Conotoxin Gene Superfamilies
Abstract
:1. Introduction
2. A-Superfamily
| Sequence | Activity | Reference | |
|---|---|---|---|
| GIA | ECCNPACGRHYSCGK | inhibits neuromuscular nAChR | [12] |
| Vc1.1 | GCCSDPRCNYDHPEIC * | inhibits neuronal nAChR (α9α10) and GABAB GPCR | [29] |
| TIA | FNWRCCLIPACRRNHKKFC * | α1-adrenoceptor modulator | [25] |
| SII | GCCCNPACGPNYGCGTSCS | inhibits neuromuscular nAChR | [22] |
| PIVA | GCCGSYONAACHOCSCKDROSYCGQ * | inhibits neuromuscular nAChR | [17] |
| SIVA | ZKSLVPSVITTCCGYDOGTMCOOCRCTNSC * | K+ channel blocker | [15] |
| CcTx # | AOWLVPSQITTCCGYNOGTMCOSCMCTNTC | activates neuronal VGSCs | [16] |
| EIVA | GCCGPYONAACHOCGCKVGROOYCDROSGG * | inhibits foetal and adult neuromuscular nAChRs | [18] |
| OIVA | CCGVONAACHOCVCKNTC * | selectively inhibits foetal neuromuscular nAChRs | [18] |
| OIVB | CCGVONAACPOCVCNKTCG * | selectively inhibits foetal neuromuscular nAChR | [18] |
| PIVE # | DCCGVKLEMCHPCLCDNSCKNYGK * | excitatory activity upon injection into fish | [21] |
| Pu14.1 | VLEKDCPPHPVPGMHKCVCLKTC | inhibits neuronal and neuromuscular nAChRs | [24] |
3. B/Conantokin-Superfamily
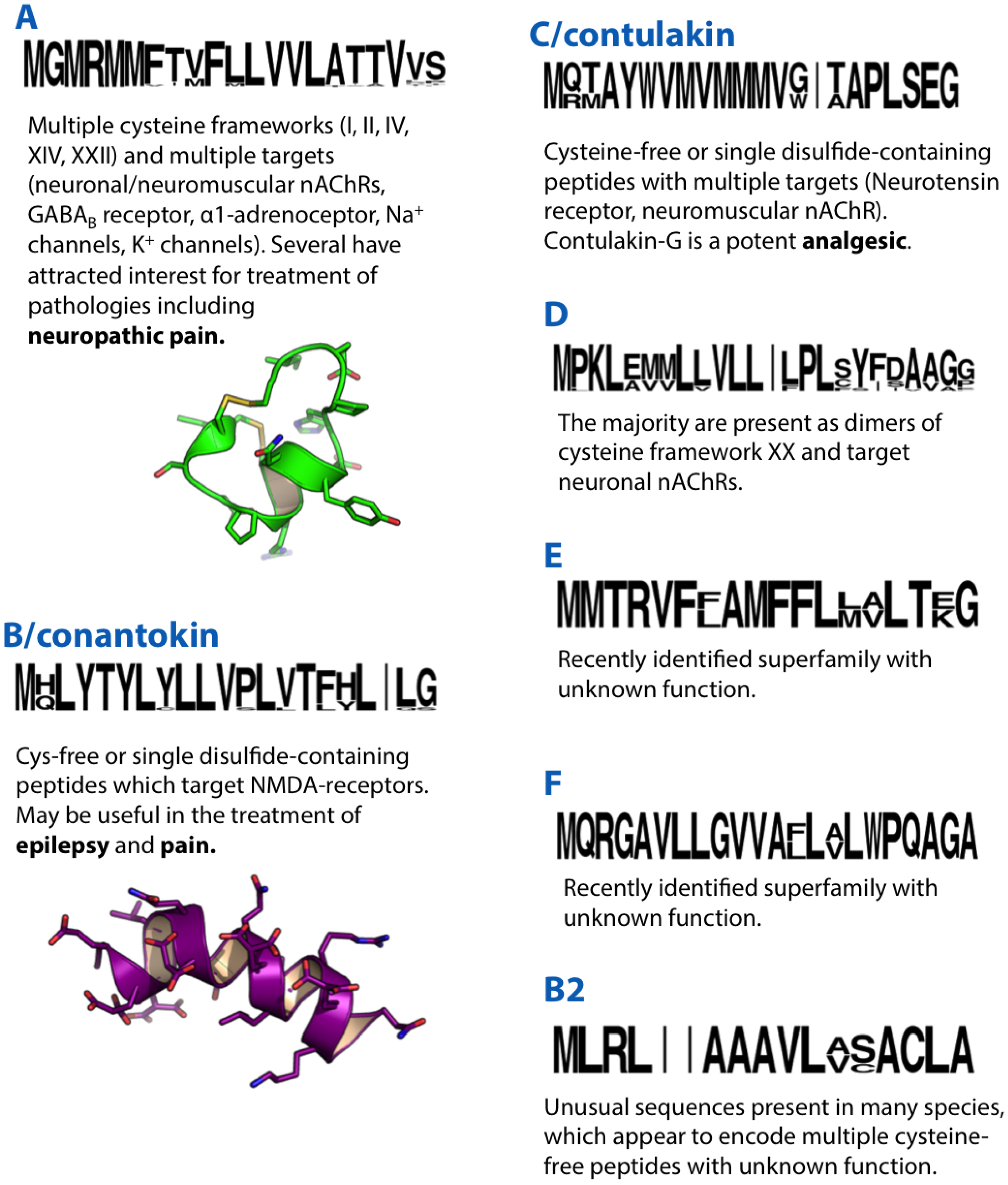
4. B2-Superfamily
5. B3-Superfamily
6. C/Contulakin-Superfamily
| Sequence | Activity | Reference | |
|---|---|---|---|
| Contulakin-G | ZSEEGGSNATKKPYIL | targets neurotensin receptor | [48] |
| αC-PrXA | TYGIYDAKPOFSCAGLRGGCVLPONLROKFKE * | inhibitor of neuromuscular nAChRs | [50] |
7. D-Superfamily
8. E-Superfamily
9. F-Superfamily
10. G-Superfamily
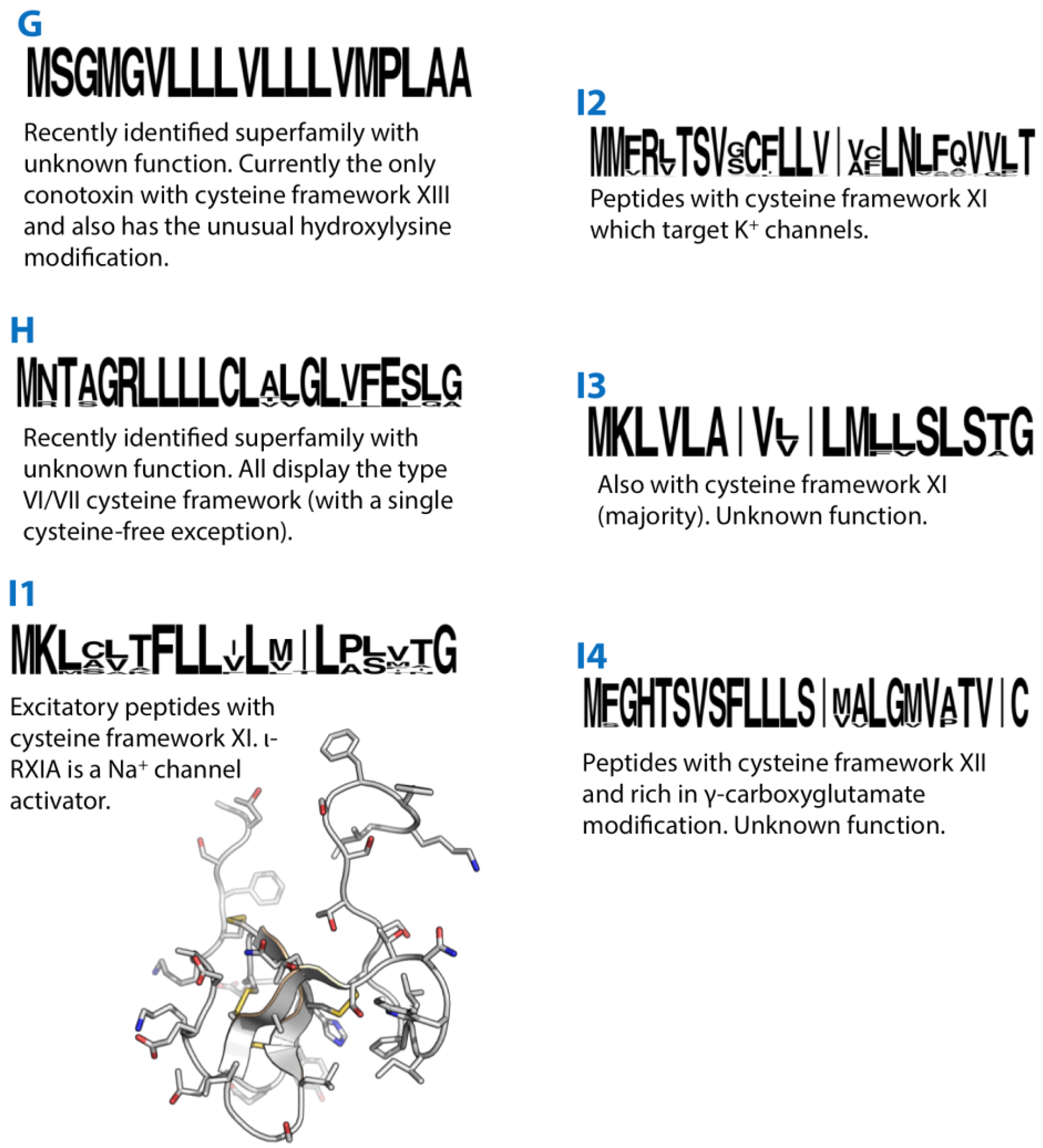
11. H-Superfamily
12. I1-Superfamily
13. I2-Superfamily
14. I3-Superfamily
15. I4-Superfamily
16. J-Superfamily
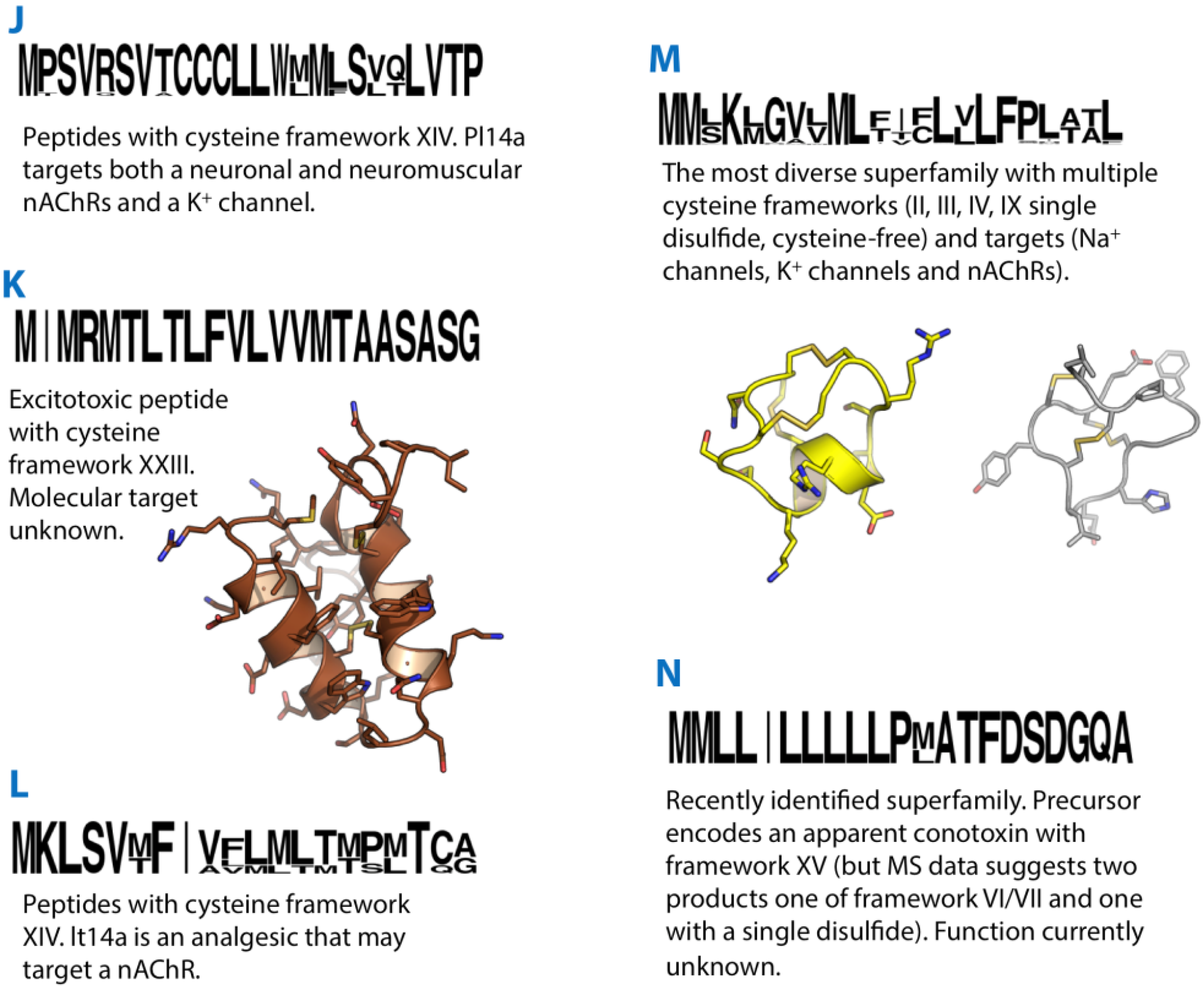
17. K-Superfamily
18. L-Superfamily
19. M/Conomarphin/CPY-Superfamily
20. N-Superfamily
| Sequence | Activity | Reference | |
|---|---|---|---|
| M4/5 | |||
| μ-KIIIA | CCNCSSKWCRDHSRCC * | blocks neuronal VGSCs | [100] |
| κ-RIIIK | LOSCCSLNLRLCOVOACKRNOCCT * | selectively blocks the KV1.2 subtype of voltage-gated K+ channels | [78] |
| ψ-PIIIE | HOOCCLYGKCRRYOGCSSASCCQR * | inhibits neuromuscular nAChRs | [82] |
| M1 | |||
| mr3e | VCCPFGGCHELCYCCD * | No effect in mice (IC injection). | [84] |
| tx3a | CCSWDVCDHPSCTCCG | excitatory behaviour in mice (hyperactivity at low doses, seizures and death at higher doses) (IC injection) | [86] |
| LtIIIA | DγCCγOQWCDGACDCCS | enhances TTX-sensitive Na+ currents | [89] |
| M2 | |||
| mr3a | GCCGSFACRFGCVOCCV | All M2-conotoxins tested show excitatory behaviour in mice (IC injection) | [90] |
| mr3b | SKQCCHLAACRFGCTOCCW | ||
| tx3b | CCPPVACNMGCKPCC * | ||
| tx3c | CCRTCFGCTOCC * | ||
| PnIVB | CCKYGWTCWLGCSPCGC | Blocks molluscan TTX-resistant Na+ channels | [95] |
| VxII | WIDPSHYCCCGGGCTDDCVNC | sedative effects, tail-stiffening and twisted jumping in mice (IC injection) | [97] |
| CPY-Pl1 | ARFLHPFQYYTLYRYLTRFLHRYPIYYIRY | Subtype-selective inhibitors of K+ channels | [99] |
| CPY-Fe1 | GTYLYPFSYYRLWRYFTRFLHKQPYYYVHI |
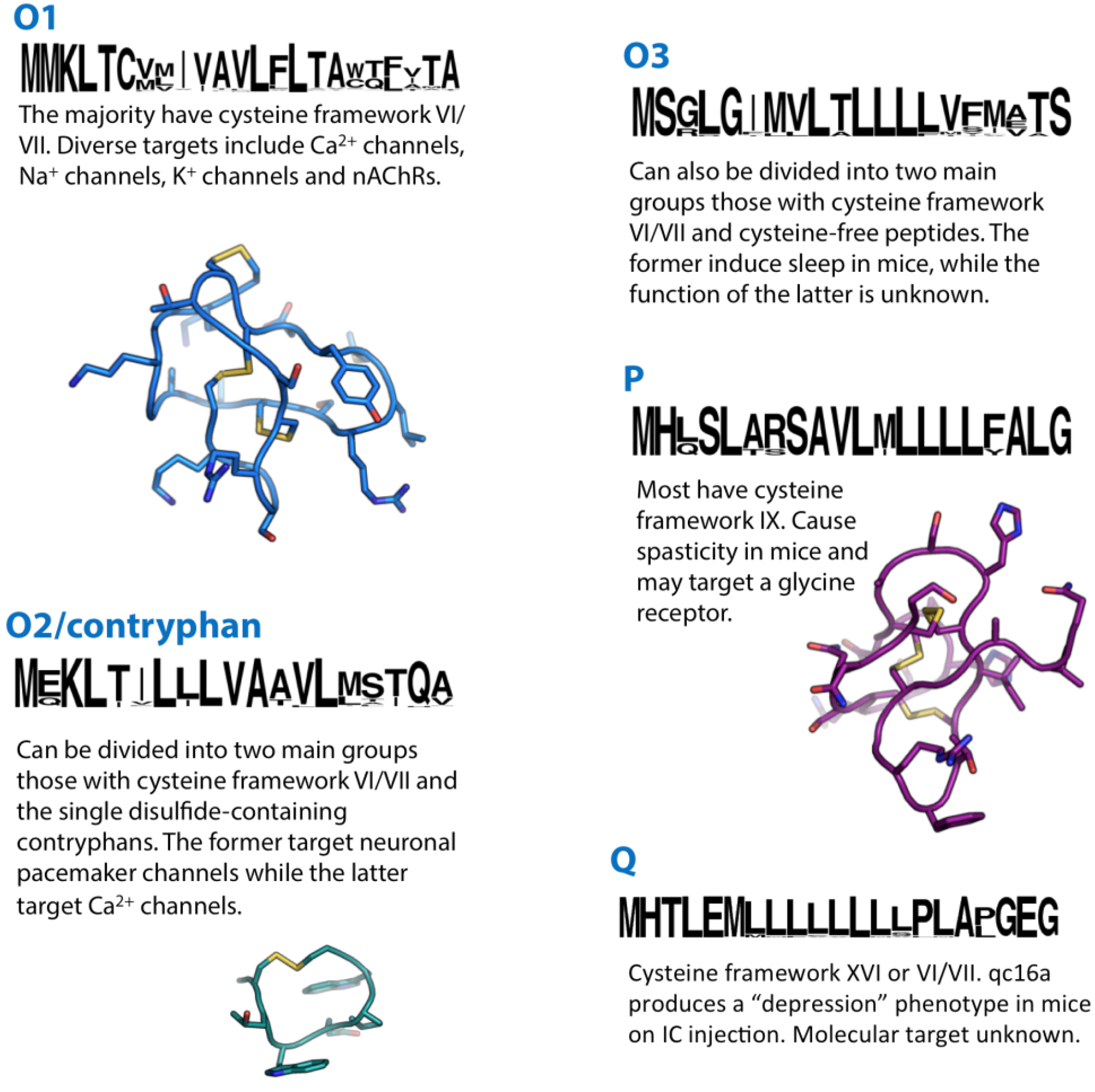
21. O1-Superfamily
| Sequence | Activity | Reference | |
|---|---|---|---|
| ω-MVIIA | CKGKGAKCSRLMYDCCTGSCRSGKC* | CaV2.2 VGCC blocker | [114] |
| μ-Conotoxin-GS | ACSGRGSRCOOQCCMGLRCGRGNPQKCIGAHγDV | VGSC inhibitor (site I) | [110] |
| δ-PVIA | EACYAOGTFCGIKOGLCCSEFCLPGVCFG* | blocks inactivation of VGSCs | [115] |
| κ-PVIIA | CRIONQKCFQHLDDCCSRKCNRFNKCV* | voltage-gated K+ channel blocker | [116] |
| μ-MrVIB | ACSKKWEYCIVPILGFVYCCPGLICGPFVCV | selective blocker of NaV1.8 subtype of VGSC | [117] |
| μ-GVIIJ | GWCDOGATCGKLRLYCCSGFCD§YTKTCKDKSSA | VGSC inhibitor (site 8) | [113] |
22. O2/Contryphan-Superfamily
| Sequence | Activity | Reference | |
|---|---|---|---|
| TxVIIA | CGGYSTYCγVDSγCCSDNCVRSYCTLF* | Targets pacemaker channels in molluscan neurons. No effect in rats (IC injection). | [118,121] |
| PnVIIA | DCTSWFGRCTVNSγCCSNSCDQTYCγLYAFOS | Targets pacemaker channels in molluscan neurons (distinct, however, from TxVIIA). No effect in fish or fly larvae. | [119] |
| as7a | TCKQKGEGCSLDVγCCSSSCKPGGPLFDFDC | Toxic effects on mollusc after IM injection. No effect in mice (IC injection). | [120] |
| Contryphan- | Sequence | Activity | Reference |
|---|---|---|---|
| R/Tx | GCOwEPWC * | “stiff-tail” syndrome in mice (IC injection). | [122,126] |
| Des[Gly1]-R | COwEPWC * | “stiff-tail” syndrome in mice (IC injection). | [122] |
| Bromo-R | GCOwEPWC * | “stiff-tail” syndrome in mice (IC injection). | [127] |
| Sm | GCOwQPWC * | “stiff-tail” syndrome in mice (IC injection). | [128] |
| Leu-P | GCVlLPWC | Body tremor and mucous secretion on fish (IM injection). Less activity than other contryphans in mice. | [129] |
| Tx | GCOwQPYC * | “stiff-tail” syndrome and paralysis of extremities in mice (IC injection). | [126] |
| Leu-Tx | CVlYPWC * | Causes folding and drooping of dorsal fins in fish (IM injection). Less activity than other contryphans in mice. | [126] |
| Vn | GDCPwKPWC * | Voltage-gated and Ca2+-activated K+ channel modulator. Mucous secretion in fish (IM injection). | [123] |
| M | NγSγCPwHPWC * | Ca2+-dependent block of L-type Ca2+ channels (mouse pancreatic β-cells). | [124] |
| P/Am975 | GCOwDPWC * | “stiff-tail” syndrome in mice (IC injection). HVA Ca2+ channel blocker (DRG neurons). | [125] |
| Lo959 | GCPwDPWC * | HVA Ca2+ channel activator (DRG neurons). | [125] |
23. O3-Superfamily
24. P-Superfamily
| Sequence | Activity | Reference | |
|---|---|---|---|
| TxIXA | GCNNSCQγHSDCγSHCICTFRGCGAVN * | Hyperactivity and spasticity in mice (IC injection). No effect in fish. May target a glycine receptor. | [135] |
| Gm9a | SCNNSCQSHSDCASHCICTFRGCGAVN * | Hyperactivity and spasticity in mice (IC injection). May target a glycine receptor. | [136] |
25. Q-Superfamily
26. S-Superfamily
| Sequence | Activity | Reference | |
|---|---|---|---|
| GVIIIA | GCTRTCGGOKCTGTCTCTNSSKCGCRYNVHPSGWGCGCACS * | Competitive inhibition of the 5-HT3 receptor. | [138] |
| RVIIIA | KCNFDKCKGTGVYNCGγSCSCγGLHSCRCTYNIGSMKSGCACICTYY | nAChR inhibition. | [139] |

27. T-Superfamily
28. U-Superfamily
| Sequence | Activity | Reference | |
|---|---|---|---|
| TxVA | γCCγDGWCCTAAO | Hyperactivity and spasticity in mice (IC injection). Suppression of gill display in fish. Reduced pre-synaptic Ca2+ influx at the Aplysia cholinergic synapse. May target a pre-synaptic Ca2+ channel or GPCR. | [146,148] |
| PVA | GCCPKQMRCCTL * | No effect in mice (IC injection). Suppression of gill display in fish. | [146] |
| AuVA | FCCPFIRYCCW | No effect in mice (IC injection). Suppression of gill display in fish. | [146] |
| SrVA | IINWCCLIFYQCC | Depressed behavioural activity in mice (IC injection). | [147] |
| LtVD | DCCPAKLLCCNP | Inhibition of TTX-sensitive Na+ currents. | [149] |
| MrIA | NGVCCGYKLCHOC | Akinesia and seizures in mice (IC injection). Non-competitive inhibition of the norepinephrine transporter. | [25,143,144] |
| MrIB | VGVCCGYKLCHOC | Non-competitive inhibition of the norepinephrine transporter. | [25] |
