Amperometric Low-Potential Detection of Malic Acid Using Single-Wall Carbon Nanotubes Based Electrodes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Apparatus and reagents
2.2. Electrode assembly
2.3. Procedure
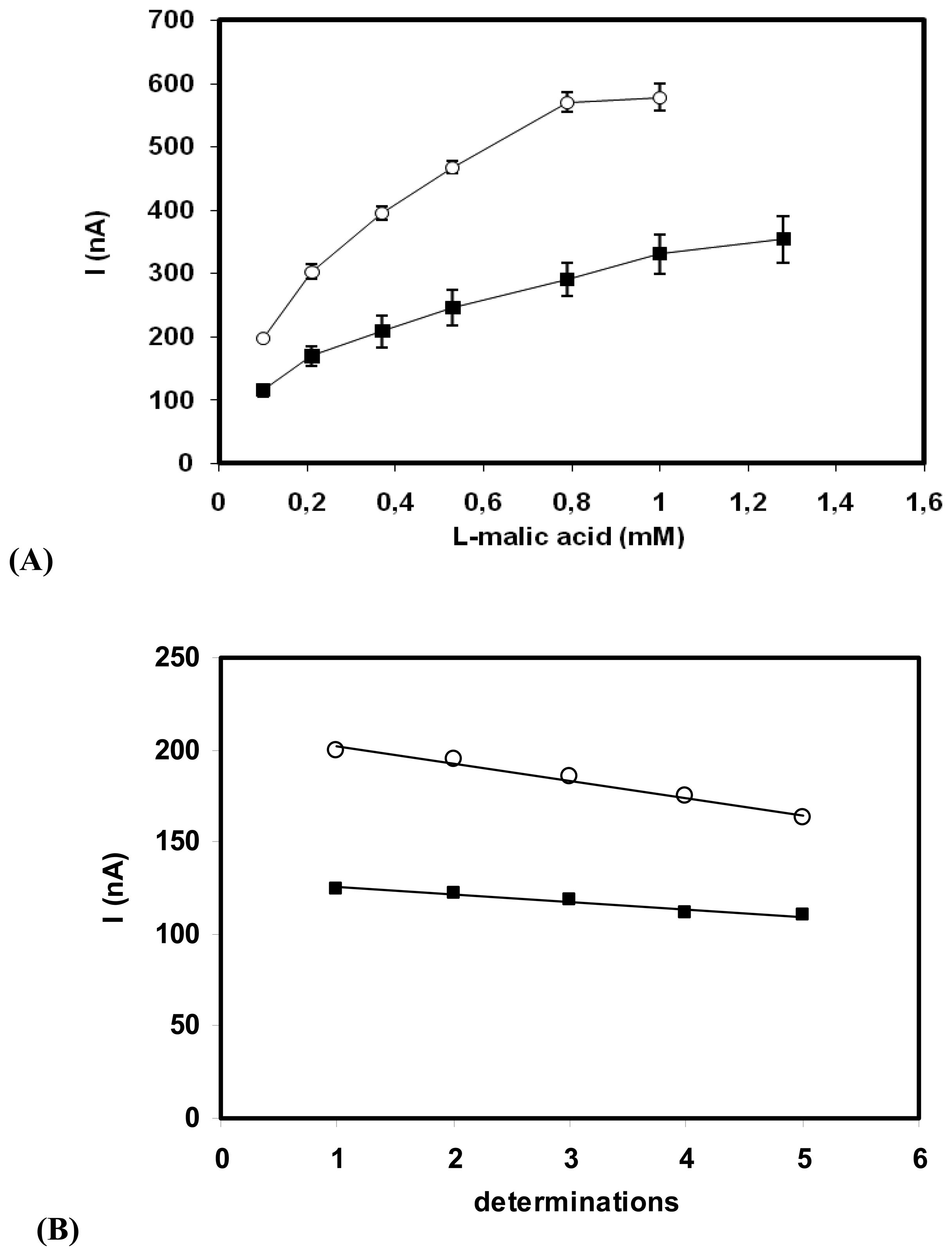
3. Results and Discussion
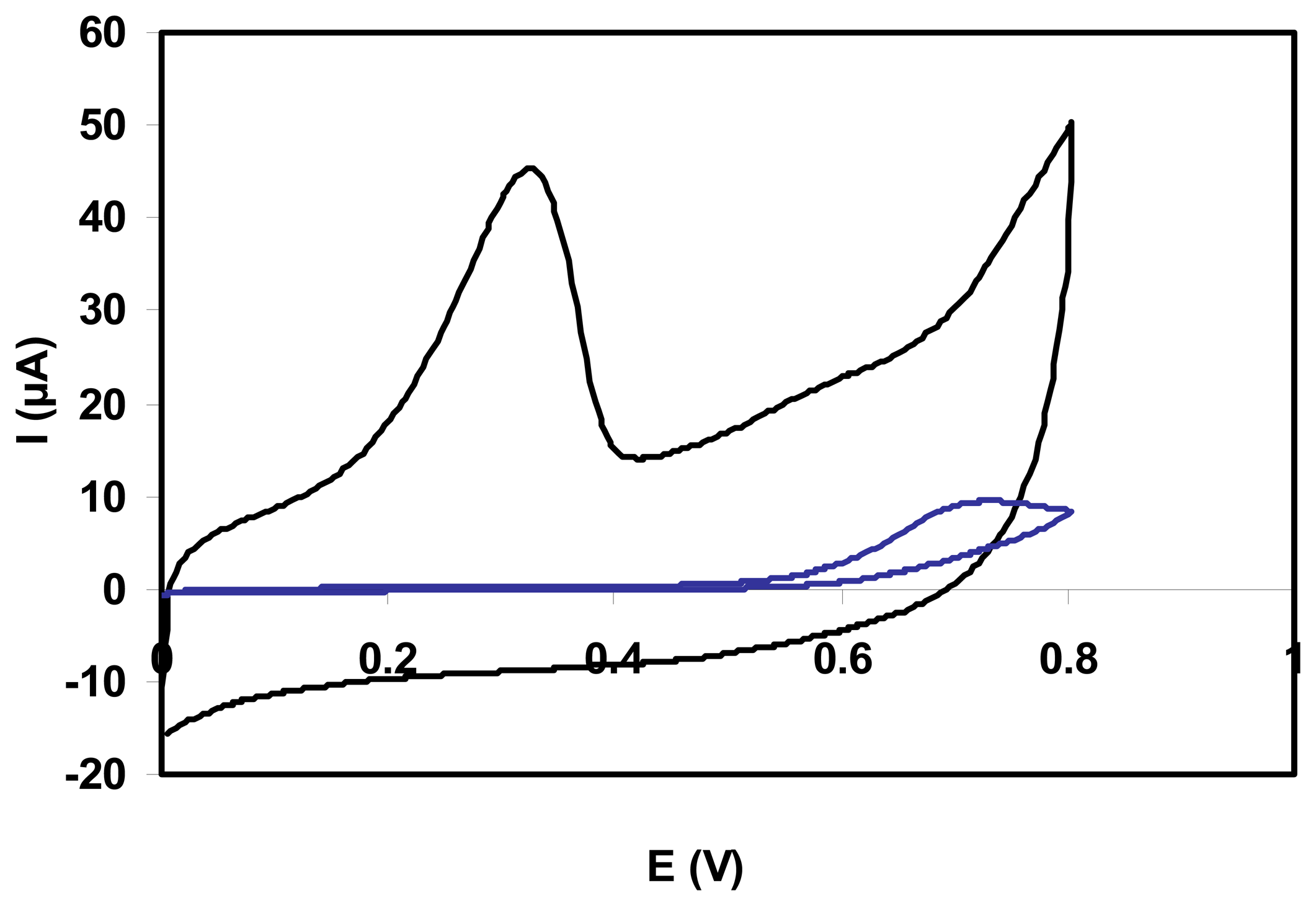
3.1. Voltammetric behaviour of the SWNT-GC sensor toward NADH
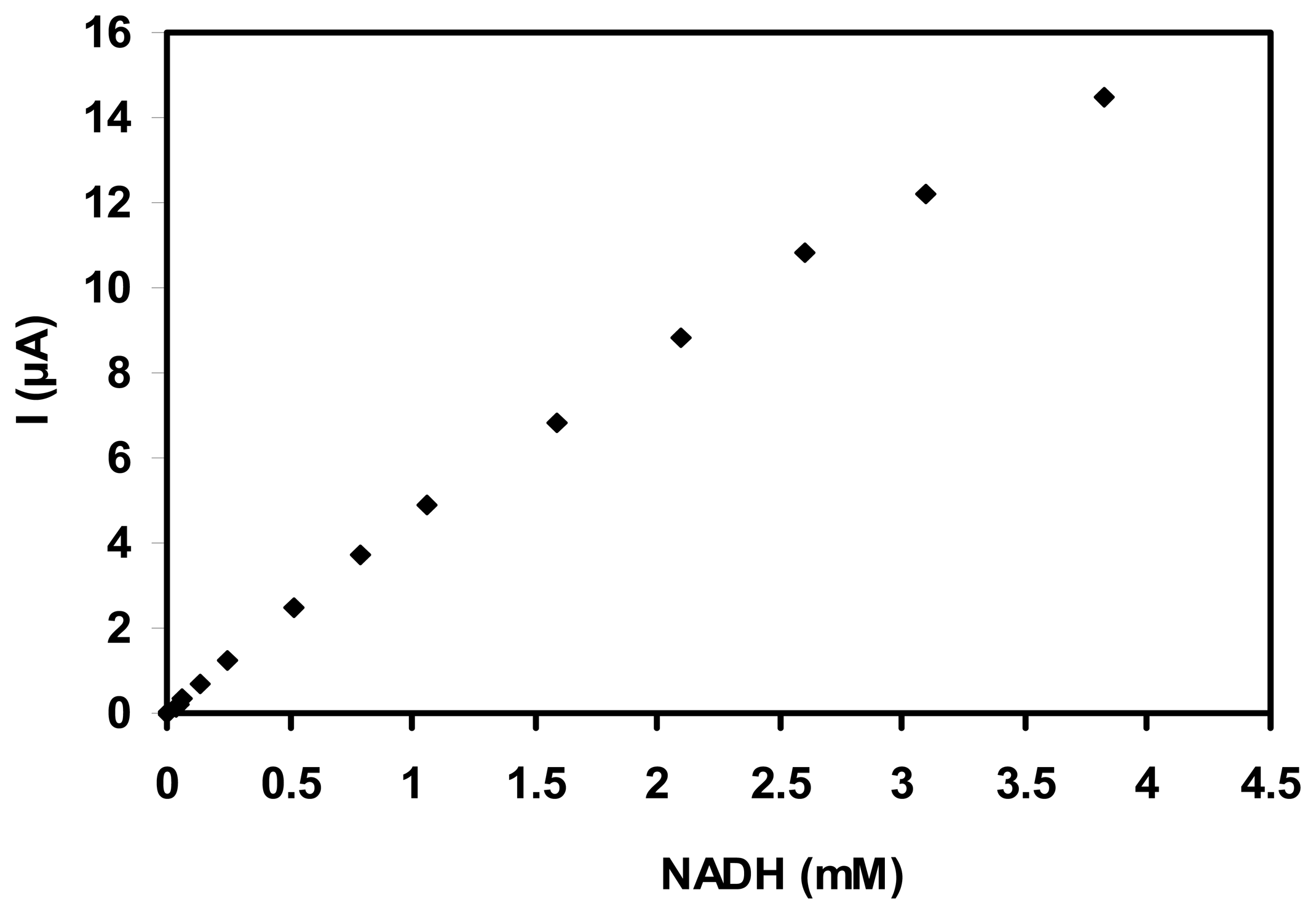
3.2. Electroanalytical performance of the NADH sensor
3.3. Biosensor for L-malic acid
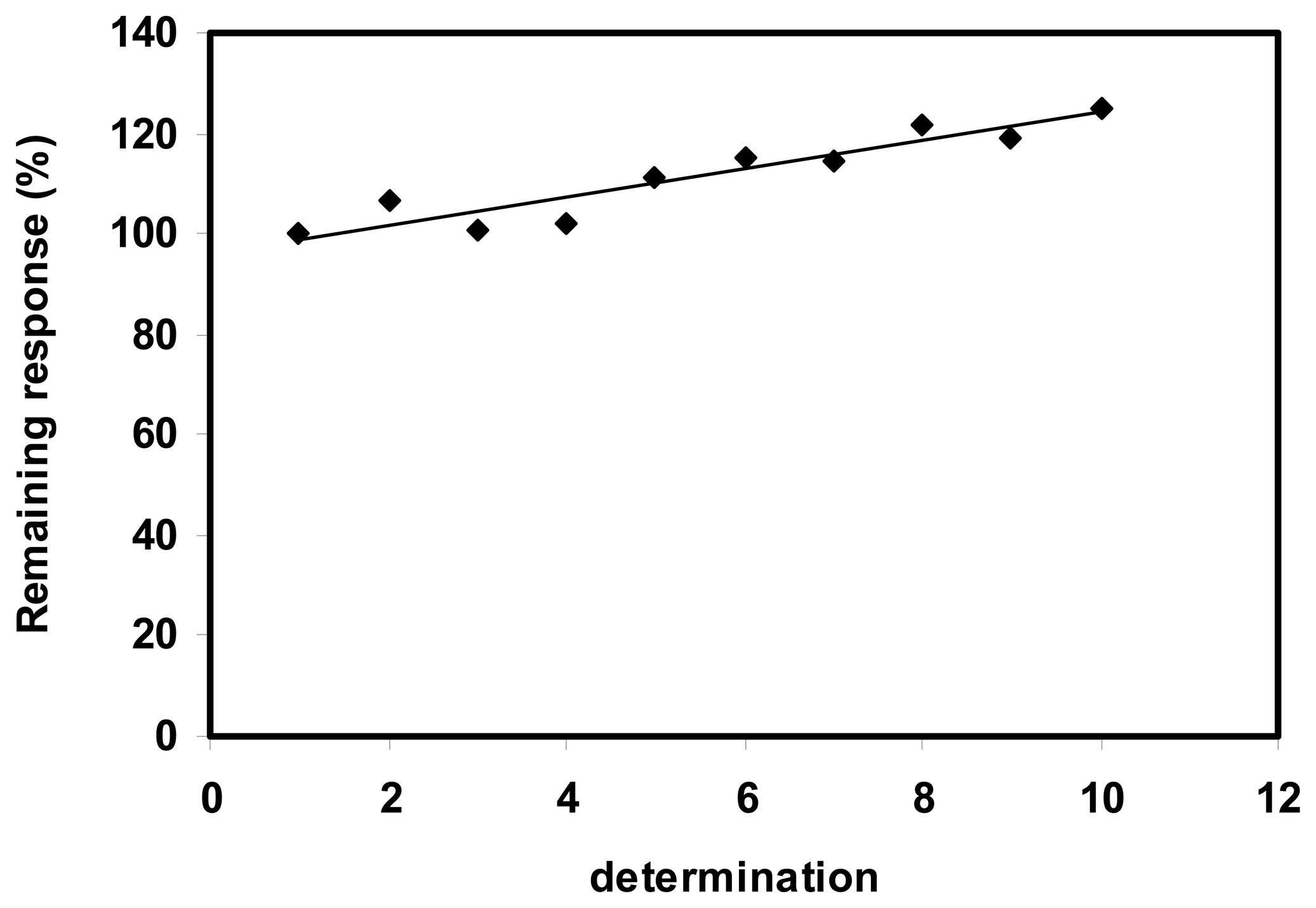
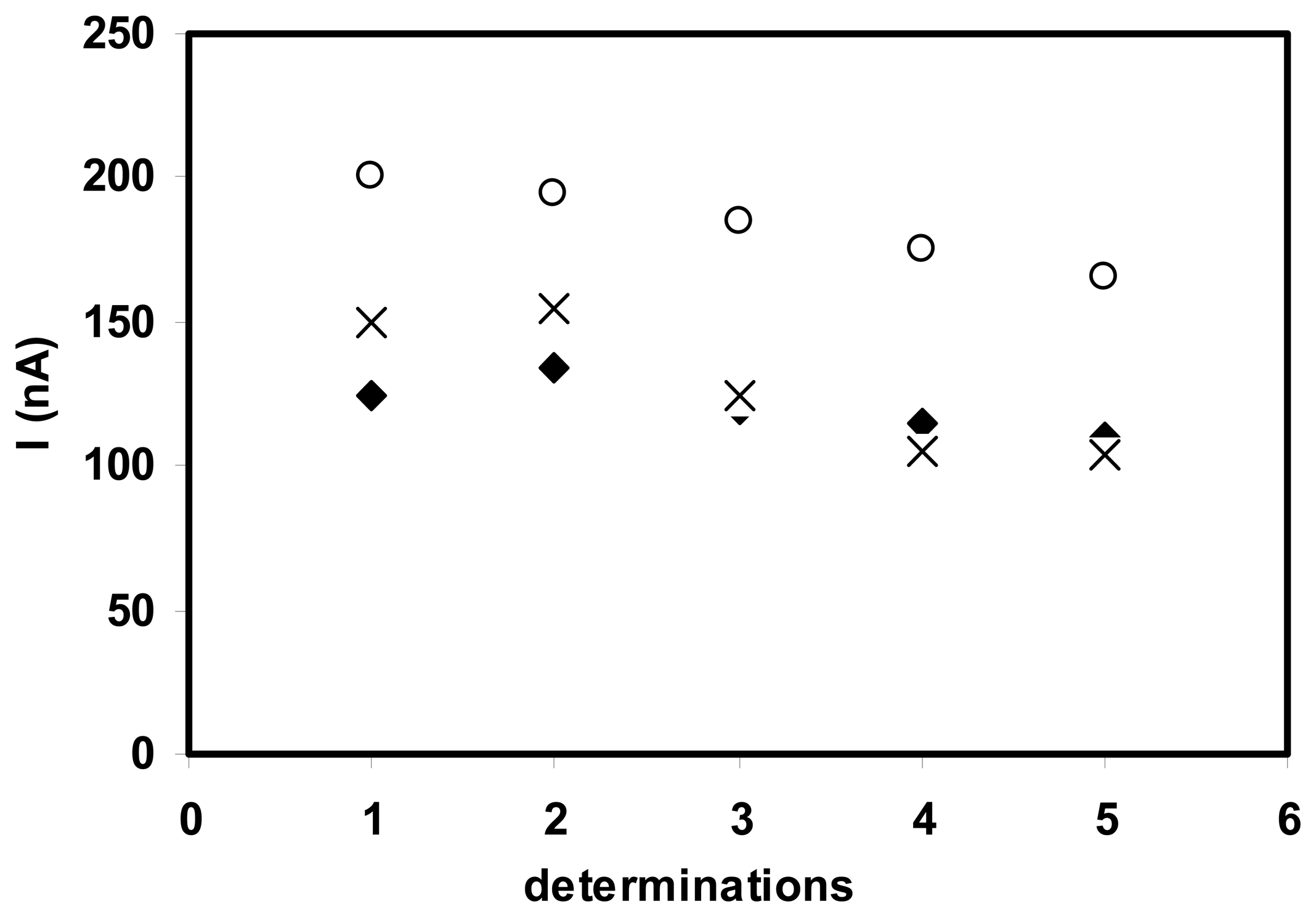
3.3.1. Adsorbed enzyme
3.3.2. Cross-linked enzyme
3.3.3. Nafion membrane - enzyme
3.4. Conclusions
Acknowledgments
References and Notes
- Zen, J.-M.; Kumar, A. S.; Tsai, D.-M. Recent Updates of Chemically Modified Electrodes in Analytical Chemistry. Electroanalysis 2003, 15(13), 1073–1087. [Google Scholar]
- Wang, J.; Musameh, M.; Lin, Y. Solubilization of Carbon Nanotubes by Nafion Towards the Preparation of Amperometric Biosensors. Journal of the American Chemical Society 2003, 125, 2408–2409. [Google Scholar]
