Using Synchrotron Radiation-Based Infrared Microspectroscopy to Reveal Microchemical Structure Characterization: Frost Damaged Wheat vs. Normal Wheat
Abstract
:1. Introduction
2. Results and Discussion
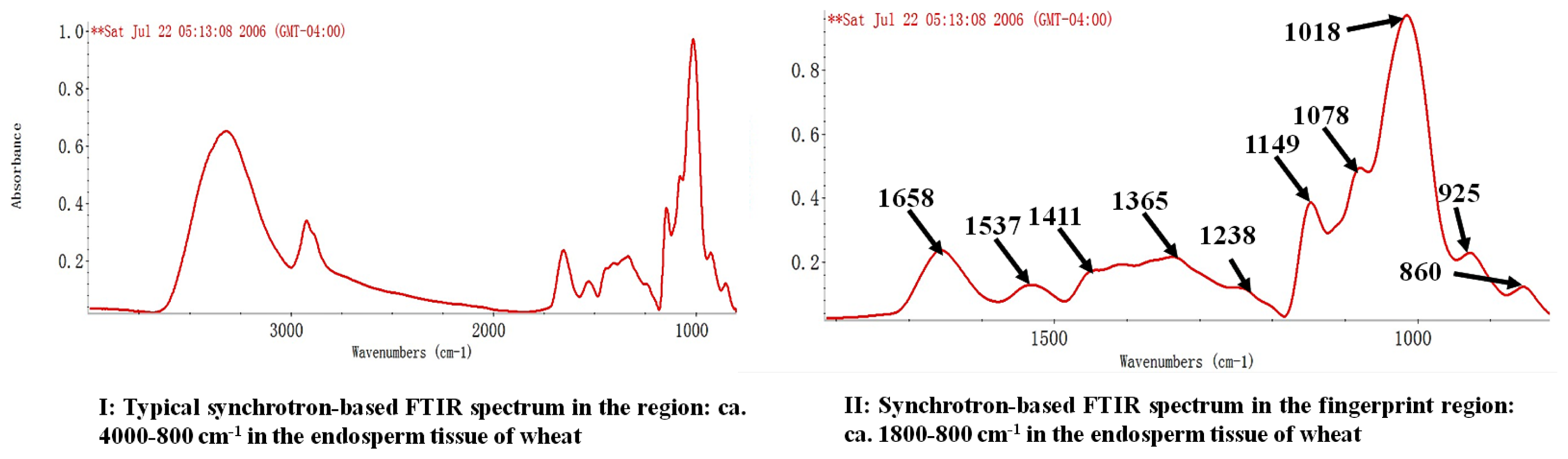
2.1. Univariate Molecular Spectral Analysis of Protein Amide and Protein Secondary Structure Profiles in Endosperm Tissue of Normal and Frost Damaged Wheat Grains
2.2. Univariate Molecular Spectral Analysis of Carbohydrate Conformation Profile in Endosperm Tissue of Normal and Frost Damaged Wheat Samples
2.3. Univariate Molecular Spectral Analysis of Spectral Ratios of Protein Amide and Individual Carbohydrate Spectral Parameter in Endosperm Tissue of Normal and Frost Damaged Wheat Samples
3. Experimental Section
3.1. Wheat Samples
3.2. Advanced Synchrotron Radiation-Based FTIR Microspectroscopy
3.3. Univariate Spectral Analysis
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Dexter, J.E.; Martin, D.G.; Preston, K.R.; Tipples, K.H.; MacGregor, A.W. The effect of frost damage on the milling and baking quality of red spring wheat. Cereal Chem 1985, 62, 75–80. [Google Scholar]
- Allen, H.M.; Pumpa, J.K.; Batten, G.D. Effect of frost on the quality of samples of Janz wheat. Aust. J. Exp. Agric 2001, 41, 641–647. [Google Scholar]
- Yu, P.; Racz, V. Rumen degradation ratios, available protein, and structural and non-structural carbohydrates: Comparison of frost-damaged wheat with normal wheat. Can. J. Anim. Sci 2007, 87, 449–454. [Google Scholar]
- Yu, P.; Racz, V. Chemical characterization, energy values, protein and carbohydrate fractions, degradation kinetics of frost damaged wheat (with severely overall weight loss) in ruminants. Anim. Sci. J 2009, 80, 140–148. [Google Scholar]
- Czerkawski, J.W. An Introduction to Rumen Studies; Pergamon Press: Oxford, UK, 1986; p. 236. [Google Scholar]
- Tamminga, S.; van Vuuren, A.M.; van der Koelen, C.J.; Ketelaar, R.S.; van der Togt, P.L. Ruminal behavior of structural carbohydrates, non-structural carbohydrates and crude protein from concentrate ingredients in dairy cows. Neth. J. Agric. Sci 1990, 38, 513–526. [Google Scholar]
- Miller, L.M.; Carlson, C.S.; Carr, G.L.; Chance, M.R. A method for examining the chemical basis for bone disease: Synchrotron infrared microspectroscopy. Cell. Mol. Biol 1998, 44, 117–127. [Google Scholar]
