Evaluation of Models Describing the Growth of Nalidixic Acid-Resistant E. coli O157:H7 in Blanched Spinach and Iceberg Lettuce as a Function of Temperature
Abstract
:1. Introduction
2. Experimental Section
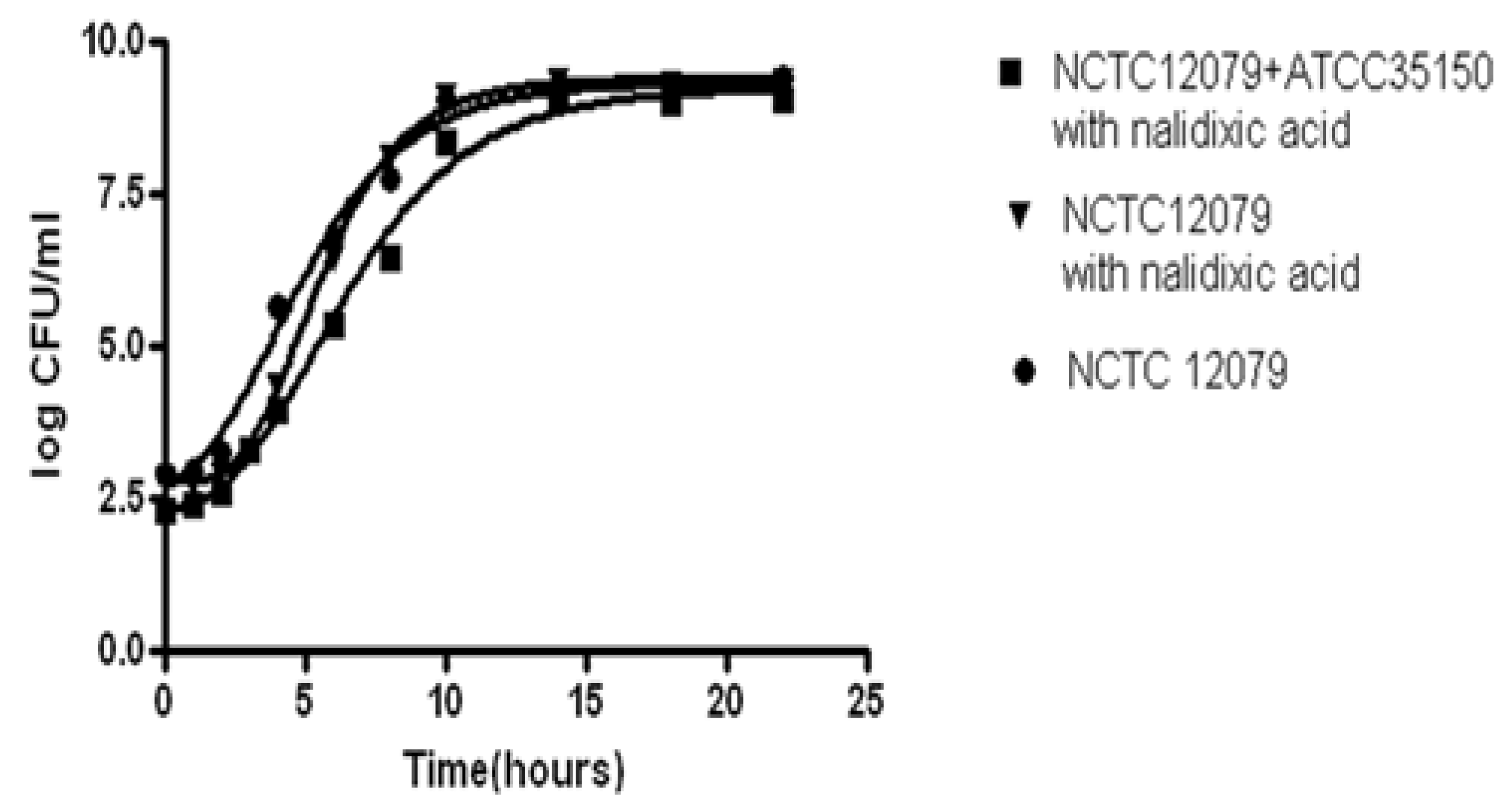
2.1. Bacterial Strains
2.2. Preparation of Inocula for the Broth Model
2.3. Preparation and Inoculation of Blanched Spinach and Fresh-Cut Iceberg Lettuce
2.4. Experimental Designs
2.5. Primary Modeling
2.6. Secondary Modeling
2.7. Performance Evaluation of Blanched Spinach and Fresh-Cut Iceberg Lettuce Models
3. Results and Discussion
3.1. Development of a Predictive Growth Model for E. coli O157:H7NR in Blanched Spinach and Fresh-Cut Iceberg Lettuce as a Function of Temperature
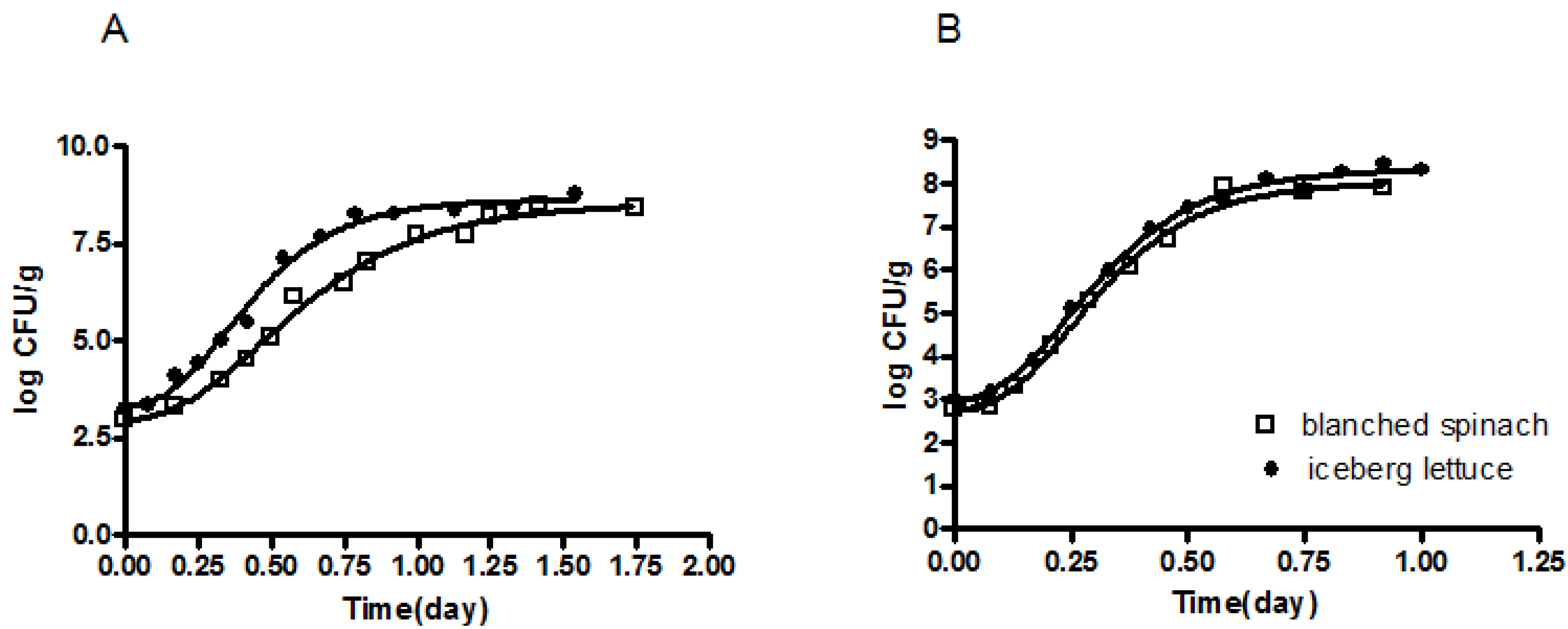
| Temperature (°C) | LT x | SGR y | MPD z | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Spinach | Lettuce | Spinach | Lettuce | Spinach | Lettuce | ||||||||