| CMrX | GICCGVSFCYOC | Paralysis in mice (IC injection). | [144] |
| τ-CnVA | ECCHRQLLCCLRFV * | somatostatin-3 receptor antagonist | [150] |
| LiC32 | LWQNTWCCRDHLRCC * | somatostatin-3 receptor antagonist | [150] |
29. V-Superfamily
30. Y-Superfamily
31. Con-Ikot-Ikots
32. ConoCAPs
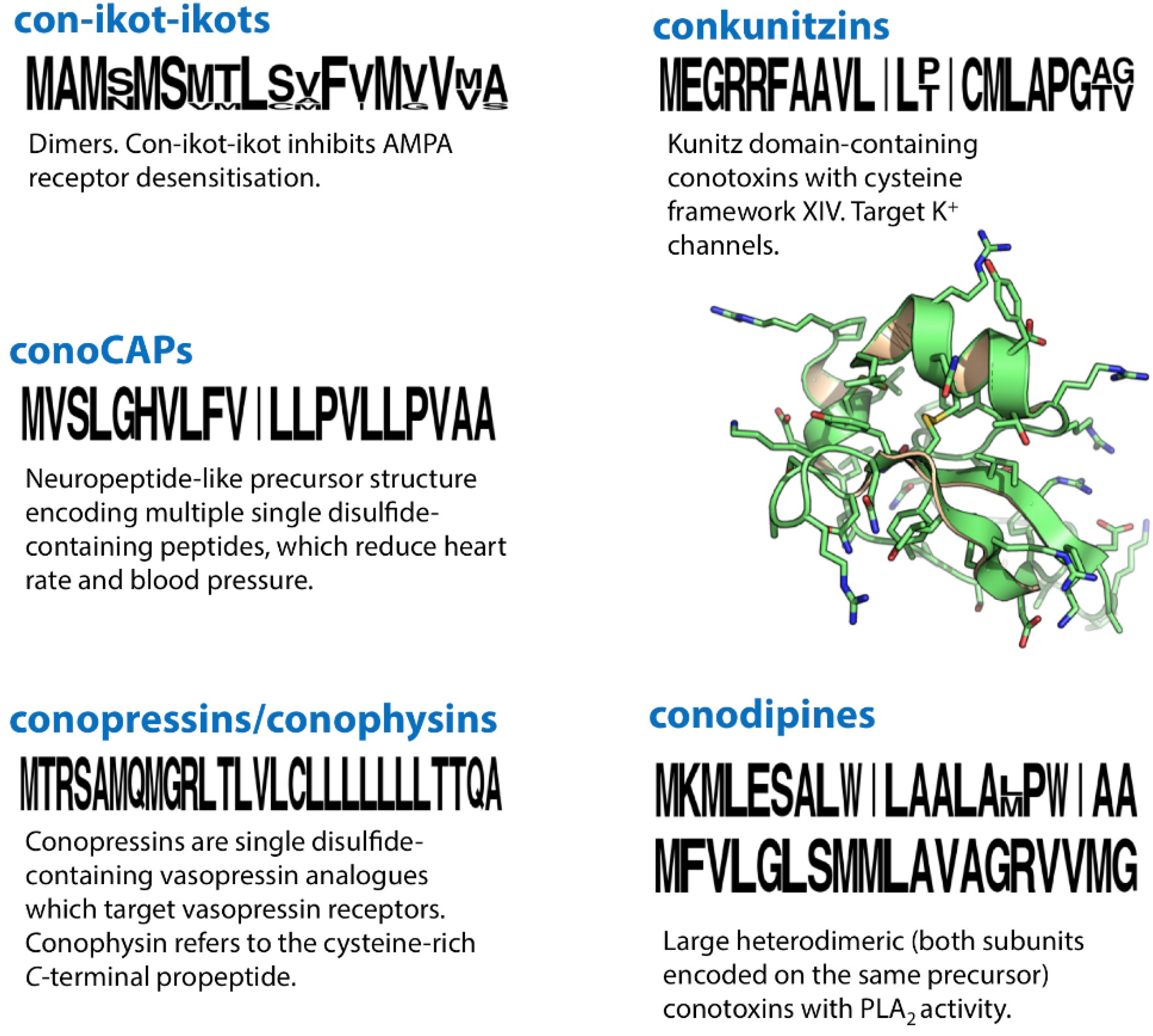
33. Conopressins/Conophysins
34. Conkunitzins
35. Conodipines
36. Unassigned Superfamilies—C. miles
37. “Divergent” Superfamilies—C. californicus
38. Conoproteins
39. Unclassified
39.1. Conolysins
39.2. Conophans
39.3. Conomap
39.4. Conorfamides
39.5. Bromoheptapeptide
39.6. as25a
39.7. RsXXIVA
39.8. vil14a
40. Concluding Remarks
| Gene Superfamily | Cysteine Frameworks | Pharmacological Family (Where Defined) |
|---|---|---|
| A | I, II, IV, VI/VII, XIV, XXII | α,κ,ρ, other |
| B | other | other |
| B2 | other | |
| B3 | XXIV | α |
| C | V, XV, other | α, other |
| D | XV, X | α |
| E | XXII | |
| F | other | |
| G | XIII | |
| H | VI/VII | |
| I1 | VI/VII, XI | ι |
| I2 | XI, XIV | κ |
| I3 | VI/VII, XI | |
| I4 | XII | |
| J | XIV | α, κ |
| K | XXIII | |
| L | XIV | α |
| M | I, II, III, IV, VI/VII, IX, XIV, XVI | α, ι, κ, μ |
| N | XV | |
| O1 | I, VI/VII, IX, XII, XIV, XVI | δ, γ, κ, μ, ω |
| O2 | VI/VII, XIV, XV | γ, other |
| O3 | VI/VII, other | |
| P | IX, XIV | |
| Q | XVI, VI/VII | |
| S | VIII | α, σ |
| T | I, V, X, XVI | χ, μ, τ |
| U | VI/VII | |
| V | XV, VI/VII | |
| Y | XVII | |
| con-ikot-ikot | XXI, other | other |
| conoCAP | other | |
| conopressin | other | other |
| conkunitzin | XIV | κ |
| conodipine | other | other |
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lewis, R.J.; Dutertre, S.; Vetter, I.; Christie, M.J. Conus venom peptide pharmacology. Pharmacol. Rev. 2012, 64, 259–298. [Google Scholar] [PubMed]
- Norton, R.S.; Olivera, B.M. Conotoxins down under. Toxicon 2006, 48, 780–798. [Google Scholar] [CrossRef] [PubMed]
- Puillandre, N.; Koua, D.; Favreau, P.; Olivera, B.; Stöcklin, R. Molecular phylogeny, classification and evolution of conopeptides. J. Mol. Evol. 2012, 74, 297–309. [Google Scholar] [CrossRef] [PubMed]
- Woodward, S.R.; Cruz, L.J.; Olivera, B.M.; Hillyard, D.R. Constant and hypervariable regions in conotoxin propeptides. EMBO J. 1990, 9, 1015–1020. [Google Scholar]
- Chang, D.; Duda, T.F. Extensive and continuous duplication facilitates rapid evolution and diversification of gene families. Mol. Biol. Evol. 2012, 29, 2019–2029. [Google Scholar] [CrossRef]
- Duda, T.F.; Palumbi, S.R. Molecular genetics of ecological diversification: Duplication and rapid evolution of toxin genes of the venomous gastropod Conus. Proc. Natl. Acad. Sci. USA 1999, 96, 6820–6823. [Google Scholar] [CrossRef] [PubMed]
- Puillandre, N.; Watkins, M.; Olivera, B. Evolution of Conus peptide genes: Duplication and positive selection in the A-superfamily. J. Mol. Evol. 2010, 70, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Azam, L.; McIntosh, J.M. Alpha-conotoxins as pharmacological probes of nicotinic acetylcholine receptors. Acta Pharmacol. Sin. 2009, 30, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Jacob, R.B.; McDougal, O.M. The M-superfamily of conotoxins: A review. Cell. Mol. Life Sci. 2010, 67, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, S.H.; Leipold, E. Conotoxins of the O-superfamily affecting voltage-gated sodium channels. Cell. Mol. Life Sci. 2007, 64, 1329–1340. [Google Scholar] [CrossRef] [PubMed]
- Kaas, Q.; Westermann, J.C.; Craik, D.J. Conopeptide characterization and classifications: An analysis using ConoServer. Toxicon 2010, 55, 1491–1509. [Google Scholar] [CrossRef] [PubMed]
- Gray, W.R.; Luque, A.; Olivera, B.M.; Barrett, J.; Cruz, L.J. Peptide toxins from Conus geographus venom. J. Biol. Chem. 1981, 256, 4734–4740. [Google Scholar] [PubMed]
- McManus, O.B.; Musick, J.R.; Gonzalez, C. Peptides isolated from the venom of Conus geographus block neuromuscular transmission. Neurosci. Lett. 1981, 25, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.D.; McIntosh, J.M.; Hillyard, D.R.; Cruz, L.J.; Olivera, B.M. The A-superfamily of conotoxins. J. Biol. Chem. 2004, 279, 17596–17606. [Google Scholar] [CrossRef] [PubMed]
- Craig, A.G.; Zafaralla, G.; Cruz, L.J.; Santos, A.D.; Hillyard, D.R.; Dykert, J.; Rivier, J.E.; Gray, W.R.; Imperial, J.; DelaCruz, R.G.; et al. An O-glycosylated neuroexcitatory Conus peptide. Biochemistry 1998, 37, 16019–16025. [Google Scholar] [CrossRef] [PubMed]
- Le Gall, F.; Favreau, P.; Benoit, E.; Mattei, C.; Bouet, F.; Menou, J.-L.; Ménez, A.; Letourneux, Y.; Molgó, J. A new conotoxin isolated from Conus consors venom acting selectively on axons and motor nerve terminals through a Na+-dependent mechanism. Eur. J. Neurosci. 1999, 11, 3134–3142. [Google Scholar] [PubMed]
- Hopkins, C.; Grilley, M.; Miller, C.; Shon, K.-J.; Cruz, L.J.; Gray, W.R.; Dykert, J.; Rivier, J.; Yoshikami, D.; Olivera, B.M. A new family of Conus peptides targeted to the nicotinic acetylcholine receptor. J. Biol. Chem. 1995, 270, 22361–22367. [Google Scholar] [CrossRef] [PubMed]
- Teichert, R.W.; López-Vera, E.; Gulyas, J.; Watkins, M.; Rivier, J.; Olivera, B.M. Definition and characterization of the short αA-conotoxins: A single residue determines dissociation kinetics from the fetal muscle nicotinic acetylcholine receptor. Biochemistry 2006, 45, 1304–1312. [Google Scholar] [CrossRef] [PubMed]
- Teichert, R.W.; Rivier, J.; Dykert, J.; Cervini, L.; Gulyas, J.; Bulaj, G.; Ellison, M.; Olivera, B.M. αA-Conotoxin OIVA defines a new αA-conotoxin subfamily of nicotinic acetylcholine receptor inhibitors. Toxicon 2004, 44, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Teichert, R.W.; Rivier, J.; Torres, J.; Dykert, J.; Miller, C.; Olivera, B.M. A uniquely selective inhibitor of the mammalian fetal neuromuscular nicotinic acetylcholine receptor. J. Neurosci. 2005, 25, 732–736. [Google Scholar] [CrossRef] [PubMed]
- Teichert, R.W.; Jacobsen, R.; Terlau, H.; Yoshikami, D.; Olivera, B.M. Discovery and characterization of the short κA-conotoxins: A novel subfamily of excitatory conotoxins. Toxicon 2007, 49, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Ramilo, C.A.; Zafaralla, G.C.; Nadasdi, L.; Hammerland, L.G.; Yoshikami, D.; Gray, W.R.; Kristipati, R.; Ramachandran, J.; Miljanich, G. Novel α- and ω-conotoxins from Conus striatus venom. Biochemistry 1992, 31, 9919–9926. [Google Scholar] [CrossRef] [PubMed]
- Bai-Song, L.; Fang, Y.; Dong, Z.; Pei-Tang, H.; Cui-Fen, H. Conopeptides from Conus striatus and Conus textile by cDNA cloning. Peptides 1999, 20, 1139–1144. [Google Scholar] [PubMed]
