Chlamydia pneumoniae Infection in Atherosclerotic Lesion Development through Oxidative Stress: A Brief Overview
Abstract
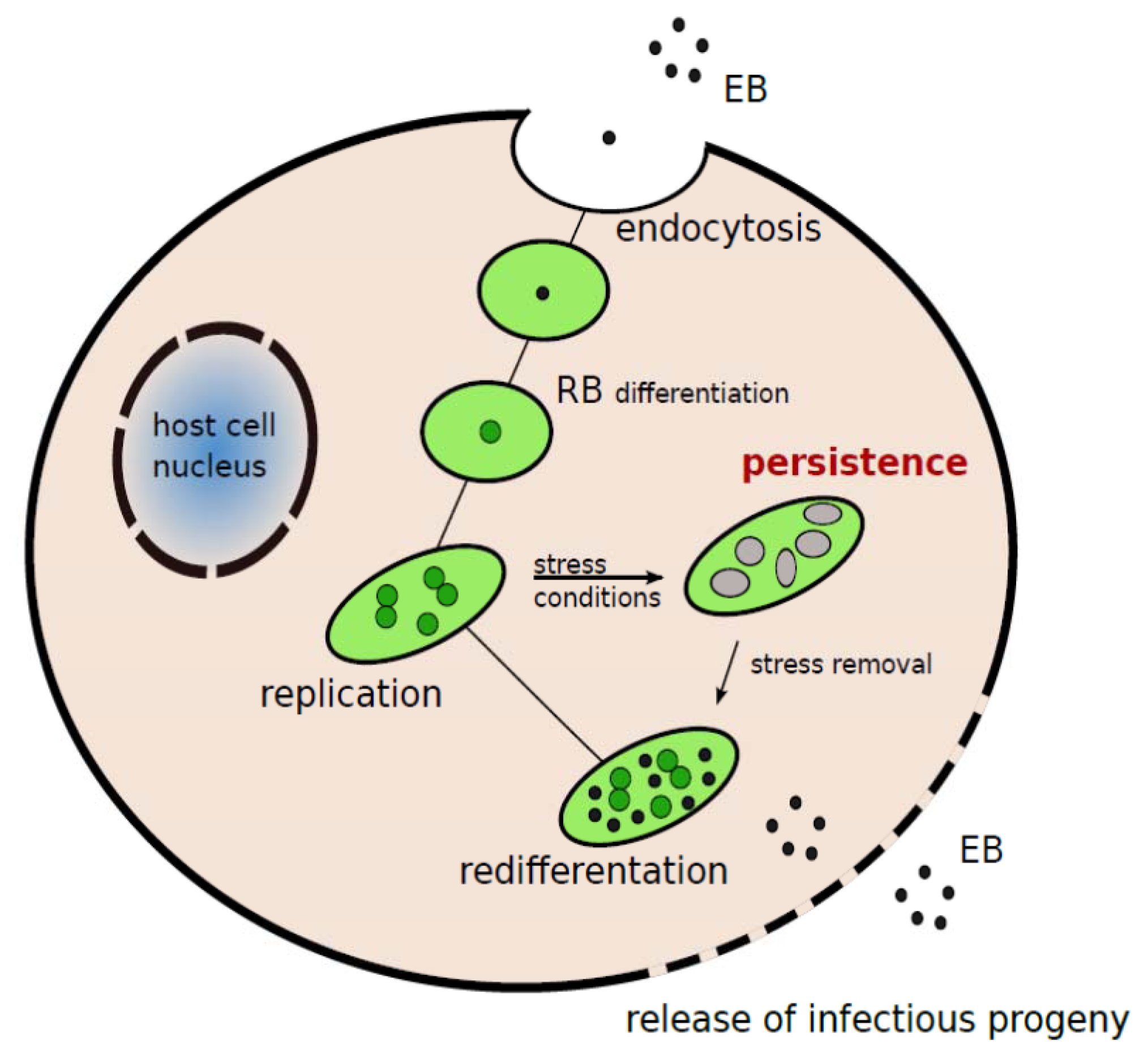
:1. Introduction
2. C. pneumoniae Infection and Atherosclerosis
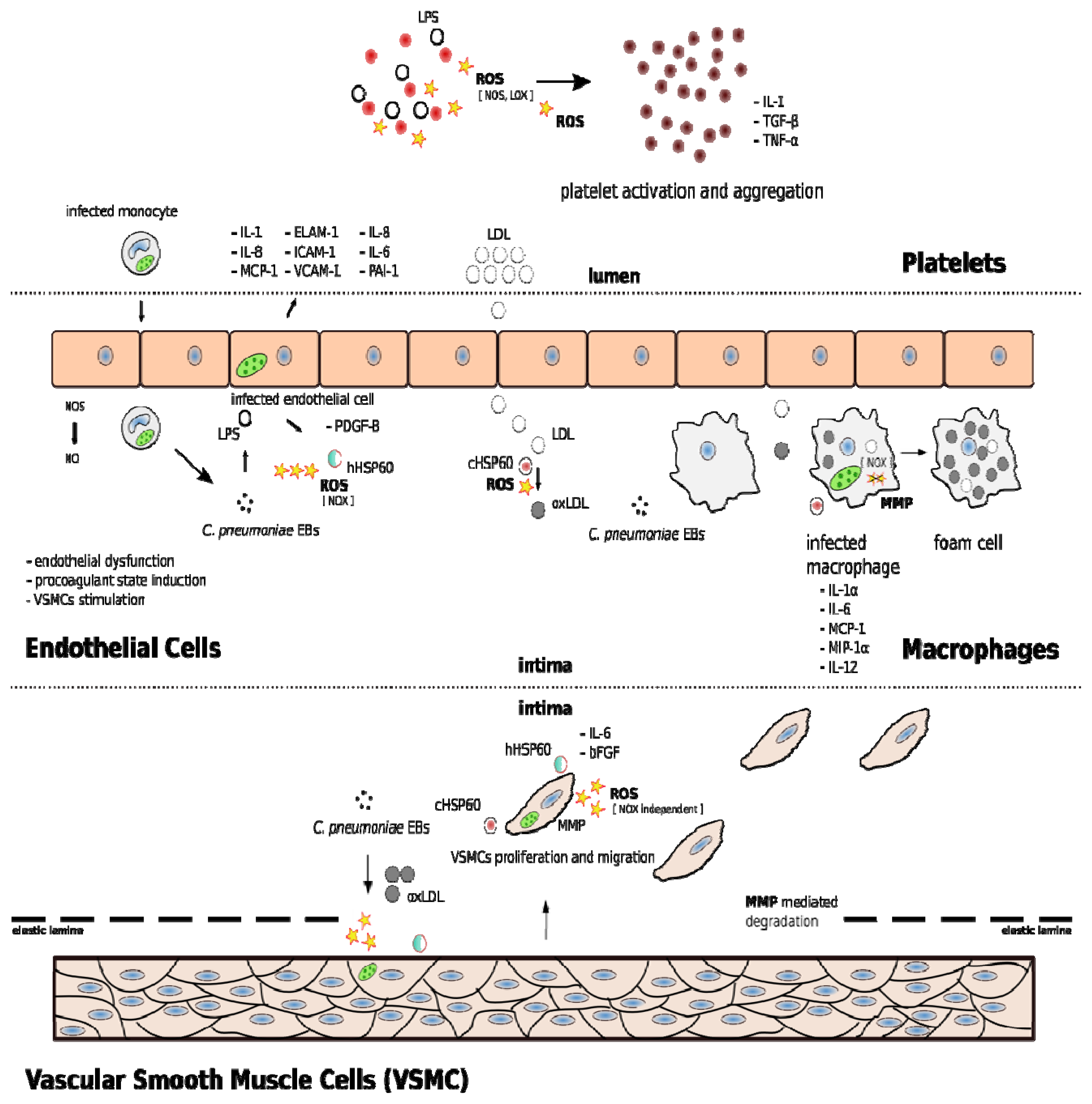
3. C. pneumoniae Induces Oxidative Stress in Vascular Cells
4. Antioxidant Strategies in C. pneumoniae-Mediated Atherosclerosis
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Wong, B.W.; Meredith, A.; Lin, D.; McManus, B.M. The biological role of inflammation in atherosclerosis. Biol. Chem 2012, 393, 1005–1011. [Google Scholar]
- Rosenfeld, M.E.; Campbell, L.A. Pathogens and atherosclerosis: Update on the potential contribution of multiple infectious organisms to the pathogenesis of atherosclerosis. Thromb. Haemost 2011, 106, 858–867. [Google Scholar]
- Ramirez, J.A. Isolation of Chlamydia pneumoniae from the coronary artery of a patient with coronary atherosclerosis. The Chlamydia pneumoniae/Atherosclerosis Study Group. Ann. Intern. Med 1996, 125, 979–982. [Google Scholar]
- Jackson, L.A.; Campbell, L.A.; Kuo, C.C.; Rodriguez, D.I.; Lee, A.; Grayston, J.T. Isolation of Chlamydia pneumoniae from a carotid endarterectomy specimen. J. Infect. Dis 1997, 176, 292–295. [Google Scholar]
- Maass, M.; Bartels, C.; Engel, P.M.; Mamat, U.; Sievers, H.H. Endovascular presence of viable Chlamydia pneumoniae is a common phenomenon in coronary artery disease. J. Am. Coll. Cardiol 1998, 31, 827–832. [Google Scholar]
- Apfalter, P.; Loidl, M.; Nadrchal, R.; Makristathis, A.; Rotter, M.; Bergmann, M.; Polterauer, P.; Hirschl, A.M. Isolation and continuous growth of Chlamydia pneumoniae from arterectomy specimens. Eur. J. Clin. Microbiol. Infect. Dis 2000, 19, 305–308. [Google Scholar]
- Schoborg, R.V. Chlamydia persistence—A tool to dissect Chlamydia-host interactions. Microbes. Infect 2011, 13, 649–662. [Google Scholar]
- Costa, C.P.; Kirschning, C.J.; Busch, D.; Dürr, S.; Jennen, L.; Heinzmann, U.; Prebeck, S.; Wagner, H.; Miethke, T. Role of chlamydial heat shock protein 60 in the stimulation of innate immune cells by Chlamydia pneumoniae. Eur. J. Immunol 2002, 32, 2460–2470. [Google Scholar]
- Kalayoglu, M.V. Chlamydial heat shock protein 60 and lipopolysaccharide: Potential virulence determinants in atherogenesis. Curr. Drug Targets Inflamm. Allergy 2002, 1, 249–255. [Google Scholar]
- Jha, H.C.; Srivastava, P.; Prasad, J.; Mittal, A. Chlamydia pneumoniae heat shock protein 60 enhances expression of ERK, TLR-4 and IL-8 in atheromatous plaques of coronary artery disease patients. Immunol. Invest 2011, 40, 206–222. [Google Scholar]
- Münzel, T.; Gori, T.; Bruno, R.M.; Taddei, S. Is oxidative stress a therapeutic target in cardiovascular disease? Eur. Heart J 2010, 31, 2741–2748. [Google Scholar]
- Chen, K.; Keaney, J.F., Jr. Evolving concepts of oxidative stress and reactive oxygen species in cardiovascular disease. Curr. Atheroscler. Rep. 2012, 14, 476–483. [Google Scholar]
- Azenabor, A.A.; Yang, S.; Job, G.; Adedokun, O.O. Elicitation of reactive oxygen species in Chlamydia pneumoniae-stimulated macrophages: A Ca2+-dependent process involving simultaneous activation of NADPH oxidase and cytochrome oxidase genes. Med. Microbiol. Immunol 2005, 194, 91–103. [Google Scholar]
