Evaluation of the Effects of Chronic Intoxication with Inorganic Mercury on Memory and Motor Control in Rats
Abstract
:1. Introduction
2. Methods
2.1. Ethics Statement
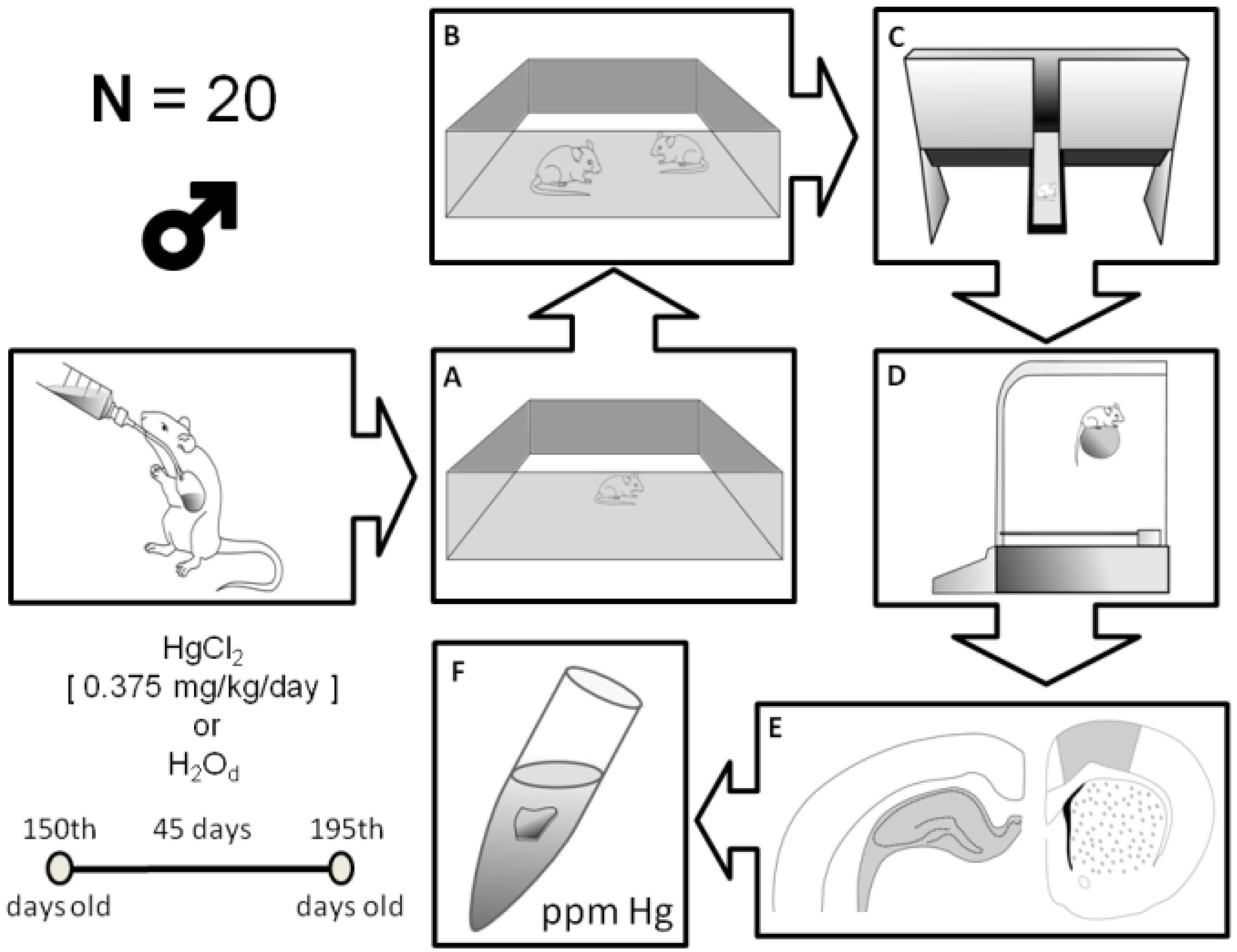
2.2. Animals and Experimental Groups
2.3. Behavioral Assays
2.4. Open Field
2.5. Social Recognition
2.6. Elevated T Maze (ETM) Test
2.7. Rotarod Test
2.8. Mercury Measurements
2.9. Statistical Analysis
3. Results
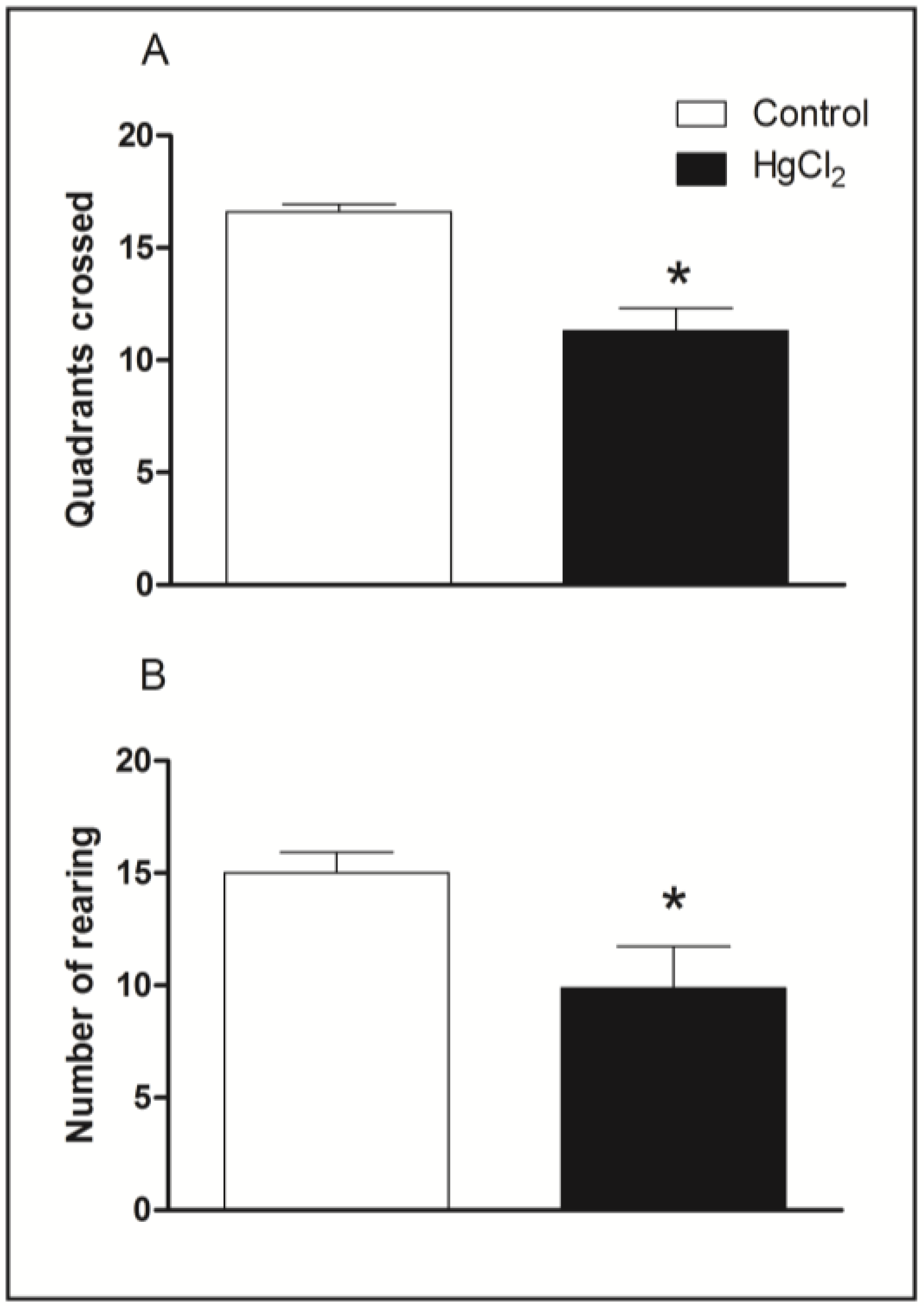
3.1. Chronic HgCl2 Exposure during Late Adulthood Induces Deficits on Spontaneous Locomotor Activity in Rats
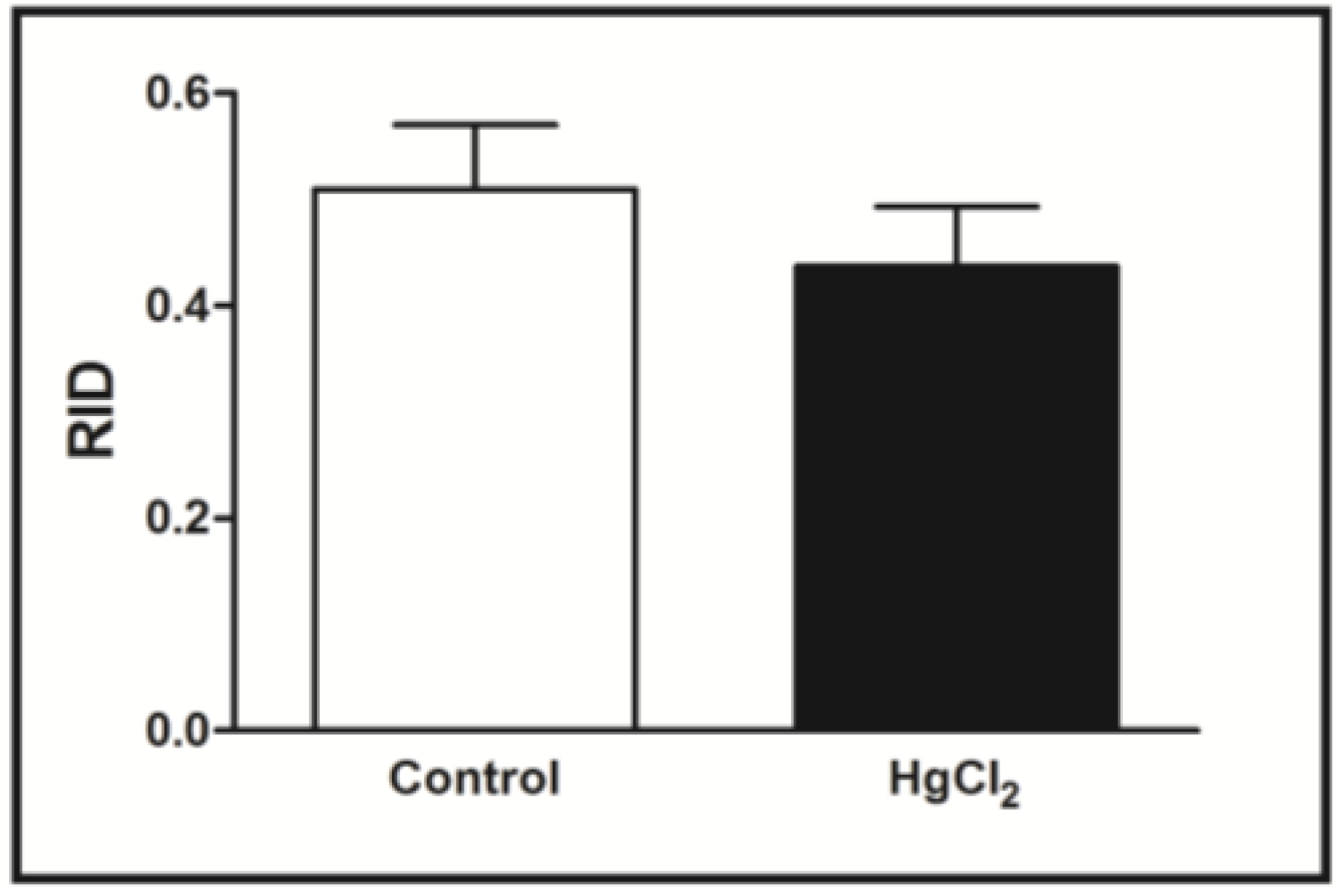
3.2. Learning, Short- and Long-Term Memory Impairments Induced by Chronic HgCl2 Exposure during Late Adulthood in Rats
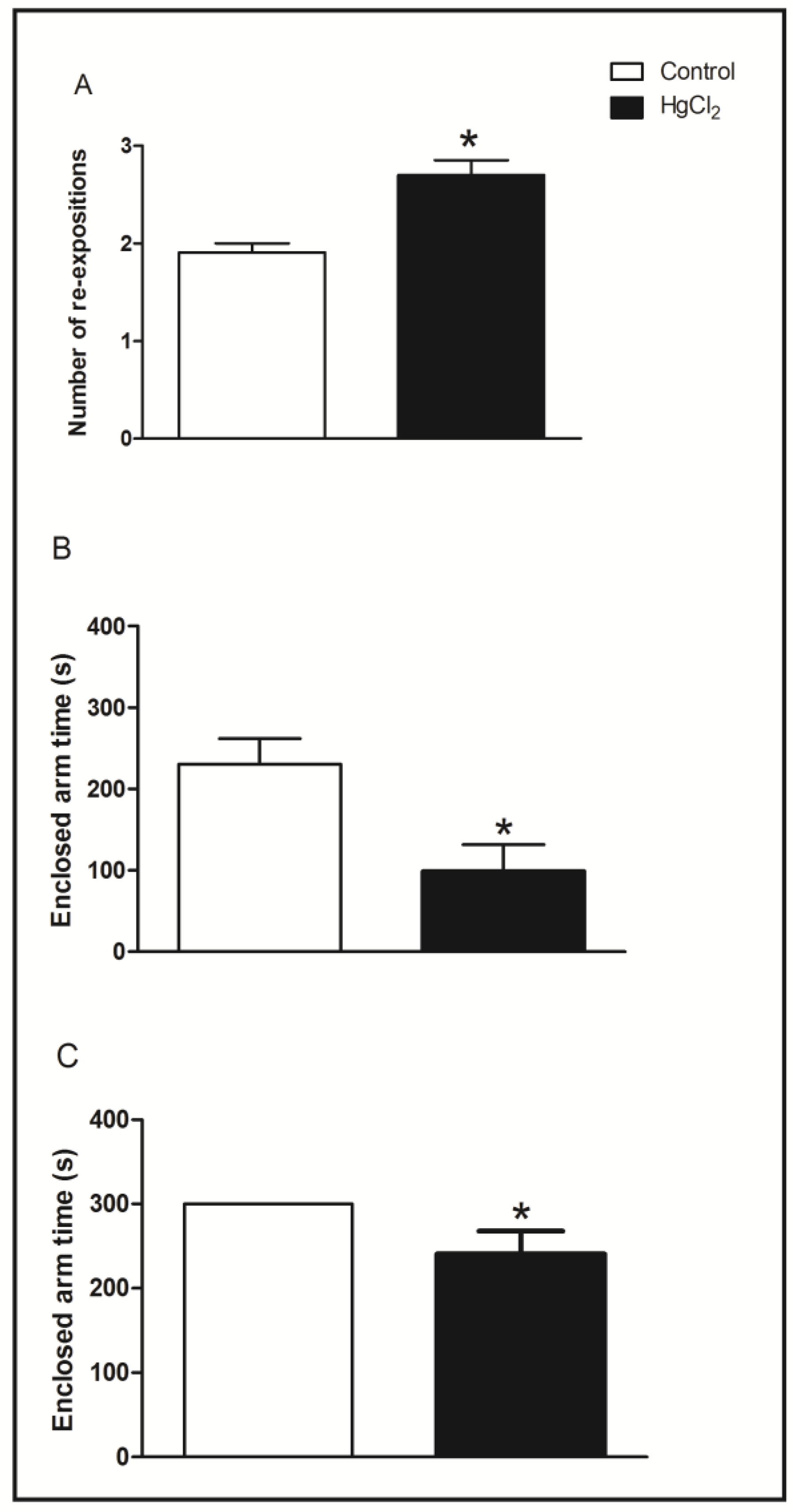
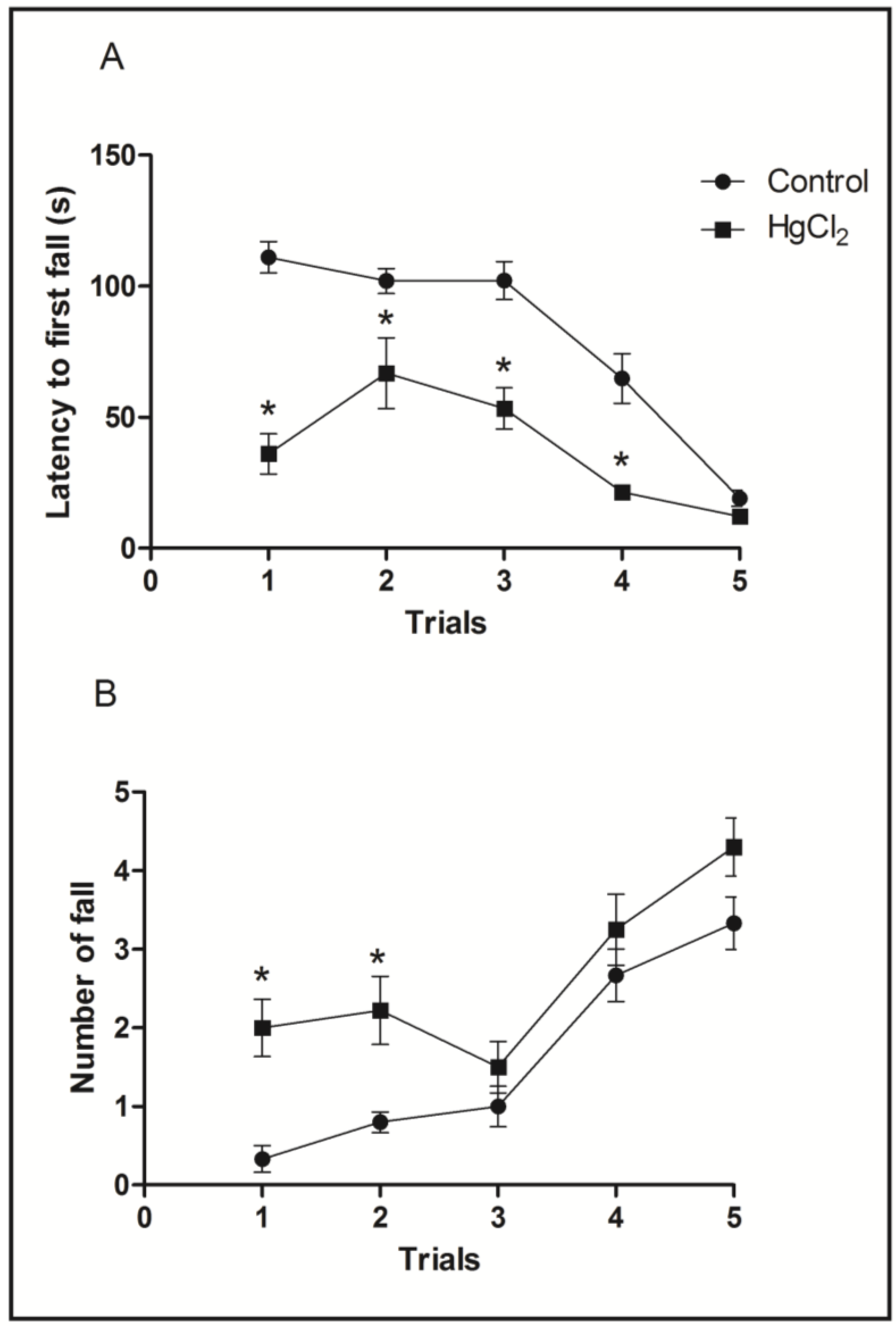
3.3. Chronic HgCl2 Exposure during Late Adulthood Promotes Alterations in Motor Function
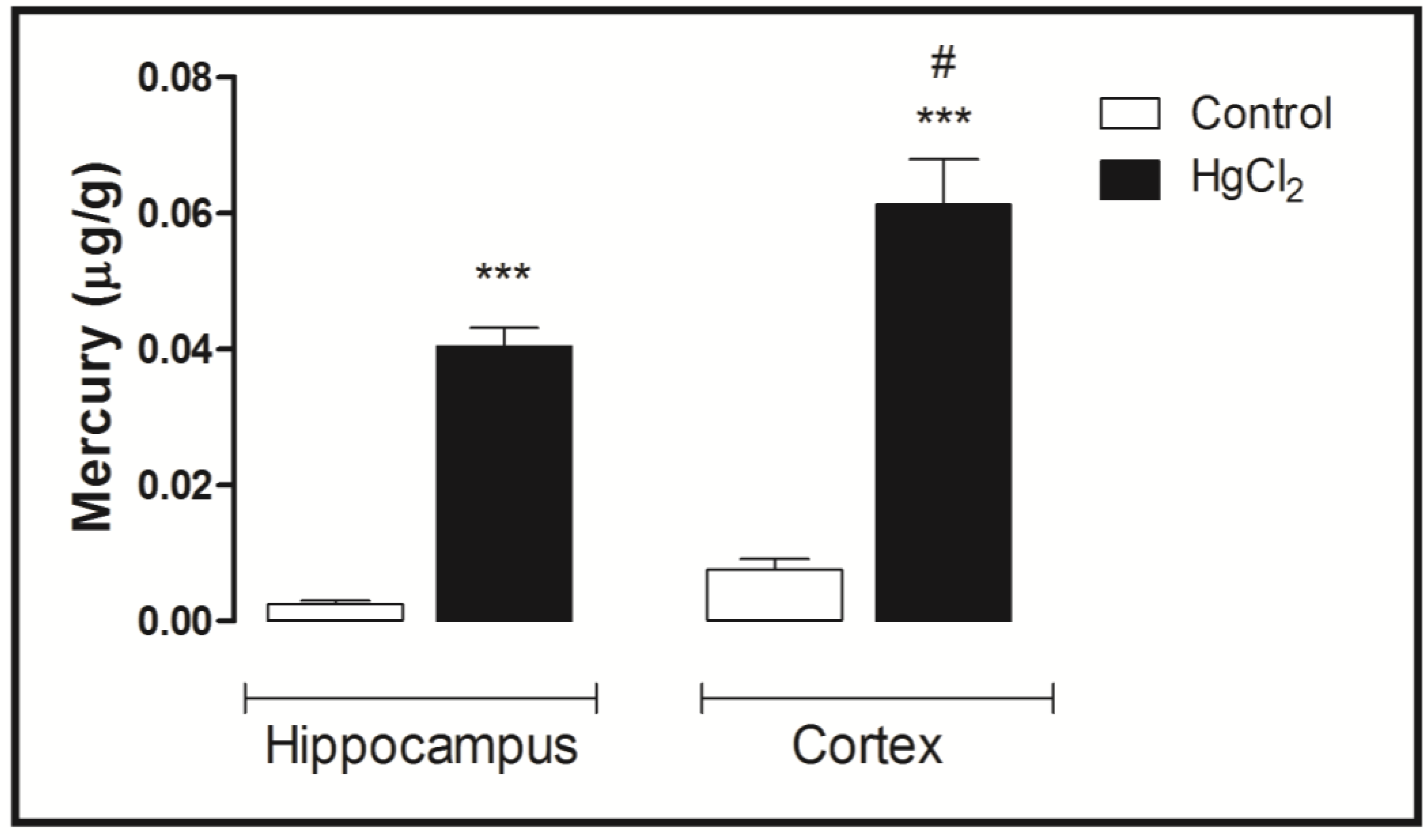
3.4. Mercury Deposition Is Higher in the Cortex than Hippocampus after Chronic Intoxication during Late Life in Rats
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bernhoft, R.A. Mercury toxicity and treatment: A review of the literature. J. Environ. Public Health 2012, 2012. [Google Scholar] [CrossRef]
- Goldman, L.R.; Shannon, M.W. American Academy of Pediatrics: Committee on Environmental Health Technical report: Mercury in the environment: Implications for pediatricians. Pediatrics 2001, 108, 197–205. [Google Scholar] [CrossRef]
- Dyall-Smith, D.J.; Scurry, J.P. Mercury pigmentation and high mercury levels from the use of a cosmetic cream. Med. J. Aust. 1990, 153, 409–415. [Google Scholar]
