [CrossRef]
- Everett, R.D. Interactions between DNA viruses, ND10 and the DNA damage response. Cell Microbiol. 2006, 8, 365–375. [Google Scholar] [CrossRef]
- Lilley, C.E.; Schwartz, R.A.; Weitzman, M.D. Using or abusing: Viruses and the cellular DNA damage response. Trends Microbiol. 2007, 15, 119–126. [Google Scholar] [CrossRef]
- Fu, L.; Gao, Y.S.; Tousson, A.; Shah, A.; Chen, T.L. Nuclear aggresomes form by fusion of PML-NB-associated aggregates. Mol. Biol. Cell 2005, 16, 4905–4917. [Google Scholar] [CrossRef]
- Iwata, A.; Christianson, J.C., Bucci; Ellerby, L.M.; Nukina, N., Forno; Kopito, R. Increased susceptibility of cytoplasmic over nuclear polyglutamine aggregates to autophagic degradation. Proc. Natl. Acad. Sci. U. S. A. 2005, 102, 13135–13140. [Google Scholar]
- Tavalai, N.; Stamminger, T. Intrinsic cellular defense mechanisms targeting human cytomegalovirus. Virus Res. 2011, 157, 128–133. [Google Scholar] [CrossRef]
- Tavalai, N.; Stamminger, T. Interplay between herpesvirus infection and host defense by PML nuclear bodies. Viruses 2009, 1, 1240–1264. [Google Scholar] [CrossRef]
- Tavalai, N.; Papior, P.; Rechter, S.; Leis, M.; Stamminger, T. Evidence for a role of the cellular ND10 protein PML in mediating intrinsic immunity against human cytomegalovirus infections. J. Virol. 2006, 80, 8006–8018. [Google Scholar] [CrossRef]
- Hutchinson, I; Whiteley, A.; Browne, H.; Elliott, G. Sequential localization of two herpes simplex virus tegument proteins to punctate nuclear dots adjacent to ICP0 domains. J. Virol. 2002, 76, 10365–10373. [Google Scholar] [CrossRef]
- Jiang, M.; Entezami, P.; Gamez, M.; Stamminger, T; Imperiale, M.J. Functional reorganization of promyelocytic leukemia nuclear bodies during BK virus infection. MBio 2011, 2, e00281–00210. [Google Scholar]
- Erickson, K.D.; Bouchet-Marquis, C.; Heiser, K.; Szomolanyi-Tsuda, E.; Mishra, R.; Lamothe, B.; Hoenger, A.; Garcea, R.L. Virion assembly factories in the nucleus of polyomavirus-infected cells. PLoS Pathog. 2012, 8, e1002630. [Google Scholar] [CrossRef]
- Cioce, M.; Lamond, A.I. Cajal bodies: A long history of discovery. Annu. Rev. Cell Dev. Biol. 2005, 21, 105–131. [Google Scholar] [CrossRef]
- James, N.J.; Howell, G.J.; Walker, J.H.; Blair, G.E. The role of Cajal bodies in the expression of late phase adenovirus proteins. Virology 2010, 399, 299–311. [Google Scholar] [CrossRef]
- Kim, S.H.; Ryabov, E.V.; Kalinina, N.O.; Rakitina, D.V.; Gillespie, T.; MacFarlane, S.; Haupt, S.; Brown, J.W.; Taliansky, M. Cajal bodies and the nucleolus are required for a plant virus systemic infection. EMBO J. 2007, 26, 2169–2179. [Google Scholar] [CrossRef]
- Weber, C.; Nover, L.; Fauth, M. Plant stress granules and mRNA processing bodies are distinct from heat stress granules. Plant J. 2008, 56, 517–530. [Google Scholar] [CrossRef]
- Lloyd, R.E. How do viruses interact with stress-associated RNA granules? PLoS Pathog. 2012, 8, e100274. [Google Scholar]
- Kulkarni, M.; Ozgur, S.; Stoecklin, G. On track with P-bodies. Biochem. Soc. Trans. 2010, 38, 242–251. [Google Scholar] [CrossRef]
- Iwasaki, S.; Takeda, A.; Motose, H.; Watanabe, Y. Characterization of Arabidopsis decapping proteins AtDCP1 and AtDCP2, which are essential for post-embryonic development. FEBS Lett. 2007, 581, 2455–2459. [Google Scholar] [CrossRef]
- Xu, J.; Chua, N.H. Processing bodies and plant development. Curr. Opin. Plant Biol. 2011, 14, 88–93. [Google Scholar] [CrossRef]
- Furtak, V.; Mulky, A.; Rawlings, S.A.; Kozhaya, L.; Lee, K.; Kewalramani, V.N.; Unutmaz, D. Perturbation of the P-body component Mov10 inhibits HIV-1 infectivity. PLoS One 2010, 5, e9081. [Google Scholar]
- Beckham, C.J.; Light, H.R.; Nissan, T.A.; Ahlquist, P.; Parker, R.; Noueiry, A. Interactions between brome mosaic virus RNAs and cytoplasmic processing bodies. J. Virol. 2007, 81, 9759–9768. [Google Scholar]