- Wang, J.-X.; Li, M.; Shi, Z.; Li, N.; Guo, Z. Direct electrochemistry of cytochrome c at a glassy carbon electrode modified with single-wall carbon nanotubes. Analytical Chemistry 2002, 74(9), 1993–1997. [Google Scholar]
- Musameh, M.; Wang, J.; Merkoci, A.; Lin, Y. Low potential stable NADH detection at carbon-nanotube-modified glassy carbon electrodes. Electrochemistry Communications 2002, 4, 743–746. [Google Scholar]
- Wang, J.; Li, M.; Shi, Z.; Li, N.; Guo, Z. Electrocatalytic oxidation of norepinephrine at a glassy carbon electrode modified with single wall carbon nanotubes. Electroanalysis 2002, 14(3), 225–230. [Google Scholar]
- Wang, Z.; Liu, J.; Liang, Q.; Wang, Y.; Luo, G. Carbon nanotube-modified electrodes for the simultaneous determination of dopamine and ascorbic acid. Analyst 2002, 127(5), 653–658. [Google Scholar]
- Sanchez-Pomales, G.; Morales-Negron, Y.; Cabrera, C.R. Study of self-assembled monolayers of DNA and DNA-carbon nanotube hybrids. Rev. Adv. Mater. Sci. 2005, 10, 261–265. [Google Scholar]