- Weldon, M.M.; Smolinski, M.S.; Maroufi, A.; Hasty, B.W.; Gilliss, D.L.; Boulanger, L.L.; Dutton, R.J. Mercury poisoning associated with a Mexican beauty cream. West. J. Med. 2000, 173, 15–18. [Google Scholar] [CrossRef]
- Clarkson, T.W.; Magos, L. The toxicology of mercury and its chemical compounds. Crit. Rev. Toxicol. 2006, 36, 609–662. [Google Scholar] [CrossRef]
- Szumafiska, G.; Gadamski, R.; Albrecht, J. Changes of the Na/K ATPase activity in the cerebral cortical microvessels of rat after single intraperitoneal administration of mercuric chloride: Histochemical demonstration with light and electron microscopy. Acta Neuropathol. 1993, 86, 65–70. [Google Scholar] [CrossRef]
- Rice, K.M.; Walker, E.M., Jr.; Gillette, M.W.C.; Blough, E.R. Environmental mercury and its toxic effects. J. Prev. Med. Public Health 2014, 47, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.C.; Allen, P.V.; Turner, M.D.; Most, B.; Fisher, H.L.; Hall, L.L. The kinetics of intravenously administered methylmercury in man. Toxicol. Appl. Pharmacol. 1994, 128, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Szasz, A.; Barna, B.; Gajda, Z.; Galbacs, G.; Kirsch-Volders, M.; Szente, M. Effects of continuous low-dose exposure to organic and inorganic mercury during development on epileptogenicity in rats. Neurotoxicology 2002, 23, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Liu, S.; Hsu, C.; Lin-Shiau, S. Neurotoxicological effects of low-dose methylmercury and mercury chloride in developing offspring mice. Toxicol. Lett. 2009, 201, 196–204. [Google Scholar] [CrossRef]
- Cai, L.; Yan, X.B.; Chen, X.N.; Meng, Q.Y.; Zhou, J.N. Chronic all-trans retinoic acid administration induced hyperactivity of HPA axis and behavioral changes in young rats. Eur. Neuropsychopharmacol. 