- Kälvegren, H.; Bylin, H.; Leanderson, P.; Richter, A.; Grenegård, M.; Bengtsson, T. Chlamydia pneumoniae induces nitric oxide synthase and lipoxygenase-dependent production of reactive oxygen species in platelets. Effects on oxidation of low density lipoproteins. Thromb. Haemost 2005, 94, 327–335. [Google Scholar]
- Chahine, M.N.; Deniset, J.; Dibrov, E.; Hirono, S.; Blackwood, D.P.; Austria, J.A.; Pierce, G.N. Oxidized LDL promotes the mitogenic actions of Chlamydia pneumoniae in vascular smooth muscle cells. Cardiovasc. Res 2011, 92, 476–483. [Google Scholar]
- Kreutmayer, S.; Csordas, A.; Kern, J.; Maass, V.; Almanzar, G.; Offterdinger, M.; Ollinger, R.; Maass, M.; Wick, G. Chlamydia pneumoniae infection acts as an endothelial stressor with the potential to initiate the earliest heat shock protein 60-dependent inflammatory stage of atherosclerosis. Cell Stress Chaperones 2013, 18, 259–268. [Google Scholar]
- Grayston, J.T. Background and current knowledge of Chlamydia pneumoniae and atherosclerosis. J. Infect. Dis 2000, 181, s402–s410. [Google Scholar]
- Sessa, R.; Di Pietro, M.; Schiavoni, G.; Petrucca, A.; Cipriani, P.; Zagaglia, C.; Nicoletti, M.; Santino, I.; del Piano, M. Measurement of Chlamydia pneumoniae bacterial load in peripheral blood mononuclear cells may be helpful to assess the state of chlamydial infection in patients with carotid atherosclerotic disease. Atherosclerosis 2007, 195, e224–e230. [Google Scholar]
- Wang, S.S.; Tondella, M.L.; Bajpai, A.; Mathew, A.G.; Mehranpour, P.; Li, W.; Kacharava, A.G.; Fields, B.S.; Austin, H.; Zafari, A.M. Circulating Chlamydia pneumoniae DNA and advanced coronary artery disease. Int. J. Cardiol 2007, 118, 215–219. [Google Scholar]
- Di Pietro, M.; Schiavoni, G.; Sessa, V.; Pallotta, F.; Costanzo, G.; Sessa, R. Chlamydia pneumoniae and osteoporosis-associated bone loss: A new risk factor? Osteoporos. Int 2013, 24, 1677–1682. [Google Scholar]
- Di Pietro, M.; Filardo, S.; Cazzavillan, S.; Segala, C.; Bevilacqua, P.; Bonoldi, E.; D’Amore, E.S.; Rassu, M.; Sessa, R. Could past Chlamydial vascular infection promote the dissemination of Chlamydia pneumoniae to the brain? J. Biol. Regul. Homeost. Agents 2013, 27, 155–164. [Google Scholar]
- Moulder, J.W. Interaction of chlamydiae and host cells in vitro. Microbiol. Rev 1991, 55, 143–190. [Google Scholar]
- Hogan, R.J.; Mathews, S.A.; Mukhopadhyay, S.; Summersgill, J.T.; Timms, P. Chlamydial persistence: Beyond the biphasic paradigm. Infect. Immun 2004, 72, 1843–1855. [Google Scholar]
- Di Pietro, M.; Tramonti, A.; de Santis, F.; de Biase, D.; Schiavoni, G.; Filardo, S.; Zagaglia, C.; Sessa, R. Analysis of gene expression in penicillin G induced persistence of Chlamydia pneumoniae. J. Biol. Regul. Homeost. Agents 2012, 26, 277–284. [Google Scholar]
- Di Pietro, M.; de Santis, F.; de Biase, D.; Sessa, R. The elusive but pathogenic peptidoglycan of Chlamydiae. Eur. J. Inflamm 2013, 11, 257–260. [Google Scholar]
- Gieffers, J.; Füllgraf, H.; Jahn, J.; Klinger, M.; Dalhoff, K.; Katus, H.A.; Solbach, W.; Maass, M. Chlamydia pneumoniae infection in circulating human monocytes is refractory to antibiotic treatment. Circulation 2001, 103, 351–356. [Google Scholar]
- Deniset, J.F.; Pierce, G.N. Possibilities for therapeutic interventions in disrupting Chlamydophila pneumoniae involvement in atherosclerosis. Fundam. Clin. Pharmacol 2010, 24, 607–617. [Google Scholar]
- Saikku, P.; Leinonen, M.; Mattila, K.; Ekman, M.R.; Nieminen, M.S.; Mäkelä, P.H.; Huttunen, J.K.; Valtonen, V. Serological evidence of an association of a novel Chlamydia, TWAR, with chronic coronary heart disease and acute myocardial infarction. Lancet 1988, 2, 983–986. [Google Scholar]
