Glucose Determination by Means of Steady-state and Time-course UV Fluorescence in Free or Immobilized Glucose Oxidase
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.1. Methods
2.2.1 Preparation of the catalytic membranes
2.2.2 Fluorescence measurements
2.2.2.1 Intrinsic fluorescence emission measurements
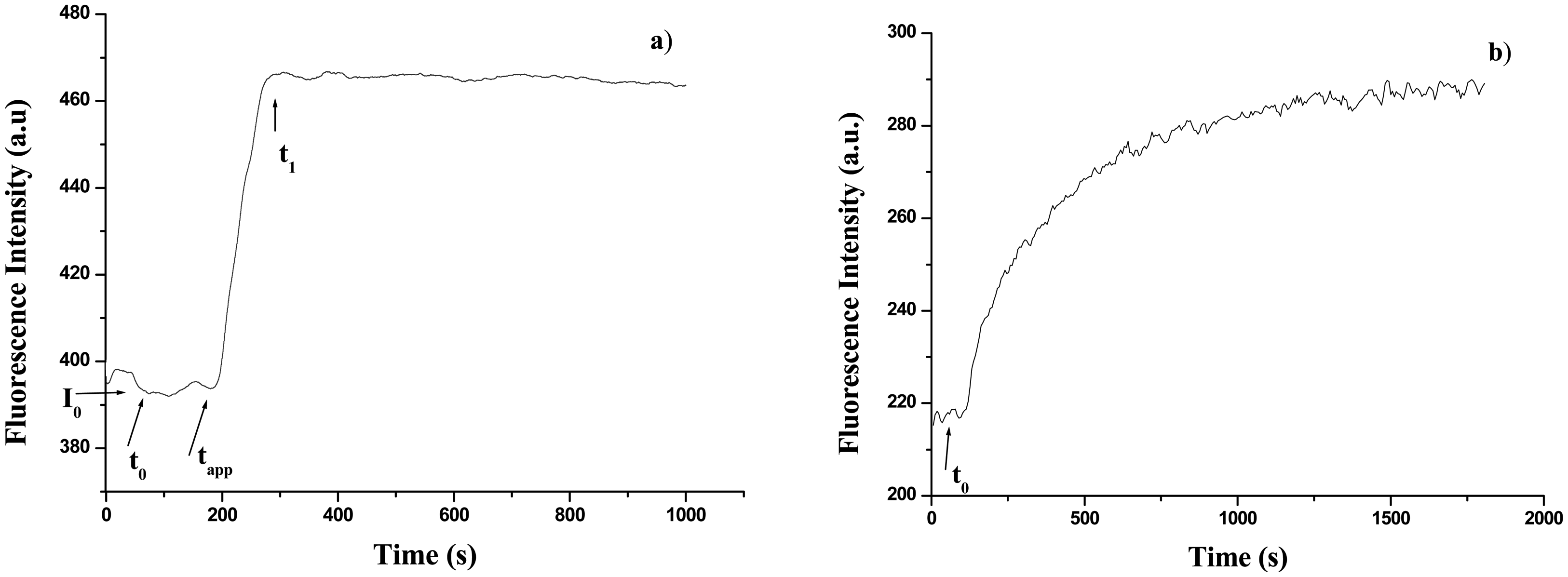
2.2.2.2 UV fluorescence time course measurements
3. Results and Discussion
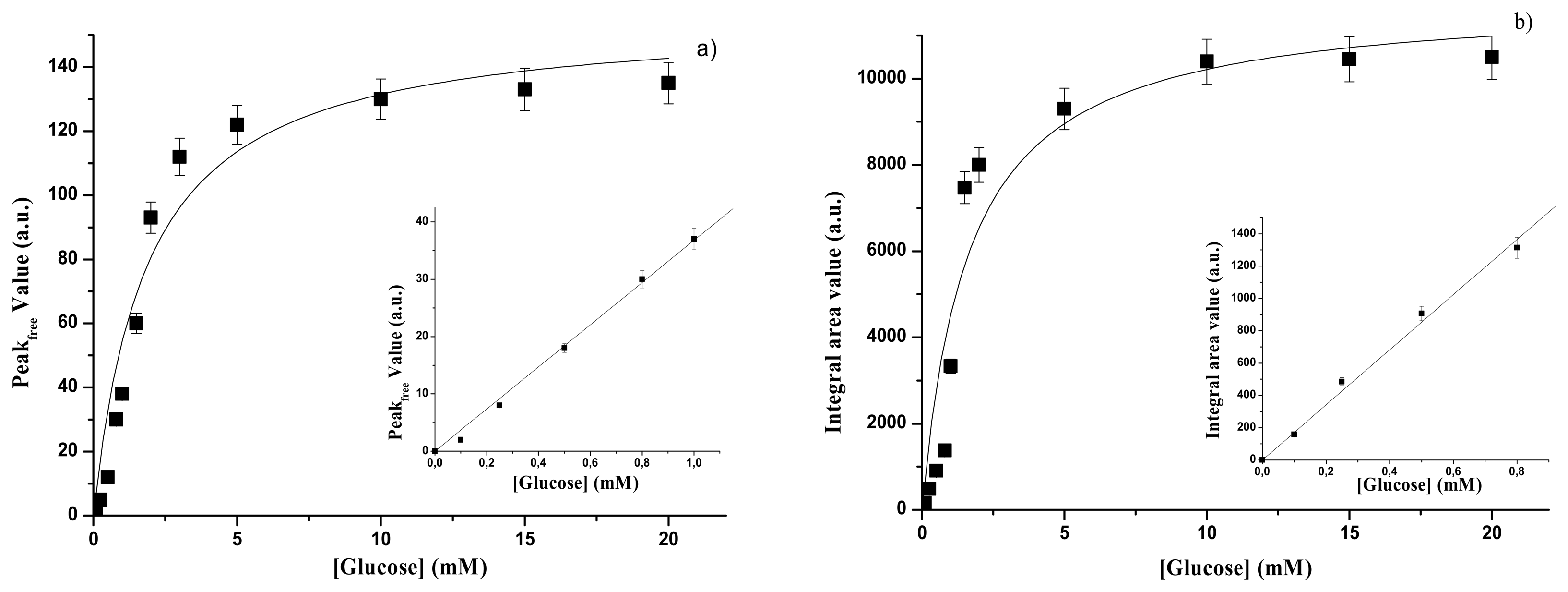
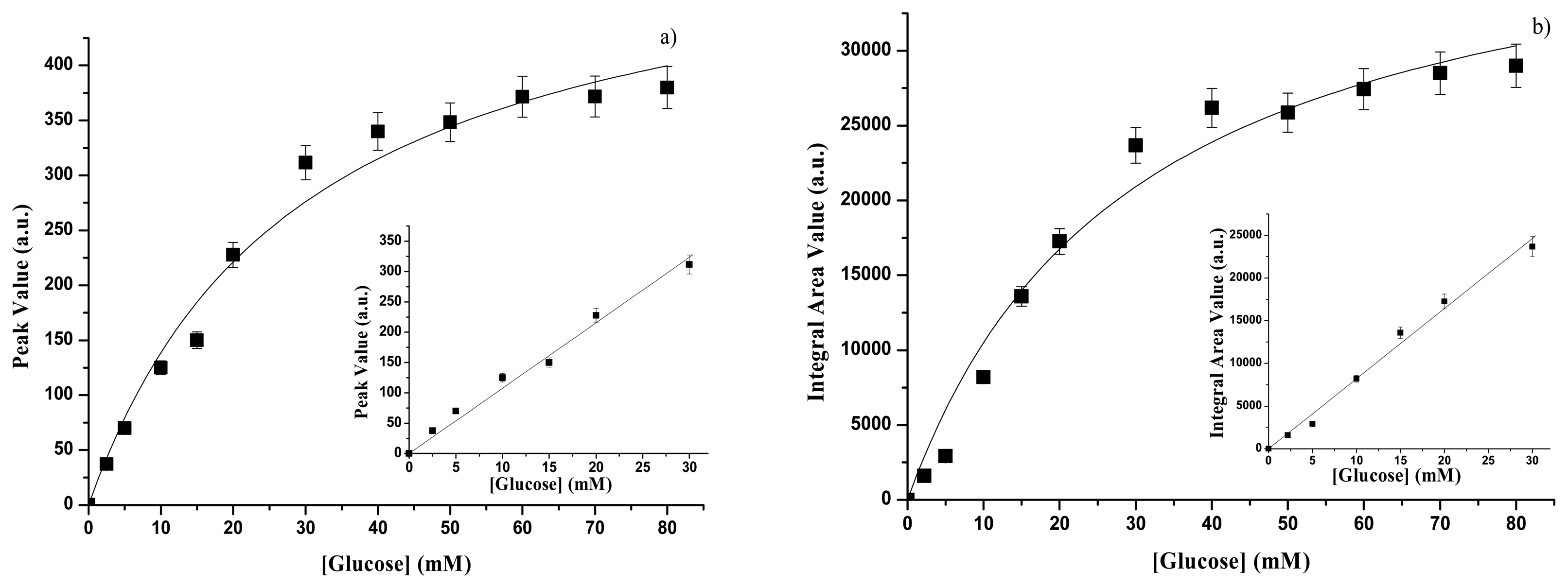
3.1. Calibration curves of glucose concentration through emission spectra
3.1.1. Free GOD
3.1.2. GOD – gelatine membrane
3.2. Calibration curves of glucose concentration through time course measurements
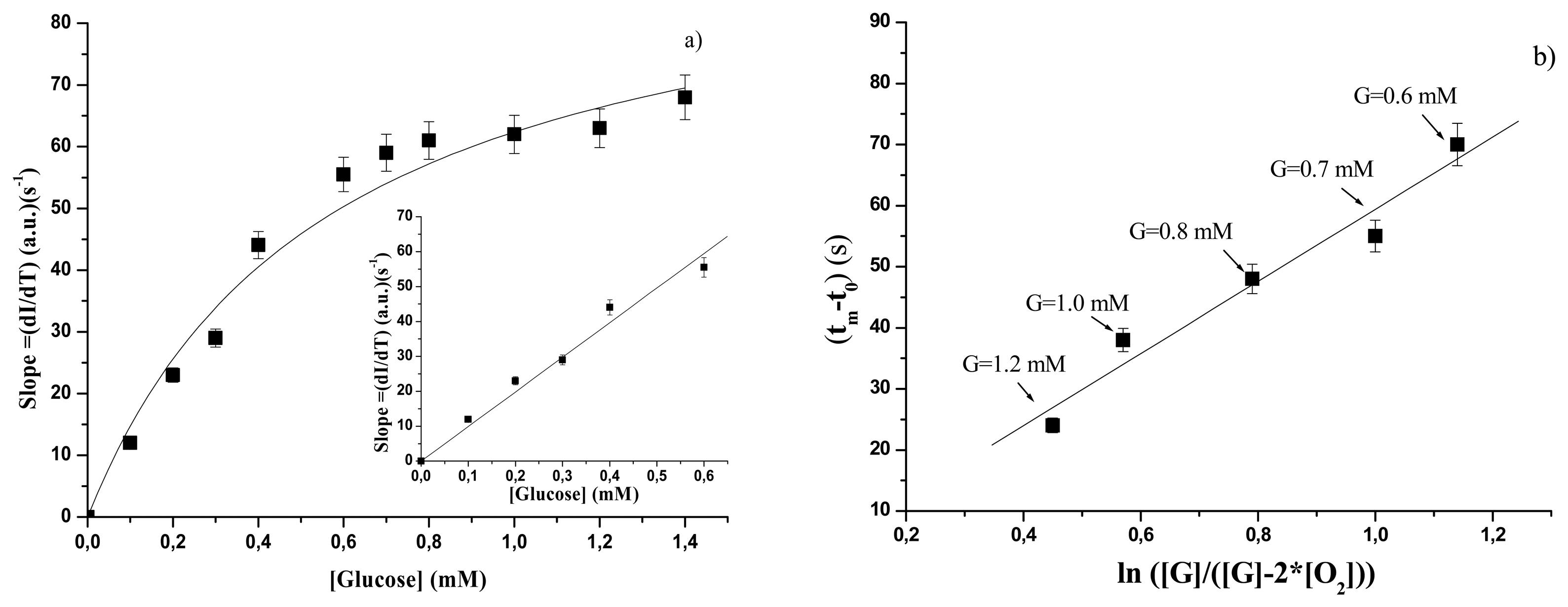
3.2.1 Free GOD
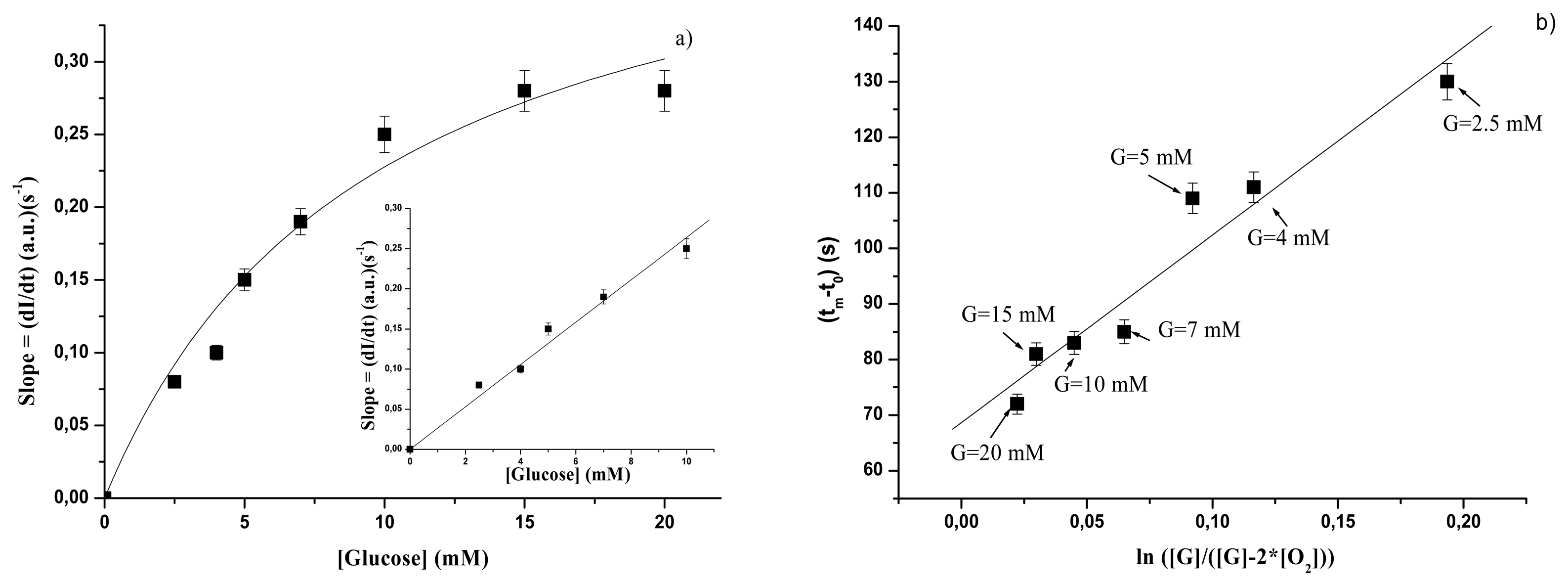
3.2.2 GOD – gelatine membrane
4. Conclusions
References and Notes