2010, 20, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, R.; Bluthe, R.M.; Koob, G.F.; le Moal, M. Modulation of social memory in male rats by neurohypophyseal peptides. Psychopharmacology 1987, 91, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Prediger, R.D.; Fernandes, D.; Takahashi, R.N. Blockade of adenosine A2A receptors reverses short-term social memory impairments in spontaneously hypertensive rats. Behav. Brain Res. 2005, 159, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, R.N.; Pamplona, F.A.; Fernandes, M.S. The cannabinoid antagonist SR141716A facilitates memory acquisition and consolidation in the mouse elevated T-maze. Neurosci. Lett. 2005, 380, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Maia, C.S.F.; Ferreira, V.M.; Diniz, J.S.; Carneiro, F.P.; de Sousa, J.B.; Costa, E.D.; Tomaz, C. Inhibitory avoidance acquisition in adult rats exposed to a combination of ethanol and methylmercury during central nervous system development. Behav. Brain Res. 2010, 211, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.J.; Sunkaria, A.; Bal, A.; Bhutia, Y.D.; Vijayaraghavan, R.; Flora, S.J.S.; Gill, K.D. Neurobehavioral impairments, generation of oxidative stress and release of pro-apoptotic factors after chronic exposure to sulphur mustard in mouse brain. Toxicol. Appl. Pharmacol. 2009, 240, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Akagi, H. Analysis of methylmercury in fish and shellfish by dithizone extraction-gas chromatography. Jpn. J. Hyg. 1985, 40, 293. [Google Scholar]
- Maia, C.S.F.; Ferreira, V.M.; Kahwage, R.L.; do Amaral, M.N.; Serra, R.B.; Santos, S.N.; Nascimento, J.L.M.; Rodrigues, L.G.; Trévia, N.; Picanço-Diniz, C.W. Adult brain nitrergic activity after concomitant prenatal exposure to ethanol and methyl mercury. Acta Histochem. 