MicroRNAs in Metal Stress: Specific Roles or Secondary Responses?
Abstract
:1. Introduction
2. Biogenesis and Working Mechanisms of miRNAs
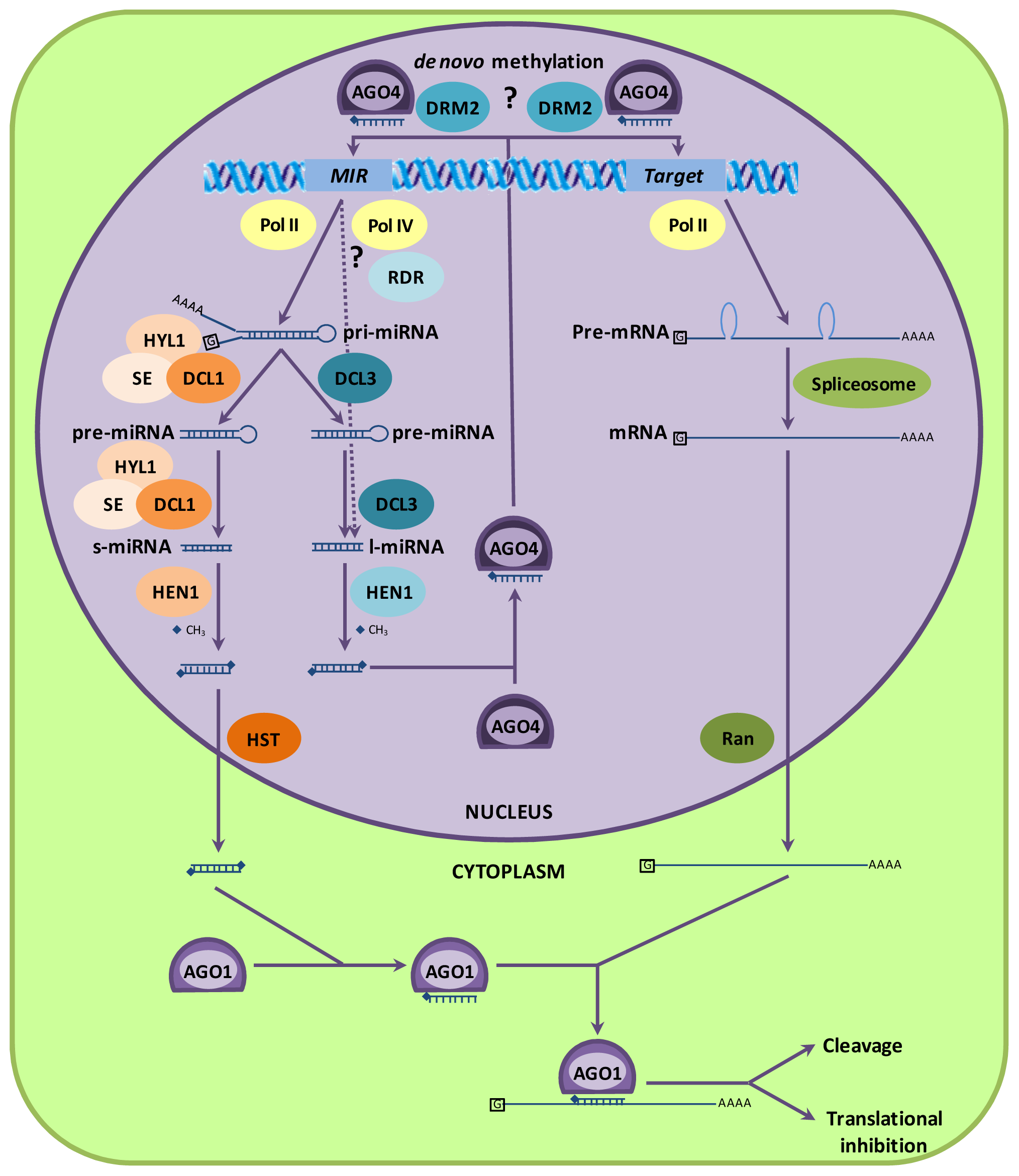
2.1. MicroRNA Biogenesis and Incorporation in Protein Complexes
2.2. MicroRNAs Can Cause DNA Methylation
2.3. Argonaute Proteins and miRNA Function
2.4. MicroRNA Biogenesis Is Regulated by a miRNA Feedback Mechanism
3. The Role of miRNAs in Regulating Their Downstream Targets
3.1. MicroRNA Functioning in Development
3.2. MicroRNA Functioning in Stress Responses
4. The Role of miRNAs in Metal Stress
4.1. Role of miRNAs in Metal Complexation
4.2. Role in Oxidative Stress
4.3. Role in Signal Transduction
4.4. Other Metal Stress Regulated miRNAs
5. Stress-Specific Regulation of miRNAs Is Not Always Straightforwardly Connected to Beneficial Target Gene Regulation
6. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Nriagu, J.O.; Pacyna, J.M. Quantitative assessment of worldwide contamination of air, water and soils by trace metals. Nature 1988, 333, 134–139. [Google Scholar]
- Vangronsveld, J.; van Assche, F.; Clijsters, H. Reclamation of a bare industrial area contaminated by non-ferrous metals: in situ metal immobilization and revegetation. Environ. Pollut 1995, 87, 51–59. [Google Scholar]
- Keunen, E.; Truyens, S.; Bruckers, L.; Remans, T.; Vangronsveld, J.; Cuypers, A. Survival of Cd-exposed Arabidopsis thaliana: Are these plants reproductively challenged? Plant Physiol. Biochem 2011, 49, 1084–1091. [Google Scholar]
- Verbruggen, N.; Hermans, C.; Schat, H. Mechanisms to cope with arsenic or cadmium excess in plants. Curr. Opin. Plant Boil 2009, 12, 364–372. [Google Scholar]
- Lahive, E.; O’Halloran, J.; Jansen, M.A.K. Frond development gradients are determinant of the impact of zinc on photosynthesis in three species of Lemnaceae. Aquat. Bot 2012, 101, 55–63. [Google Scholar]
- Dalcorso, G.; Farinati, S.; Maistri, S.; Furini, A. How plants cope with cadmium: Staking all on metabolism and gene expression. J. Integr. Plant Biol 2008, 50, 1268–1280. [Google Scholar]
- Yruela, I. Copper in plants: Acquisition, transport and interactions. Funct. Plant Biol 2009, 36, 409–430. [Google Scholar]
- Sharma, S.S.; Dietz, K.J. The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci 2009, 14, 43–50. [Google Scholar]
- Hall, J.L. Cellular mechanisms for heavy metal detoxification and tolerance. J. Exp. Bot 2002, 53, 1–11. [Google Scholar]
- Zhang, H.Y.; Xu, W.Z.; Guo, J.B.; He, Z.Y.; Ma, M. Coordinated responses of phytochelatins and metallothioneins to heavy metals in garlic seedlings. Plant Sci 2005, 169, 1059–1065. [Google Scholar]
- Semane, B; Dupae, J.; Cuypers, A.; Noben, J.P.; Tuomainen, M.; Tervahauta, A.; Kärenlampi, S.; van Belleghem, F.; Smeets, K.; Vangronsveld, J. Leaf proteome responses of Arabidopsis thaliana exposed to mild cadmium stress. J. Plant Physiol. 2007, 167, 247–254. [Google Scholar]