- Sessa, R.; Di Pietro, M.; Santino, I.; del Piano, M.; Varveri, A.; Dagianti, A.; Penco, M. Chlamydia pneumoniae infection and atherosclerotic coronary disease. Am. Heart J 1999, 137, 1116–1119. [Google Scholar]
- Boman, J.; Hammerschlag, M.R. Chlamydia pneumoniae and atherosclerosis: Critical assessment of diagnostic methods and relevance to treatment studies. Clin. Microbiol. Rev 2002, 15, 1–20. [Google Scholar]
- Voorend, M.; Faber, C.G.; van der Ven, A.J.; Kessels, F.; Bruggeman, C.A.; Lodder, J. Chlamydia pneumoniae is a likely risk factor for ischemic stroke in young patients. J. Stroke Cerebrovasc. Dis 2004, 13, 85–91. [Google Scholar]
- Jha, H.C.; Srivastava, P.; Sarkar, R.; Prasad, J.; Mittal, A. Chlamydia pneumoniae IgA and elevated level of IL-6 may synergize to accelerate coronary artery disease. J. Cardiol 2008, 52, 140–145. [Google Scholar]
- Swierszcz, J.; Jacek, D.S.; Milewicz, T.; Krzysiek, J.; Sztefko, K.; Galicka-Latała, D. One-year observation of inflammatory markers in patients with aortic valve stenosis who expressed high or low Chlamydia pneumoniae antibody titers. J. Heart Valve Dis 2012, 21, 599–607. [Google Scholar]
- Joshi, R.; Khandelwal, B.; Joshi, D.; Gupta, O.P. Chlamydophila pneumoniae infection and cardiovascular disease. N. Am. J. Med. Sci 2013, 5, 169–181. [Google Scholar]
- Dowell, S.F.; Peeling, R.W.; Boman, J.; Carlone, G.M.; Fields, B.S.; Guarner, J.; Hammerschlag, M.R.; Jackson, L.A.; Kuo, C.C.; Maass, M.; et al. C. pneumoniae Workshop Participants. Standardizing Chlamydia pneumoniae assays: Recommendations from the Centers for Disease Control and Prevention (USA) and the Laboratory Centre for Disease Control (Canada). Clin. Infect. Dis. 2001, 33, 492–503. [Google Scholar]
- Shor, A.; Phillips, J.I. Histological and ultrastructural findings suggesting an initiating role for Chlamydia pneumoniae in the pathogenesis of atherosclerosis. Cardiovasc. J. S. Afr 2000, 11, 16–23. [Google Scholar]
- Sessa, R.; Di Pietro, M.; Schiavoni, G.; Nicoletti, M.; Soda, G.; Nardoni, S.; Bosco, D.; Santino, I.; Cipriani, P.; del Piano, M. Detection of Chlamydia pneumoniae in atherosclerotic coronary arteries. Int. J. Immunopathol. Pharmacol 2004, 17, 301–306. [Google Scholar]
- Virok, D.; Kis, Z.; Kari, L.; Barzo, P.; Sipka, R.; Burian, K.; Nelson, D.E.; Jackel, M.; Kerenyi, T.; Bodosi, M.; et al. Chlamydophila pneumoniae and human cytomegalovirus in atherosclerotic carotid plaques–combined presence and possible interactions. Acta Microbiol. Immunol. Hung 2006, 53, 35–50. [Google Scholar]
- Sessa, R.; di Pietro, M.; Schiavoni, G.; Galdiero, M.; Cipriani, P.; Romano, S.; Zagaglia, C.; Santino, I.; Faccilongo, S.; del Piano, M. Chlamydia pneumoniae in asymptomatic carotid atherosclerosis. Int. J. Immunopathol. Pharmacol 2006, 19, 111–118. [Google Scholar]
- Jha, H.C.; Srivastava, P.; Divya, A.; Prasad, J.; Mittal, A. Prevalence of Chlamydophila pneumoniae is higher in aorta and coronary artery than in carotid artery of coronary artery disease patients. APMIS 2009, 117, 905–911. [Google Scholar]
- Atik, B.; Johnston, S.C.; Dean, D. Association of carotid plaque Lp-PLA(2) with macrophages and Chlamydia pneumoniae infection among patients at risk for stroke. PLoS One 2010, 5. [Google Scholar] [CrossRef]
