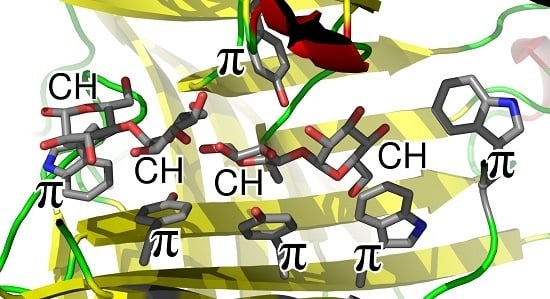
CH/π Interactions in Carbohydrate Recognition
Abstract
:1. Carbohydrate–Aromatic Interactions
2. Physical Nature of Carbohydrate–Aromatic Interactions—CH/π Bond
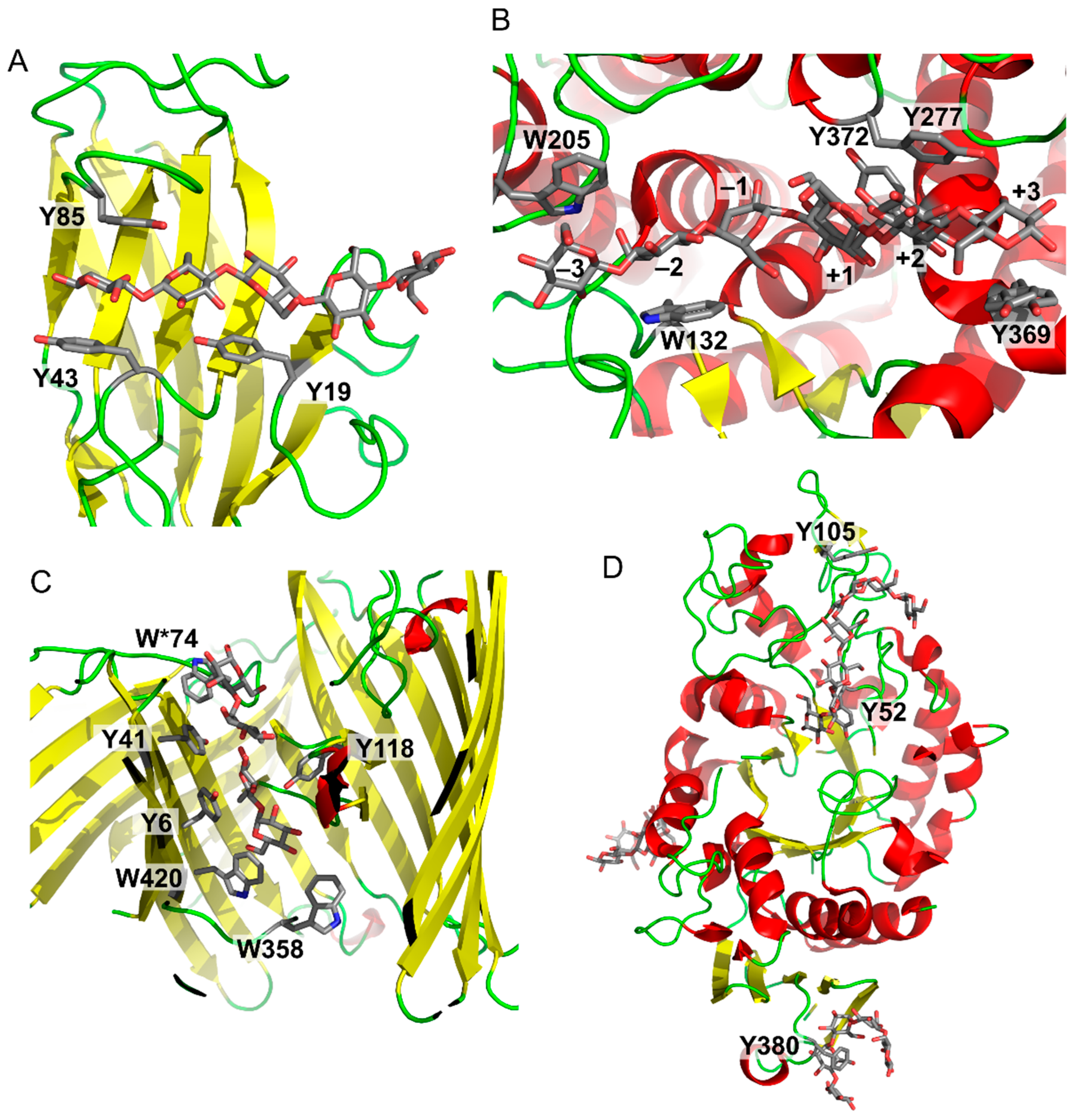
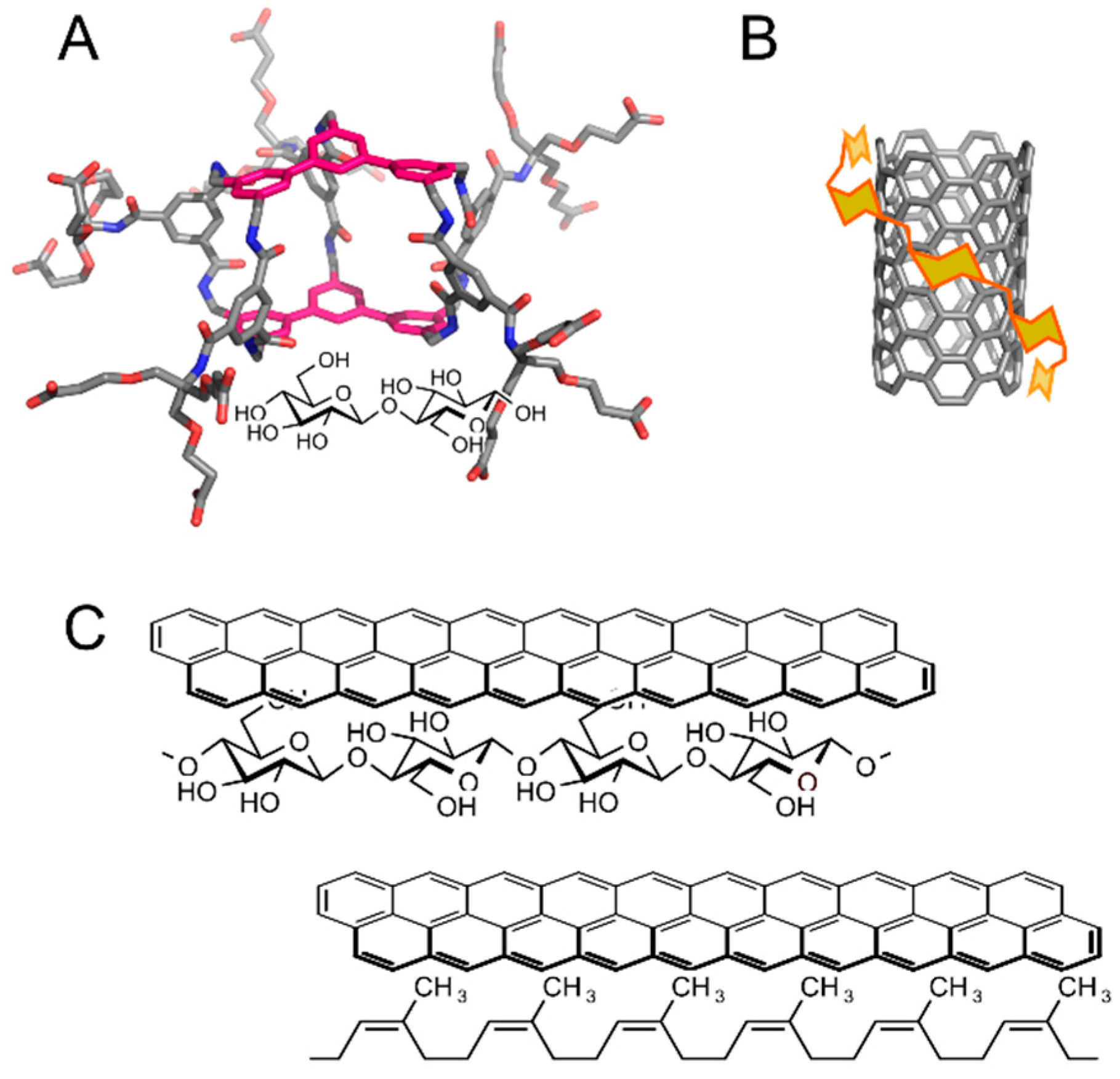
3. Applications
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Karasová-Lipovová, P.; Strnad, H.; Spiwok, V.; Malá, Š.; Kralová, B.; Russell, N.J. The Cloning, Purification and Characterisation of a Cold-Active β-Galactosidase from the Psychrotolerant Antarctic Bacterium Arthrobacter sp. C2-2. Enzym. Microb. Technol. 2003, 33, 836–844. [Google Scholar] [CrossRef]
- Petroková, H.; Vondráčková, E.; Skálová, T.; Dohnálek, J.; Lipovová, P.; Spiwok, V.; Strnad, H.; Králová, B.; Hašek, J. Crystallization and Preliminary X-Ray Diffraction Analysis of Cold-Active β-Galactosidase from Arthrobacter sp. C2-2. Collect. Czech. Chem. Commun. 2005, 70, 124–132. [Google Scholar] [CrossRef]