- Seth, C.S.; Remans, T.; Keunen, E.; Jozefczak, M.; Gielen, H.; Opdenakker, K.; Weyens, N.; Vangronsveld, J.; Cuypers, A. Phytoextraction of toxic metals: A central role for glutathione. Plant Cell Environ 2012, 35, 334–346. [Google Scholar]
- Smeets, K.; Opdenakker, K.; Remans, T.; van Sanden, S.; van Belleghem, F.; Semane, B.; Horemans, N.; Guisez, Y.; Vangronsveld, J.; Cuypers, A. Oxidative stress-related responses at transcriptional and enzymatic levels after exposure to Cd or Cu in a multipollution context. J. Plant Physiol 2009, 166, 1982–1992. [Google Scholar]
- Cuypers, A.; Smeets, K.; Ruytinx, J.; Opdenakker, K.; Keunen, E.; Remans, T.; Horemans, N.; Vanhoudt, N.; van Sanden, S.; van Belleghem, F.; et al. The cellular redox state as a modulator in cadmium and copper responses in Arabidopsis thaliana seedlings. J. Plant Physiol 2011, 168, 309–316. [Google Scholar]
- Seki, M.; Narusaka, M.; Ishida, J.; Nanjo, T.; Fujita, M.; Oono, Y.; Kamiya, A.; Nakajima, M.; Enju, A.; Sakurai, T.; et al. Monitoring the expression profiles of 7000 Arabidopsis genes under drought, cold and high-salinity stresses using a full-length cDNA microarray. Plant J 2002, 31, 279–292. [Google Scholar]
- Shinozaki, K.; Yamaguch-Shinozaki, K. Gene networks involved in drought stress response and tolerance. J. Exp. Bot 2007, 58, 221–227. [Google Scholar]
- Dos Reis, S.P.; Lima, A.M.; de Souza, C.R.B. Recent molecular advances on downstream plant responses to abiotic stress. Int. J. Mol. Sci 2012, 13, 8628–8647. [Google Scholar]
- Zhang, H.; Zhang, F.; Xia, Y.; Wang, G.; Chen, Z. Excess copper induces production of hydrogen peroxide in the leaf of Elsholtzia haichowensis through apoplastic and symplastic CuZn-superoxide dismutase. J. Hazard. Mater 2010, 178, 834–843. [Google Scholar]
- Drazkiewicz, M.; Skórzynska-Polit, E.; Krupa, Z. Effect of BSO-supplemented heavy metals on atioxidant enzymes in Arabidopsis thaliana. Ecotoxicol. Environ. Saf 2010, 73, 1362–1369. [Google Scholar]
- Remans, T.; Opdenakker, K.; Guisez, Y.; Carleer, R.; Schat, H.; Vangronsveld, J.; Cuypers, A. Exposure of Arabidopsis thaliana to excess Zn reveals a Zn-specific oxidative stress signature. Environ. Exp. Bot 2012, 84, 61–71. [Google Scholar]
- Sunkar, R.; Kapoor, A.; Zhu, J.K. Posttranscriptional induction of two Cu/Zn superoxide dismutase genes in Arabidopsis is mediated by downregulation of miR398 and important for oxidative stress tolerance. Plant Cell 2006, 18, 2051–2065. [Google Scholar]
- Wu, L; Zhou, H.; Zhang, Q.; Zhang, J.; Ni, F.; Liu, C.; Qi, Y. DNA methylation mediated by a microRNA pathway. Mol. Cell 2010, 38, 465–475. [Google Scholar]
- Khraiwesh, B.; Zhu, J.K.; Zhu, J. Role of miRNAs and siRNAs in biotic and abiotic stress responses of plants. Biochim. Biophys. Acta 2012, 1819, 137–148. [Google Scholar]
- Jamalkandi, S.A.; Masoudi-Nejad, A. Reconstruction of Arabidopsis thaliana fully integrated small RNA pathway. Funct. Integr. Genomics 2009, 9, 419–432. [Google Scholar]
