Effects of a Ruthenium Schiff Base Complex on Glucose Homeostasis in Diet-Induced Pre-Diabetic Rats
Abstract
:1. Introduction
2. Results
2.1. Caloric Intake
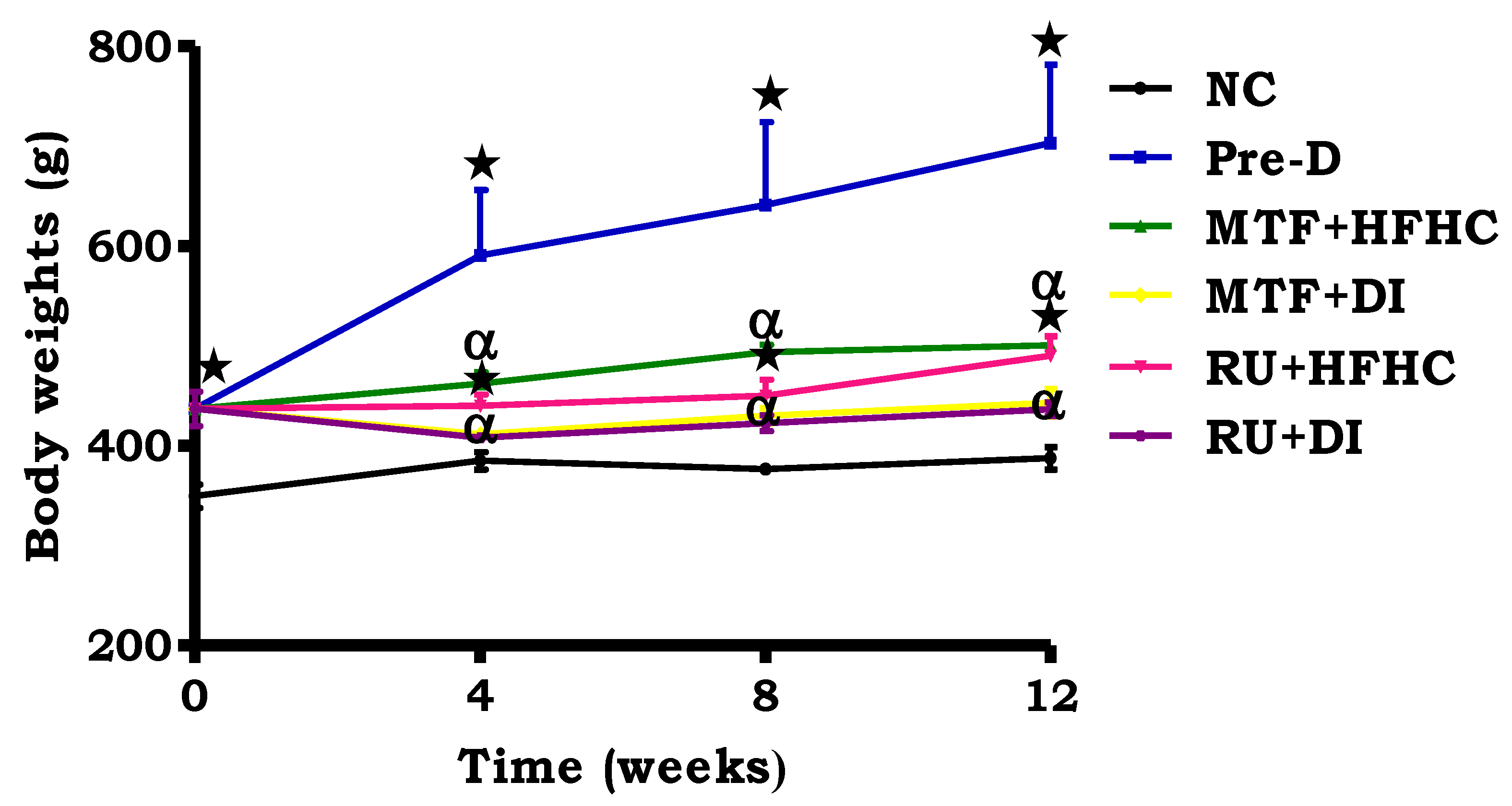
2.2. Body Weight Change
2.3. Plasma Ghrelin Concentration
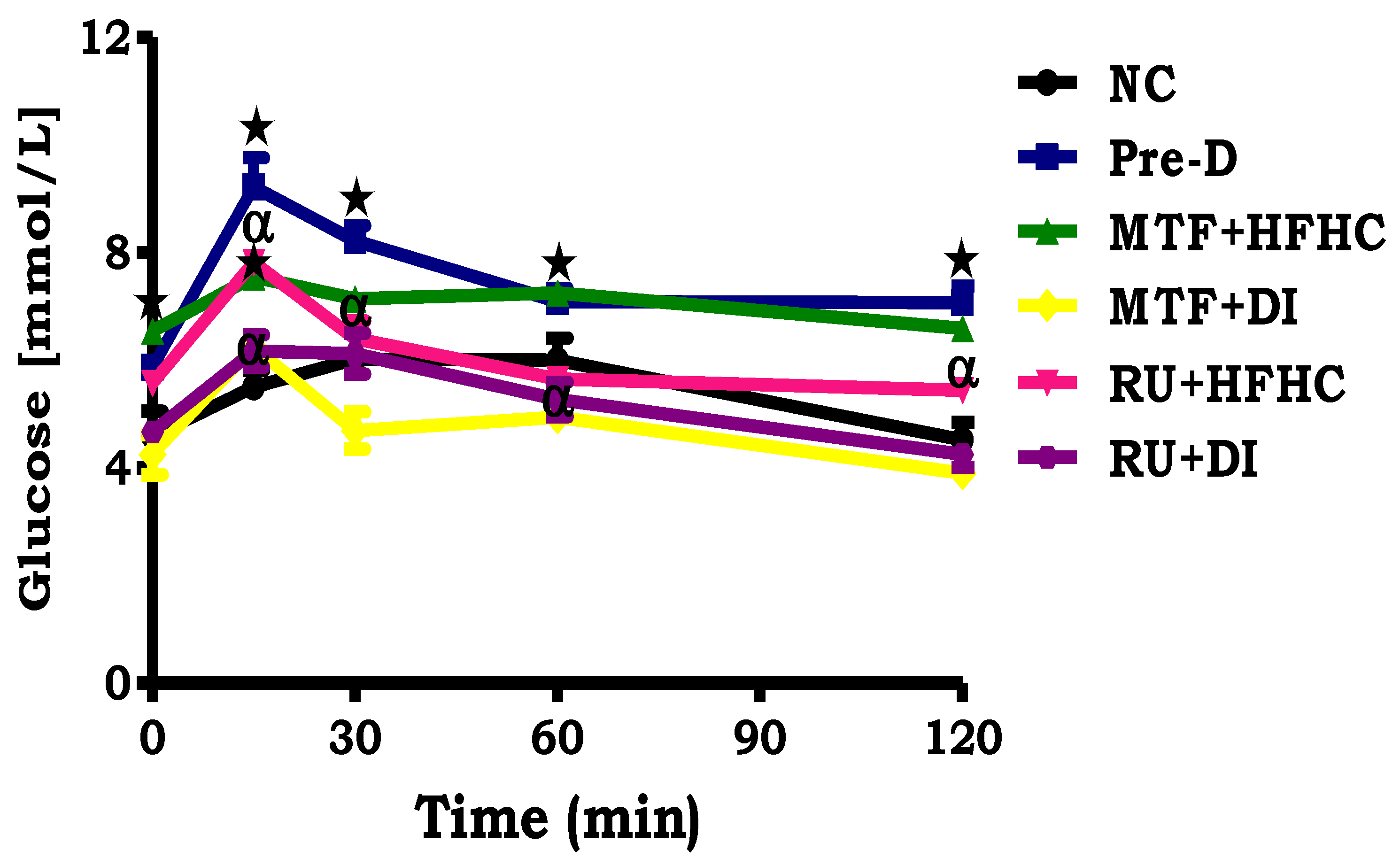
2.4. OGT Response
2.5. Gastrocnemius Muscle Glycogen Concentration
2.6. HOMA2-IR Index
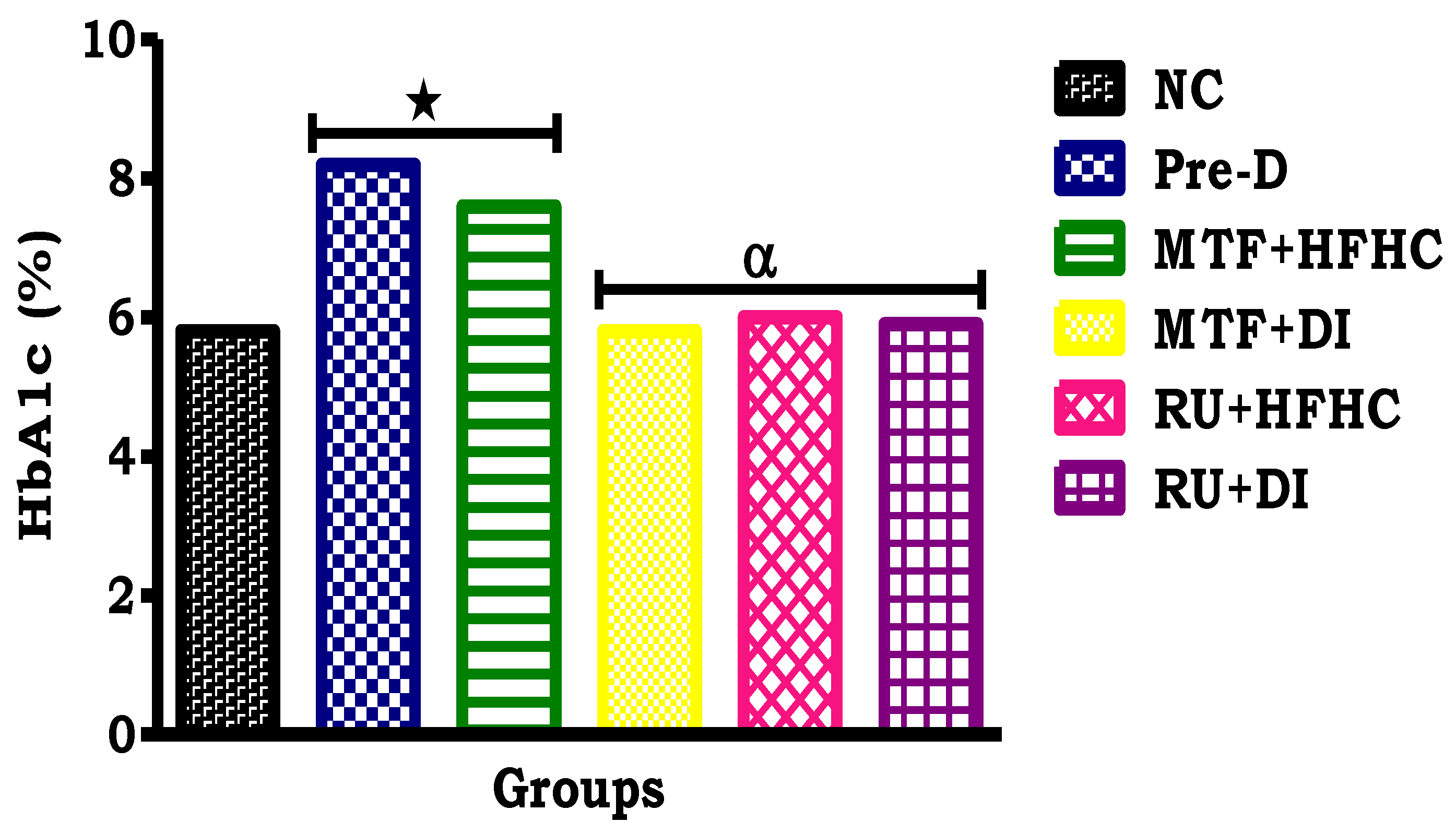
2.7. Glycated Haemoglobin (HbA1c) Concentration
3. Discussion
4. Materials and Methods
4.1. Chemicals and Drugs
4.2. Synthesis of Ruthenium (II) Complex
4.3. Animals and Housing
4.3.1. Induction of Pre-Diabetes
4.3.2. Experimental Design
4.3.3. Treatment of Pre-Diabetic Animals
4.3.4. Blood Collection and Tissue Harvesting
4.4. Biochemical Analysis
4.5. Glycogen Assay
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kumar, P.; Clark, M. Textbook of Clinical Medicine; Elsevier Health Sciences: Edinburgh, UK, 2002; pp. 1099–1121. [Google Scholar]
- Zimmet, P.; Alberti, K.; Shaw, J. Global and societal implications of the diabetes epidemic. Nature 2001, 414, 782–787. [Google Scholar] [CrossRef] [PubMed]
- Bornfeldt, K.; Tabas, I. Insulin resistance, hyperglycemia, and atherosclerosis. Cell Metab. 2011, 14, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Belgardt, B.; Brüning, J. CNS leptin and insulin action in the control of energy homeostasis. Ann. NY Acad. Sci. 2010, 1212, 97–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhillon, S.; McFadden, S.; Chalmers, J.; Centeno, M.; Kim, G.; Belsham, D. Cellular leptin resistance impairs the leptin-mediated suppression of neuropeptide Y secretion in hypothalamic neurons. Endocrinology 2011, 152, 4138–4147. [Google Scholar] [CrossRef] [PubMed]