- Skálová, T.; Dohnálek, J.; Spiwok, V.; Lipovová, P.; Vondráčková, E.; Petroková, H.; Dušková, J.; Strnad, H.; Králová, B.; Hašek, J. Cold-active β-Galactosidase from Arthrobacter sp. C2-2 Forms Compact 660 kDa Hexamers: Crystal Structure at 1.9 Å Resolution. J. Mol. Biol. 2005, 353, 282–294. [Google Scholar] [CrossRef] [PubMed]
- Juers, D.H.; Heightman, T.D.; Vasella, A.; McCarter, J.D.; Mackenzie, L.; Withers, S.G.; Matthews, B.W. A structural view of the action of Escherichia coli (LacZ) β-galactosidase. Biochemistry 2001, 40, 14781–14794. [Google Scholar] [CrossRef] [PubMed]
- Huber, R.E.; Hakda, S.; Cheng, C.; Cupples, C.G.; Edwards, R.A. Trp-999 of β-galactosidase (Escherichia coli) is a key residue for binding, catalysis, and synthesis of allolactose, the natural lac operon inducer. Biochemistry 2003, 42, 1796–1803. [Google Scholar] [CrossRef] [PubMed]
- Muraki, M. The importance of CH/π interactions to the function of carbohydrate binding proteins. Protein Peptide Lett. 2002, 9, 195–209. [Google Scholar] [CrossRef]
- Balaji, P.V. Contribution of C-H...π Interactions to the Affinity and Specificity of Carbohydrate Binding Sites. Mini-Rev. Org. Chem. 2011, 8, 222–228. [Google Scholar] [CrossRef]
- Asensio, J.L.; Ardá, A.; Cañada, F.J.; Jiménez-Barbero, J. Carbohydrate-aromatic interactions. Acc. Chem. Res. 2013, 46, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Macias, M.A.; Lütteke, T. Statistical analysis of amino acids in the vicinity of carbohydrate residues performed by GlyVicinity. Methods Mol. Biol. 2015, 1273, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Codon Usage Database. Available online: http://www.kazusa.or.jp/codon/ (accessed on 9 April 2017).