- Lee, Y.; Kim, M.; Han, J.; Yeom, K.H.; Lee, S.; Baek, S.H.; Kim, V.N. MicroRNA genes are transcribed by polymerase II. EMBO J 2004, 23, 4051–4060. [Google Scholar]
- Yu, B.; Yang, Z.; Li, J.; Minakhina, S.; Yang, M.; Padgett, R.W.; Steward, R.; Chen, X. Methylation as a crucial step in plant miRNA biogenesis. Science 2005, 307, 932–935. [Google Scholar]
- Kurihara, Y.; Takashi, Y.; Watanabe, Y. The interaction between DCL1 and HYL1 is important for efficient and precise processing of pri-miRNA in plant microRNA biogenesis. RNA 2006, 12, 206–212. [Google Scholar]
- Park, M.Y.; Wu, G.; Gonzalez-Sulser, A.; Vaucheret, H.; Poethig, R.S. Nuclear processing and export of microRNAs in Arabidopsis. Proc. Natl. Acad. Sci. USA 2005, 102, 3691–3696. [Google Scholar]
- Vaucheret, H. Plant argonautes. Trends Plant Sci 2008, 13, 350–358. [Google Scholar]
- Rhoades, M.W.; Reinhart, B.J.; Lim, L.P.; Burge, C.B.; Bartel, B.; Bartel, D.P. Prediction of plant miRNA targets. Cell 2002, 110, 513–520. [Google Scholar]
- Mallory, A.C.; Bouché, N. MicroRNA-directed regulation: To cleave or not to cleave. Trends Plant Sci 2008, 13, 359–367. [Google Scholar]
- Vazquez, F.; Blevins, T.; Ailhas, J.; Boller, T.; Meins, F., Jr. Evolution of Arabidopsis MIR genes generates novel microRNA classes. Nucleic Acids Res. 2008, 36, 6429–6438. [Google Scholar]
- Chellappan, P.; Xia, J.; Zhou, X.; Gao, S.; Zhang, X.; Coutino, G.; Vazquez, F.; Zhang, W.; Jin, H. siRNAs from miRNA sites mediate DNA methylation of target genes. Nucleic Acids Res 2010, 38, 6883–6894. [Google Scholar]
- Matzke, M.; Kanno, T.; Daxinger, L.; Huettel, B.; Matzke, A.J. RNA-mediated chromatin-based silencing in plants. Curr. Opin. Cell Biol 2009, 21, 367–376. [Google Scholar]
- Bao, N.; Lye, K.W.; Barton, M.K. MicroRNA binding sites in Arabidopsis class III HD-ZIP mRNAs are required for methylation of the template chromosome. Dev. Cell 2004, 7, 653–662. [Google Scholar]
- Chinnusamy, V.; Zhu, J.K. Epigenetic regulation of stress responses in plants. Curr. Opin. Plant Biol 2009, 12, 133–139. [Google Scholar]
- Boyko, A.; Kovalchuk, I. Epigenetic control of plant stress response. Environ. Mol. Mutagen 2008, 49, 61–72. [Google Scholar]
- Luo, M.; Liu, X.; Singh, P.; Cui, Y.; Zimmerli, L.; Wu, K. Chromatin modifications and remodeling in plant abiotic stress responses. Biochim. Biophys. Acta 2012, 1819, 129–136. [Google Scholar]
- Aina, R.; Sgorbati, S.; Santagostino, A.; Labra, M.; Ghiani, A.; Citterio, S. Specific hypomethylation of DNA is induced by heavy metals in white clover and industrial hamp. Physiol. Plant 2004, 121, 472–480. [Google Scholar]
- Cao, D.; Gao, X.; Liu, J.; Wang, X.; Geng, S.; Yang, C.; Liu, B.; Shi, D. Root-specific DNA methylation in Chloris virgata, a natural alkaline-resistant halophyte, in response to salt and alkaline stresses. Plant Mol. Biol. Rep 2012, 30, 1102–1109. [Google Scholar]