- Kalra, S. Pivotal role of leptin-hypothalamus signaling in the etiology of diabetes uncovered by gene therapy, a new therapeutic intervention? Gene Ther. 2011, 18, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Mantzoros, C.; Magkos, F.; Brinkoetter, M.; Sienkiewicz, E.; Dardeno, T.; Kim, S. Leptin in human physiology and pathophysiology. Am. J. Physiol.-Endoc. Metb. 2011, 301, E567–E584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lim, A. Diabetic nephropathy–complications and treatment. Intern. J. Nephr. Renov. Dis. 2014, 7, 361. [Google Scholar] [CrossRef] [PubMed]
- Polińska, B.; Matowicka-Karna, J.; Kemona, H. The role of ghrelin in the organism. Endocr. Rev. 2011, 65, 1–7. [Google Scholar] [CrossRef]
- Pusztai, P.; Sarman, B.; Ruzicska, E.; Toke, J.; Racz, K.; Somogyi, A. Ghrelin: A new peptide regulating the neurohormonal system, energy homeostasis and glucose metabolism. Diab. Metab. Res. Rev. 2008, 24, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Evert, A.; Boucher, J.; Cypress, M.; Dunbar, S.; Franz, M.; Mayer-Davis, E. Nutrition therapy recommendations for the management of adults with diabetes. Diab. Care 2014, 37, S120–S143. [Google Scholar] [CrossRef] [PubMed]
- Giacco, R.; Vetrani, C.; Griffo, E.; Rivellese, A. Role of Diet and Diet Interventions in Diabetic Patients: Physiological and Metabolic Changes and Reduction in Morbidity and Mortality. Curr. Nutr. Rep. 2013, 2, 174–180. [Google Scholar] [CrossRef] [Green Version]