2010, 112, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Maia, C.S.F.; Lucena, G.M.; Correa, P.B.; Serra, R.B.; Matos, R.W.; Menezes, F.C.; Santos, S.N.; Sousa, J.B.; Costa, E.T.; Ferreira, V.M.M. Interference of ethanol and methylmercury in the developing central nervous system. Neurotoxicology 2009, 30, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, P.J.; Mitchell, J.; Wheal, H.V. Identity of ultrastructural effects of mercuric chloride and methyl mercury after intracerebral injection. Toxicology 1982, 23, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Maier, W.E.; Costa, L.G. Na+/K+ ATPase as a and marker, respectively, for neurotoxicity: Studies with chlordecone, organotins and mercury compounds. Toxicol. Lett. 1990, 51, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Moller-Madsen, B. Localization of mercury in CNS of the rat. II. Intraperitoneal injection of methylmercuric chloride (CH3HgCl) and mercuric chloride (HgC12). Toxicol. Appl. Pharmacol. 1990, 103, 303–323. [Google Scholar]
- Moller-Madsen, B.; Danscher, G. Localization of mercury in CNS of the rat I. Mercuric Chloride (HgCl2) per os. Environ. Res. 1986, 41, 29–43. [Google Scholar]
- Pamphlett, R.; Jew, S.K. Uptake of inorganic mercury by human locus ceruleus and corticomotor neurons: Implications for amyotrophic lateral sclerosis. Acta Neuropathol. Commun. 2013, 1. [Google Scholar] [CrossRef]
- Arvidson, B. A review of axonal transport of metals. Toxicology 1994, 88, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Brookes, N.; Kristt, D.A. Inhibition of amino acid transport and protein synthesis by HgC12 and methylmercury in astrocytes: Selectivity and reversibility. J. Neurochem. 1989, 53, 1228–1237. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.H.; Kim, R.C. The comparative effects of methylmercuric chloride and mercuric chloride upon DNA synthesis in mouse fetal astrocytes in vitro. Exp. Mol. Pathol. 1984, 41, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Franciscato, C.; Goulart, F.R.; Lovatto, N.M.; Duarte, F.A.; Flores, E.M.; Dressler, V.L.; Peixoto, N.C.; Pereira, M.E. ZnCl2 exposure protects against behavioral and acetylcholinesterase changes induced by HgCl2. Int. J. Dev. Neurosci. 2009, 27, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Franco, J.L.; Braga, H.C.; Nunes, A.K.; Ribas, C.M.; Stringari, J.; Silva, A.P.; Garcia Pomblum, S.C.; Moro, A.M.; Bohrer, D.; Santos, A.R.; et al. Lactational exposure to inorganic mercury: Evidence of neurotoxic effects. Neurotoxicol. Teratol. 2007, 29, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Yasutake, A.; Marumoto, M.; Yoshida, M. Neurotoxic action of inorganic mercury injected in the intraventricular space of mouse cerebrum. J. Toxicol. Sci. 2010, 35, 767–771. [Google Scholar] [CrossRef] [PubMed]
- Skinner, H.B.; Barrack, R.L.; Cook, S.D. Age-related decline in proprioception. Clin. Orthop. Relat. Res. 1984, 184, 208–211. [Google Scholar] [PubMed]
- Sturnieks, D.L.; St George, R.; Lord, S.R. Balance disorders in the elderly. Neurophysiol. Clin. 2008, 38, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Asanuma, H. Cerebral cortical control of movement. Physiologist 1973, 16, 143–166. [Google Scholar] [PubMed]
- Evarts, E.V.; Fromm, C.; Kroller, J.; Jennings, V.A. Motor cortex control of finely graded forces. J. Neurophysiol. 1983, 49, 1199–1215. [Google Scholar] [PubMed]
- Sanes, J.N.; Donoghue, J.P. Plasticity and primary motor cortex. Annu. Rev. Neurosci. 2000, 23, 393–415. [Google Scholar] [CrossRef] [PubMed]
