Assessment of Benzo(a)pyrene-equivalent Carcinogenicity and Mutagenicity of Residential Indoor versus Outdoor Polycyclic Aromatic Hydrocarbons Exposing Young Children in New York City
Abstract
:1. Introduction
2. Experimental Section
- (BaP-TEQ)Σ8PAH = [BaA] × 0.1 + [Chry] × 0.01 + [BbFA] × 0.1 + [BkFA] × 0.1 + [BaP] × 1 + [IP] × 0.1 + [DahA] × 5 + [BghiP] × 0.01.
- (BaP-MEQ)Σ8PAH = [BaA] × 0.082 + [Chry] × 0.017 + [BbFA] × 0.25 + [BkFA] × 0.11 + [BaP] × 1 + [IP] × 0.31 + [DahA] × 0.29 + [BghiP] × 0.19.
3. Results
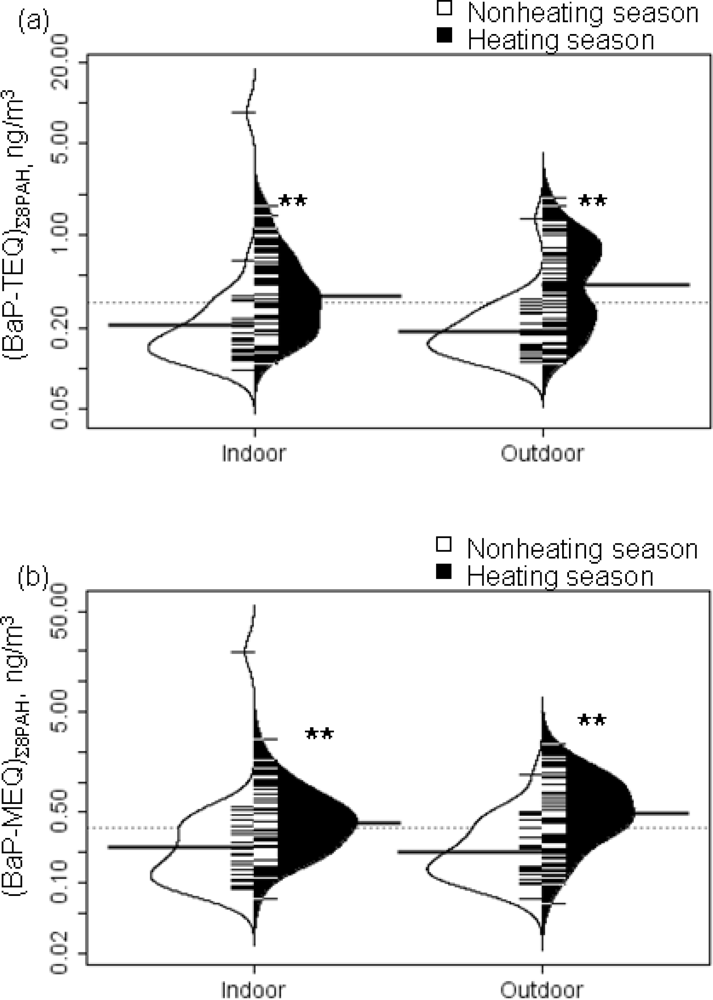
Residential indoor and outdoor BaP-equivalents
Seasonal variations in BaP-equivalents
4. Discussion and Conclusions
Acknowledgments
References
- Durant, J; Lafleur, A; Busby, W; Donhoffner, L; Penman, B; Crespi, C. Mutagenicity of C24H14 PAH in human cells expressing CYP1A1. Mutat. Res.-Genet. Toxicol. E. M 1999, 446, 1–14. [Google Scholar]
- Durant, J; Busby, W; Lafleur, A; Penman, B; Crespi, C. Human cell mutagenicity of oxygenated, nitrated and unsubstituted polycyclic aromatic hydrocarbons associated with urban aerosols. Mutat. Res.-Genet. Tox 1996, 371, 123–157. [Google Scholar]
- Nisbet, I; LaGoy, P. Toxic equivalency factors (TEFs) for polycyclic aromatic hydrocarbons (PAHs). Regul. Toxicol. Pharmacol. RTP 1992, 16, 290–300. [Google Scholar]
- Marty, MA; Alexeeff, GV; Collins, JF; Blaisdell, RJ; Rosenbaum, J; Lee, L. Airborne emissions from industrial point sources and associated cancer risks of selected carcinogens in California. In The Emissions Inventory: Perception and Reality Proceedings of an International Specialty Conference; Air & Waste Management Association: Pittsburgh, PA, USA, 1994; pp. 1086–1097. [Google Scholar]
- Wynder, E; Hoffmann, D. A study of tobacco carcinogenesis. X. Tumor promoting activity. CA-Cancer J. Clin 1959, 24, 289–301. [Google Scholar]
- Chu, MML; Chen, CW. Evaluation and estimation of potential carcinogenic risks of polynuclear aromatic hydrocarbons. In Presented at the Symposium on Polycyclic Aromatic Hydrocarbons in the Workplace; Pacific Rim Risk Conference: Honolulu, HI, USA, 1984. [Google Scholar]
- Clement, Comparative potency approach for estimating the cancer risk associated with exposure to mixtures of polycyclic aromatic hydrocarbons (Interim Final Report). In Prepared for EPA under Contract 68-02-4403; ICF-Clement Associates: Fairfax, VA, USA, April 1988.