- Holman, H.N.; Bjornstad, K.A.; McNamara, M.P.; Martin, M.C.; McKinney, W.R.; Blakely, E.A. Synchrotron infrared spectromicroscopy as a novel bioanalytical microprobe for individual living cells: Cytotoxicity considerations. J. Biomed. Opt 2002, 7, 1–10. [Google Scholar]
- Wetzel, D.L.; Eilert, A.J.; Pietrzak, L.N.; Miller, S.S.; Sweat, J.A. Ultraspatially resolved synchrotron infrared microspectroscopy of plant tissue in situ. Cell. Mol. Biol 1998, 44, 145–167. [Google Scholar]
- Miller, L.M. The impact of infrared synchrotron radiation on biology: Past, present, and future. Synchrotron Rad. News 2000, 13, 31–37. [Google Scholar]
- Marinkovic, N.S.; Huang, R.; Bromberg, P.; Sullivan, M.; Toomey, J.; Miller, L.M.; Sperber, E.; Moshe, S.; Jones, K.W.; Chouparova, E. Center for Synchrotron Biosciences’ U2B beamline: An international resource for biological infrared spectroscopy. J. Synchrotron Rad 2002, 9, 189–197. [Google Scholar]
- Yu, P.; Christensen, D.A.; Christensen, C.R.; Drew, M.D.; Rossnagel, B.G.; McKinnon, J.J. Use of synchrotron FTIR microspectroscopy to identify chemical differences in barley endosperm tissue in relation to rumen degradation characteristics. Can. J. Anim. Sci 2004, 84, 523–527. [Google Scholar]
- Severcan, F.; Haris, P.I. Vibrational Spectroscopy in Diagnosis and Screening-Advances in Biomedical Spectroscopy; IOS Press: Amsterdam, The Netherlands, 2012; pp. 386–418. [Google Scholar]
- Liu, B.; McKinnon, J.J.; Thacker, P.; Yu, P. Molecular structure and metabolic characteristics of the proteins and energy in triticale grains and dried distillers grains with solubles for dairy cattle. J. Agric. Food Chem 2012, 60, 10064–10074. [Google Scholar]
- Yu, P. Short communication: Relationship of carbohydrate molecular spectroscopic features to carbohydrate nutrient profiles in co-products from bioethanol production. J. Dairy Sci 2012, 95, 2091–2096. [Google Scholar]
- Yu, P.; Nuez-Ortín, W.G. Relationship of protein molecular structure to metabolisable proteins in different types of dried distillers grains with solubles: A novel approach. Br. J. Nutr 2010, 104, 1429–1437. [Google Scholar]
- Dyson, H.J.; Wright, P.E. Peptide conformation and protein folding. Curr. Opin. Struct. Biol 1990, 3, 60–64. [Google Scholar]
- Carey, F.A. Organic Chemistry, 3rd ed; Mcgraw-Hill: New York, NY, USA; p. 1996.