- Shim, M.; Kam, N.W.S.; Chen, R.J.; Li, Y.; Dai, H. Functionalization of carbon nanotubes for biocompatibility and biomolecular recognition. Nano Letters 2002, 2(4), 285–288. [Google Scholar]
- Wang, J.; Liu, G.; Jan, M. Ultrasensitive Electrical Biosensing of Proteins and DNA: Carbon-Nanotube Derived Amplification of the Recognition and Transduction Events. Journal of the American Chemical Society 2004, 126, 3010–3011. [Google Scholar]
- Ferancová, A.; Ovádeková, R.; Vaníčková, M.; Šatka, A.; Viglaský, R.; Zima, J.; Barek, J.; Labuda, J. DNA-modified screen-printed electrodes with nanostructured films of multiwall carbon nanotubes, hydroxyapatite and montmorillonite. Electroanalysis 2006, 18(2), 163–168. [Google Scholar]
- Kim, S. CNT Sensors for Detecting Gases with Low Adsorption Energy by Ionization. Sensors 2006, 6, 503–51. [Google Scholar]
- Banks, C.E.; Davies, T.J.; Wildgoose, G.G.; Compton, R.G. Electrocatalysis at graphite and carbon nanotube modified electrodes: edge-plane sites and tube ends are the reactive sites. Chemistry Communications 2005, 7, 829–841. [Google Scholar]