- Collins, J; Brown, J; Alexeeff, G; Salmon, A. Potency equivalency factors for some polycyclic aromatic hydrocarbons and polycyclic aromatic hydrocarbon derivatives. Regul. Toxicol. Pharmacol 1998, 28, 45–54. [Google Scholar]
- Pfeiffer, EH. Oncogenic interaction of carcinogenic and non-carcinogenic polycyclic aromatic hydrocarbons. In Air Pollution and Cancer in Man; Mohr, V, Schmahl, D, Tomatis, L, Eds.; IARC Scientific Publication 16World Health Organization: Lyon, France, 1977. [Google Scholar]
- Kier, L; Brusick, D; Auletta, A; Von Halle, E; Brown, M; Simmon, V; Dunkel, V; McCann, J; Mortelmans, K. The Salmonella typhimurium/mammalian microsomal assay. A report of the US Environmental Protection Agency Gene-Tox Program. Mutat. Res 1986, 168, 67–238. [Google Scholar]
- Zeiger, E. Mutagens that are not carcinogens: faulty theory or faulty tests? Mutat. Res.-Genet. Toxicol. E. M 2001, 492, 29–38. [Google Scholar]
- Zeiger, E. Identification of rodent carcinogens and noncarcinogens using genetic toxicity tests: premises, promises, and performance. Regul. Toxicol. Pharmacol 1998, 28, 85–95. [Google Scholar]
- DeMarini, D; Brooks, L; Warren, S; Kobayashi, T; Gilmour, M; Singh, P. Bioassay-directed fractionation and salmonella mutagenicity of automobile and forklift diesel exhaust particles. Environ. Health Perspect 2004, 112, 814. [Google Scholar]
- Seagrave, J; McDonald, J; Gigliotti, A; Nikula, K; Seilkop, S; Gurevich, M; Mauderly, J. Mutagenicity and in vivo toxicity of combined particulate and semivolatile organic fractions of gasoline and diesel engine emissions. Toxicol. Sci 2002, 70, 212. [Google Scholar]
- Ohura, T; Amagai, T; Fusaya, M; Matsushita, H. Polycyclic aromatic hydrocarbons in indoor and outdoor environments and factors affecting their concentrations. Environ. Sci. Technol 2004, 38, 77–83. [Google Scholar]
- Ohura, T; Noda, T; Amagai, T; Fusaya, M. Prediction of personal exposure to PM 2. 5 aid carcinogenic polycyclic aromatic hydrocarbons by their concentrations in residential microenvironments. Environ. Sci. Technol 2005, 39, 5592–5599. [Google Scholar]
- Nielsen, T; Jørgensen, H; Larsen, J; Poulsen, M. City air pollution of polycyclic aromatic hydrocarbons and other mutagens: occurrence, sources and health effects. Sci. Total Environ 1996, 189, 41–49. [Google Scholar]
- Miller, R; Garfinkel, R; Horton, M; Camann, D; Perera, F; Whyatt, R; Kinney, P. Polycyclic aromatic hydrocarbons, environmental tobacco smoke, and respiratory symptoms in an inner-city birth cohort. Chest 2004, 126, 1071–1078. [Google Scholar]
- Perera, F; Illman, S; Kinney, P; Whyatt, R; Kelvin, E; Shepard, P; Evans, D; Fullilove, M; Ford, J; Miller, R. The challenge of preventing environmentally related disease in young children: community-based research in New York City. Environ. Health Perspect 2002, 110, 197–204. [Google Scholar]