- Stigger, F.; Lovatel, G.; Marques, M.; Bertoldi, K.; Moysés, F.; Elsner, V.; Siqueira, I.R.; Achaval, R.; Marcuzzo, S. Inflammatory response and oxidative stress in developing rat brain and its consequences on motor behavior following maternal administration of LPS and perinatal anoxia. Int. J. Dev. Neurosci. 2013, 31, 820–827. [Google Scholar] [CrossRef] [PubMed]
- Carmel, J.B.; Martin, J.H. Motor cortex electrical stimulation augmentes sprouting of the corticospinal tract and promotes recovery of motor function. Front. Integr. Neurosci. 2014, 8. [Google Scholar] [CrossRef]
- Karl, T.; Pabst, R.; Von Hörsten, S. Behavioral phenotyping of mice in pharmacological and toxicological research. Exp. Toxicol. Pathol. 2003, 55, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Helfer, J.L.; Calizo, L.H.; Dong, W.K.; Goodlett, C.R.; Greenough, W.T.; Klintsova, A.Y. Binge-like postnatal alcohol exposure triggers cortical gliogenesis in adolescent rats. J. Comp. Neurol. 2009, 514, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, G.B.; Fontes, E.D.; de Carvalho, S.; da Silva, J.B.; Fernandes, L.M.; Oliveira, M.C.; Prediger, R.D.; Gomes-Leal, W.; Lima, R.R.; Maia, C.S. Mynocycline mitigates motor impairments and cortical neuronal loss induced by focal ischemia in rats chronically exposed to ethanol during adolescence. Brain Res. 2014, 1561, 23–34. [Google Scholar] [CrossRef]
- Teixeira, F.B.; Santana, L.N.; Bezerra, F.R.; de Carvalho, S.; Fontes-Júnior, E.A.; Prediger, R.D.; Crespo-López, M.E.; Maia, C.S.; Lima, R.R. Chronic ethanol exposure during adolescence in rats induces motor impairments and cerebral cortex damage associated with oxidative stress. PLoS One 2014, 26. [Google Scholar] [CrossRef]