- Valiente-Echeverría, F.; Melnychuk, L.; Mouland, A.J. Viral modulation of stress granules. Virus Res. 2012, 20, 175–183. [Google Scholar]
- Linero, F.N.; Thomas, M.G.; Boccaccio, G.L.; Scolaro, L.A. Junin virus infection impairs stress-granule formation in Vero cells treated with arsenite via inhibition of eIF2α phosphorylation. J. Gen. Virol. 2011, 92, 2889–2899. [Google Scholar] [CrossRef]
- Simpson-Holley, M.; Kedersha, N.; Dower, K.; Rubins, K.H.; Anderson, P.; Hensley, L.E.; Connor, J.H. Formation of antiviral cytoplasmic granules during orthopoxvirus infection. J. Virol. 2011, 85, 1581–1593. [Google Scholar] [CrossRef]
- Nover, L.; Scharf, K.D.; Neumann, D. Formation of cytoplasmic heat shock granules in tomato cell cultures and leaves. Mol. Cell Biol. 1983, 3, 1648–1655. [Google Scholar]
- Kirschner, M.; Winkelhaus, S.; Thierfelder, J.M.; Nover, L. Transient expression and heat-stress-induced co-aggregation of endogenous and heterologous small heat-stress proteins in tobacco protoplasts. Plant J. 2000, 24, 397–411. [Google Scholar] [CrossRef]
- Kumakura, N.; Takeda, A.; Fujioka, Y.; Motose, H.; Takano, R.; Watanabe, Y. SGS3 and RDR6 interact and colocalize in cytoplasmic SGS3/RDR6-bodies. FEBS Lett. 2009, 583, 1261–1266. [Google Scholar] [CrossRef]
- Jouannet, V.; Moreno, A.B.; Elmayan, T.; Vaucheret, H.; Crespi, M.D.; Maizel, A. Cytoplasmic Arabidopsis AGO7 accumulates in membrane-associated siRNA bodies and is required for ta-siRNA biogenesis. EMBO J. 2012, 31, 1704–1713. [Google Scholar] [CrossRef]
- Du, Z.; Xiao, D.; Wu, J.; Jia, D.; Yuan, Z.; Liu, Y.; Hu, L.; Han, Z.; Wei, T.; Lin, Q.; Wu, Z.; Xie, L. p2 of rice stripe virus (RSV) interacts with OsSGS3 and is a silencing suppressor. Mol. Plant. Pathol. 2011, 12, 808–814. [Google Scholar] [CrossRef]
- Zrachya, A.; Glick, E.; Levy, Y.; Arazi, T.; Citovsky, V.; Gafni, Y. Suppressor of RNA silencing encoded by Tomato yellow leaf curl virus-Israel. Virology 2007, 358, 159–165. [Google Scholar] [CrossRef]
- Glick, E.; Zrachya, A.; Levy, Y.; Mett, A.; Gidoni, D.; Belausov, E.; Citovsky, V.; Gafni, Y. Interaction with host SGS3 is required for suppression of RNA silencing by tomato yellow leaf curl virus V2 protein. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 157–161. [Google Scholar]
- Russo, M.; Cohen, S.; Martelli, G.P. Virus-like particles in tomato plants affected by the yellow leaf curl disease. J. Gen. Virol. 1980, 49, 209–213. [Google Scholar] [CrossRef]
- Rothenstein, D.; Krenz, B.; Selchow, O.; Jeske, H. Tissue and cell tropism of Indian cassava mosaic virus (ICMV) and its AV2 (precoat) gene product. Virology 2007, 359, 137–145. [Google Scholar]
- Abouzid, A.M.; Barth, A.; Jeske, H. Immunogold labeling of the Abutilon mosaic virus in ultrathin sections of epoxy resin embedded leaf tissue. J. Ultrastruct. Res. 1988, 99, 39–47. [Google Scholar]
- Drucker, M.; Froissart, R.; Hébrard, E.; Uzest, M.; Ravallec, M.; Espérandieu, P.; Mani, J.C.; Pugnière, M.; Roquet, F.; Fereres, A.; Blanc, S. Intracellular distribution of viral gene products regulates a complex mechanism of cauliflower mosaic virus acquisition by its aphid vector. Proc. Natl. Acad. Sci. U. S. A. 2002, 99, 2422–2427. [Google Scholar]
- Khelifa, M.; Journou, S.; Krishnan, K.; Gargani, D.; Esperandieu, P.; Blanc, S.; Drucker, M. Electron-lucent inclusion bodies are structures specialized for aphid transmission of cauliflower mosaic virus. J. Gen. Virol. 2007, 88, 2872–2880. [Google Scholar] [CrossRef]
- Martinière, A.; Gargani, D.; Uzest, M.; Lautredou, N.; Blanc, S.; Drucker, M. A role for plant microtubules in the formation of transmission-specific inclusion bodies of Cauliflower mosaic virus. Plant J. 2009, 58, 135–146. [Google Scholar] [CrossRef]