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | ||
| 7 | 63.36 | 1.67 | 52.8 | 0.80 | 0.004 | 0.00 | 0.004 | 0.04 | 7.20 | 0.02 | 7.14 | 0.06 | |
| 10 | 28.32 | 0.26 | 40.08 | 0.06 | 0.005 | 0.00 | 0.005 | 0.03 | 7.31 | 0.25 | 7.23 | 0.20 | |
| 17 | 10.80 | 0.87 | 8.88 | 0.09 | 0.006 | 0.00 | 0.008 | 0.00 | 7.81 | 0.03 | 8.04 | 0.02 | |
| 24 | 3.60 | 0.13 | 4.28 | 0.21 | 0.019 | 0.01 | 0.015 | 0.05 | 8.06 | 0.04 | 8.18 | 0.08 | |
| 30 | 2.64 | 0.05 | 2.64 | 0.34 | 0.049 | 0.00 | 0.051 | 0.00 | 8.04 | 0.03 | 8.27 | 0.13 | |
| 36 | 2.40 | 0.19 | 2.40 | 0.02 | 0.110 | 0.02 | 0.092 | 0.02 | 8.80 | 0.14 | 8.41 | 0.17 | |

3.2. Evaluation of Model Performance of Secondary Spinach Models for Interpolation and Extrapolation (Model Extension)
| Dataset | Model | Bf a | MRE b | Af c | MARE d | %RE e |
|---|---|---|---|---|---|---|