- Peng, C.; Ye, M.; Wang, Y.; Shao, X.; Yuan, D.; Liu, J.; Hawrot, E.; Wang, C.; Chi, C. A new subfamily of conotoxins belonging to the A-superfamily. Peptides 2010, 31, 2009–2016. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, I.A.; Gehrmann, J.; Loughnan, M.L.; Thomas, L.; Adams, D.A.; Atkins, A.; Palant, E.; Craik, D.J.; Adams, D.J.; Alewood, P.F.; et al. Two new classes of conopeptides inhibit the α1-adrenoceptor and noradrenaline transporter. Nat. Neurosci. 2001, 4, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.J.; Callaghan, B.; Berecki, G. Analgesic conotoxins: block and G protein-coupled receptor modulation of N-type (CaV2.2) calcium channels. Br. J. Pharmacol. 2012, 166, 486–500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, S.D.; Safavi-Hemami, H.; McIntosh, L.D.; Purcell, A.W.; Norton, R.S.; Papenfuss, A.T. Diversity of conotoxin gene superfamilies in the venomous snail, Conus victoriae. PLoS One 2014, 9, e87648. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.; Yang, L.; Xu, S.; Wang, C. Various conotoxin diversifications revealed by a venomic study of Conus flavidus. Mol. Cell. Proteomics 2013, 13, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Callaghan, B.; Haythornthwaite, A.; Berecki, G.; Clark, R.J.; Craik, D.J.; Adams, D.J. Analgesic α-conotoxins Vc1.1 and RgIA inhibit N-type calcium channels in rat sensory neurons via GABAB receptor activation. J. Neurosci. 2008, 28, 10943–10951. [Google Scholar] [CrossRef] [PubMed]
- Olivera, B.M.; McIntosh, J.M.; Clark, C.; Middlemas, D.; Gray, W.R.; Cruz, L.J. A sleep-inducing peptide from Conus geographus venom. Toxicon 1985, 23, 277–282. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, J.M.; Olivera, B.M.; Cruz, L.J.; Gray, W.R. γ-carboxyglutamate in a neuroactive toxin. J. Biol. Chem. 1984, 259, 14343–14346. [Google Scholar] [PubMed]
- Skjærbæk, N.; Nielsen, K.J.; Lewis, R.J.; Alewood, P.; Craik, D.J. Determination of the solution structures of conantokin-G and conantokin-T by CD and NMR spectroscopy. J. Biol. Chem. 1997, 272, 2291–2299. [Google Scholar] [CrossRef] [PubMed]
- Rivier, J.; Galyean, R.; Simon, L.; Cruz, L.J.; Olivera, B.M.; Gray, W.R. Total synthesis and further characterization of the γ-carboxyglutamate-containing “sleeper” peptide from Conus geographus venom. Biochemistry 1987, 26, 8508–8512. [Google Scholar] [CrossRef] [PubMed]
- Mena, E.E.; Gullak, M.F.; Pagnozzi, M.J.; Richter, K.E.; Rivier, J.; Cruz, L.J.; Olivera, B.M. Conantokin-G: A novel peptide antagonist to the N-methyl-d-aspartic acid (NMDA) receptor. Neurosci. Lett. 1990, 118, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Dingledine, R.; Borges, K.; Bowie, D.; Traynelis, S.F. The glutamate receptor ion channels. Pharmacol. Rev. 1999, 51, 7–62. [Google Scholar] [PubMed]
- Chatterton, J.E.; Awobuluyi, M.; Premkumar, L.S.; Takahashi, H.; Talantova, M.; Shin, Y.; Cui, J.; Tu, S.; Sevarino, K.A.; Nakanishi, N.; et al. Excitatory glycine receptors containing the NR3 family of NMDA receptor subunits. Nature 2002, 415, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Donevan, S.D.; McCabe, R.T. Conantokin-G Is an NR2B-selective competitive antagonist of N-Methyl-d-aspartate receptors. Mol. Pharmacol. 2000, 58, 614–623. [Google Scholar]
- Teichert, R.W.; Jimenez, E.C.; Twede, V.; Watkins, M.; Hollmann, M.; Bulaj, G.; Olivera, B.M. Novel conantokins from Conus parius venom are specific antagonists of N-methyl-d-aspartate receptors. J. Biol. Chem. 2007, 282, 36905–36913. [Google Scholar] [CrossRef] [PubMed]
- Gowd, K.H.; Twede, V.; Watkins, M.; Krishnan, K.S.; Teichert, R.W.; Bulaj, G.; Olivera, B.M. Conantokin-P, an unusual conantokin with a long disulfide loop. Toxicon 2008, 52, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Twede, V.D.; Teichert, R.W.; Walker, C.S.; Gruszczyński, P.; Kaźmierkiewicz, R.; Bulaj, G.; Olivera, B.M. Conantokin-Br from Conus brettinghami and selectivity determinants for the NR2D subunit of the NMDA receptor. Biochemistry 2009, 48, 4063–4073. [Google Scholar] [CrossRef] [PubMed]
- Gowd, K.H.; Han, T.S.; Twede, V.; Gajewiak, J.; Smith, M.D.; Watkins, M.; Platt, R.J.; Toledo, G.; White, H.S.; Olivera, B.M.; et al. Conantokins derived from the asprella clade impart conRl-B, an N-methyl-d-aspartate receptor antagonist with a unique selectivity profile for NR2B subunits. Biochemistry 2012, 51, 4685–4692. [Google Scholar] [CrossRef] [PubMed]
- Abogadie, F.C.; Cruz, L.J.; Olivera, B.M.; Walker, C.; Colledge, C.; Hillyard, D.R.; Jimenez, E.; Layer, R.T.; Zhou, L.-M.; Shen, G.S.; et al. Conantokins. U.S. Patent 6,515,103, 2003. [Google Scholar]
- Malmberg, A.B.; Gilbert, H.; McCabe, R.T.; Basbaum, A.I. Powerful antinociceptive effects of the cone snail venom-derived subtype-selective NMDA receptor antagonists conantokins G and T. Pain 2003, 101, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, E.C.; Donevan, S.; Walker, C.; Zhou, L.-M.; Nielsen, J.; Cruz, L.J.; Armstrong, H.; White, H.S.; Olivera, B.M. Conantokin-L, a new NMDA receptor antagonist: determinants for anticonvulsant potency. Epilepsy Res. 2002, 51, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Pi, C.; Liu, J.; Peng, C.; Liu, Y.; Jiang, X.; Zhao, Y.; Tang, S.; Wang, L.; Dong, M.; Chen, S.; et al. Diversity and evolution of conotoxins based on gene expression profiling of Conus litteratus. Genomics 2006, 88, 809–819. [Google Scholar] [CrossRef]
- Violette, A.; Biass, D.; Dutertre, S.; Koua, D.; Piquemal, D.; Pierrat, F.; Stöcklin, R.; Favreau, P. Large-scale discovery of conopeptides and conoproteins in the injectable venom of a fish-hunting cone snail using a combined proteomic and transcriptomic approach. J. Proteomics 2012, 75, 5215–5225. [Google Scholar] [PubMed]
- Luo, S.; Christensen, S.; Zhangsun, D.; Wu, Y.; Hu, Y.; Zhu, X.; Chhabra, S.; Norton, R.S.; McIntosh, J.M. A novel inhibitor of α9α10 nicotinic acetylcholine receptors from Conus vexillum delineates a new conotoxin superfamily. PLoS One 2013, 8, e54648. [Google Scholar] [CrossRef] [PubMed]
- Craig, A.G.; Norberg, T.; Griffin, D.; Hoeger, C.; Akhtar, M.; Schmidt, K.; Low, W.; Dykert, J.; Richelson, E.; Navarro, V.; et al. Contulakin-G, an O-glycosylated invertebrate neurotensin. J. Biol. Chem. 1999, 274, 13752–13759. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.W.; Hofer, K.; McCumber, D.; Wagstaff, J.D.; Layer, R.T.; McCabe, R.T.; Yaksh, T.L. An assessment of the antinociceptive efficacy of intrathecal and epidural contulakin-G in rats and dogs. Anesth. Analg. 2007, 104, 1505–1513. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, E.C.; Olivera, B.M.; Teichert, R.W. αC-conotoxin PrXA: A new family of nicotinic acetylcholine receptor antagonists. Biochemistry 2007, 46, 8717–8724. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.M.; Olivera, B.M.; Watkins, M.G.; James, E. B-Superfamily conotoxins. U.S. Patent Appl. WO02060923A1, 2005. [Google Scholar]
- Lluisma, A.O.; Milash, B.A.; Moore, B.; Olivera, B.M.; Bandyopadhyay, P.K. Novel venom peptides from the cone snail Conus pulicarius discovered through next-generation sequencing of its venom duct transcriptome. Mar. Genomics 2012, 5, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Loughnan, M.; Nicke, A.; Jones, A.; Schroeder, C.I.; Nevin, S.T.; Adams, D.J.; Alewood, P.F.; Lewis, R.J. Identification of a novel class of nicotinic receptor antagonists: Dimeric conotoxins VxXIIA, VxXIIB, and VxXIIC from Conus vexillum. J. Biol. Chem. 2006, 281, 24745–24755. [Google Scholar] [CrossRef] [PubMed]