- Hudson, K.L.; Bartlett, G.J.; Diehl, R.C.; Agirre, J.; Gallagher, T.; Kiessling, L.L.; Woolfson, D.N. Carbohydrate-Aromatic Interactions in Proteins. J. Am. Chem. Soc. 2015, 137, 15152–15160. [Google Scholar] [CrossRef] [PubMed]
- Rose, I.A.; Hanson, K.R.; Wilkinson, K.D.; Wimmer, M.J. A suggestion for naming faces of ring compounds. Proc. Natl. Acad. Sci. USA 1980, 77, 2439–2441. [Google Scholar] [CrossRef] [PubMed]
- Pérez, S.; Tvaroška, I. Carbohydrate-protein interactions: Molecular modeling insights. Adv. Carbohyd. Chem. Biochem. 2014, 71, 9–136. [Google Scholar] [CrossRef]
- Eschenmoser, A. Etiology of Potentially Primordial Biomolecular Structures: From Vitamin B12 to the Nucleic Acids and an Inquiry into the Chemistry of Life’s Origin: A Retrospective. Angew. Chem. Int. Ed. 2011, 50, 12412–12472. [Google Scholar] [CrossRef] [PubMed]
- Boraston, A.B.; Nurizzo, D.; Notenboom, V.; Ducros, V.; Rose, D.R.; Kilburn, D.G.; Davies, G.J. Differential Oligosaccharide Recognition by Evolutionarily-Related β-1,4 and β-1,3 Glucan-Binding Modules. J. Mol. Biol. 2002, 319, 1143–1156. [Google Scholar] [CrossRef]
- Guerin, D.M.; Lascombe, M.B.; Costabel, M.; Souchon, H.; Lamzin, V.; Beguin, P.; Alzari, P.M. Atomic (0.94 Å) resolution structure of an inverting glycosidase in complex with substrate. J. Mol. Biol. 2002, 316, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Merritt, E.A.; Kuhn, P.; Sarfaty, S.; Erbe, J.L.; Holmes, R.K.; Hol, W.G. The 1.25 Å resolution refinement of the cholera toxin B-pentamer: Evidence of peptide backbone strain at the receptor-binding site. J. Mol. Biol. 1998, 282, 1043–1059. [Google Scholar] [CrossRef] [PubMed]
- Dutzler, R.; Wang, Y.F.; Rizkallah, P.J.; Rosenbusch, J.P.; Schirmer, T. Crystal structures of various maltooligosaccharides bound to maltoporin reveal a specific sugar translocation pathway. Structure 1996, 4, 127–134. [Google Scholar] [CrossRef]
- Denker, K.; Orlik, F.; Schiffler, B.; Benz, R. Site-directed mutagenesis of the greasy slide aromatic residues within the LamB (maltoporin) channel of Escherichia coli: Effect on ion and maltopentaose transport. J. Mol. Biol. 2005, 352, 534–550. [Google Scholar] [CrossRef] [PubMed]
- Robert, X.; Haser, R.; Gottschalk, T.E.; Ratajczak, F.; Driguez, H.; Svensson, B.; Aghajari, N. The structure of barley alpha-amylase isozyme 1 reveals a novel role of domain C in substrate recognition and binding: A pair of sugar tongs. Structure 2003, 11, 973–984. [Google Scholar] [CrossRef]
- Bozonnet, S.; Jensen, M.T.; Nielsen, M.M.; Aghajari, N.; Jensen, M.H.; Kramhøft, B.; Willemoës, M.; Tranier, S.; Haser, R.; Svensson, B. The ‘pair of sugar tongs’ site on the non-catalytic domain C of barley alpha-amylase participates in substrate binding and activity. FEBS J. 2007, 274, 5055–5067. [Google Scholar] [CrossRef] [PubMed]
- Besombes, S.; Mazeau, K. Molecular dynamics simulations of a guaiacyl β-O-4 lignin model compound: Examination of intramolecular hydrogen bonding and conformational flexibility. Biopolymers 2004, 73, 301–315. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Wareham, R.S. Carbohydrate Recognition through Noncovalent Interactions: A Challenge for Biomimetic and Supramolecular Chemistry. Angew. Chem. Int. Edit. 1999, 38, 2978–2996. [Google Scholar] [CrossRef]