| Spinach | LT f | 1.01 | 0.01 | 1.16 | 0.14 | 90 |
| SGR g | 0.89 | −0.13 | 1.15 | 0.12 | 100 | |
| MPD h | 0.97 | −0.02 | 1.04 | 0.03 | 100 | |
| Lettuce | LT i | 0.95 | 0.01 | 1.13 | 0.13 | 100 |
| SGR j | 0.96 | −0.02 | 1.09 | 0.08 | 100 | |
| MPD k | 1.00 | 0.00 | 1.02 | 0.02 | 100 |

| Dataset | Model | Bf a | MRE b | Af c | MARE d | % RE e |
|---|---|---|---|---|---|---|
| Lettuce | LT f | 1.01 | 0.03 | 1.12 | 0.12 | 100 |
| SGR g | 0.95 | −3.04 | 1.09 | 0.08 | 100 | |
| Broth | LT | 0.77 | −0.37 | 1.40 | 0.43 | 33 |
| SGR | 0.69 | −24.68 | 1.51 | 0.31 | 50 |
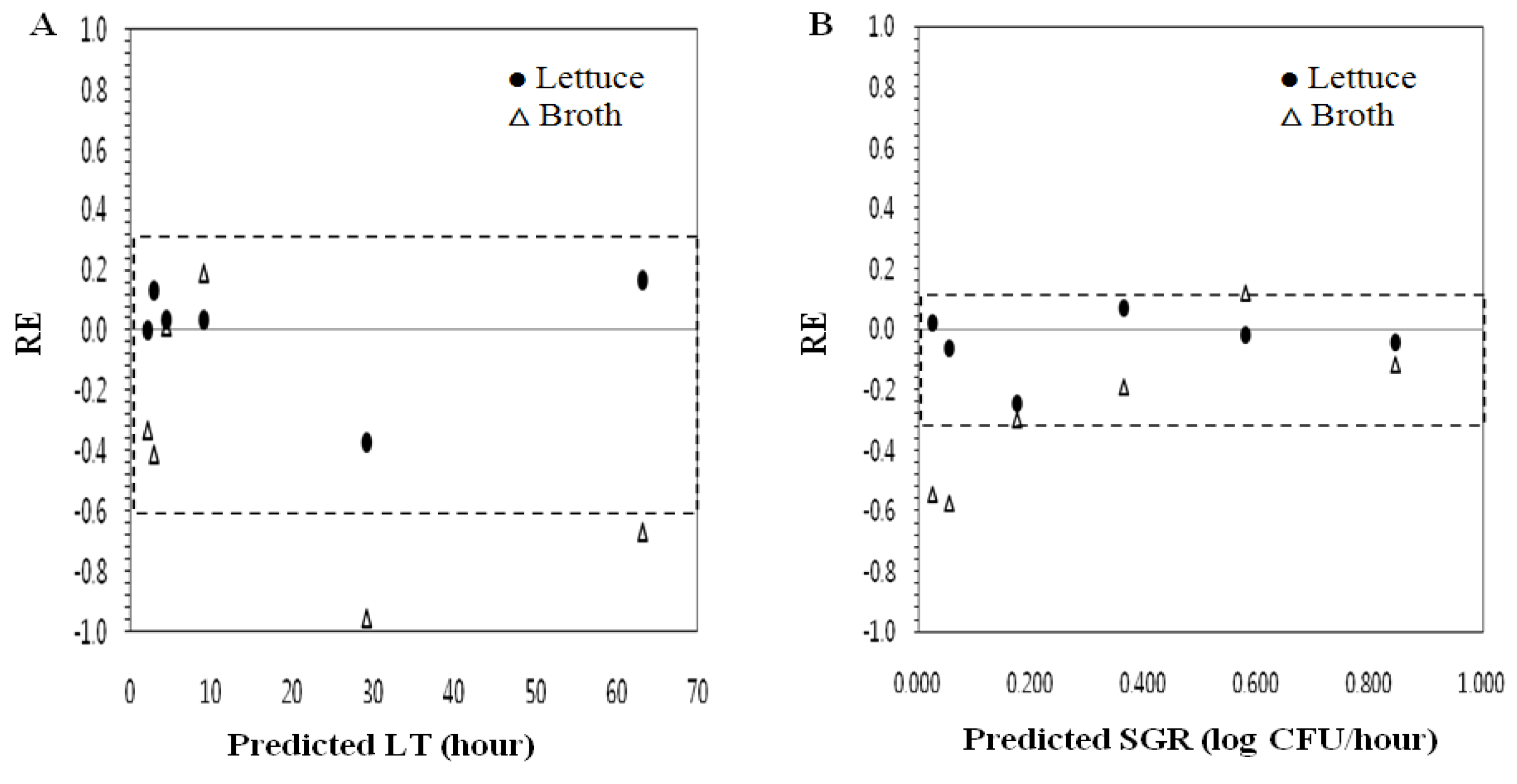
4. Conclusions
Acknowledgments
Conflict of Interest
References
- USDA Microbiological Data Program-Progress Update and 2009 Data Summary. Available online: http://www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELPRDC5088761 (accessed on 27 May 2013).
- Berger, C.N.; Sodha, S.V.; Shaw, R.K.; Griffin, P.M.; Pink, D.; Hand, P.; Frankel, G. Fresh fruit and vegetables as vehicles for the transmission of human pathogens. Environ. Microbiol. 2010, 12, 2385–2397. [Google Scholar] [CrossRef]
- CDC Investigation Update: Multistate Outbreak of E. coli O157:H7 Infections Linked to Romaine Lettuce. Available online: http://www.cdc.gov/ecoli/2011/ecoliO157/romainelettuce/032312/index.html (accessed on 23 March 2013).
- Harrington, R. Germany Finally Confirms Source of Deadly E. coli Outbreak. Available online: http://www.foodproductiondaily.com/Quality-Safety/Germany-finally-confirms-source-of-deadly-E.coli-outbreak (accessed on 13 June 2013).
- CDC Multistate Outbreak of Shiga Toxin-producing Escherichia coli O157:H7 Infections Linked to Organic Spinach and Spring Mix Blend. Available online: http://www.cdc.gov/ecoli/2012/O157H7-11-12/index.html (accessed on 10 December 2012).