- Perera, FP; Rauh, V; Whyatt, RM; Tsai, WY; Tang, D; Diaz, D; Hoepner, L; Barr, D; Tu, YH; Camann, D; Kinney, P. Effect of prenatal exposure to airborne polycyclic aromatic hydrocarbons on neurodevelopment in the first 3 years of life among inner-city children. Environ. Health Perspect 2006, 114, 1287–1292. [Google Scholar]
- Jung, KH; Patel, MM; Kinney, PL; Chillrud, SN; Whyatt, R; Hoepner, L; Yan, B; Garfinkel, R; Ross, J; Camann, D; Perera, FP; Miller, RL. Effects of season and indoor heating on indoor and outdoor residential levels of airborne polycyclic aromatic hydrocarbons, absorbance and particulate matter 2.5 in an inner city cohort of young children. J Allergy Clin Immunol 2010, 125, AB81. [Google Scholar]
- Whyatt, R; Garfinkel, R; Hoepner, L; Holmes, D; Borjas, M; Williams, M; Reyes, A; Rauh, V; Perera, FP; Camann, DE. Within-and between-home variability in indoor-air insecticide levels during pregnancy among an inner-city cohort from New York City. Environ. Health Perspect 2007, 115, 383–389. [Google Scholar]
- Tonne, C; Whyatt, R; Camann, D; Perera, F; Kinney, P. Predictors of personal polycyclic aromatic hydrocarbon exposures among pregnant minority women in New York City. Environ. Health Perspect 2004, 112, 754–760. [Google Scholar]
- Boffetta, P. Cancer risk from occupational and environmental exposure to polycyclic aromatic hydrocarbons. Cancer Causes Control 1997, 8, 442–472. [Google Scholar]
- Choi, H; Jedrychowski, W; Spengler, J; Camann, D; Whyatt, R; Rauh, V; Tsai, W; Perera, F. International studies of prenatal exposure to polycyclic aromatic hydrocarbons and fetal growth. Environ. Health Perspect 2006, 114, 1744. [Google Scholar]
- Miguel, A; Pereira, P. Benzo (k) fluoranthene, benzo (ghi) perylene and indeno (1, 2, 3-cd) pyrene. New tracers of automotive emissions in receptor modeling. Aerosol. Sci. Tech 1989, 10, 292–295. [Google Scholar]
- Kinney, PL; Chillrud, SN; Ramstrom, S; Ross, J; Spengler, JD. Exposures to multiple air toxics in New York City. Environ. Health Perspect 2002, 110, 539–546. [Google Scholar]
- Schauer, C; Niessner, R; Poschl, U. Polycyclic aromatic hydrocarbons in urban air particulate matter: decadal and seasonal trends, chemical degradation, and sampling artifacts. Environ. Sci. Technol 2003, 37, 2861–2868. [Google Scholar]
- Ohura, T; Amagai, T; Sugiyama, T; Fusaya, M; Matsushita, H. Characteristics of particle matter and associated polycyclic aromatic hydrocarbons in indoor and outdoor air in two cities in Shizuoka, Japan. Atmos. Environ 2004, 38, 2045–2054. [Google Scholar]
- WHO, Air quality guidelines for Europe. WHO Regional Publications, European Series, No. 23, 1987.