- Yu, P.; Block, H.C.; Doiron, K. Understanding the differences in molecular conformation of carbohydrate and protein in endosperm tissues of grains with different biodegradation kinetics using advanced synchrotron technology. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc 2009, 71, 1837–1844. [Google Scholar]
- Preston, K.R.; Kilborn, R.H.; Morgan, B.C.; Babb, J.C. Effects of frost and immaturity on the quality of a Canadian hard red spring wheat. Cereal Chem 1991, 68, 133–138. [Google Scholar]
- Mathloutyhi, M.; Koening, J.L. Vibrational spectra of carbohydrates. Adv. Carbohydr. Chem. Biochem 1986, 44, 7–89. [Google Scholar]
- Cromey, M.G.; Wright, D.S.C.; Boddington, H.J. Effects of frost during grain filling on wheat yield and grain structure. N. Z. J. Crop Horticult. Sci 1998, 26, 279–290. [Google Scholar]
- Nocek, J.E.; Tamminga, S. Site of digestion of starch in the gastrointestinal tract of dairy cows and its effect on milk yield and composition. J. Dairy Sci 1991, 74, 3598–3629. [Google Scholar]
- Newton, R.; McCalla, A.G. Effect of frost on wheat at progressive stages of maturity. III. Milling and baking quality. Can. J. Res. Sect. C 1935, 13, 263–282. [Google Scholar]
- Liu, N.; Yu, P. Characterization of the microchemical structure of seed endosperm within a cellular dimension among six barley varieties with distinct degradation kinetics, using ultraspatially resolved synchrotron-based infrared microspectroscopy. J. Agric. Food Chem 2010, 58, 7801–7810. [Google Scholar]

| Baseline (cm−1) | Peak (cm−1) | Normal wheat | Frost damaged wheat | SEM c | p | |
|---|---|---|---|---|---|---|
| Protein amides profile a | ||||||
| Amide I height | 1774–1475 | ~1658 | 0.13 | 0.11 | 0.012 | 0.50 |
| Amide II height | 1774–1475 | ~1537 | 0.04 | 0.05 | 0.004 | 0.28 |
| Height ratio of amide I to II | 1774–1475 | 3.34 a | 2.50 b | 0.173 | 0.01 | |
| Amide I area | 1774–1475 | ~1658 | 8.81 | 8.77 | 0.910 | 0.97 |
| Amide II area | 1774–1475 | ~1537 | 2.13 | 2.77 | 0.254 | 0.11 |
| Total amide (amide I + II) area | 1774–1475 | 10.94 | 11.54 | 1.128 | 0.72 | |
| Area ratio of amide I to II | 1774–1475 | 4.56 a | 3.28 b | 0.353 | 0.03 | |
| Protein 2nd structure profile b | ||||||
| α-helix | 1774–1475 | ~1658 | 0.12 | 0.11 | 0.012 | 0.55 |
| β-sheet | 1774–1475 | ~1627 | 0.09 | 0.08 | 0.007 | 0.74 |
| Height ratio of α-helix to β-sheet | 1774–1475 | 1.43 | 1.36 | 0.041 | 0.21 | |
| Baseline (cm−1) | Peak (cm−1) | Normal wheat | Frost damaged wheat | SEM f | p | |
|---|---|---|---|---|---|---|
| Structural carbohydrates (SCHO) profile a | ||||||
| SCHO peak 1 height | 1498–1176 | ~1411 | 0.11 a | 0.09 b | 0.006 | 0.03 |
| SCHO peak 2 height | 1498–1176 | ~1365 | 0.13 a | 0.11 b | 0.007 | 0.03 |
| SCHO peak 3 height | 1498–1176 | ~1238 | 0.08 | 0.07 | 0.002 | 0.10 |
| SCHO area | 1498–1176 | 26.19 a | 22.19 b | 1.168 | 0.04 | |
| Cellulosic compounds (CELC) profile b | ||||||
| CELC height | 1295–1176 | ~1236 | 0.03 | 0.03 | 0.001 | 0.25 |
| CELC area | 1295–1176 | ~1236 | 1.32 | 1.28 | 0.060 | 0.60 |
| Total carbohydrates (CHO) profile c | ||||||
| CHO peak 1 height | 1191–906 | ~1149 | 0.34 a | 0.24 b | 0.026 | 0.02 |
| CHO peak 2 height | 1191–906 | ~1078 | 0.45 a | 0.30 b | 0.037 | 0.02 |
| CHO peak 3 height | 1191–906 | ~1018 | 0.79 a | 0.50 b | 0.070 | 0.02 |
| CHO peak1 area | 1191–906 | ~1149 | 13.53 a | 9.41 b | 1.024 | 0.02 |
| CHO peak 2 area | 1191–906 | ~1078 | 15.80 a | 10.57 b | 1.256 | 0.02 |
| CHO peak 3 area | 1191–906 | ~1018 | 53.23 a | 37.06 b | 4.099 | 0.02 |