- Jiang, H.; Kohler, C. Evolution, function and regulation of genomic imprinting in plant seed development. J. Exp. Bot 2012, 63, 4713–4722. [Google Scholar]
- Kuhlmann, M.; Mette, M.F. Developmentally non-redundant SET domain proteins SUVH2 and SUVH9 are required for transcriptional gene silencing in Arabidopsis thaliana. Plant Mol. Biol 2012, 79, 623–633. [Google Scholar]
- Berr, A.; Shafiq, S.; Shen, W.H. Histone modifications in transcriptional activation during plant development. BBA Gene Regul. Mech 2011, 1809, 567–576. [Google Scholar]
- Mallory, A.; Vaucheret, H. Form, function and regulation of ARGONAUTE proteins. Plant Cell 2010, 22, 3879–3889. [Google Scholar]
- Mi, S.; Cai, T.; Hu, Y.; Chen, Y.; Hodges, E.; Ni, F.; Wu, L.; Li, S.; Zhou, H.; Long, C.; et al. Sorting of small RNAs into Arabidopsis Argonaute complexes is directed by the 5′ terminal nucleotide. Cell 2008, 133, 116–127. [Google Scholar]
- Wang, H.; Zhang, X.; Liu, J.; Kiba, T.; Woo, J.; Ojo, T.; Hafner, M.; Tuschl, T.; Chua, N.H.; Wang, X.J. Deep sequencing of small RNAs specifically associated with Arabidopsis AGO1 and AGO4 uncovers new AGO functions. Plant J 2011, 67, 292–304. [Google Scholar]
- Xie, Z.; Kasschau, K.D.; Carrington, J.C. Negative feedback regulation of Dicer-like1 in Arabidopsis by miRNA-guided mRNA degradation. Curr. Biol 2003, 13, 784–789. [Google Scholar]
- Vaucheret, H.; Vazquez, F.; Crété, P.; Bartel, D.P. The action of ARGONAUTE1 in the miRNA pathway and its regulation by the miRNA pathway are crucial for plant development. Gene Dev 2004, 18, 1187–1197. [Google Scholar]
- Jones-Rhoades, M.W.; Bartel, D.P. Computational identification of plant microRNAs and their targets, including a stress-induced miRNA. Mol. Cell 2004, 14, 787–799. [Google Scholar]
- Zhang, B.; Pan, X.; Wang, Q.; Cobb, G.P.; Anderson, T.A. Computational identification of miRNAs and their targets. Comput. Biol. Chem 2006, 30, 395–407. [Google Scholar]
- Addo-Quaye, C.; Eshoo, T.F.; Bartel, D.P.; Axtell, M.J. Endogenous siRNA and miRNA targets identified by sequencing of the Arabidopsis degradome. Curr. Biol 2008, 18, 758–762. [Google Scholar]
- Liu, B.; Li, P.C.; Li, X.; Liu, C.Y.; Cao, S.Y.; Chu, C.C.; Cao, X.F. Loss of function of OsDCL1 affects miRNA accumulation and causes developmental defects in rice. Plant Physiol 2005, 139, 296–305. [Google Scholar]
- Laufs, P.; Peaucelle, A.; Morin, H.; Traas, J. MicroRNA regulation of the CUC genes is required for boundary size control in Arabidopsis meristems. Development 2004, 131, 4311–4322. [Google Scholar]
- Palatnik, J.F.; Allen, E.; Wu, X.; Schommer, C.; Schwab, R.; Carrington, J.C.; Weigel, D. Control of leaf morphogenesis by microRNAs. Nature 2003, 425, 257–263. [Google Scholar]
- Emery, J.F.; Floyd, S.K.; Alvarez, J.; Eshed, Y.; Hawker, N.P.; Izhaki, A.; Baum, S.F.; Bowman, J.L. Radical patterning of Arabidopsis shoots by class III HD-ZIP and KANADI genes. Curr. Biol 2003, 13, 1768–1774. [Google Scholar]