- Packard, M.G.; McGaugh, J.L. Double dissociation of fornix and caudate nucleus lesions on acquisition of two water maze tasks: Further evidence for multiple memory systems. Behav. Neurosci. 1992, 106, 439–446. [Google Scholar] [CrossRef] [PubMed]
- McDonald, R.J.; White, N.M. A triple dissociation of memory systems: Hippocampus, amygdala, and dorsal striatum. Behav. Neurosci. 1993, 107, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Middleton, F.A.; Strick, P.L. Basal-ganglia “projections” to the prefrontal cortex of the primate. Cereb. Cortex 2002, 12, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Moser, E.; Moser, M.B.; Andersen, P. Spatial learning impairment parallels the magnitude of dorsal hippocampal lesions, but is hardly present following ventral lesions. J. Neurosci. 1993, 13, 3916–3925. [Google Scholar] [PubMed]
- Bannerman, D.M.; Rawlins, J.N.; McHugh, S.B.; Deacon, R.M.; Yee, B.K.; Bast, T.; Zhang, W.N.; Pothuizen, H.H.; Feldon, J. Regional dissociations within the hippocampus: Memory and anxiety. Neurosci Biobehav Rev. 2004, 28, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Bondy, S.C.; Agrawal, A.K. The inhibition of cerebral high affinity receptor sites by lead and mercury compounds. Arch. Toxicol. 1980, 46, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Castoldi, A.F.; Candura, S.M.; Costa, P.; Manzo, L.; Costa, L.G. Interaction of mercury compounds with muscarinic receptor subtypes in the rat brain. Neurotoxicology 1996, 17, 735–742. [Google Scholar] [PubMed]
- Arakawa, O.; Nakahiro, M.; Narahashi, T. Mercury modulation of GABA-activated chloride channels and non-specific cation channels in rat dorsal root ganglion neurons. Brain Res. 1991, 551, 58–63. [Google Scholar]