| Total CHO area | 1191–906 | 82.56 a | 57.05 b | 6.364 | 0.02 | |
| Non-structural carbohydrates (NSCHO) profile d | ||||||
| NSCHO peak 1 height | 954–869 | ~925 | 0.06 a | 0.04 b | 0.004 | 0.01 |
| NSCHO peak 2 height | 890–809 | ~860 | 0.09 | 0.05 | 0.018 | 0.18 |
| NSCHO peak 1 area | 954–869 | ~925 | 2.01 a | 1.37 b | 0.176 | 0.03 |
| NSCHO peak 2 area | 890–809 | ~860 | 2.01 a | 1.16 b | 0.161 | 0.0002 |
| Total NSCHO area | 954–809 | 3.69 a | 2.44 b | 0.277 | 0.01 | |
| Spectral ratio profile e | ||||||
| Height ratio of SCHO peak 1 : 2 | 1498–1176 | 0.83 | 0.83 | 0.013 | 0.90 | |
| Height ratio of SCHO peak 2 : 3 | 1498–1176 | 1.78 a | 1.51 b | 0.062 | 0.01 | |
| Height ratio of SCHO peak 1 : 3 | 1498–1176 | 1.48 a | 1.25b | 0.057 | 0.02 | |
| Height ratio of CHO peak 1 : 2 | 1191–906 | 0.81 | 0.80 | 0.040 | 0.89 | |
| Height ratio of CHO peak 2 : 3 | 1191–906 | 0.58 | 0.60 | 0.011 | 0.18 | |
| Height ratio of CHO peak 1 : 3 | 1191–906 | 0.44 | 0.48 | 0.014 | 0.08 | |
| Area ratio of SCHO : CELC | 1498–1176/1295–1176 | 23.41 | 17.63 | 2.506 | 0.14 | |
| Area ratio of SCHO : CHO | 1498–1176/1191–906 | 0.33 b | 0.40 a | 0.019 | 0.02 | |
| Area ratio of CELC : CHO | 1295–1176/1191–906 | 0.02 b | 0.03 a | 0.002 | 0.01 | |
| Area ratio of CHO peak 1 : 2 | 1191–906 | 0.86 | 0.89 | 0.011 | 0.09 | |
| Area ratio of CHO peak 2 : 3 | 1191–906 | 0.30 | 0.28 | 0.004 | 0.054 | |
| Area ratio of CHO peak 1 : 3 | 1191–906 | 0.25 | 0.25 | 0.004 | 0.68 | |
| Area ratio of SCHO : NSCHO | 1498–1176/954–809 | 7.28 b | 9.19 a | 0.338 | 0.004 | |
| Area ratio of CHO : NSCHO | 1191–906/954–809 | 22.27 | 23.52 | 0.726 | 0.26 | |
| Area ratio of CELC : NSCHO | 1295–1176/954–809 | 0.69 | 0.62 | 0.167 | 0.77 | |
| Baseline (cm−1) | Peak (cm−1) | Normal wheat | Frost damaged wheat | SEM a | p | |
|---|---|---|---|---|---|---|
| Height ratio of amide I : CEL | 1774–1475/1295–1176 | ~1658/~1236 | 4.63 | 4.80 | 0.457 | 0.81 |
| Height ratio of amide I : CHO peak 3 | 1774–1475/1191–906 | ~1658/~1018 | 0.18 b | 0.27 a | 0.023 | 0.03 |
| Height ratio of amide I : NSCHO peak 1 | 1774–1475/954–869 | ~1658/~925 | 2.20 | 3.32 | 0.367 | 0.06 |
| Height ratio of amide I : NSCHO peak 2 | 1774–1475/890–809 | ~1658/~860 | 2.65 | 3.22 | 0.331 | 0.25 |
| Area ratio of total amide : SCHO | 1774–1475/1498–1176 | 0.54 | 0.65 | 0.102 | 0.47 | |
| Area ratio of total amide : CEL | 1774–1475/1295–1176 | 11.81 | 10.46 | 2.647 | 0.73 | |
| Area ratio of total amide : CHO | 1774–1475/1191–906 | 0.15 b | 0.23 a | 0.018 | 0.01 | |
| Area ratio of total amide : NSCHO | 1774–1475/954–809 | 3.22 b | 5.66 a | 0.513 | 0.01 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xin, H.; Zhang, X.; Yu, P. Using Synchrotron Radiation-Based Infrared Microspectroscopy to Reveal Microchemical Structure Characterization: Frost Damaged Wheat vs. Normal Wheat. Int. J. Mol. Sci. 2013, 14, 16706-16718. https://doi.org/10.3390/ijms140816706
Xin H, Zhang X, Yu P. Using Synchrotron Radiation-Based Infrared Microspectroscopy to Reveal Microchemical Structure Characterization: Frost Damaged Wheat vs. Normal Wheat. International Journal of Molecular Sciences. 2013; 14(8):16706-16718. https://doi.org/10.3390/ijms140816706
Chicago/Turabian StyleXin, Hangshu, Xuewei Zhang, and Peiqiang Yu. 2013. "Using Synchrotron Radiation-Based Infrared Microspectroscopy to Reveal Microchemical Structure Characterization: Frost Damaged Wheat vs. Normal Wheat" International Journal of Molecular Sciences 14, no. 8: 16706-16718. https://doi.org/10.3390/ijms140816706