- Banks, C.E.; Compton, R.G. Exploring the electrocatalytic sites of carbon nanotubes for NADH detection: an edge plane pyrolytic graphite electrode study. Analyst 2005, 130(9), 1232–1239. [Google Scholar]
- Yin, Y.; Lü, Y.; Wu, P.; Cai, C. Direct Electrochemistry of Redox Proteins and Enzymes Promoted by Carbon Nanotubes. Sensors 2005, 5, 220–234. [Google Scholar]
- Zhao, Q.; Gan, Z.; Zhuang, Q. Electrochemical sensors based on carbon nanotubes. Electroanalysis 2002, 14, 1609–1613. [Google Scholar]
- Wang, J. Carbon-Nanotube Based Electrochemical Biosensors: A Review. Electroanalysis 2005, 17, 7–14. [Google Scholar]
- Tangkuaram, T.; Wang, J.; Rodríguez, M. C.; Laocharoensuk, R.; Veerasai, W. Highly Stable Amplified Low-Potential Electrocatalytic Detection of NAD+ at Azure-Chitosan Modified Carbon Electrodes. Sensors and Actuators B 2007, 121, 277–281. [Google Scholar]
- Wang, J.; Musameh, M. Carbon Nanotube/Teflon Composite Electrochemical Sensors and Biosensors. Analytical Chemistry 2003, 75(9), 2075–2079. [Google Scholar]