- Raba, J.; Mottola, H.A. Glucose oxidase as an analytical reagents. Crit. Rev. Anal. Chem. 1995, 25, 1–42. [Google Scholar]
- Wolfbeis, O.S.; Bohmer, M.; Durkop, A.; Enderlein, J.; Gruber, M.; Klimant, I.; Krause, C.; Kurner, J.; Liebsch, G.; Lin, Z.; Ostwald, B.; Lu, M. Fluorescence Spectroscopy, Imaging and Probes; Kraayenhof, R., Visser, A.J.W.G., Gerritsen, H.G, Eds.; Springer Verlag: Heidelberg, 2002. [Google Scholar]
- McShane, M.J. Potential for glucose monitoring with nanoengineered fluorescent biosensors. Diabetes Technol. Ther. 2002, 4, 533–538. [Google Scholar]
- Srivastava, R.; Brown, J.Q.; Zhu, H.; McShane, M.J. Stable encapsulation of active enzyme by application of multiplayer nanofilm coatings to alginate microspheres. Macromol. Biosci. 2005, 5, 717–727. [Google Scholar]
- Brown, J.Q.; Srivastava, R.; Zhu, H.; McShane, J. Enzymatic Fluorescent Microsfere Glucose Sensors: evaluation of response under dynamic conditions. Diabetes Technol. Ther. 2006, 8, 288–295. [Google Scholar]