- Goriaux, M; Jourdain, B; Temime, B; Besombes, J; Marchand, N; Albinet, A; Leoz-Garziandia, E; Wortham, H. Field comparison of particulate PAH measurements using a low-flow denuder device and conventional sampling systems. Environ. Sci. Technol 2006, 40, 6398–6404. [Google Scholar]
- Lodovici, M; Venturini, M; Marini, E; Grechi, D; Dolara, P. Polycyclic aromatic hydrocarbons air levels in Florence, Italy, and their correlation with other air pollutants. Chemosphere 2003, 50, 377–382. [Google Scholar]
- Marr, L; Dzepina, K; Jimenez, J; Reisen, F; Bethel, H; Arey, J; Gaffney, J; Marley, N; Molina, L; Molina, M. Sources and transformations of particle-bound polycyclic aromatic hydrocarbons in Mexico City. Atmos. Chem. Phys 2006, 6, 1733–1745. [Google Scholar]
- Brorström, E; Grennfelt, P; Lindskog, A. The effect of nitrogen dioxide and ozone on the decomposition of particle-associated polycyclic aromatic hydrocarbons during sampling from the atmosphere. Atmos. Environ 1983, 17, 601–605. [Google Scholar]
- Pitts, J, Jr; Lokensgard, D; Ripley, P; van Cauwenberghe, K; van Vaeck, L; Shaffer, S; Thill, A; Belser, W, Jr. “Atmospheric” epoxidation of benzo[a]pyrene by ozone: formation of the metabolite benzo[a]pyrene-4,5-oxide. Science 1980, 210, 1347–1349. [Google Scholar]
- Tsapakis, M; Stephanou, E. Collection of gas and particle semi-volatile organic compounds: use of an oxidant denuder to minimize polycyclic aromatic hydrocarbons degradation during high-volume air sampling. Atmos. Environ 2003, 37, 4935–4944. [Google Scholar]
- Busby, W; Stevens, E; Martin, C; Chow, F; Garner, R. Comparative lung tumorigenicity of parent and mononitro-polynuclear aromatic hydrocarbons in the BLU: Ha newborn mouse assay. Toxicol. Appl. Pharmacol 1989, 99, 555–563. [Google Scholar]
- Wislocki, P; Bagan, E; Lu, A; Dooley, K; Fu, P; Han-Hsu, H; Beland, F; Kadlubar, F. Tumorigenicity of nitrated derivatives of pyrene, benz[a]anthracene, chrysene and benzo[a]pyrene in the newborn mouse assay. Carcinogenesis 1986, 7, 1317–1322. [Google Scholar]
- Chuang, J; Callahan, P; Lyu, C; Wilson, N. Polycyclic aromatic hydrocarbon exposures of children in low-income families. J. Expo. Anal. Env. Epid 1999, 9, 85–98. [Google Scholar]
- Vyskocil, A; Fiala, Z; Chéier, V; Krajak, L; Ettlerova, E; Bukac, J; Viau, C; Emminger, S. Assessment of multipathway exposure of small children to PAH. Environ. Toxicol. Pharmacol 2000, 8, 111–118. [Google Scholar]
| BaP-TEQ Levels, ng/m3 | BaP-MEQ Levels, ng/m3 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Analyte | n | aTEF | Median | Mean | SD | Range | bMEF | Median | Mean | SD | Range |
| Σ8PAH | 255 | NA | 0.299 | 0.478 | 0.709 | 0.098–8.348 | NA | 0.376 | 0.590 | 1.325 | 0.069–19.72 |
| BaP | 255 | 1 | 0.121 | 0.198 | 0.363 | 0.015–4.494 | 1 | 0.121 | 0.198 | 0.363 | 0.015–4.494 |
| BaA | 255 | 0.1 | 0.006 | 0.009 | 0.010 | 0.002–0.132 | 0.082 | 0.005 | 0.007 | 0.009 | 0.001–0.108 |
| Chry | 255 | 0.01 | 0.001 | 0.001 | 0.002 | 0.000–0.021 | 0.017 | 0.001 | 0.002 | 0.003 | 0.000–0.036 |
| BbFA | 255 | 0.1 | 0.019 | 0.028 | 0.034 | 0.004–0.346 | 0.25 | 0.047 | 0.069 | 0.084 | 0.010–0.865 |
| BkFA | 255 | 0.1 | 0.006 | 0.010 | 0.013 | 0.002–0.140 | 0.11 | 0.007 | 0.010 | 0.014 | 0.002–0.154 |
| IP | 255 | 0.1 | 0.028 | 0.044 | 0.094 | 0.002–1.400 | 0.31 | 0.087 | 0.136 | 0.291 | 0.006–4.340 |