- Luque, A.; Turu, M.M.; Rovira, N.; Juan-Babot, J.O.; Slevin, M.; Krupinski, J. Early atherosclerotic plaques show evidence of infection by Chlamydia pneumoniae. Front. Biosci 2012, 4, 2423–2432. [Google Scholar]
- Fong, I.W.; Chiu, B.; Viira, E.; Fong, M.W.; Jang, D.; Mahony, J. Rabbit model for Chlamydia pneumoniae infection. J. Clin. Microbiol 1997, 35, 48–52. [Google Scholar]
- Muhlestein, J.B. Chlamydia pneumoniae-induced atherosclerosis in a rabbit model. J. Infect. Dis 2000, 181, S505–S507. [Google Scholar]
- Blessing, E.; Campbell, L.A.; Rosenfeld, M.E.; Chough, N.; Kuo, C.C. Chlamydia pneumoniae infection accelerates hyperlipidemia induced atherosclerotic lesion development in C57BL/6J mice. Atherosclerosis 2001, 158, 13–17. [Google Scholar]
- Ezzahiri, R.; Stassen, F.R.; Kurvers, H.A.; van Pul, M.M.; Kitslaar, P.J.; Bruggeman, C.A. Chlamydia pneumoniae infection induces an unstable atherosclerotic plaque phenotype in LDL-receptor, ApoE double knockout mice. Eur. J. Vasc. Endovasc. Surg 2003, 26, 88–95. [Google Scholar]
- Chen, S.; Shimada, K.; Zhang, W.; Huang, G.; Crother, T.R.; Arditi, M. IL-17A is proatherogenic in high-fat diet-induced and Chlamydia pneumoniae infection-accelerated atherosclerosis in mice. J. Immunol 2010, 185, 5619–5627. [Google Scholar]
- Lin, F.Y.; Lin, Y.W.; Huang, C.Y.; Chang, Y.J.; Tsao, N.W.; Chang, N.C.; Ou, K.L.; Chen, T.L.; Shih, C.M.; Chen, Y.H. GroEL1, a heat shock protein 60 of Chlamydia pneumoniae, induces lectin-like oxidized low-density lipoprotein receptor 1 expression in endothelial cells and enhances atherogenesis in hypercholesterolemic rabbits. J. Immunol 2011, 186, 4405–4414. [Google Scholar]
- Huang, C.Y.; Shih, C.M.; Tsao, N.W.; Chen, Y.H.; Li, C.Y.; Chang, Y.J.; Chang, N.C.; Ou, K.L.; Lin, C.Y.; Lin, Y.W.; et al. GroEL1, from Chlamydia pneumoniae, induces vascular adhesion molecule 1 expression by p37(AUF1) in endothelial cells and hypercholesterolemic rabbit. PLoS One 2012, 7. [Google Scholar] [CrossRef]
- Moazed, T.C.; Kuo, C.; Grayston, J.T.; Campbell, L.A. Murine models of Chlamydia pneumoniae infection and atherosclerosis. J. Infect. Dis 1997, 175, 883–890. [Google Scholar]
- Moazed, T.C.; Kuo, C.C.; Grayston, J.T.; Campbell, L.A. Evidence of systemic dissemination of Chlamydia pneumoniae via macrophages in the mouse. J. Infect. Dis 1998, 177, 1322–1325. [Google Scholar]
- De Kruif, M.D.; van Gorp, E.C.; Keller, T.T.; Ossewaarde, J.M.; ten Cate, H. Chlamydia pneumoniae infections in mouse models: Relevance for atherosclerosis research. Cardiovasc. Res 2005, 65, 317–327. [Google Scholar]
- Little, C.S.; Bowe, A.; Lin, R.; Litsky, J.; Fogel, R.M.; Balin, B.J.; Fresa-Dillon, K.L. Age alterations in extent and severity of experimental intranasal infection with Chlamydophila pneumoniae in BALB/c mice. Infect. Immun 2005, 73, 1723–1734. [Google Scholar]
- Boman, J.; Söderberg, S.; Forsberg, J.; Birgander, L.S.; Allard, A.; Persson, K.; Jidell, E.; Kumlin, U.; Juto, P.; Waldenström, A.; Wadell, G. High prevalence of Chlamydia pneumoniae DNA in peripheral blood mononuclear cells in patients with cardiovascular disease and in middle-aged blood donors. J. Infect. Dis 1998, 178, 274–277. [Google Scholar]
- Sessa, R.; Di Pietro, M.; Schiavoni, G.; Santino, I.; Cipriani, P.; Romano, S.; Penco, M.; del Piano, M. Prevalence of Chlamydia pneumoniae in peripheral blood mononuclear cells in Italian patients with acute ischaemic heart disease. Atherosclerosis 2001, 159, 521–525. [Google Scholar]