- Ferrand, Y.; Crump, M.P.; Davis, A.P. A Synthetic Lectin Analogue for Biomimetic Disaccharide Recognition. Science 2007, 318, 619–622. [Google Scholar] [CrossRef] [PubMed]
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeersch, T.; Zurek, E.; Hutchison, G.R. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 2012, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Gabbanini, S.; Lucchi, E.; Guidugli, F.; Matera, R.; Valgimigli, L. Anomeric discrimination and rapid analysis of underivatized lactose, maltose, and sucrose in vegetable matrices by U-HPLC-ESI-MS/MS using porous graphitic carbon. J. Mass. Spectrom. 2010, 45, 1012–1018. [Google Scholar] [CrossRef] [PubMed]
- Kim, O.K.; Je, J.; Baldwin, J.W.; Kooi, S.; Pehrsson, P.E.; Buckley, L.J. Solubilization of single-wall carbon nanotubes by supramolecular encapsulation of helical amylose. J. Am. Chem. Soc. 2003, 125, 4426–4427. [Google Scholar] [CrossRef] [PubMed]
- Bhoi, V.I.; Kumar, S.; Murthy, C.N. The self-assembly and aqueous solubilization of [60]fullerene with disaccharides. Carbohyd. Res. 2012, 359, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Birchall, L.S.; Roy, S.; Jayawarna, V.; Hughes, M.; Irvine, E.; Okorogheye, G.T.; Saudi, N.; De Santis, E.; Tuttle, T.; Edwards, A.A.; et al. Exploiting CH-π interactions in supramolecular hydrogels of aromatic carbohydrate amphiphiles. Chem. Sci. 2011, 2, 1349–1355. [Google Scholar] [CrossRef]
- Nishio, M.; Hirota, M.; Umezawa, Y. The CH-Pi Interaction: Evidence, Nature, and Consequences, 1st ed.; Wiley-VCH: New York, NY, USA, 1998. [Google Scholar]
- Spiwok, V.; Lipovová, P.; Skálová, T.; Buchtelová, E.; Hašek, J.; Králová, B. Role of CH/π Interactions in Substrate Binding by Escherichia coli β-Galactosidase. Carbohyd. Res. 2004, 339, 2275–2280. [Google Scholar] [CrossRef] [PubMed]
- Spiwok, V.; Lipovová, P.; Skálová, T.; Vondráčková, E.; Dohnálek, J.; Hašek, J.; Králová, B. Modelling of Carbohydrate-Aromatic Interactions: Ab Initio Energetics and Force Field Performance. J. Comput. Aided Mol. Des. 2005, 19, 887–901. [Google Scholar] [CrossRef] [PubMed]
- Kozmon, S.; Matuška, R.; Spiwok, V.; Koča, J. Three-Dimensional Potential Energy Surface of Selected Carbohydrates' CH/π Dispersion Interactions Calculated by High-Level Quantum Mechanical Methods. Chem. Eur. J. 2011, 17, 5680–5690. [Google Scholar] [CrossRef] [PubMed]
- Kozmon, S.; Matuška, R.; Spiwok, V.; Koča, J. Dispersion Interactions of Carbohydrates with Condensate Aromatic Moieties: Theoretical Study on the CH-π Interaction Additive Properties. Phys. Chem. Chem. Phys. 2011, 13, 14215–14222. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Alonso, M.C.; Canada, F.J.; Jimenez-Barbero, J.; Cuevas, G. Molecular recognition of saccharides by proteins. Insights on the origin of the carbohydrate-aromatic interactions. J. Am. Chem. Soc. 2005, 127, 7379–7386. [Google Scholar] [CrossRef] [PubMed]
- Sujatha, M.S.; Sasidhar, Y.U.; Balaji, P.V. Insights into the role of the aromatic residue in galactose-binding sites: MP2/6-311G++** study on galactose- and glucose-aromatic residue analogue complexes. Biochemistry 2005, 44, 8554–8562. [Google Scholar] [CrossRef] [PubMed]