- Loughnan, M.L.; Nicke, A.; Lawrence, N.; Lewis, R.J. Novel αD-conopeptides and their precursors identified by cDNA cloning define the D-conotoxin superfamily. Biochemistry 2009, 48, 3717–3729. [Google Scholar] [CrossRef] [PubMed]
- Dutertre, S.; Jin, A.-h.; Kaas, Q.; Jones, A.; Alewood, P.F.; Lewis, R.J. Deep venomics reveals the mechanism for expanded peptide diversity in cone snail venom. Mol. Cell. Proteomics 2012, 12, 312–329. [Google Scholar] [PubMed]
- Aguilar, M.B.; López-Vera, E.; Ortiz, E.; Becerril, B.; Possani, L.D.; Olivera, B.M.; Heimer de la Cotera, E.P. A Novel conotoxin from Conus delessertii with posttranslationally modified lysine residues. Biochemistry 2005, 44, 11130–11136. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, M.B.; Ortiz, E.; Kaas, Q.; López-Vera, E.; Becerril, B.; Possani, L.D.; de la Cotera, E.P.H. Precursor De13.1 from Conus delessertii defines the novel G gene superfamily. Peptides 2013, 41, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Puillandre, N.; Bouchet, P.; Duda, T.F., Jr.; Kauferstein, S.; Kohn, A.J.; Olivera, B.M.; Watkins, M.; Meyer, C. Molecular phylogeny and evolution of the cone snails (Gastropoda, Conoidea). Mol. Phylogen. Evol. 2014, 78, 290–303. [Google Scholar] [CrossRef]
- Jimenez, E.C.; Shetty, R.P.; Lirazan, M.; Rivier, J.; Walker, C.; Abogadie, F.C.; Yoshikami, D.; Cruz, L.J.; Olivera, B.M. Novel excitatory Conus peptides define a new conotoxin superfamily. J. Neurochem. 2003, 85, 610–621. [Google Scholar] [CrossRef] [PubMed]
- Buczek, O.; Wei, D.; Babon, J.J.; Yang, X.; Fiedler, B.; Chen, P.; Yoshikami, D.; Olivera, B.M.; Bulaj, G.; Norton, R.S. Structure and sodium channel activity of an excitatory I1-superfamily conotoxin. Biochemistry 2007, 46, 9929–9940. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, B.; Zhang, M.-M.; Buczek, O.; Azam, L.; Bulaj, G.; Norton, R.S.; Olivera, B.M.; Yoshikami, D. Specificity, affinity and efficacy of iota-conotoxin RXIA, an agonist of voltage-gated sodium channels NaV1.2, 1.6 and 1.7. Biochem. Pharmacol. 2008, 75, 2334–2344. [Google Scholar] [CrossRef] [PubMed]
- Buczek, O.; Jimenez, E.C.; Yoshikami, D.; Imperial, J.S.; Watkins, M.; Morrison, A.; Olivera, B.M. I1-superfamily conotoxins and prediction of single d-amino acid occurrence. Toxicon 2008, 51, 218–229. [Google Scholar] [CrossRef]
- Kauferstein, S.; Huys, I.; Lamthanh, H.; Stöcklin, R.; Sotto, F.; Menez, A.; Tytgat, J.; Mebs, D. A novel conotoxin inhibiting vertebrate voltage-sensitive potassium channels. Toxicon 2003, 42, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, M.B.; Pérez-Reyes, L.I.; López, Z.; de la Cotera, E.P.H.; Falcón, A.; Ayala, C.; Galván, M.; Salvador, C.; Escobar, L.I. Peptide sr11a from Conus spurius is a novel peptide blocker for KV1 potassium channels. Peptides 2010, 31, 1287–1291. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.-X.; Chen, X.-K.; Zhang, C.; Wang, L.-X.; Duan, K.-L.; He, L.-L.; Cao, Y.; Liu, S.-Y.; Zhong, M.-N.; Ulens, C.; et al. A novel conotoxin from Conus betulinus, κ-BtX, unique in cysteine pattern and in function as a specific BK channel modulator. J. Biol. Chem. 2003, 278, 12624–12633. [Google Scholar] [CrossRef] [PubMed]
- Yuan, D.-D.; Liu, L.; Shao, X.-X.; Peng, C.; Chi, C.-W.; Guo, Z.-Y. New conotoxins define the novel I3-superfamily. Peptides 2009, 30, 861–865. [Google Scholar] [CrossRef] [PubMed]
- Hansson, K.; Furie, B.; Furie, B.C.; Stenflo, J. Isolation and characterization of three novel Gla-containing Conus marmoreus venom peptides, one with a novel cysteine pattern. Biochem. Biophys. Res. Commun. 2004, 319, 1081–1087. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, Z.; Liu, N.; Zhao, C.; Hu, J.; Dai, Q. cDNA cloning of conotoxins with framework XII from several Conus species. Acta Biochim. Biophys. Sin. 2010, 42, 656–661. [Google Scholar] [CrossRef]
- Imperial, J.S.; Bansal, P.S.; Alewood, P.F.; Daly, N.L.; Craik, D.J.; Sporning, A.; Terlau, H.; López-Vera, E.; Bandyopadhyay, P.K.; Olivera, B.M. A novel conotoxin inhibitor of KV1.6 channel and nAChR subtypes defines a new superfamily of conotoxins. Biochemistry 2006, 45, 8331–8340. [Google Scholar] [CrossRef]
- Liu, Z.; Li, H.; Liu, N.; Wu, C.; Jiang, J.; Yue, J.; Jing, Y.; Dai, Q. Diversity and evolution of conotoxins in Conus virgo, Conus eburneus, Conus imperialis and Conus marmoreus from the South China Sea. Toxicon 2012, 60, 982–989. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Khoo, K.K.; Xu, S.; Zhou, M.; Boonyalai, N.; Perugini, M.A.; Shao, X.; Chi, C.; Galea, C.A.; Wang, C.; et al. A helical conotoxin from Conus imperialis has a novel cysteine framework and defiines a new superfamily. J. Biol. Chem. 2012, 287, 14973–14983. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; Tang, S.; Pi, C.; Liu, J.; Wang, F.; Wang, L.; Zhou, W.; Xu, A. Discovery of a novel class of conotoxin from Conus litteratus, lt14a, with a unique cysteine pattern. Peptides 2006, 27, 2174–2181. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Bandyopadhyay, P.; Olivera, B.; Yandell, M. Elucidation of the molecular envenomation strategy of the cone snail Conus geographus through transcriptome sequencing of its venom duct. BMC Genomics 2012, 13, 284. [Google Scholar] [CrossRef] [PubMed]
- Jin, A.-H.; Dutertre, S.; Kaas, Q.; Lavergne, V.; Kubala, P.; Lewis, R.J.; Alewood, P.F. Transcriptomic messiness in the venom duct of Conus miles contributes to conotoxin diversity. Mol. Cell. Proteomics 2013, 12, 3824–3833. [Google Scholar] [CrossRef] [PubMed]
- Biggs, J.S.; Watkins, M.; Puillandre, N.; Ownby, J.-P.; Lopez-Vera, E.; Christensen, S.; Moreno, K.J.; Bernaldez, J.; Licea-Navarro, A.; Corneli, P.S.; et al. Evolution of Conus peptide toxins: Analysis of Conus californicus Reeve, 1844. Mol. Phylogen. Evol. 2010, 56, 1–12. [Google Scholar] [CrossRef]
- Norton, R.S. µ-conotoxins as leads in the development of new analgesics. Molecules 2010, 15, 2825–2844. [Google Scholar] [CrossRef] [PubMed]
- Green, B.R.; Bulaj, G.; Norton, R.S. Structure and function of μ-conotoxins, peptide-based sodium channel blockers with analgesic activity. Future Med. Chem. 2014, 6, 1677–1698. [Google Scholar] [CrossRef] [PubMed]
- Al-Sabi, A.; Lennartz, D.; Ferber, M.; Gulyas, J.; Rivier, J.E.F.; Olivera, B.M.; Carlomagno, T.; Terlau, H. κM-conotoxin RIIIK, structural and functional novelty in a K+ channel antagonist. Biochemistry 2004, 43, 8625–8635. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Dendorfer, A.; Finol-Urdaneta, R.K.; Terlau, H.; Olivera, B.M. Biochemical characterization of κM-RIIIJ, a KV1.2 channel blocker. J. Biol. Chem. 2010, 285, 14882–14889. [Google Scholar] [CrossRef] [PubMed]