- Froissart, R.; Uzest, M.; Ruiz-Ferrer, V.; Drucker, M.; Hebrard, E.; Hohn, T.; Blanc, S. Splicing of Cauliflower mosaic virus 35S RNA serves to downregulate a toxic gene product. J. Gen. Virol. 2004, 85, 2719–2725. [Google Scholar] [CrossRef]
- Gorovits, R.; Moshe, A.; Kolot, M.; Sobol, I.; Czosnek, H. Progressive aggregation of Tomato yellow leaf curl virus coat protein in systemically infected tomato plants, susceptible and resistant to the virus. Virus Res. 2012, in press. [Google Scholar]
- Gorovits, R. Personal communication. The Otto Warburg Minerva Center for Agricultural Biotechnology, The Robert H. Smith Faculty of Agriculture, Food and Environment, The Hebrew University of Jerusalem: Rehovot, Israel, 2012. [Google Scholar]
- Gorovits, R.; Czosnek, H. Biotic and abiotic stress response in tomato breeding lines resistant and susceptible to Tomato Yellow Leaf Curl Virus. In The Tomato Yellow Leaf Curl Virus Disease: Management, Molecular Biology and Breeding for Resistanc; Czosnek, H., Ed.; Springer: Dordrecht, The Netherlands, 2007; pp. 23–37, Part III, Chapter 6. [Google Scholar]
- Moshe, A.; Pfannstiel, J.; Brotman, Y.; Kolot, M.; Sobol, I.; Czosnek, H.; Gorovits, R. Stress responses to Tomato Yellow (Leaf Curl Virus TYLCV) infection of Resistant and Susceptible tomato plants are different. Metabolomics 2012. [Google Scholar] [CrossRef]
- Vidavsky, F.; Leviatov, S.; Milo, J.; Rabinowitch, H.D.; Kedar, N.; Czosnek, H. Behavior of tolerant tomato breeding lines (Lycopersicon esculentum) originating from three different sources (L. peruvianum, L. pimpinellifolium and L. chilense) upon early controlled inoculation by tomato yellow leaf curl virus. Plant Breed. 1998, 117, 165–169. [Google Scholar]
- Edwardson, J.R. Cylindrical inclusions in the cytoplasm of leaf cells infected with tobacco etch virus. Science 1966, 153, 883–884. [Google Scholar]
- Willison, J.H. The hexagonal lattice spacing of intracellular crystalline tobacco mosaic virus. J. Ultrastruct. Res. 1976, 54, 176–182. [Google Scholar] [CrossRef]
- Florida Plant Diagnostic Network (FPDN). Available online: http://fpdn.ifas.ufl.edu (accessed on 6 August 2012).
- Kim, K.S. An ultrastructural study of inclusions and disease development in plant cells infected by cowpea chlorotic mottle virus. J. Gen. Virol. 1977, 35, 535–543. [Google Scholar] [CrossRef]
- Russo, M.; Di Franco, A.; Martelli, G.P. The fine structure of Cymbidium ringspot virus infections in host tissues. III. Role of peroxisomes in the genesis of multivesicular bodies. J. Ultrastruct. Res. 1983, 82, 52–63. [Google Scholar] [CrossRef]
- van der Scheer, C.; Groenewegen, J. Structure in cells of Vigna unguiculata infected with cowpea mosaic virus. Virology 1971, 46, 493–497. [Google Scholar] [CrossRef]
- Laliberte, J.-F.; Sanfacon, H. Cellular remodeling during plant virus infection. Ann. Rev. Phytopathol. 2010, 48, 69–91. [Google Scholar] [CrossRef]
- Netherton, C.; Simpson, J.; Haller, O.; Wileman, T.E.; Takamatsu, H.H.; Monaghan, P.; Taylor, G. Inhibition of a large double-stranded DNA virus by MxA protein. J. Virol. 2009, 83, 2310–2320. [Google Scholar] [CrossRef]
- Haller, O.; Kochs, G. Human MxA protein: An interferon-induced dynamin-like GTPase with broad antiviral activity. J. Interfer. Cytokine Res. 2011, 31, 79–87. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Moshe, A.; Gorovits, R. Virus-Induced Aggregates in Infected Cells. Viruses 2012, 4, 2218-2232. https://doi.org/10.3390/v4102218
Moshe A, Gorovits R. Virus-Induced Aggregates in Infected Cells. Viruses. 2012; 4(10):2218-2232. https://doi.org/10.3390/v4102218
Chicago/Turabian StyleMoshe, Adi, and Rena Gorovits. 2012. "Virus-Induced Aggregates in Infected Cells" Viruses 4, no. 10: 2218-2232. https://doi.org/10.3390/v4102218