- Mallory, A.C.; Reinhart, B.J.; Jones-Rhoades, M.W.; Tang, G.; Zamore, P.D.; Kathryn Barton, M.; Bartel, D.P. MicroRNA control of PHABULOSA in leaf development: Importance of pairing to the microRNA 5′ region. EMBO J 2004, 23, 3356–3364. [Google Scholar]
- Williams, L.; Grigg, S.P.; Xie, M.; Christensen, S.; Fletcher, J.C. Regulation of Arabidopsis shoot apical meristem and lateral organ formation by microRNA miR166g and its AtHD-ZIP target genes. Development 2005, 132, 3657–3668. [Google Scholar]
- Guo, H.S.; Xie, Q.; Fei, J.F.; Chua, N.H. MicroRNA directs mRNA cleavage of the transcription factor NAC1 to downregulate auxin signals for Arabidopsis lateral root development. Plant Cell 2005, 17, 1376–1386. [Google Scholar]
- Mallory, A.C.; Bartel, D.P.; Bartel, B. MicroRNA-directed regulation of Arabidopsis AUXIN RESPONSE FACTOR17 is essential for proper development and modulates expression of early auxin response genes. Plant Cell 2005, 17, 1360–1375. [Google Scholar]
- Wu, M.F.; Tian, Q.; Reed, J.W. Arabidopsis microRNA167 controls patterns of ARF6 and ARF8 expression, and regulates both female and male reproduction. Development 2006, 133, 4211–4218. [Google Scholar]
- Wu, G.; Poethig, R.S. Temporal regulation of shoot development in Arabidopsis thaliana by miR156 and its target SPL3. Development 2006, 133, 3539–3547. [Google Scholar]
- Lauter, N.; Kampani, A.; Carlson, S.; Goebel, M.; Moose, S.P. microRNA172 down-regulates glossy15 to promote vegetative phase change in maize. Proc. Natl. Acad. Sci. USA 2005, 102, 9412–9417. [Google Scholar]
- Allen, E.; Xie, Z.; Gustafson, A.M.; Carrington, J.C. microRNA-directed phasing during trans-acting siRNA biogenesis in plants. Cell 2005, 121, 207–221. [Google Scholar]
- Kawashima, C.G.; Yoshimoto, M.; Maruyama-Nakashita, A.; Tsuchiya, Y.N.; Saito, K.; Takahashi, H.; Dalmay, T. Sulphur starvation induces the expression of microRNA-395 and one of its targets genes but in different cell types. Plant J 2009, 57, 313–321. [Google Scholar]
- Liang, G.; Yang, F.; Yu, D. microRNA395 mediates regulation of sulfate accumulation and allocation in Arabidopsis thaliana. Plant J 2010, 62, 1046–1057. [Google Scholar]
- Fujii, H.; Chiou, T.J.; Lin, S.I.; Aung, K.; Zhu, J.K. A miRNA involved in phosphate starvation response in Arabidopsis. Curr. Biol 2005, 15, 2038–2043. [Google Scholar]
- Chiou, T.J.; Aung, K.; Lin, S.I.; Wu, C.C.; Chiang, S.F.; Su, C.I. Regulation of phosphate homeostasis by microRNA in Arabidopsis. Plant Cell 2006, 18, 412–421. [Google Scholar]
- Zhou, X.; Wang, G.; Zhang, W. UV-B responsive microRNA genes in Arabidopsis thaliana. Mol. Syst. Biol 2007, 3, 103. [Google Scholar]
- Liu, H.H.; Tian, X.; Li, Y.J.; Wu, C.A.; Zheng, C.C. Microarray-based analysis of stress-regulated microRNAs in Arabidopsis thaliana. RNA 2008, 14, 836–843. [Google Scholar]
- Sunkar, R.; Zhu, J.K. Novel and stress-regulated microRNAs and other small RNAs from Arabidopsis. Plant Cell 2004, 16, 2001–2019. [Google Scholar]