- Wang, J.; Musameh, M. A Reagentless Amperometric Alcohol Biosensor Based on Carbon-Nanotube/Teflon Composite Electrodes. Analytical Letters 2003, 36(9), 2041–2048. [Google Scholar]
- Valentini, F.; Amine, A.; Orlanducci, S.; Terranova, M. L.; Palleschi, G. Carbon Nanotube Purification: Preparation and Characterization of Carbon Nanotube Paste Electrodes. Analytical Chemistry 2003, 75, 5413–5421. [Google Scholar]
- Moore, R. R.; Banks, C. E.; Compton, R. G. Basal plane pyrolytic graphite modified electrodes: comparison of carbon nanotubes and graphite powder as electrocatalysts. Analytical Chemistry 2004, 76(10), 2677–2682. [Google Scholar]
- Santos, A.S.; Pereira, A.C.; Durán, N.; Kubota, L.T. Amperometric biosensor for ethanol based on co-immobilization of alcohol dehydrogenase and Meldola's Blue on multi-wall carbon nanotube. Electrochimica Acta 2006, 52(1), 215–220. [Google Scholar]
- Pereira, A.C.; Aguiar, M.R.; Kisner, A.; Macedo, D.V.; Kubota, L.T. Amperometric biosensor for lactate based on lactate dehydrogenase and Meldola Blue coimmobilized on multi-wall carbon-nanotube. Sensors and Actuators B 2007, 124(1), 269–276. [Google Scholar]
- Antiochia, R.; Lavagnini, I.; Pastore, P. A comparison between the use of a redox mediator in solution and of surface modified electrodes in the electrocatalytic oxidation of nicotinamide adenine dinucleotide. Bioelectrochemistry 2004, 64, 157–163. [Google Scholar]
- Zhang, M.; Smith, A.; Gorski, W. Carbon nanotube-chitosan system for electrochemical sensing based on dehydrogenase enzymes. Analytical Chemistry 2004, 76, 5045–5050. [Google Scholar]
- Gardner, W.H. Handbook of Food Additives, 2nd ed.; Furia, T.E., Ed.; CRC Press: Cleveland, OH, 1973; p. 239. [Google Scholar]
- Segundo, M. A.; Rangel, A. O. S. S. Kinetic determination of L(-)malic acid in wines using sequential injection analysis. Analytica Chimica Acta 2003, 499, 99–106. [Google Scholar]
- Gallarta, F.; Sáinz, F. J.; Sáenz, C. Fluorescent sensing layer for the determination of L -malic acid in wine. Analytical and Bioanalytical Chemistry 2007, 387(6), 2297–2305. [Google Scholar]