- Rajanna, B.; Rajanna, S.; Hall, E.; Yallapragada, P.R. In vitro metal inhibition of N-methyl-d-aspartate specific glutamate receptor binding in neonatal and adult rat brain. Drug Chem. Toxicol. 1997, 20, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Scheuhammer, A.M.; Cherian, M.G. Effects of heavy metal cations sulfhydryl reagents and other chemical agents on striatal D2 dopamine receptors. Biochem. Pharmacol. 1985, 34, 3405–3413. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Teixeira, F.B.; Fernandes, R.M.; Farias-Junior, P.M.A.; Costa, N.M.M.; Fernandes, L.M.P.; Santana, L.N.S.; Silva-Junior, A.F.; Silva, M.C.F.; Maia, C.S.F.; Lima, R.R. Evaluation of the Effects of Chronic Intoxication with Inorganic Mercury on Memory and Motor Control in Rats. Int. J. Environ. Res. Public Health 2014, 11, 9171-9185. https://doi.org/10.3390/ijerph110909171
Teixeira FB, Fernandes RM, Farias-Junior PMA, Costa NMM, Fernandes LMP, Santana LNS, Silva-Junior AF, Silva MCF, Maia CSF, Lima RR. Evaluation of the Effects of Chronic Intoxication with Inorganic Mercury on Memory and Motor Control in Rats. International Journal of Environmental Research and Public Health. 2014; 11(9):9171-9185. https://doi.org/10.3390/ijerph110909171
Chicago/Turabian StyleTeixeira, Francisco B., Rafael M. Fernandes, Paulo M. A. Farias-Junior, Natacha M. M. Costa, Luanna M. P. Fernandes, Luana N. S. Santana, Ademir F. Silva-Junior, Marcia C. F. Silva, Cristiane S. F. Maia, and Rafael R. Lima. 2014. "Evaluation of the Effects of Chronic Intoxication with Inorganic Mercury on Memory and Motor Control in Rats" International Journal of Environmental Research and Public Health 11, no. 9: 9171-9185. https://doi.org/10.3390/ijerph110909171