- Sujatha, M.S.; Sasidhar, Y.U.; Balaji, P.V. MP2/6-311++G(d,p) study on galactose–aromatic residue analogue complexes in different position-orientations of the saccharide relative to aromatic residue. J. Mol. Struct. THEOCHEM 2007, 814, 11–24. [Google Scholar] [CrossRef]
- Sharma, R.; McNamara, J.P.; Raju, R.K.; Vincent, M.A.; Hillier, I.A.; Morgado, C.A. The interaction of carbohydrates and amino acids with aromatic systems studied by density functional and semi-empirical molecular orbital calculations with dispersion corrections. Phys. Chem. Chem. Phys. 2008, 10, 2767–2774. [Google Scholar] [CrossRef] [PubMed]
- Raju, R.K.; Ramraj, A.; Hillier, I.H.; Vincent, M.A.; Burton, N.A. Carbohydrate-aromatic π interactions: A test of density functionals and the DFT-D method. Phys. Chem. Chem. Phys. 2009, 11, 3411–3416. [Google Scholar] [CrossRef] [PubMed]
- Tsuzuki, S.; Uchimaru, T.; Mikami, M. Magnitude and nature of carbohydrate-aromatic interactions: Ab initio calculations of fucose-benzene complex. J. Phys. Chem. B 2009, 113, 5617–5621. [Google Scholar] [CrossRef] [PubMed]
- Hobza, P.; Muller-Dethlefs, K. Non-Covalent Interactions: Theory and Experiment, 1st ed.; Royal Society of Chemistry: Cambridge, UK, 2009. [Google Scholar]
- Phipps, M.J.; Fox, T.; Tautermann, C.S.; Skylaris, C.K. Energy decomposition analysis approaches and their evaluation on prototypical protein-drug interaction patterns. Chem. Soc. Rev. 2015, 44, 3177–3211. [Google Scholar] [CrossRef] [PubMed]
- Screen, J.; Stanca-Kaposta, E.C.; Gamblin, D.P.; Liu, B.; Macleod, N.A.; Snoek, L.C.; Davis, B.G.; Simons, J.P. IR-spectral signatures of aromatic-sugar complexes: Probing carbohydrate-protein interactions. Angew. Chem. Int. Ed. 2007, 46, 3644–3648. [Google Scholar] [CrossRef] [PubMed]
- Wohlert, J.; Schnupf, U.; Brady, J.W. Free energy surfaces for the interaction of d-glucose with planar aromatic groups in aqueous solution. J. Chem. Phys. 2010, 133, 155103. [Google Scholar] [CrossRef] [PubMed]
- Mareška, V.; Tvaroška, I.; Králová, B.; Spiwok, V. Molecular Simulations of Hevein/(GlcNAc)3 Complex with Weakened OH/O and CH/π Hydrogen Bonds: Implications for their Role in Complex Stabilization. Carbohyd. Res. 2015, 408, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Colombo, G.; Meli, M.; Cañada, J.; Asensio, J.L.; Jimenez-Barbero, J. A dynamic perspective on the molecular recognition of chitooligosaccharide ligands by hevein domains. Carbohyd. Res. 2005, 340, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- Asensio, J.L.; Siebert, H.C.; von Der Lieth, C.W.; Laynez, J.; Bruix, M.; Soedjanaamadja, U.M.; Beintema, J.J.; Cañada, F.J.; Gabius, H.J.; Jiménez-Barbero, J. NMR investigations of protein-carbohydrate interactions: Studies on the relevance of Trp/Tyr variations in lectin binding sites as deduced from titration microcalorimetry and NMR studies on hevein domains. Determination of the NMR structure of the complex between pseudohevein and N,N′,N′′-triacetylchitotriose. Proteins 2000, 40, 218–236. [Google Scholar] [CrossRef] [PubMed]