- Rafique, S.; Idrees, M.; Nasim, A.; Akbar, H.; Athar, A. Transition metal complexes as potential therapeutic Agents. Biotech. Mol. Bio. Rev. 2010, 5, 1538–2273. [Google Scholar]
- Antonyan, A.; De, A.; Vitali, L.; Pettinari, R.; Marchetti, F.; Gigliobianco, M. Evaluation of (arene) Ru (II) complexes of curcumin as inhibitors of dipeptidyl peptidase IV. Biochimie 2014, 99, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Komers, R.; Anderson, S. Paradoxes of nitric oxide in the diabetic kidney. Am. J. Phys. Ren. Phys. 2003, 284, 1121–1137. [Google Scholar] [CrossRef] [PubMed]
- Booysen, I.; Maikoo, S.; Akerman, M.; Xulu, B. Novel ruthenium (II) and (III) compounds with multidentate Schiff base chelates bearing biologically significant moieties. Polyhedron 2014, 79, 250–257. [Google Scholar] [CrossRef]
- Gallagher, E.; Le Roith, D.; Bloomgarden, Z. Review of hemoglobin A1c in the management of diabetes. J. Diab. 2009, 1, 9–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Syed, I. Glycated haemoglobin; past, present, and future are we ready for the change. Diabetes 2011, 11, 12. [Google Scholar]
- American Diabites Association. Foundations of care and comprehensive medical evaluation. Diab. Care 2016, 39, S23–S35. [Google Scholar]
- Franz, M. The glycemic index. Diab. Care 2003, 26, 2466–2468. [Google Scholar] [CrossRef]