- Ferber, M.; Sporning, A.; Jeserich, G.; DeLaCruz, R.; Watkins, M.; Olivera, B.M.; Terlau, H. A novel Conus peptide ligand for K+ channels. J. Biol. Chem. 2003, 278, 2177–2183. [Google Scholar] [CrossRef] [PubMed]
- Lluisma, A.O.; López-Vera, E.; Bulaj, G.; Watkins, M.; Olivera, B.M. Characterization of a novel ψ-conotoxin from Conus parius Reeve. Toxicon 2008, 51, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Shon, K.-J.; Grilley, M.; Jacobsen, R.; Cartier, G.E.; Hopkins, C.; Gray, W.R.; Watkins, M.; Hillyard, D.R.; Rivier, J.; Torres, J.; et al. A noncompetitive peptide inhibitor of the nicotinic acetylcholine receptor from Conus purpurascens venom. Biochemistry 1997, 36, 9581–9587. [Google Scholar] [CrossRef]
- Van Wagoner, R.M.; Jacobsen, R.B.; Olivera, B.M.; Ireland, C.M. Characterization and three-dimensional structure determination of ψ-conotoxin PIIIF, a novel noncompetitive antagonist of nicotinic acetylcholine receptors. Biochemistry 2003, 42, 6353–6362. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-H.; Wang, Q.; Jiang, H.; Liu, L.; Xiao, C.; Yuan, D.-D.; Shao, X.-X.; Dai, Q.-Y.; Cheng, J.-S.; Chi, C.-W. Characterization of novel M-superfamily conotoxins with new disulfide linkage. FEBS J. 2006, 273, 4972–4982. [Google Scholar] [CrossRef] [PubMed]
- McDougal, O.M.; Poulter, C.D. Three-dimensional structure of the mini-M conotoxin mr3a. Biochemistry 2003, 43, 425–429. [Google Scholar] [CrossRef]
- McDougal, O.M.; Turner, M.W.; Ormond, A.J.; Poulter, C.D. Three-dimensional structure of conotoxin tx3a: An M-1 branch peptide of the M-superfamily. Biochemistry 2008, 47, 2826–2832. [Google Scholar] [CrossRef] [PubMed]
- Poppe, L.; Hui, J.O.; Ligutti, J.; Murray, J.K.; Schnier, P.D. PADLOC: A powerful tool to assign disulfide bond connectivities in peptides and proteins by NMR spectroscopy. Anal. Chem. 2011, 84, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Du, W.-H.; Han, Y.-H.; Huang, F.-j.; Li, J.; Chi, C.-W.; Fang, W.-H. Solution structure of an M-1 conotoxin with a novel disulfide linkage. FEBS J. 2007, 274, 2596–2602. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, J.; Pi, C.; Zeng, X.; Zhou, M.; Jiang, X.; Chen, S.; Ren, Z.; Xu, A. Identification of a novel M-superfamily conotoxin with the ability to enhance tetrodotoxin sensitive sodium currents. Arch. Toxicol. 2009, 83, 925–932. [Google Scholar] [CrossRef]
- Corpuz, G.P.; Jacobsen, R.B.; Jimenez, E.C.; Watkins, M.; Walker, C.; Colledge, C.; Garrett, J.E.; McDougal, O.; Li, W.; Gray, W.R.; et al. Definition of the M-conotoxin superfamily: Characterization of novel peptides from molluscivorous Conus venoms. Biochemistry 2005, 44, 8176–8186. [Google Scholar] [CrossRef] [PubMed]
- Akcan, M.; Cao, Y.; Chongxu, F.; Craik, D.J. The three-dimensional solution structure of mini-M conotoxin BtIIIA reveals a disconnection between disulfide connectivity and peptide fold. Biorg. Med. Chem. 2013, 21, 3590–3596. [Google Scholar] [CrossRef]
- Khoo, K.K.; Gupta, K.; Green, B.R.; Zhang, M.-M.; Watkins, M.; Olivera, B.M.; Balaram, P.; Yoshikami, D.; Bulaj, G.; Norton, R.S. Distinct disulfide isomers of μ-conotoxins KIIIA and KIIIB block voltage-gated sodium channels. Biochemistry 2012, 51, 9826–9835. [Google Scholar] [PubMed]
- Han, Y.; Huang, F.; Jiang, H.; Liu, L.; Wang, Q.; Wang, Y.; Shao, X.; Chi, C.; Du, W.; Wang, C. Purification and structural characterization of a d-amino acid-containing conopeptide, conomarphin, from Conus marmoreus. FEBS J. 2008, 275, 1976–1987. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.-C.; Zhou, M.; Peng, C.; Shao, X.-X.; Guo, Z.-Y.; Chi, C.-W. Novel conopeptides in a form of disulfide-crosslinked dimer. Peptides 2010, 31, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Fainzilber, M.; Nakamura, T.; Gaathon, A.; Lodder, J.C.; Kits, K.S.; Burlingame, A.L.; Zlotkin, E. A new cysteine framework in sodium channel blocking conotoxins. Biochemistry 1995, 34, 8649–8656. [Google Scholar] [CrossRef] [PubMed]
- Conticello, S.G.; Gilad, Y.; Avidan, N.; Ben-Asher, E.; Levy, Z.; Fainzilber, M. Mechanisms for evolving hypervariability: The case of conopeptides. Mol. Biol. Evol. 2001, 18, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Wang, C.-Z.; Xu, C.-Q.; Fan, C.-X.; Dai, X.-D.; Chen, J.-S.; Chi, C.-W. A novel M-superfamily conotoxin with a unique motif from Conus vexillum. Peptides 2006, 27, 682–689. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Chang, S.; Yang, L.; Shi, J.; McFarland, K.; Yang, X.; Moller, A.; Wang, C.; Zou, X.; Chi, C.; et al. Conopeptide Vt3.1 preferentially inhibits BK potassium channels containing β4 subunits via electrostatic interactions. J. Biol. Chem. 2014, 289, 4735–4742. [Google Scholar] [CrossRef] [PubMed]
- Imperial, J.S.; Chen, P.; Sporning, A.; Terlau, H.; Daly, N.L.; Craik, D.J.; Alewood, P.F.; Olivera, B.M. Tyrosine-rich conopeptides affect voltage-gated K+ channels. J. Biol. Chem. 2008, 283, 23026–23032. [Google Scholar] [CrossRef] [PubMed]
- Bulaj, G.; West, P.J.; Garrett, J.E.; Marsh, M.; Zhang, M.M.; Norton, R.S.; Smith, B.J.; Yoshikami, D.; Olivera, B.M. Novel conotoxins from Conus striatus and Conus kinoshitai selectively block TTX-resistant sodium channels. Biochemistry 2005, 44, 7259–7265. [Google Scholar] [CrossRef]
- Daly, N.L.; Ekberg, J.A.; Thomas, L.; Adams, D.J.; Lewis, R.J.; Craik, D.J. Structures of μO-conotoxins from Conus marmoreus: Inhibitors of tetrodotoxin (TTX)-sensitive and TTX-resistant sodium channels in mammalian sensory neurons. J. Biol. Chem. 2004, 279, 25774–25782. [Google Scholar] [CrossRef] [PubMed]
- Kohno, T.; Kim, J.I.; Kobayashi, K.; Kodera, Y.; Maeda, T.; Sato, K. Three-dimensional structure in solution of the calcium channel blocker ω-conotoxin MVIIA. Biochemistry 1995, 34, 10256–10265. [Google Scholar] [CrossRef] [PubMed]
- Kohno, T.; Sasaki, T.; Kobayashi, K.; Fainzilber, M.; Sato, K. Three-dimensional solution structure of the sodium channel agonist/antagonist δ-conotoxin TxVIA. J. Biol. Chem. 2002, 277, 36387–36391. [Google Scholar] [CrossRef]
- Savarin, P.; Guenneugues, M.; Gilquin, B.; Lamthanh, H.; Gasparini, S.; Zinn-Justin, S.; Ménez, A. Three-dimensional structure of κ-conotoxin PVIIA, a novel potassium channel-blocking toxin from cone snails. Biochemistry 1998, 37, 5407–5416. [Google Scholar] [CrossRef] [PubMed]
- Pallaghy, P.K.; Nielsen, K.J.; Craik, D.J.; Norton, R.S. A common structural motif incorporating a cystine knot and a triple-stranded β-sheet in toxic and inhibitory polypeptides. Protein Sci. 1994, 3, 1833–1839. [Google Scholar] [CrossRef] [PubMed]
- Norton, R.S.; Pallaghy, P.K. The cystine knot structure of ion channel toxins and related polypeptides. Toxicon 1998, 36, 1573–1583. [Google Scholar] [CrossRef] [PubMed]
- Ekberg, J.; Jayamanne, A.; Vaughan, C.W.; Aslan, S.; Thomas, L.; Mould, J.; Drinkwater, R.; Baker, M.D.; Abrahamsen, B.; Wood, J.N.; et al. μO-conotoxin MrVIB selectively blocks NaV1.8 sensory neuron specific sodium channels and chronic pain behavior without motor deficits. Proc. Natl. Acad. Sci. USA 2006, 103, 17030–17035. [Google Scholar] [CrossRef] [PubMed]