- Smieja, M.; Mahony, J.; Petrich, A.; Boman, J.; Chernesky, M. Association of circulating Chlamydia pneumoniae DNA with cardiovascular disease: A systematic review. BMC Infect. Dis. 2002, 2. [Google Scholar] [CrossRef] [Green Version]
- Kern, J.M.; Maass, V.; Maass, M. Chlamydia pneumoniae adversely modulates vascular cell properties by direct interaction with signalling cascades. Thromb. Haemost 2009, 102, 1064–1070. [Google Scholar]
- Di Pietro, M.; Schiavoni, G.; del Piano, M.; Shaik, Y.; Boscolo, P.; Caraffa, A.; Grano, M.; Teté, S.; Conti, F.; Sessa, R. Chlamydia pneumoniae and atherosclerosis: The role of mast cells. J. Biol. Regul. Homeost. Agents 2009, 23, 65–69. [Google Scholar]
- Oksaharju, A.; Lappalainen, J.; Tuomainen, A.M.; Pussinen, P.J.; Puolakkainen, M.; Kovanen, P.T.; Lindstedt, K.A. Pro-atherogenic lung and oral pathogens induce an inflammatory response in human and mouse mast cells. J. Cell. Mol. Med 2009, 13, 103–113. [Google Scholar]
- Kalayoglu, M.V.; Miranpuri, G.S.; Golenbock, D.T.; Byrne, G.I. Characterization of low-density lipoprotein uptake by murine macrophages exposed to Chlamydia pneumoniae. Microbes Infect 1999, 1, 409–418. [Google Scholar]
- Cao, F.; Castrillo, A.; Tontonoz, P.; Re, F.; Byrne, G.I. Chlamydia pneumoniae-induced macrophage foam cell formation is mediated by Toll-like receptor 2. Infect. Immun 2007, 75, 753–759. [Google Scholar]
- Kitazawa, T.; Fukushima, A.; Okugawa, S.; Yanagimoto, S.; Tsukada, K.; Tatsuno, K.; Koike, K.; Kimura, S.; Kishimoto, T.; Shibasaki, Y.; et al. Chlamydophilal antigens induce foam cell formation via c-Jun NH2-terminal kinase. Microbes Infect 2007, 9, 1410–1414. [Google Scholar]
- He, P.; Mei, C.; Cheng, B.; Liu, W.; Wang, Y.; Wan, J. Chlamydia pneumoniae induces macrophage-derived foam cell formation by up-regulating acyl-coenzyme A: Cholesterol acyltransferase 1. Microbes Infect 2009, 11, 157–163. [Google Scholar]
- Liu, W.; He, P.; Cheng, B.; Mei, C.L.; Wang, Y.F.; Wan, J.J. Chlamydia pneumoniae disturbs cholesterol homeostasis in human THP-1 macrophages via JNK-PPARγ dependent signal transduction pathways. Microbes Infect 2010, 12, 1226–1235. [Google Scholar]
- Netea, M.G.; Selzman, C.H.; Kullberg, B.J.; Galama, J.M.; Weinberg, A.; Stalenhoef, A.F.; van der Meer, J.W.; Dinarello, C.A. Acellular components of Chlamydia pneumoniae stimulate cytokine production in human blood mononuclear cells. Eur. J. Immunol 2000, 30, 541–549. [Google Scholar]
- Gaydos, C.A. Growth in vascular cells and cytokine production by Chlamydia pneumoniae. J. Infect. Dis 2000, 181, S473–478. [Google Scholar]
- Kol, A.; Sukhova, G.K.; Lichtman, A.H.; Libby, P. Chlamydial heat shock protein 60 localizes in human atheroma and regulates macrophage tumor necrosis factor-alpha and matrix metalloproteinase expression. Circulation 1998, 98, 300–307. [Google Scholar]
- Di Pietro, M.; de Santis, F.; Schiavoni, G.; Filardo, S.; Sessa, R. Resveratrol in Chlamydia pneumoniae induced foam cell formationand interleukin-17A sythesis. J. Biol. Regul. Homeost. Agents 2013, 27. in press. [Google Scholar]
- Krüll, M.; Klucken, A.C.; Wuppermann, F.N.; Fuhrmann, O.; Magerl, C.; Seybold, J.; Hippenstiel, S.; Hegemann, J.H.; Jantos, C.A.; Suttorp, N. Signal transduction pathways activated in endothelial cells following infection with Chlamydia pneumoniae. J. Immunol 1999, 162, 4834–4841. [Google Scholar]