- Chinnayelka, S.; McShane, M.J. Glucose-sensitive nanoassemblies comprisingaffinity-binding complexes trapped in fuzzy microshells. J. Fluorescence 2004, 14, 585–595. [Google Scholar]
- Brown, J.Q.; McShane, J.M. Modeling of spherical fluorescent glucose microsensor systems: design of enzymatic smart tattoos. Biosens. Bioelectron. 2004, 21, 212–216. [Google Scholar]
- Koo, Y.E.L.; Cao, Y.; Kopelman, R.; Koo, S.M.; Brasuel, M.; Philbert, M.A. Real-time measurement of dissolved oxygen inside live cells by organically modified silicate fluorescent nanosensors. Anal.Chem. 2004, 76, 2498–2505. [Google Scholar]
- Kuswandi, B.; Andres, R.; Narayanaswamy, A.R. Optical fibre biosensors based on immobilised enzymes. Analyst 2001, 126, 1469–1491. [Google Scholar]
- Marangoni, A.G. Enzyme Kinetics: A Modern Approach; J. Wiley & Sons: New York, 2003. [Google Scholar]
- Cao, L. Carrier-bond Immobilized Enzymes: Principles, Application & Design; J. Wiley & Sons: New York, 2003. [Google Scholar]
- Pickup, J.; Hussain, F.; Evans, N.D.; Rolinski, O.J.; Birch, D.J.S. Fluorescence-based glucose sensors. Biosens. Bioelectron. 2005, 20, 2555–2565. [Google Scholar]
- Pickup, J.; Hussain, F.; Evans, N.D.; Sachedina, N. In vivo glucose monitoring: the clinical reality and the promise. Biosens. Bioelectron. 2005, 20, 1897–1902. [Google Scholar]
- Trettnak, W.; Leiner, M.J.P.; Wolfbeis, O.S. Optical sensors. Part 34. Fibre optic glucose biosensor with an oxygen optrode as the transducer. Analyst 1988, 113, 1519–1523. [Google Scholar]
- Trettnak, W.; Wolfbeis, O.S. Fully reversible fibre-optic glucose biosensor based on the intrinsic fluorescence of glucose oxidase. Analytica Chimica Acta 1989, 211, 196–203. [Google Scholar]