- Kozak, G.K.; MacDonald, D.; Landry, L.; Farber, J.M. Foodborne outbreaks in Canada linked to produce: 2001 through 2009. J. Food Prot. 2013, 76, 173–183. [Google Scholar]
- Ross, T.; Ratkowsky, D.A.; Mellefont, L.A.; McMeekin, T.A. Modelling the effects of temperature, water activity, pH and lactic acid concentration on the growth rate of Escherichia coli. Int. J. Food Microbiol. 2003, 82, 33–43. [Google Scholar] [CrossRef]
- Presser, K.A.; Ratkowsky, D.A.; Ross, T. Modelling the growth rate of Escherichia coli as a function of pH and lactic acid concentration. Appl. Environ. Microbiol. 1997, 63, 2355–2360. [Google Scholar]
- Sutherland, J.P.; Bayliss, A.J.; Braxton, D.S.; Beaumont, A.L. Predictive modelling of Escherichia coli O157:H7: Inclusion of carbon dioxide as a fourth factor in a pre-existing model. Int. J. Food Microbiol. 1997, 37, 113–120. [Google Scholar] [CrossRef]
- Kovarova, K.; Zehnder, A.J.; Egli, T. Temperature-dependent growth kinetics of Escherichia coli ML 30 in glucose-limited continuous culture. J. Bacteriol. 1996, 178, 4530–4539. [Google Scholar]
- Sutherland, J.P.; Bayliss, A.J.; Braxton, D.S. Predictive modelling of growth of Escherichia coli O157:H7: The effects of temperature, pH and sodium chloride. Int. J. Food Microbiol. 1995, 25, 29–49. [Google Scholar] [CrossRef]
- Buchanan, R.L.; Bagi, L.K.; Goins, R.V.; Phillips, J.G. Response surface models for the growth kinetics of Escherichia coli O157:H7. Food Microbiol. 1993, 10, 303–315. [Google Scholar] [CrossRef]
- Koseki, S.; Isobe, S. Prediction of pathogen growth on iceberg lettuce under real temperature history during distribution from farm to table. Int. J. Food Microbiol. 2005, 104, 239–248. [Google Scholar] [CrossRef]
- McKellar, R.C.; Delaquis, P. Development of a dynamic growth-death model for Escherichia coli O157:H7 in minimally processed leafy green vegetables. Int. J. Food Microbiol. 2011, 151, 7–14. [Google Scholar] [CrossRef]
- Oscar, T.P. Development and validation of primary, secondary, and tertiary models for growth of Salmonella typhimurium on sterile chicken. J. Food Prot. 2005, 68, 2606–2613. [Google Scholar]
- Ross, T. Indices for performance evaluation of predictive models in food microbiology. J. Appl. Bacteriol. 1996, 81, 501–508. [Google Scholar]
- Oscar, T.P. Validation of lag time and growth rate models for Salmonella typhimurium: Acceptable prediction zone method. Food Microbiol. Saf. 2005, 70, M129–M137. [Google Scholar]
- Inatsu, Y.; Bari, M.L.; Kawasaki, S.; Isshiki, K.; Kawamoto, S. Efficacy of acidified sodium chlorite treatments in reducing Escherichia coli O157:H7 on Chinese cabbage. J. Food Prot. 2005, 68, 251–255. [Google Scholar]
- Yoon, K.S.; Burnette, C.N.; Abou-Zeid, K.A.; Whiting, R.C. Control of growth and survival of Listeria monocytogenes on smoked salmon by combined potassium lactate and sodium diacetate and freezing stress during refrigeration and frozen storage. J. Food Prot. 2004, 67, 2465–2471. [Google Scholar]
- Daughtry, B.J.; Davey, K.R.; King, K.D. Temperature dependence of growth kinetics of food bacteria. Food Microbiol. 1997, 14, 21–30. [Google Scholar] [CrossRef]
- Oscar, T.P. Development and validation of a tertiary simulation model for predicting the potential growth of Salmonella typhimurium on cooked chicken. Int. J. Food Microbiol. 2002, 76, 177–190. [Google Scholar] [CrossRef]
- Ratkowsky, D.A.; Olley, J.; McMeekin, T.A.; Ball, A. Relationship between temperature and growth rate of bacterial cultures. J. Bacteriol. 1982, 149, 1–5. [Google Scholar]