- Molestina, R.E.; Miller, R.D.; Ramirez, J.A.; Summersgill, J.T. Infection of human endothelial cells with Chlamydia pneumoniae stimulates transendothelial migration of neutrophils and monocytes. Infect. Immun 1999, 67, 1323–1330. [Google Scholar]
- Kothe, H.; Dalhoff, K.; Rupp, J.; Müller, A.; Kreuzer, J.; Maass, M.; Katus, H.A. Hydroxymethylglutaryl coenzyme A reductase inhibitors modify the inflammatory response of human macrophages and endothelial cells infected with Chlamydia pneumoniae. Circulation 2000, 101, 1760–1763. [Google Scholar]
- Högdahl, M.; Söderlund, G.; Kihlström, E. Expression of chemokines and adhesion molecules in human coronary artery endothelial cells infected with Chlamydia (Chlamydophila) pneumoniae. APMIS 2008, 116, 1082–1088. [Google Scholar]
- Fryer, R.H.; Schwobe, E.P.; Woods, M.L.; Rodgers, G.M. Chlamydia species infect human vascular endothelial cells and induce procoagulant activity. J. Investig. Med 1997, 45, 168–174. [Google Scholar]
- Dechend, R.; Maass, M.; Gieffers, J.; Dietz, R.; Scheidereit, C.; Leutz, A.; Gulba, D.C. Chlamydia pneumoniae infection of vascular smooth muscle and endothelial cells activates NF-kappaB and induces tissue factor and PAI-1 expression: A potential link to accelerated arteriosclerosis. Circulation 1999, 100, 1369–1373. [Google Scholar]
- Summersgill, J.T.; Molestina, R.E.; Miller, R.D.; Ramirez, J.A. Interactions of Chlamydia pneumoniae with human endothelial cells. J. Infect. Dis 2000, 181, s479–s482. [Google Scholar]
- Wang, A.; Al-Kuhlani, M.; Johnston, S.C.; Ojcius, D.M.; Chou, J.; Dean, D. Transcription factor complex AP-1 mediates inflammation initiated by Chlamydia pneumoniae infection. Cell Microbiol 2013, 15, 779–794. [Google Scholar]
- Netea, M.G.; Kullberg, B.J.; Galama, J.M.; Stalenhoef, A.F.; Dinarello, C.A.; van der Meer, J.W. Non-LPS components of Chlamydia pneumoniae stimulate cytokine production through Toll-like receptor 2-dependent pathways. Eur. J. Immunol 2002, 32, 1188–1195. [Google Scholar]
- Yang, X.; Coriolan, D.; Schultz, K.; Golenbock, D.T.; Beasley, D. Toll-like receptor 2 mediates persistent chemokine release by Chlamydia pneumoniae-infected vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol 2005, 25, 2308–2314. [Google Scholar]
- Touyz, R.M.; Briones, A.M.; Sedeek, M.; Burger, D.; Montezano, A.C. NOX isoforms and reactive oxygen species in vascular health. Mol. Interv 2011, 11, 27–35. [Google Scholar]
- Azenabor, A.A.; Muili, K.; Akoachere, J.F.; Chaudhry, A. Macrophage antioxidant enzymes regulate Chlamydia pneumoniae chronicity: Evidence of the effect of redox balance on host-pathogen relationship. Immunobiology 2006, 211, 325–339. [Google Scholar]
- Airenne, S.; Surcel, H.M.; Alakärppä, H.; Laitinen, K.; Paavonen, J.; Saikku, P.; Laurila, A. Chlamydia pneumoniae infection in human monocytes. Infect. Immun 1999, 67, 1445–1449. [Google Scholar]
- Mannonen, L.; Kamping, E.; Penttilä, T.; Puolakkainen, M. IFN-gamma induced persistent Chlamydia pneumoniae infection in HL and Mono Mac 6 cells: Characterization by real-time quantitative PCR and culture. Microb. Pathog 2004, 36, 41–50. [Google Scholar]
- Peters, J.; Hess, S.; Endlich, K.; Thalmann, J.; Holzberg, D.; Kracht, M.; Schaefer, M.; Bartling, G.; Klos, A. Silencing or permanent activation: Host-cell responses in models of persistent Chlamydia pneumoniae infection. Cell Microbiol 2005, 7, 1099–1108. [Google Scholar]