| Enzyme loading (UI/electrode) | Sensitivity (nA/mM) | Linear range (mM) | LOD (μM) | Linear regression equation | R2 |
|---|---|---|---|---|---|
| 0.7 | 455 | 0.2 – 0.8 | 33 | y = 455x + 217 | 0.9916 |
| 1.4 | 283 | 0.2 - 0.8 | 53 | y = 283x + 145 | 0.9956 |
| 2.8 | 233 | 0.1 – 0.6 | 50 | y = 299x + 85 | 0.9936 |
© 2008 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Arvinte, A.; Rotariu, L.; Bala, C. Amperometric Low-Potential Detection of Malic Acid Using Single-Wall Carbon Nanotubes Based Electrodes. Sensors 2008, 8, 1497-1507. https://doi.org/10.3390/s8031497
Arvinte A, Rotariu L, Bala C. Amperometric Low-Potential Detection of Malic Acid Using Single-Wall Carbon Nanotubes Based Electrodes. Sensors. 2008; 8(3):1497-1507. https://doi.org/10.3390/s8031497
Chicago/Turabian StyleArvinte, Adina, Lucian Rotariu, and Camelia Bala. 2008. "Amperometric Low-Potential Detection of Malic Acid Using Single-Wall Carbon Nanotubes Based Electrodes" Sensors 8, no. 3: 1497-1507. https://doi.org/10.3390/s8031497