- McMeekin, T.A.; Olley, J.; Ross, T. Predictive Microbiology: Theory and Application; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 1993. [Google Scholar]
- Abou-Zeid, K.A.; Oscar, T.P.; Schwarz, J.G.; Hashem, F.M.; Whiting, R.C.; Yoon, K. Development and validation of a predictive model for Listeria monocytogenes Scott A as a function of temperature, pH, and commercial mixture of potassium lactate and sodium diacetate. J. Microbiol. Biotechnol. 2009, 19, 718–726. [Google Scholar]
- Delignette-Muller, M.L.; Rosso, L.; Flandrois, J.P. Accuracy of microbial growth predictions with square root and polynomial models. Int. J. Food Microbiol. 1995, 27, 139–146. [Google Scholar] [CrossRef]
- Bharathi, S.; Ramesh, M.N.; Varadaraj, M.C. Predicting the behavioural pattern of Escherichia coli in minimally processed vegetables. Food Control 2001, 12, 275–284. [Google Scholar] [CrossRef]
- Khalil, R.K.; Frank, J.F. Behavior of Escherichia coli O157:H7 on damaged leaves of spinach, lettuce, cilantro, and parsley stored at abusive temperatures. J. Food Prot. 2010, 73, 212–220. [Google Scholar]
- Palumbo, S.A.; Call, J.E.; Schultz, F.J.; Williams, A.C. Minimum and maximum temperatures for growth and verotoxin production by hemorrhagic strains of Escherichia coli. J. Food Prot. 1995, 58, 352–356. [Google Scholar]
- Rosso, L.; Lobry, J.R.; Flandrois, J.P. An unexpected correlation between cardinal temperatures of microbial growth highlighted by a new model. J. Theor. Biol. 1993, 162, 447–463. [Google Scholar] [CrossRef]
- Tamplin, M.L. Growth of Escherichia coli O157:H7 in raw ground beef stored at 10 °C and the influence of competitive bacterial flora, strain variation, and fat level. J. Food Prot. 2002, 65, 1535–1540. [Google Scholar]
- Ross, T. Predictive Food Microbiology Models in the Meat Industry; Meat and Livestock Australia (MLA): North Sydney, Australia, 1999; p. 196. [Google Scholar]
- Walls, I.; Scott, V.N. Validation of predictive mathematical models describing the growth of Escherichia coli O157:H7 in raw ground beef. J. Food Prot. 1996, 59, 1331–1335. [Google Scholar]
- Rasch, M. Experimental Design and Data Collection. Modelling Microbial Responses in Food; CRC Press: London, UK, 2004. [Google Scholar]
- Salter, M.A.; Ross, T.; McMeekin, T.A. McMeekin, Applicability of a model for non-pathogenic Escherichia coli for predicting the growth of pathogenic Escherichia coli. J. Appl. Microbiol. 1998, 85, 357–364. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kim, J.; Chung, H.; Cho, J.; Yoon, K. Evaluation of Models Describing the Growth of Nalidixic Acid-Resistant E. coli O157:H7 in Blanched Spinach and Iceberg Lettuce as a Function of Temperature. Int. J. Environ. Res. Public Health 2013, 10, 2857-2870. https://doi.org/10.3390/ijerph10072857
Kim J, Chung H, Cho J, Yoon K. Evaluation of Models Describing the Growth of Nalidixic Acid-Resistant E. coli O157:H7 in Blanched Spinach and Iceberg Lettuce as a Function of Temperature. International Journal of Environmental Research and Public Health. 2013; 10(7):2857-2870. https://doi.org/10.3390/ijerph10072857
Chicago/Turabian StyleKim, Juhui, Hyunjung Chung, Joonil Cho, and Kisun Yoon. 2013. "Evaluation of Models Describing the Growth of Nalidixic Acid-Resistant E. coli O157:H7 in Blanched Spinach and Iceberg Lettuce as a Function of Temperature" International Journal of Environmental Research and Public Health 10, no. 7: 2857-2870. https://doi.org/10.3390/ijerph10072857