- Huang, S.Q.; Xiang, A.L.; Che, L.L.; Chen, S.; Li, H.; Song, J.B.; Yang, Z.M. A set of miRNAs from Brassica napus in response to sulphate deficiency and cadmium stress. Plant Biotechnol. J 2010, 8, 887–899. [Google Scholar]
- Zhou, Z.S.; Song, J.B.; Yang, Z.M. Genome-wide identification of Brassica napus microRNAs and their targets in response to cadmium. J. Exp. Bot 2012, 63, 4597–4613. [Google Scholar]
- Zhou, Z.S.; Zeng, H.Q.; Liu, Z.P.; Yang, Z.M. Genome-wide identification of Medicago truncatula microRNAs and their targets reveals their differential regulation by heavy metal. Plant Cell Environ 2012, 35, 86–99. [Google Scholar]
- Zhou, Z.S.; Huang, S.Q.; Yang, Z.M. Bioinformatic identification and expression analysis of new microRNAs from Mecicago truncatula. Biochem. Biophys. Res. Commun 2008, 374, 538–542. [Google Scholar]
- Valdes-Lopez, O.; Yang, S.S.; Aparicio-Fabre, R.; Graham, P.H.; Reyes, J.L.; Vance, C.P.; Hernández, G. MicroRNA expression profile in common bean (Phaseolus vulgaris) under nutrient deficiency stresses and manganese toxicity. New Phytol 2010, 187, 805–818. [Google Scholar]
- Burklew, C.E.; Ashlock, J.; Winfrey, W.B.; Zhang, B. Effects of aluminum oxide nanoparticles on the growth, development, and microRNA expression of tobacco (Nicotiana tabacum). PLoS One 2012, 7, e34783. [Google Scholar]
- Xie, F.L.; Huang, S.Q.; Guo, K.; Xiang, A.L.; Zhu, Y.Y.; Nie, L.; Yang, Z.M. Computational identification of novel microRNAs and targets in Brassica napus. FEBS Lett 2007, 581, 1464–1474. [Google Scholar]
- Lima, J.C.; Arenhart, R.A.; Margis-Pinheiro, M.; Margis, R. Aluminum triggers broad changes in microRNA expression in rice roots. Genet. Mol. Res 2011, 10, 2817–2832. [Google Scholar]
- Ding, Y.; Chen, Z.; Zhu, C. Microarray-based analysis of cadmium-responsive microRNAs in rice (Oryza sativa). J. Exp. Bot 2011, 62, 3563–3573. [Google Scholar]
- Liu, Q.; Zhang, H. Molecular identification and analysis of arsenite stress-responsive miRNAs in rice. J. Agric. Food Chem 2012, 60, 6524–6536. [Google Scholar]
- Huang, S.Q.; Peng, J.; Qiu, C.X.; Yang, Z.M. Heavy metal-regulated new miRNAs from rice. J. Inorg. Biochem 2009, 103, 282–287. [Google Scholar]
- Kopriva, S. Regulation of sulfate assimilation in Arabidopsis and beyond. Ann. Bot 2006, 97, 479–495. [Google Scholar]
- Carrasco-Gil, L.; Álvarez-Fernández, A.; Sobrino-Plata, J.; Milán, R.; Carpena-Ruiz, R.O.; Leduc, D.L.; Andrews, J.C.; Abadía, J.; Hernández, L.E. Complexation of Hg with phytochelatins is important for plant Hg tolerance. Plant Cell Environ 2011, 34, 778–791. [Google Scholar]
- Cobbett, C.; Goldsbrough, P. Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu. Rev. Plant Biol 2002, 53, 159–182. [Google Scholar]
- Howden, R.; Goldsbrough, P.B.; Andersen, C.R.; Cobbett, C.S. Cadmium-sensitive, cad1 mutants of Arabidopsis thaliana are phytochelatin deficient. Plant Physiol 1995, 107, 1059–1066. [Google Scholar]