- Klos, A.; Thalmann, J.; Peters, J.; Gérard, H.C.; Hudson, A.P. The transcript profile of persistent Chlamydophila (Chlamydia) pneumoniae in vitro depends on the means by which persistence is induced. FEMS Microbiol. Lett 2009, 291, 120–126. [Google Scholar]
- Azenabor, A.A.; Job, G.; Yang, S. Induction of lipoprotein lipase gene expression in Chlamydia pneumoniae-infected macrophages is dependent on Ca2+ signaling events. Biol. Chem 2004, 385, 67–74. [Google Scholar]
- Mei, C.L.; He, P.; Cheng, B.; Liu, W.; Wang, Y.F.; Wan, J.J. Chlamydia pneumoniae induces macrophage-derived foam cell formation via PPAR alpha and PPAR gamma-dependent pathways. Cell Biol. Int 2009, 33, 301–308. [Google Scholar]
- Yaraei, K.; Campbell, L.A.; Zhu, X.; Liles, W.C.; Kuo, C.C.; Rosenfeld, M.E. Chlamydia pneumoniae augments the oxidized low-density lipoprotein-induced death of mouse macrophages by a caspase-independent pathway. Infect. Immun 2005, 73, 4315–4322. [Google Scholar]
- Bouwman, J.J.; Visseren, F.L.; Bevers, L.M.; van der Vlist, W.E.; Bouter, K.P.; Diepersloot, R.J. Azithromycin reduces Chlamydia pneumoniae-induced attenuation of eNOS and cGMP production by endothelial cells. Eur. J. Clin. Invest 2005, 35, 573–582. [Google Scholar]
- Projahn, D.; Koenen, R.R. Platelets: Key players in vascular inflammation. J. Leukoc. Biol 2012, 92, 1167–1175. [Google Scholar]
- Kälvegren, H.; Andersson, J.; Grenegård, M.; Bengtsson, T. Platelet activation triggered by Chlamydia pneumoniae is antagonized by 12-lipoxygenase inhibitors but not cyclooxygenase inhibitors. Eur. J. Pharmacol 2007, 566, 20–27. [Google Scholar]
- Al-Bannawi, A.; Al-Wesebai, K.; Taha, S.; Bakhiet, M. Chlamydia pneumoniae induces chemokine expression by platelets in patients with atherosclerosis. Med. Princ. Pract 2011, 20, 438–443. [Google Scholar]
- Fleissner, F.; Thum, T. Critical role of the nitric oxide/reactive oxygen species balance in endothelial progenitor dysfunction. Antioxid. Redox. Signal 2011, 15, 933–948. [Google Scholar]
- Chen, C.; Chai, H.; Wang, X.; Lin, P.H.; Yao, Q. Chlamydia heat shock protein 60 decreases expression of endothelial nitric oxide synthase in human and porcine coronary artery endothelial cells. Cardiovasc. Res 2009, 83, 768–777. [Google Scholar]
- Wick, G.; Knoflach, M.; Xu, Q. Autoimmune and inflammatory mechanisms in atherosclerosis. Annu. Rev. Immunol 2004, 22, 361–403. [Google Scholar]
- Nazzal, D.; Cantero, A.V.; Therville, N.; Segui, B.; Negre-Salvayre, A.; Thomsen, M.; Benoist, H. Chlamydia pneumoniae alters mildly oxidized low-density lipoprotein-induced cell death in human endothelial cells, leading to necrosis rather than apoptosis. J. Infect. Dis 2006, 193, 136–145. [Google Scholar]
- Rivera, J.; Walduck, A.K.; Strugnell, R.A.; Sobey, C.G.; Drummond, G.R. Chlamydia pneumoniae induces a pro-inflammatory phenotype in murine vascular smooth muscle cells independently of elevating reactive oxygen species. Clin. Exp. Pharmacol. Physiol 2012, 39, 218–226. [Google Scholar]
- Hirono, S.; Dibrov, E.; Hurtado, C.; Kostenuk, A.; Ducas, R.; Pierce, G.N. Chlamydia pneumoniae stimulates proliferation of vascular smooth muscle cells through induction of endogenous heat shock protein 60. Circ. Res 2003, 93, 710–716. [Google Scholar]
- Zhang, L.; Li, X.; Zhang, L.; Wang, B.; Zhang, T.; Ye, J. Chlamydophila (Chlamydia) pneumoniae infection promotes vascular smooth muscle cell adhesion and migration through IQ domain GTPase-activating protein 1. Microb. Pathog 2012, 53, 207–213. [Google Scholar]