- Černý, J.; Kabeláč, M.; Hobza, P. Double-helical→ladder structural transition in the B-DNA is induced by a loss of dispersion energy. J. Am. Chem. Soc. 2008, 130, 16055–16059. [Google Scholar] [CrossRef] [PubMed]
- Černý, J.; Vondrášek, J.; Hobza, P. Loss of dispersion energy changes the stability and folding/unfolding equilibrium of the Trp-cage protein. J. Phys. Chem. B 2009, 113, 5657–5660. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Enck, S.; Price, J.L.; Powers, D.L.; Powers, E.T.; Wong, C.H.; Dyson, H.J.; Kelly, J.W. Structural and energetic basis of carbohydrate-aromatic packing interactions in proteins. J. Am. Chem. Soc. 2013, 135, 9877–9884. [Google Scholar] [CrossRef] [PubMed]
- Santana, A.G.; Jiménez-Moreno, E.; Gómez, A.M.; Corzana, F.; González, C.; Jiménez-Oses, G.; Jiménez-Barbero, J.; Asensio, J.L. A dynamic combinatorial approach for the analysis of weak carbohydrate/aromatic complexes: Dissecting facial selectivity in CH/π stacking interactions. J. Am. Chem. Soc. 2013, 135, 3347–3350. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Moreno, E.; Gómez, A.M.; Bastida, A.; Corzana, F.; Jiménez-Oses, G.; Jiménez-Barbero, J.; Asensio, J.L. Modulating weak interactions for molecular recognition: A dynamic combinatorial analysis for assessing the contribution of electrostatics to the stability of CH-π bonds in water. Angew. Chem. Int. Ed. 2015, 54, 4344–4348. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Moreno, E.; Jiménez-Osés, G.; Gómez, A.M.; Santana, A.G.; Corzana, F.; Bastida, A.; Jiménez-Barbero, J.; Asensio, J.L. A thorough experimental study of CH/π interactions in water: Quantitative structure-stability relationships for carbohydrate/aromatic complexes. Chem. Sci. 2015, 6, 6076–6085. [Google Scholar] [CrossRef]
- Murray, A.N.; Chen, W.; Antonopoulos, A.; Hanson, S.R.; Wiseman, R.L.; Dell, A.; Haslam, S.M.; Powers, D.L.; Powers, E.T.; Kelly, J.W. Enhanced aromatic sequons increase oligosaccharyltransferase glycosylation efficiency and glycan homogeneity. Chem. Biol. 2015, 22, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Wyss, D.F.; Choi, J.S.; Li, J.; Knoppers, M.H.; Willis, K.J.; Arulanandam, A.R.; Smolyar, A.; Reinherz, E.L.; Wagner, G. Conformation and function of the N-linked glycan in the adhesion domain of human CD2. Science 1995, 269, 1273–1278. [Google Scholar] [CrossRef] [PubMed]
- Doxey, A.C.; Cheng, Z.; Moffatt, B.A.; McConkey, B.J. Structural motif screening reveals a novel, conserved carbohydrate-binding surface in the pathogenesis-related protein PR-5d. BMC Struct. Biol. 2010, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- Kerzmann, A.; Fuhrmann, J.; Kohlbacher, O.; Neumann, D. BALLDock/SLICK: A new method for protein-carbohydrate docking. J. Chem. Inf. Model. 2008, 48, 1616–1625. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spiwok, V. CH/π Interactions in Carbohydrate Recognition. Molecules 2017, 22, 1038. https://doi.org/10.3390/molecules22071038
Spiwok V. CH/π Interactions in Carbohydrate Recognition. Molecules. 2017; 22(7):1038. https://doi.org/10.3390/molecules22071038
Chicago/Turabian StyleSpiwok, Vojtěch. 2017. "CH/π Interactions in Carbohydrate Recognition" Molecules 22, no. 7: 1038. https://doi.org/10.3390/molecules22071038