- Lepore, M.; Portaccio, M.; De Tommasi, E.; De Luca, P.; Bencivenga, U.; Maiuri, P.; Mita, D.G. Glucose concentration determination by means of fluorescence emission of soluble and insoluble glucose oxidase: some useful indications for optical fibre-based sensors. J. Mol. Catal. B: Enzym. 2004, 31, 151–158. [Google Scholar]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy; Kluwer Academic Plenum Publisher: New York, 1999. [Google Scholar]
- D'Auria, S.; Lakowicz, J.R. Enzyme fluorescence as a sensing tool: new perspectives in biotechnology. Current Opinion in Biotechnology 2001, 12, 99–104. [Google Scholar]
- Sierra, J.F.; Galban, J.; Castello, J.R. Determination of glucose in blood based on the intrinsic fluorescence of glucose oxidase. Anal. Chem. 1997, 69, 1471–1476. [Google Scholar]
- Haouz, A.; Twist, C.; Zentz, C.; de Kersabiec, A.M.; Pin, S.; Alpert, B. Forster Energy transfer from trytophan to flavin in glucose oxydase enzymes. Chem. Phys. Lett. 1998, 294, 197–203. [Google Scholar]
- Haouz, A.; Glandières, J.M.; Alpert, B. Involvement of protein dynamics in enzyme stability. The case of glucose oxidase. FEBS Letters 2001, 506, 216–220. [Google Scholar]
- White, B.J.; Harmon, H.J. Novel optical solid-state glucose sensor using immobilized glucose oxidase. Biochem. Biophys. Res. Comm. 2002, 296, 1069–1071. [Google Scholar]
- Wolfbeis, O.S.; Oheme, I.; Papkovskaya, N.; Klimant, I. Sol–gel based glucose biosensors employing optical oxygen transducers, and a method for compensating for variable oxygen background. Biosens. Bioelectron. 2000, 15, 69–76. [Google Scholar]
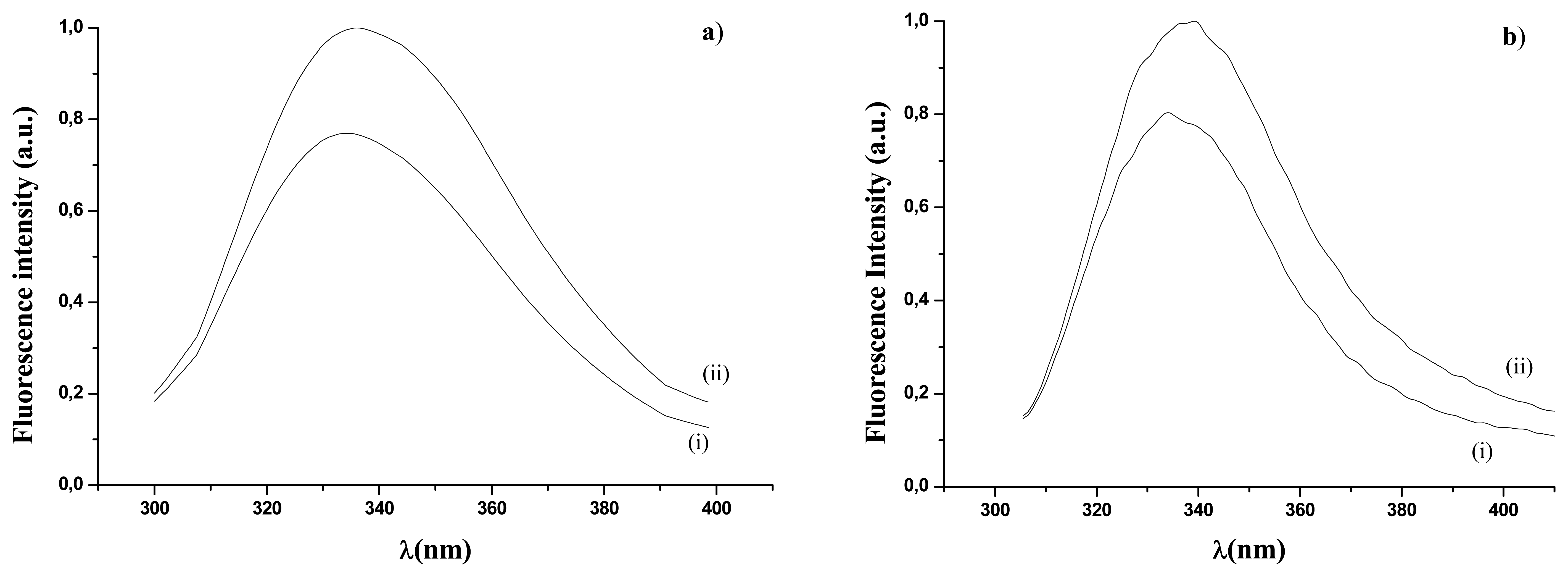
 ), in UV range (□). Fluorescence emission of immobilized GOD in visible range (●), in UV range (▪).Empty and full dots refer to results in reference 16.