- Yamasaki, H.; Abdel-Ghany, S.E.; Bohu, C.M.; Kobayashi, Y.; Shikanai, T.; Pilon, M. Regulation of copper homeostasis bu micro-RNA in Arabidopsis. J. Biol. Chem 2007, 282, 16369–16378. [Google Scholar]
- Yamasaki, H.; Hayashi, M.; Fukazawa, M.; Kobayashi, Y.; Shikanai, T. SQUAMOSA promoter binding protein-like7 is a central regulator for copper homeostasis in Arabidopsis. Plant Cell 2009, 21, 347–361. [Google Scholar]
- Abdel-Ghany, S.E.; Burkhead, J.L.; Gogolin, K.A.; Andrés-Colás, N.; Bodecker, J.R.; Puig, S.; Peñarrubia, L.; Pilon, M. AtCCS is a functional homolog of the yeast copper chaperone Ccs1/Lys7. FEBS Lett 2005, 579, 2307–2312. [Google Scholar]
- Abdel-Ghany, S.E.; Pilon, M. MicroRNA-mediated systemic down-regulation of copper protein expression in response to low copper availability in Arabidopsis. J. Biol. Chem 2008, 283, 15932–15945. [Google Scholar]
- Opdenakker, K.; Remans, T.; Keunen, E.; Vangronsveld, J.; Cuypers, A. Exposure of Arabidopsis thaliana to Cd or Cu excess leads to oxidative stress mediated alterations in MAPKinase transcript levels. Environ. Exp. Bot 2012, 83, 53–61. [Google Scholar]
- Jonak, C.; Nakagami, H.; Hirt, H. Heavy metal stress. Activation of distinct mitogen-activated protein kinase pathways by copper and cadmium. Plant Physiol 2004, 136, 3276–3283. [Google Scholar]
- Rao, K.P.; Vani, G.; Kumar, K.; Wankhede, D.P.; Misra, M.; Gupta, M.; Sinha, A.K. Arsenic stress activates MAP kinase in rice roots and leaves. Arch. Biochem. Biophys 2011, 506, 73–82. [Google Scholar]
- Smeets, K.; Opdenakker, K.; Remans, T.; Forzani, C.; Hirt, H.; Vangronsveld, J.; Cuypers, C. The role of the kinase OXI1 in cadmium and copper induced molecular responses inArabidopsis thaliana. Plant Cell Environ 2012, in press. [Google Scholar]
- Chen, M.; Meng, Y.; Mao, C.; Chen, D.; Wu, P. Methodological framework for functional characterization of plant microRNAs. J. Exp. Bot 2010, 61, 2271–2280. [Google Scholar]
- Sandmann, G.; Böger, P. Copper-mediated lipid peroxidation processes in photosynthetic membranes. Plant Physiol 1980, 66, 797–800. [Google Scholar]
- Maksymiec, W.; Wianowska, D.; Dawidowicz, A.L.; Radkiewicz, S.; Mardarowicz, M.; Krupa, Z. The level of jasmonic acid in Arabidopsis thaliana and Phaseolus coccineus plants under heavy metal stress. J. Plant Physiol 2005, 162, 1338–1346. [Google Scholar]
- Peto, A.; Lehotai, N.; Lozano-Juste, J.; León, J.; Tari, I.; Erdei, L.; Kolbert, Z. Involvement of nitric oxide and auxin signal transduction of copper-induced morphological responses in Arabidopsis seedlings. Ann. Bot 2011, 108, 449–457. [Google Scholar]
- Maksymiec, W. Signaling responses in plants to heavy metal stress. Acta Physiol. Plant 2007, 29, 177–187. [Google Scholar]
- Schommer, C.; Palatnik, J.F.; Aggarwal, P.; Chételat, A.; Cubas, P.; Farmer, E.E.; Nath, U.; Weigel, D. Control of jasmonate biosynthesis and senescense by miR319 targets. PLoS Biol 2008, 6, e230. [Google Scholar]