- Lindström, J.; Peltonen, M.; Eriksson, J.; Louheranta, A.; Fogelholm, M.; Uusitupa, M. High-fibre, low-fat diet predicts long-term weight loss and decreased type 2 diabetes risk: The Finnish Diabetes Prevention Study. Diabetologia 2006, 49, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Panchal, S.; Poudyal, H.; Iyer, A.; Nazer, R.; Alam, A.; Diwan, V. High-carbohydrate high-fat diet–induced metabolic syndrome and cardiovascular remodeling in rats. J. Card. Pharm. 2011, 57, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Lozano, I.; Van der Werf, R.; Bietiger, W.; Seyfritz, E.; Peronet, C.; Pinget, M. High-fructose and high-fat diet-induced disorders in rats: Impact on diabetes risk, hepatic and vascular complications. Nutr. Metab. 2016, 13, 15. [Google Scholar] [CrossRef] [PubMed]
- Gamede, M.; Mabuza, L.; Ngubane, P.; Khathi, A. The Effects of Plant-Derived Oleanolic Acid on Selected Parameters of Glucose Homeostasis in a Diet-Induced Pre-Diabetic Rat Model. Molecules 2018, 23, 794. [Google Scholar] [CrossRef] [PubMed]
- Luvuno, M.; Mbongwa, H.; Khathi, A. Development of a novel prediabetes animal model using a high fat high carbohydrate diet: Implications for type 2 diabetes. PLoS ONE 2017, 13, 8–14. [Google Scholar]
- Sinitskaya, N.; Gourmelen, S.; Schuster-Klein, C.; Guardiola-Lemaitre, B.; Pévet, P.; Challet, E. Increasing the fat-to-carbohydrate ratio in a high-fat diet prevents the development of obesity but not a prediabetic state in rats. Clin. Sci. 2007, 113, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Le, K.; Faeh, D.; Stettler, R.; Ith, M.; Kreis, R.; Vermathen, P. A 4-wk high-fructose diet alters lipid metabolism without affecting insulin sensitivity or ectopic lipids in healthy humans. Am. J. Clin. Nutr. 2006, 84, 1374–1379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hostalek, U.; Gwilt, M.; Hildemann, S. Therapeutic use of metformin in prediabetes and diabetes prevention. Drugs 2015, 75, 1071–1094. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.; Kola, B.; Korbonits, M.; Grossman, A. Ghrelin’s role as a major regulator of appetite and its other functions in neuroendocrinology. Prog. Brain Res. 2010, 182, 189–205. [Google Scholar] [PubMed]
- Bastaki, A. Diabetes mellitus and its treatment. Int. J. Diab. Metab. 2005, 13, 111. [Google Scholar]
- He, L.; Wang, X.; Zhao, C.; Wang, H.; Du, W. Ruthenium complexes as novel inhibitors of human islet amyloid polypeptide fibril formation. Metallomics 2013, 5, 1599–1603. [Google Scholar] [CrossRef] [PubMed]
- Cusi, K.; Cukier, S.; DeFronzo, R.; Torres, M.; Puchulu, F.; Redondo, J. Vanadyl sulfate improves hepatic and muscle insulin sensitivity in type 2 diabetes. J. Clin. Endoc. Metab. 2001, 86, 1410–1417. [Google Scholar] [CrossRef]
- Wallace, T.; Levy, J.; Matthews, D. Use and abuse of HOMA modeling. Diab. Care 2004, 27, 1487–1495. [Google Scholar] [CrossRef]
- Jesudason, D.; Dunstan, K.; Leong, D.; Wittert, G. Macrovascular risk and diagnostic criteria for type 2 diabetes. Diab. Care 2003, 26, 485–490. [Google Scholar] [CrossRef]
- Bennett, C.; Guo, M.; Dharmage, S. HbA1c as a screening tool for detection of type 2 diabetes: A systematic review. Diab. Med. 2007, 24, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.; Silva, D.; Petrovsky, N. Case study: Potential pitfalls of using hemoglobin A1c as the sole measure of glycemic control. Clin. Diab. 2004, 22, 141–143. [Google Scholar] [CrossRef]