), in UV range (□). Fluorescence emission of immobilized GOD in visible range (●), in UV range (▪).Empty and full dots refer to results in reference 16.
 ), in UV range (□). Fluorescence emission of immobilized GOD in visible range (●), in UV range (▪).Empty and full dots refer to results in reference 16.
), in UV range (□). Fluorescence emission of immobilized GOD in visible range (●), in UV range (▪).Empty and full dots refer to results in reference 16.
| GOD form | Spectral range | Kp(mM) | Psat(a.u.) | Linear range | SP (a.u)(mM-1) | Reference |
|---|---|---|---|---|---|---|
| free | UV | 1.87±0.50 | 156±11 | Up to 1.0 mM | 36.3±0.9 | This paper |
| free | VIS | 0.47±0.10 | 46.8±4.7 | Up to 0.5 mM | 54±2 | Ref. 16 |
| immobilized | UV | 27.8±4.9 | 546±37 | Up to 30 mM | 10.7±0.3 | This paper |
| immobilized | VIS | 8.68±1.61 | 90.9±6.2 | Up to 8 mM | 5.6±0.2 | Ref. 16 |
| GOD form | Spectral range | KA(mM) | Asat(a.u) | Linear range | SA (a.u)(mM-1) | Reference. |
|---|---|---|---|---|---|---|
| free | UV | 1.6±0.5 | 11880±960 | Up to 0.8 mM | 1780±29 | This paper |
| free | VIS | 0.47±0.15 | 2962±230 | Up to 0.5 mM | 3530±57 | Ref.16 |
| immobilized | UV | 29.5±7.3 | 41526±4033 | Up to 30 mM | 808.5±22.1 | This paper |
| immobilized | VIS | 8.68±2.15 | 5406±520 | Up to 8 mM | 355±10 | Ref. 16 |
| GOD forms | KSl (s-1 mM) | Slsat (a.u.)(s-1) | SSl (a.u)(s-1mM-1) | Linear range |
|---|---|---|---|---|
| free | 0.56±0.10 | 97±8 | 99.1±3.9 | Up to 0.6 mM |
| immobilized | 9.69±2.38 | 0.44±0.05 | 0.027±0.001 | Up to 10 mM |
| GOD forms | STC (s) | TC Linear range |
|---|---|---|
| free | 59±8 | 0.6 – 1.2 mM |
| immobilized | 337.8±43.6 | 2.5 – 20 mM |
© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
De Luca, P.; Lepore, M.; Portaccio, M.; Esposito, R.; Rossi, S.; Bencivenga, U.; Mita, D.G. Glucose Determination by Means of Steady-state and Time-course UV Fluorescence in Free or Immobilized Glucose Oxidase. Sensors 2007, 7, 2612-2625. https://doi.org/10.3390/s7112612
De Luca P, Lepore M, Portaccio M, Esposito R, Rossi S, Bencivenga U, Mita DG. Glucose Determination by Means of Steady-state and Time-course UV Fluorescence in Free or Immobilized Glucose Oxidase. Sensors. 2007; 7(11):2612-2625. https://doi.org/10.3390/s7112612
Chicago/Turabian StyleDe Luca, Paola, Maria Lepore, Marianna Portaccio, Rosario Esposito, Sergio Rossi, Umberto Bencivenga, and Damiano Gustavo Mita. 2007. "Glucose Determination by Means of Steady-state and Time-course UV Fluorescence in Free or Immobilized Glucose Oxidase" Sensors 7, no. 11: 2612-2625. https://doi.org/10.3390/s7112612
APA StyleDe Luca, P., Lepore, M., Portaccio, M., Esposito, R., Rossi, S., Bencivenga, U., & Mita, D. G. (2007). Glucose Determination by Means of Steady-state and Time-course UV Fluorescence in Free or Immobilized Glucose Oxidase. Sensors, 7(11), 2612-2625. https://doi.org/10.3390/s7112612