- Sasu, S.; LaVerda, D.; Qureshi, N.; Golenbock, D.T.; Beasley, D. Chlamydia pneumoniae and chlamydial heat shock protein 60 stimulate proliferation of human vascular smooth muscle cells via toll-like receptor 4 and p44/p42 mitogen-activated protein kinase activation. Circ. Res 2001, 89, 244–250. [Google Scholar]
- Deby-Dupont, G.; Mouithys-Mickalad, A.; Serteyn, D.; Lamy, M.; Deby, C. Resveratrol and curcumin reduce the respiratory burst of Chlamydia-primed THP-1 cells. Biochem. Biophys. Res. Commun 2005, 333, 21–27. [Google Scholar]
- Mouithys-Mickalad, A.; Deby-Dupont, G.; Dogne, J.M.; de Leval, X.; Kohnen, S.; Navet, R.; Sluse, F.; Hoebeke, M.; Pirotte, B.; Lamy, M. Effects of COX-2 inhibitors on ROS produced by Chlamydia pneumoniae-primed human promonocytic cells (THP-1). Biochem. Biophys. Res. Commun 2004, 325, 1122–1130. [Google Scholar]
- Dechend, R.; Gieffers, J.; Dietz, R.; Joerres, A.; Rupp, J.; Luft, F.C.; Maass, M. Hydroxymethylglutaryl coenzyme A reductase inhibition reduces Chlamydia pneumoniae-induced cell interaction and activation. Circulation 2003, 108, 261–265. [Google Scholar]
- Prochnau, D.; Rödel, J.; Prager, K.; Kuersten, D.; Heller, R.; Straube, E.; Figulla, H.R. Induced expression of lectin-like oxidized ldl receptor-1 in vascular smooth muscle cells following Chlamydia pneumoniae infection and its down-regulation by fluvastatin. Acta Microbiol. Immunol. Hung 2010, 57, 147–155. [Google Scholar]
- Li, H.; Horke, S.; Förstermann, U. Oxidative stress in vascular disease and its pharmacological prevention. Trends Pharmacol. Sci 2013, 34, 313–319. [Google Scholar]
- Cannon, C.P.; Braunwald, E.; McCabe, C.H.; Rader, D.J.; Rouleau, J.L.; Belder, R.; Joyal, S.V.; Hill, K.A.; Pfeffer, M.A.; Skene, A.M. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N. Engl. J. Med 2004, 350, 1495–1504. [Google Scholar]
- Murphy, S.A.; Cannon, C.P.; Wiviott, S.D.; McCabe, C.H.; Braunwald, E. Reduction in recurrent cardiovascular events with intensive lipid-lowering statin therapy compared with moderate lipid-lowering statin therapy after acute coronary syndromes from the PROVE IT-TIMI 22 (Pravastatin or Atorvastatin Evaluation and Infection Therapy-Thrombolysis In Myocardial Infarction 22) trial. J. Am. Coll. Cardiol 2009, 54, 2358–2362. [Google Scholar]
- Sadowitz, B.; Seymour, K.; Costanza, M.J.; Gahtan, V. Statin therapy—Part II: Clinical considerations for cardiovascular disease. Vasc. Endovascular. Surg 2010, 44, 421–433. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Di Pietro, M.; Filardo, S.; De Santis, F.; Sessa, R. Chlamydia pneumoniae Infection in Atherosclerotic Lesion Development through Oxidative Stress: A Brief Overview. Int. J. Mol. Sci. 2013, 14, 15105-15120. https://doi.org/10.3390/ijms140715105
Di Pietro M, Filardo S, De Santis F, Sessa R. Chlamydia pneumoniae Infection in Atherosclerotic Lesion Development through Oxidative Stress: A Brief Overview. International Journal of Molecular Sciences. 2013; 14(7):15105-15120. https://doi.org/10.3390/ijms140715105
Chicago/Turabian StyleDi Pietro, Marisa, Simone Filardo, Fiorenzo De Santis, and Rosa Sessa. 2013. "Chlamydia pneumoniae Infection in Atherosclerotic Lesion Development through Oxidative Stress: A Brief Overview" International Journal of Molecular Sciences 14, no. 7: 15105-15120. https://doi.org/10.3390/ijms140715105