- Ciftci, O.; Beytur, A.; Vardi, N.; Ozdemir, I. Evaluation of reproductive toxicity in male rats treated with novel synthesized ruthenium(II) and gold(I.)-NHC complexes. Drug Dev. Ind. Pharm. 2012, 381, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Khathi, A.; Serumula, M.; Myburg, R.; Van Heerden, F.; Musabayane, C. Effects of Syzygium aromaticum-derived triterpenes on postprandial blood glucose in streptozotocin-induced diabetic rats following carbohydrate challenge. PLoS ONE 2013, 8, e81632. [Google Scholar] [CrossRef] [PubMed]
- Musabayane, C.; Xozwa, K.; Ojewole, J. Effects of Hypoxis hemerocallidea (Fisch. & C.A. Mey.) [Hypoxidaceae] corm (African Potato) aqueous extract on renal electrolyte and fluid handling in the rat. Ren. Fail. 2005, 27, 763–770. [Google Scholar] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
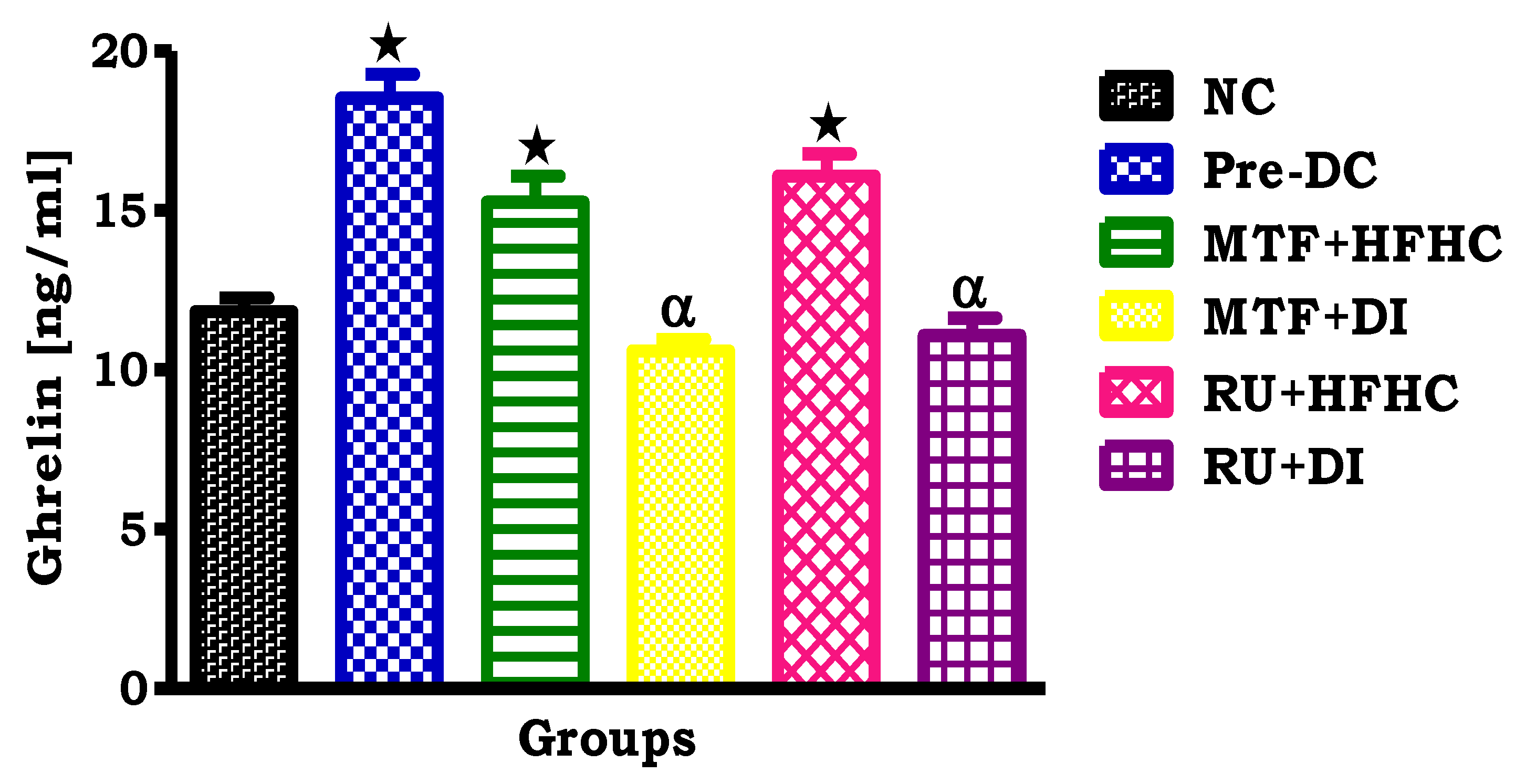
| Groups | Caloric Intake (kcal/g) | |||
|---|---|---|---|---|
| Week 0 | Week 4 | Week 8 | Week 12 | |
| NC | 106.18 ± 1.90 | 115.74 ± 2.34 | 123.96 ± 1.61 | 138.40 ± 0.87 |
| Pre-D | 120.72 ± 1.11 * | 158.20 ± 0.64 * | 184.88 ± 0.84 * | 205.84 ± 0.85 * |
| MTF + HFHC | 118.09 ± 0.51 * | 100.54 ± 0.98 * | 99.51 ± 1.52 *α | 151.66 ± 0.69 *,α |
| MTF + DI | 115.12 ± 0.67 | 102.69 ± 1.17 α | 120.51 ± 0.75 α | 144.72 ± 1.64 α |
| RU + HFHC | 119.17 ± 0.65 * | 124.73 ± 0.31 *,α | 155.58 ± 0.99 α,* | 167.44 ± 0.87 *,α |
| RU + DI | 113.73 ± 0.98 | 118.94 ± 0.85 α | 127.09 ± 0.89 α | 138.82 ± 0.76 α |