- Llave, C.; Kasschau, K.D.; Rector, M.A.; Carrington, J.C. Endogenous and silencing-associated small RNAs in plants. Plant Cell 2002, 14, 1605–1619. [Google Scholar]
- Zhang, Z.L.; Ogawa, M.; Fleet, C.M.; Zentella, R.; Hu, J.; Heo, J.O.; Lim, J.; Kamiya, Y.; Yamaguchi, S.; Sun, T.P. SCARECROW-LIKE3 promotes gibberellin signaling by antagonizing master growth repressor DELLA in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 2160–2165. [Google Scholar]
- Si-Ammour, A.; Windels, D.; Arn-Bouloires, E.; Kutter, C.; Ailhas, J.; Meins, F., Jr; Vazquez, F. MiR393 and secondary siRNAs regulate expression of the TIR1/AFB2 auxin receptor clade and auxin-related development of Arabidopsis leaves. Plant Physiol 2011, 157, 683–691. [Google Scholar]
- Turlapati, P.V.; Kim, K.W.; Davin, L.B.; Lewis, N.G. The laccase multigene family in Arabidopsis thaliana: towards addressing the mystery of their gene function(s). Planta 2011, 233, 439–470. [Google Scholar]
- Liang, M.X.; Davis, E.; Gardner, D.; Cai, X.N.; Wu, Y.J. Involvement of AtLAC15 in lignin synthesis in seeds and in root elongation of Arabidopsis. Planta 2006, 5, 1185–1196. [Google Scholar]
- Berthet, S.; Demont-Caulet, N.; Pollet, B.; Bidzinsky, P.; Cézard, L.; Le Bris, P.; Borrega, N.; Hervé, J.; Blondet, E.; Balzergue, S.; et al. Disruption of LACCASE4 and 17 results in tissue-specific alterations to lignifications of Arabidopsis thaliana stems. Plant Cell 2011, 23, 1124–1137. [Google Scholar]
- Marmiroli, M.; Antonioli, G.; Maestri, E.; Marmiroli, N. Evidence of the involvement of plant lingo-cellulosic structure in the sequestration of Pb: An X-ray spectroscopy-based analysis. Environ. Pollut 2005, 134, 217–227. [Google Scholar]
- Elobeid, M.; Gobel, C.; Feussner, I.; Polle, A. Cadmium interferes with auxin physiology and lignifications in poplar. J. Exp. Bot 2012, 63, 1413–1421. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gielen, H.; Remans, T.; Vangronsveld, J.; Cuypers, A. MicroRNAs in Metal Stress: Specific Roles or Secondary Responses? Int. J. Mol. Sci. 2012, 13, 15826-15847. https://doi.org/10.3390/ijms131215826
Gielen H, Remans T, Vangronsveld J, Cuypers A. MicroRNAs in Metal Stress: Specific Roles or Secondary Responses? International Journal of Molecular Sciences. 2012; 13(12):15826-15847. https://doi.org/10.3390/ijms131215826
Chicago/Turabian StyleGielen, Heidi, Tony Remans, Jaco Vangronsveld, and Ann Cuypers. 2012. "MicroRNAs in Metal Stress: Specific Roles or Secondary Responses?" International Journal of Molecular Sciences 13, no. 12: 15826-15847. https://doi.org/10.3390/ijms131215826
APA StyleGielen, H., Remans, T., Vangronsveld, J., & Cuypers, A. (2012). MicroRNAs in Metal Stress: Specific Roles or Secondary Responses? International Journal of Molecular Sciences, 13(12), 15826-15847. https://doi.org/10.3390/ijms131215826