- Lubbers, N.L.; Campbell, T.J.; Polakowski, J.S.; Bulaj, G.; Layer, R.T.; Moore, J.; Gross, G.J.; Cox, B.F. Postischemic administration of CGX-1051, a peptide from cone snail venom, reduces infarct size in both rat and dog models of myocardial ischemia and reperfusion. J. Cardiovasc. Pharmacol. 2005, 46, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Miljanich, G.P. Ziconotide: neuronal calcium channel blocker for treating severe chronic pain. Curr. Med. Chem. 2004, 11, 3029–3040. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, Y.; Abe, T.; Satake, M.; Odani, S.; Suzuki, J.; Ishikawa, K. A novel sodium channel inhibitor from Conus geographus: purification, structure, and pharmacological properties. Biochemistry 1988, 27, 6256–6262. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.M.; Atkins, A.R.; Loughnan, M.L.; Jones, A.; Adams, D.A.; Martin, R.C.; Lewis, R.J.; Craik, D.J.; Alewood, P.F. Conotoxin TVIIA, a novel peptide from the venom of Conus tulipa. Eur. J. Biochem. 2000, 267, 4642–4648. [Google Scholar] [CrossRef] [PubMed]
- Elliger, C.A.; Richmond, T.A.; Lebaric, Z.N.; Pierce, N.T.; Sweedler, J.V.; Gilly, W.F. Diversity of conotoxin types from Conus californicus reflects a diversity of prey types and a novel evolutionary history. Toxicon 2011, 57, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Gajewiak, J.; Azam, L.; Imperial, J.; Walewska, A.; Green, B.R.; Bandyopadhyay, P.K.; Raghuraman, S.; Ueberheide, B.; Bern, M.; Zhou, H.M.; et al. A disulfide tether stabilizes the block of sodium channels by the conotoxin μO§-GVIIJ. Proc. Natl. Acad. Sci. USA 2014, 111, 2758–2763. [Google Scholar] [CrossRef] [PubMed]
- Olivera, B.; Gray, W.; Zeikus, R.; McIntosh, J.; Varga, J.; Rivier, J.; de Santos, V.; Cruz, L. Peptide neurotoxins from fish-hunting cone snails. Science 1985, 230, 1338–1343. [Google Scholar] [CrossRef] [PubMed]
- Shon, K.-J.; Grilley, M.M.; Marsh, M.; Yoshikami, D.; Hall, A.R.; Kurz, B.; Gray, W.R.; Imperial, J.S.; Hillyard, D.R.; Olivera, B.M. Purification, characterization, synthesis, and cloning of the lockjaw peptide from Conus purpurascens venom. Biochemistry 1995, 34, 4913–4918. [Google Scholar] [CrossRef] [PubMed]
- Terlau, H.; Shon, K.-J.; Grilley, M.; Stocker, M.; Stuhmer, W.; Olivera, B.M. Strategy for rapid immobilization of prey by a fish-hunting marine snail. Nature 1996, 381, 148–151. [Google Scholar] [CrossRef] [PubMed]
- Bulaj, G.; Zhang, M.-M.; Green, B.R.; Fiedler, B.; Layer, R.T.; Wei, S.; Nielsen, J.S.; Low, S.J.; Klein, B.D.; Wagstaff, J.D.; et al. Synthetic μO-conotoxin MrVIB blocks TTX-resistant sodium channel NaV1.8 and has a long-lasting analgesic activity. Biochemistry 2006, 45, 7404–7414. [Google Scholar] [CrossRef] [PubMed]
- Fainzilber, M.; Gordon, D.; Hasson, A.; Spira, M.E.; Zlotkin, E. Mollusc-specific toxins from the venom of Conus textile neovicarius. Eur. J. Biochem. 1991, 202, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Fainzilber, M.; Nakamura, T.; Lodder, J.C.; Zlotkin, E.; Kits, K.S.; Burlingame, A.L. γ-conotoxin-PnVIIA, a γ-carboxyglutamate-containing peptide agonist of neuronal pacemaker cation currents. Biochemistry 1998, 37, 1470–1477. [Google Scholar] [CrossRef] [PubMed]
- Zugasti-Cruz, A.; Maillo, M.; López-Vera, E.; Falcón, A.; Cotera, E.P.H.d.l.; Olivera, B.M.; Aguilar, M.B. Amino acid sequence and biological activity of a γ-conotoxin-like peptide from the worm-hunting snail Conus austini. Peptides 2006, 27, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Yu, Z.; Fainzilber, M.; Burlingame, A.L. Mass spectrometric-based revision of the structure of a cysteine-rich peptide toxin with γ-carboxyglutamic acid, TxVIIA, from the sea snail, Conus textile. Protein Sci. 1996, 5, 524–530. [Google Scholar] [CrossRef] [PubMed]
- Jimenéz, E.C.; Olivera, B.M.; Gray, W.R.; Cruz, L.J. Contryphan is a D-tryptophan-containing Conus peptide. J. Biol. Chem. 1996, 271, 28002–28005. [Google Scholar] [CrossRef] [PubMed]
- Massilia, G.R.; Eliseo, T.; Grolleau, F.; Lapied, B.; Barbier, J.; Bournaud, R.; Molgó, J.; Cicero, D.O.; Paci, M.; Eugenia Schininà, M.; et al. Contryphan-Vn: a modulator of Ca2+-dependent K+ channels. Biochem. Biophys. Res. Commun. 2003, 303, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Hansson, K.; Ma, X.; Eliasson, L.; Czerwiec, E.; Furie, B.; Furie, B.C.; Rorsman, P.; Stenflo, J. The first γ-carboxyglutamic acid-containing contryphan. J. Biol. Chem. 2004, 279, 32453–32463. [Google Scholar] [CrossRef] [PubMed]
- Sabareesh, V.; Gowd, K.H.; Ramasamy, P.; Sudarslal, S.; Krishnan, K.S.; Sikdar, S.K.; Balaram, P. Characterization of contryphans from Conus loroisii and Conus amadis that target calcium channels. Peptides 2006, 27, 2647–2654. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, E.C.; Watkins, M.; Juszczak, L.J.; Cruz, L.J.; Olivera, B.M. Contryphans from Conus textile venom ducts. Toxicon 2001, 39, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, E.C.; Craig, A.G.; Watkins, M.; Hillyard, D.R.; Gray, W.R.; Gulyas, J.; Rivier, J.E.; Cruz, L.J.; Olivera, B.M. Bromocontryphan: Post-translational bromination of tryptophan. Biochemistry 1997, 36, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, R.; Jimenez, E.C.; Grilley, M.; Watkins, M.; Hillyard, D.; Cruz, L.J.; Olivera, B.M. The contryphans, a d-tryptophan-containing family of Conus peptides: interconversion between conformers. J. Pept. Res. 1998, 51, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, R.B.; Jimenez, E.C.; De la Cruz, R.G.C.; Gray, W.R.; Cruz, L.J.; Olivera, B.M. A novel d-leucine-containing Conus peptide: Diverse conformational dynamics in the contryphan family. J. Pept. Res. 1999, 54, 93–99. [Google Scholar] [CrossRef]
- Eliseo, T.; Cicero, D.O.; Romeo, C.; Schininà, M.E.; Massilia, G.R.; Polticelli, F.; Ascenzi, P.; Paci, M. Solution structure of the cyclic peptide contryphan-Vn, a Ca2+-dependent K+ channel modulator. Biopolymers 2004, 74, 189–198. [Google Scholar] [CrossRef]
- Grant, M.A.; Hansson, K.; Furie, B.C.; Furie, B.; Stenflo, J.; Rigby, A.C. The metal-free and calcium-bound structures of a γ-carboxyglutamic acid-containing contryphan from Conus marmoreus, glacontryphan-M. J. Biol. Chem. 2004, 279, 32464–32473. [Google Scholar] [CrossRef] [PubMed]
- Pallaghy, P.K.; He, W.; Jimenez, E.C.; Olivera, B.M.; Norton, R.S. Structures of the contryphan family of cyclic peptides. Role of electrostatic interactions in cis−trans isomerism. Biochemistry 2000, 39, 12845–12852. [Google Scholar] [CrossRef] [PubMed]
- Zhangsun, D.; Luo, S.; Wu, Y.; Zhu, X.; Hu, Y.; Xie, L. Novel O-superfamily conotoxins identified by cDNA cloning from three vermivorous Conus species. Chem. Biol. Drug Des. 2006, 68, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Craig, A.G.; Jimenez, E.C.; Dykert, J.; Nielsen, D.B.; Gulyas, J.; Abogadie, F.C.; Porter, J.; Rivier, J.E.; Cruz, L.J.; Olivera, B.M.; et al. A novel post-translational modification involving bromination of tryptophan. J. Biol. Chem. 1997, 272, 4689–4698. [Google Scholar] [CrossRef] [PubMed]