| Groups | Muscle Glycogen (nmol/g Protein) |
|---|---|
| NC | 0.19 ± 0.03 |
| Pre-D | 0.73 ± 0.01 * |
| MTF + HFHC | 0.43 ± 0.11 *,α |
| MTF + DI | 0.24 ± 0.02 α |
| RU + HFHC | 0.31 ± 0.03 *,α |
| RU + DI | 0.21 ± 0.01 α |
| Groups | Plasma Glucose (mmol/L) | Plasma Insulin (mIU/L) | HOMA-IR Values |
|---|---|---|---|
| NC | 4.60 ± 0.09 | 5.89 ± 0.90 | 1.20 ± 0.61 |
| Pre-D | 5.60 ± 0.32 * | 20.92 ± 3.45 * | 5.20 ± 2.98 * |
| MTF + HFHC | 6.60 ± 0.81 *,α | 18.00 ±3.12 * | 5.30 ± 1.02 * |
| MTF + DI | 4.30 ± 0.90 α | 7.57 ±1.32 *,α | 1.30 ±1.15 *,α |
| RU + HFHC | 5.60 ±1.02 *,α | 16.17± 1.88 *,α | 4.00 ±1.05 * |
| RU + DI | 4.70 ± 0.82 α | 8.29 ± 2.00 α | 1.70 ± 0.99 α |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mabuza, L.P.; Gamede, M.W.; Maikoo, S.; Booysen, I.N.; Ngubane, P.S.; Khathi, A. Effects of a Ruthenium Schiff Base Complex on Glucose Homeostasis in Diet-Induced Pre-Diabetic Rats. Molecules 2018, 23, 1721. https://doi.org/10.3390/molecules23071721
Mabuza LP, Gamede MW, Maikoo S, Booysen IN, Ngubane PS, Khathi A. Effects of a Ruthenium Schiff Base Complex on Glucose Homeostasis in Diet-Induced Pre-Diabetic Rats. Molecules. 2018; 23(7):1721. https://doi.org/10.3390/molecules23071721
Chicago/Turabian StyleMabuza, Lindokuhle Patience, Mlindeli Wilkinson Gamede, Sanam Maikoo, Irvin Noel Booysen, Phikelelani Siphosethu Ngubane, and Andile Khathi. 2018. "Effects of a Ruthenium Schiff Base Complex on Glucose Homeostasis in Diet-Induced Pre-Diabetic Rats" Molecules 23, no. 7: 1721. https://doi.org/10.3390/molecules23071721