| DahA | 255 | 5 | 0.083 | 0.180 | 0.212 | 0.055–1.741 | 0.29 | 0.005 | 0.010 | 0.012 | 0.003–0.101 |
| BghiP | 255 | 0.01 | 0.004 | 0.008 | 0.033 | 0.001–0.517 | 0.19 | 0.084 | 0.156 | 0.624 | 0.016–9.828 |
| Outdoor | |||||||||||
| Σ8PAH | 82 | NA | 0.277 | 0.450 | 0.389 | 0.109–1.932 | NA | 0.360 | 0.528 | 0.454 | 0.062–2.394 |
| BaP | 82 | 1 | 0.091 | 0.133 | 0.133 | 0.016–0.748 | 1 | 0.091 | 0.133 | 0.133 | 0.016–0.748 |
| BaA | 82 | 0.1 | 0.007 | 0.011 | 0.011 | 0.001–0.059 | 0.082 | 0.005 | 0.009 | 0.009 | 0.001–0.048 |
| Chry | 82 | 0.01 | 0.001 | 0.002 | 0.002 | 0.0003–0.008 | 0.017 | 0.002 | 0.004 | 0.003 | 0.001–0.013 |
| BbFA | 82 | 0.1 | 0.039 | 0.048 | 0.043 | 0.006–0.237 | 0.25 | 0.097 | 0.121 | 0.108 | 0.015–0.592 |
| BkFA | 82 | 0.1 | 0.010 | 0.015 | 0.017 | 0.002–0.114 | 0.11 | 0.011 | 0.017 | 0.018 | 0.002–0.125 |
| IP | 82 | 0.1 | 0.027 | 0.035 | 0.031 | 0.003–0.197 | 0.31 | 0.085 | 0.107 | 0.095 | 0.010–0.611 |
| DahA | 82 | 5 | 0.083 | 0.199 | 0.188 | 0.075–1.021 | 0.29 | 0.005 | 0.012 | 0.011 | 0.004–0.059 |
| BghiP | 82 | 0.01 | 0.005 | 0.007 | 0.007 | 0.001–0.039 | 0.19 | 0.087 | 0.126 | 0.126 | 0.010–0.739 |
| Season | Measure | Indoor, ng/m3 (na = 55; nb = 26) | Outdoor, ng/m3 (na = 55; nb = 26) | |||
|---|---|---|---|---|---|---|
| Average ± SD | Median | Average ± SD | Median | p | ||
| Heating | (BaP-TEQ)∑8PAH | 0.460 ± 0.365 | 0.346 | 0.558 ± 0.407 | 0.485 | 0.002** |
| (BaP-MEQ)∑8PAH | 0.521 ± 0.448 | 0.388 | 0.660 ± 0.480 | 0.500 | 0.001** | |
| Nonheating | (BaP-TEQ)∑8PAH | 0.515 ± 1.602 | 0.163 | 0.232 ± 0.233 | 0.167 | 0.989 |
| (BaP-MEQ)∑8PAH | 0.975 ± 3.826 | 0.174 | 0.260 ± 0.230 | 0.173 | 0.989 | |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jung, K.H.; Yan, B.; Chillrud, S.N.; Perera, F.P.; Whyatt, R.; Camann, D.; Kinney, P.L.; Miller, R.L. Assessment of Benzo(a)pyrene-equivalent Carcinogenicity and Mutagenicity of Residential Indoor versus Outdoor Polycyclic Aromatic Hydrocarbons Exposing Young Children in New York City. Int. J. Environ. Res. Public Health 2010, 7, 1889-1900. https://doi.org/10.3390/ijerph7051889
Jung KH, Yan B, Chillrud SN, Perera FP, Whyatt R, Camann D, Kinney PL, Miller RL. Assessment of Benzo(a)pyrene-equivalent Carcinogenicity and Mutagenicity of Residential Indoor versus Outdoor Polycyclic Aromatic Hydrocarbons Exposing Young Children in New York City. International Journal of Environmental Research and Public Health. 2010; 7(5):1889-1900. https://doi.org/10.3390/ijerph7051889
Chicago/Turabian StyleJung, Kyung Hwa, Beizhan Yan, Steven N. Chillrud, Frederica P. Perera, Robin Whyatt, David Camann, Patrick L. Kinney, and Rachel L. Miller. 2010. "Assessment of Benzo(a)pyrene-equivalent Carcinogenicity and Mutagenicity of Residential Indoor versus Outdoor Polycyclic Aromatic Hydrocarbons Exposing Young Children in New York City" International Journal of Environmental Research and Public Health 7, no. 5: 1889-1900. https://doi.org/10.3390/ijerph7051889
APA StyleJung, K. H., Yan, B., Chillrud, S. N., Perera, F. P., Whyatt, R., Camann, D., Kinney, P. L., & Miller, R. L. (2010). Assessment of Benzo(a)pyrene-equivalent Carcinogenicity and Mutagenicity of Residential Indoor versus Outdoor Polycyclic Aromatic Hydrocarbons Exposing Young Children in New York City. International Journal of Environmental Research and Public Health, 7(5), 1889-1900. https://doi.org/10.3390/ijerph7051889