- Lirazan, M.B.; Hooper, D.; Corpuz, G.P.; Ramilo, C.A.; Bandyopadhyay, P.; Cruz, L.J.; Olivera, B.M. The spasmodic peptide defines a new conotoxin superfamily. Biochemistry 2000, 39, 1583–1588. [Google Scholar] [CrossRef] [PubMed]
- Miles, L.A.; Dy, C.Y.; Nielsen, J.; Barnham, K.J.; Hinds, M.G.; Olivera, B.M.; Bulaj, G.; Norton, R.S. Structure of a novel P-superfamily spasmodic conotoxin reveals an inhibitory cystine knot motif. J. Biol. Chem. 2002, 277, 43033–43040. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Hong, J.; Zhou, M.; Huang, L.; Shao, X.; Yang, Y.; Sigworth, F.J.; Chi, C.; Lin, D.; Wang, C. A novel conotoxin, qc16a, with a unique cysteine framework and folding. Peptides 2011, 32, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- England, L.J.; Imperial, J.; Jacobsen, R.; Craig, A.G.; Gulyas, J.; Akhtar, M.; Rivier, J.; Julius, D.; Olivera, B.M. Inactivation of a serotonin-gated ion channel by a polypeptide toxin from marine snails. Science 1998, 281, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Teichert, R.W.; Jimenez, E.C.; Olivera, B.M. αS-Conotoxin RVIIIA: A structurally unique conotoxin that broadly targets nicotinic acetylcholine receptors. Biochemistry 2005, 44, 7897–7902. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wu, X.; Yuan, D.; Chi, C.; Wang, C. Identification of a novel S-superfamily conotoxin from vermivorous Conus caracteristicus. Toxicon 2008, 51, 1331–1337. [Google Scholar] [CrossRef] [PubMed]
- Quinton, L.; Gilles, N.; De Pauw, E. TxXIIIA, an atypical homodimeric conotoxin found in the Conus textile venom. J. Proteomics 2009, 72, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Pi, C.; Liu, Y.; Peng, C.; Jiang, X.; Liu, J.; Xu, B.; Yu, X.; Yu, Y.; Jiang, X.; Wang, L.; et al. Analysis of expressed sequence tags from the venom ducts of Conus striatus: focusing on the expression profile of conotoxins. Biochimie 2006, 88, 131–140. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, J.M.; Corpuz, G.O.; Layer, R.T.; Garrett, J.E.; Wagstaff, J.D.; Bulaj, G.; Vyazovkina, A.; Yoshikami, D.; Cruz, L.J.; Olivera, B.M. Isolation and characterization of a novel Conus peptide with apparent antinociceptive activity. J. Biol. Chem. 2000, 275, 32391–32397. [Google Scholar] [CrossRef] [PubMed]
- Balaji, R.A.; Ohtake, A.; Sato, K.; Gopalakrishnakone, P.; Kini, R.M.; Seow, K.T.; Bay, B.H. λ-conotoxins, a new family of conotoxins with unique disulfide pattern and protein folding: Isolation and characterization from the venom of Conus marmoreus. J. Biol. Chem. 2000, 275, 39516–39522. [Google Scholar] [PubMed]
- Ueberheide, B.M.; Fenyö, D.; Alewood, P.F.; Chait, B.T. Rapid sensitive analysis of cysteine rich peptide venom components. Proc. Natl. Acad. Sci. USA 2009, 106, 6910–6915. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.S.; Steel, D.; Jacobsen, R.B.; Lirazan, M.B.; Cruz, L.J.; Hooper, D.; Shetty, R.; DelaCruz, R.C.; Nielsen, J.S.; Zhou, L.M.; et al. The T-superfamily of conotoxins. J. Biol. Chem. 1999, 274, 30664–30671. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, M.B.; Lezama-Monfil, L.; Maillo, M.; Pedraza-Lara, H.; López-Vera, E.; Heimer de la Cotera, E.P. A biologically active hydrophobic T-1-conotoxin from the venom of Conus spurius. Peptides 2006, 27, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Rigby, A.C.; Lucas-Meunier, E.; Kalume, D.E.; Czerwiec, E.; Hambe, B.; Dahlqvist, I.; Fossier, P.; Baux, G.; Roepstorff, P.; Baleja, J.D.; et al. A conotoxin from Conus textile with unusual posttranslational modifications reduces presynaptic Ca2+ influx. Proc. Natl. Acad. Sci. USA 1999, 96, 5758–5763. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, Q.; Pi, C.; Zhao, Y.; Zhou, M.; Wang, L.; Chen, S.; Xu, A. Isolation and characterization of a T-superfamily conotoxin from Conus litteratus with targeting tetrodotoxin sensitive sodium channels. Peptides 2007, 28, 2313–2319. [Google Scholar] [CrossRef] [PubMed]
- Petrel, C.; Hocking, H.G.; Reynaud, M.; Upert, G.; Favreau, P.; Biass, D.; Paolini-Bertrand, M.; Peigneur, S.; Tytgat, J.; Gilles, N.; et al. Identification, structural and pharmacological characterization of τ-CnVA, a conopeptide that selectively interacts with somatostatin sst3 receptor. Biochem. Pharmacol. 2013, 85, 1663–1671. [Google Scholar] [CrossRef] [PubMed]
- Cruz, L.J.; Ramilo, C.A.; Corpuz, G.P.; Olivera, B.M. Conus peptides: Phylogenetic range of biological activity. Biol. Bull. 1992, 183, 159–164. [Google Scholar] [CrossRef]
- Peng, C.; Liu, L.; Shao, X.; Chi, C.; Wang, C. Identification of a novel class of conotoxins defined as V-conotoxins with a unique cysteine pattern and signal peptide sequence. Peptides 2008, 29, 985–991. [Google Scholar] [CrossRef] [PubMed]
- Yuan, D.-D.; Liu, L.; Shao, X.-X.; Peng, C.; Chi, C.-W.; Guo, Z.-Y. Isolation and cloning of a conotoxin with a novel cysteine pattern from Conus caracteristicus. Peptides 2008, 29, 1521–1525. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.S.; Jensen, S.; Ellison, M.; Matta, J.A.; Lee, W.Y.; Imperial, J.S.; Duclos, N.; Brockie, P.J.; Madsen, D.M.; Isaac, J.T.R.; et al. A novel Conus snail polypeptide causes excitotoxicity by blocking desensitization of AMPA receptors. Curr. Biol. 2009, 19, 900–908. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Dürr, K.L.; Gouaux, E. X-ray structures of AMPA receptor–cone snail toxin complexes illuminate activation mechanism. Science 2014, 345, 1021–1026. [Google Scholar] [CrossRef] [PubMed]
- Möller, C.; Marí, F. 9.3 KDa components of the injected venom of Conus purpurascens define a new five-disulfide conotoxin framework. Biopolymers 2011, 96, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Safavi-Hemami, H.; Hu, H.; Gorasia, D.G.; Bandyopadhyay, P.K.; Veith, P.D.; Young, N.D.; Reynolds, E.C.; Yandell, M.; Olivera, B.M.; Purcell, A.W. Combined proteomic and transcriptomic interrogation of the venom gland of Conus geographus uncovers novel components and functional compartmentalization. Mol. Cell. Proteomics 2014, 13, 938–953. [Google Scholar] [CrossRef]
- Möller, C.; Melaun, C.; Castillo, C.; Díaz, M.E.; Renzelman, C.M.; Estrada, O.; Kuch, U.; Lokey, S.; Marí, F. Functional hypervariability and gene diversity of cardioactive neuropeptides. J. Biol. Chem. 2010, 285, 40673–40680. [Google Scholar] [CrossRef] [PubMed]
- Lirazan, M.; Jimenez, E.C.; Grey Craig, A.; Olivera, B.M.; Cruz, L.J. Conophysin-R, a Conus radiatus venom peptide belonging to the neurophysin family. Toxicon 2002, 40, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Cruz, L.J.; de Santos, V.; Zafaralla, G.C.; Ramilo, C.A.; Zeikus, R.; Gray, W.R.; Olivera, B.M. Invertebrate vasopressin/oxytocin homologs. Characterization of peptides from Conus geographus and Conus straitus venoms. J. Biol. Chem. 1987, 262, 15821–15824. [Google Scholar] [PubMed]
- Dutertre, S.; Croker, D.; Daly, N.L.; Andersson, Å.; Muttenthaler, M.; Lumsden, N.G.; Craik, D.J.; Alewood, P.F.; Guillon, G.; Lewis, R.J. Conopressin-T from Conus tulipa reveals an antagonist switch in vasopressin-like peptides. J. Biol. Chem. 2008, 283, 7100–7108. [Google Scholar] [CrossRef] [PubMed]
- Möller, C.; Marí, F. A vasopressin/oxytocin-related conopeptide with γ-carboxyglutamate at position 8. Biochem. J. 2007, 404, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Bayrhuber, M.; Vijayan, V.; Ferber, M.; Graf, R.; Korukottu, J.; Imperial, J.; Garrett, J.E.; Olivera, B.M.; Terlau, H.; Zweckstetter, M.; et al. Conkunitzin-S1 is the first member of a new kunitz-type neurotoxin family. J. Biol. Chem. 2005, 280, 23766–23770. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Bandyopadhyay, P.; Olivera, B.; Yandell, M. Characterization of the Conus bullatus genome and its venom-duct transcriptome. BMC Genomics 2011, 12, 60. [Google Scholar] [CrossRef] [PubMed]
- Terrat, Y.; Biass, D.; Dutertre, S.; Favreau, P.; Remm, M.; Stöcklin, R.; Piquemal, D.; Ducancel, F. High-resolution picture of a venom gland transcriptome: Case study with the marine snail Conus consors. Toxicon 2012, 59, 34–46. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, J.M.; Ghomashchi, F.; Gelb, M.H.; Dooley, D.J.; Stoehr, S.J.; Giordani, A.B.; Naisbitt, S.R.; Olivera, B.M. Conodipine-M, a novel phospholipase A isolated from the venom of the marine snail Conus magus. J. Biol. Chem. 1995, 270, 3518–3526. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Garrett, J.E.; Watkins, M.; Olivera, B.M. Purification and characterization of a novel excitatory peptide from Conus distans venom that defines a novel gene superfamily of conotoxins. Toxicon 2008, 52, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, J.i.; Nakamura, H.; Hirata, Y.; Ohizumi, Y. Isolation of a cardiotonic glycoprotein, striatoxin, from the venom of the marine snail Conus striatus. Biochem. Biophys. Res. Commun. 1982, 105, 1389–1395. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, J.i.; Nakamura, H.; Hirata, Y.; Ohizumi, Y. Isolation of eburnetoxin, a vasoactive substance from the Conus eburneus venom. Life Sci. 1982, 31, 1085–1091. [Google Scholar] [CrossRef] [PubMed]
- Schweitz, H.; Renaud, J.-F.; Randimbivololona, N.; Préau, C.; Schmid, A.; Romey, G.; Rakotovao, L.; Lazdunski, M. Purification, subunit structure and pharmacological effects on cardiac and smooth muscle cells of a polypeptide toxin isolated from the marine snail Conus tessulatus. Eur. J. Biochem. 1986, 161, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, A.; Biass, D.; Kordiš, D.; Stöcklin, R.; Favreau, P.; Križaj, I. Conus consors snail venom proteomics proposes functions, pathways and novel families involved in its venomic system. J. Proteome Res. 2012, 11, 5046–5058. [Google Scholar] [CrossRef]
- Violette, A.; Leonardi, A.; Piquemal, D.; Terrat, Y.; Biass, D.; Dutertre, S.; Noguier, F.; Ducancel, F.; Stöcklin, R.; Križaj, I.; et al. Recruitment of glycosyl hydrolase proteins in a cone snail venomous arsenal: Further insights into biomolecular features of Conus venoms. Mar. Drugs 2012, 10, 258–280. [Google Scholar] [CrossRef] [PubMed]
- Milne, T.J.; Abbenante, G.; Tyndall, J.D.A.; Halliday, J.; Lewis, R.J. Isolation and characterization of a cone snail protease with homology to CRISP proteins of the pathogenesis-related protein superfamily. J. Biol. Chem. 2003, 278, 31105–31110. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Guo, Z.-y.; Chi, C.-w. Cloning and isolation of a Conus cysteine-rich protein homologous to Tex31 but without proteolytic activity. Acta Biochim. Biophys. Sin. 2008, 40, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Hansson, K.; Thämlitz, A.-M.; Furie, B.; Furie, B.C.; Stenflo, J. A single γ-carboxyglutamic acid residue in a novel cysteine-rich secretory protein without propeptide. Biochemistry 2006, 45, 12828–12839. [Google Scholar] [CrossRef] [PubMed]
- Safavi-Hemami, H.; Möller, C.; Marí, F.; Purcell, A.W. High molecular weight components of the injected venom of fish-hunting cone snails target the vascular system. J. Proteomics 2013, 91, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Biggs, J.S.; Rosenfeld, Y.; Shai, Y.; Olivera, B.M. Conolysin-Mt: A Conus peptide that disrupts cellular membranes. Biochemistry 2007, 46, 12586–12593. [Google Scholar] [CrossRef]
- Pisarewicz, K.; Mora, D.; Pflueger, F.C.; Fields, G.B.; Marí, F. Polypeptide chains containing d-γ-hydroxyvaline. J. Am. Chem. Soc. 2005, 127, 6207–6215. [Google Scholar] [CrossRef] [PubMed]
- Dutertre, S.; Lumsden, N.G.; Alewood, P.F.; Lewis, R.J. Isolation and characterisation of conomap-Vt, a D-amino acid containing excitatory peptide from the venom of a vermivorous cone snail. FEBS Lett. 2006, 580, 3860–3866. [Google Scholar] [CrossRef] [PubMed]
- Maillo, M.; Aguilar, M.B.; Lopéz-Vera, E.; Craig, A.G.; Bulaj, G.; Olivera, B.M.; Heimer de la Cotera, E.P. Conorfamide, a Conus venom peptide belonging to the RFamide family of neuropeptides. Toxicon 2002, 40, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.; Papaioannou, S.; Holden-Dye, L. A review of FMRFamide- and RFamide-like peptides in metazoa. Invertebr. Neurosci. 2009, 9, 111–153. [Google Scholar] [CrossRef]
- Aguilar, M.B.; Zugasti-Cruz, A.; Falcón, A.; Batista, C.V.F.; Olivera, B.M.; Heimer de la Cotera, E.P. A novel arrangement of Cys residues in a paralytic peptide of Conus cancellatus (jr. syn.: Conus austini), a worm-hunting snail from the Gulf of Mexico. Peptides 2013, 41, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Bernaĺdez, J.; Román-Gonźalez, S.A.; Martińez, O.; Jimeńez, S.; Vivas, O.; Arenas, I.; Corzo, G.; Arreguiń, R.; García, D.E.; Possani, L.D.; et al. A Conus regularis conotoxin with a novel eight-cysteine framework inhibits CaV2.2 channels and displays an anti-nociceptive activity. Mar. Drugs 2013, 11, 1188–1202. [Google Scholar] [CrossRef] [PubMed]
- Möller, C.; Rahmankhah, S.; Lauer-Fields, J.; Bubis, J.; Fields, G.B.; Marí, F. A novel conotoxin framework with a helix-loop-helix (cs α/α) fold. Biochemistry 2005, 44, 15986–15996. [Google Scholar] [CrossRef] [PubMed]
- Zugasti-Cruz, A.; Aguilar, M.B.; Falcón, A.; Olivera, B.M.; Heimer de la Cotera, E.P. Two new 4-Cys conotoxins (framework 14) of the vermivorous snail Conus austini from the Gulf of Mexico with activity in the central nervous system of mice. Peptides 2008, 29, 179–185. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robinson, S.D.; Norton, R.S. Conotoxin Gene Superfamilies. Mar. Drugs 2014, 12, 6058-6101. https://doi.org/10.3390/md12126058
Robinson SD, Norton RS. Conotoxin Gene Superfamilies. Marine Drugs. 2014; 12(12):6058-6101. https://doi.org/10.3390/md12126058
Chicago/Turabian StyleRobinson, Samuel D., and Raymond S. Norton. 2014. "Conotoxin Gene Superfamilies" Marine Drugs 12, no. 12: 6058-6101. https://doi.org/10.3390/md12126058







