Response of Ustilago maydis against the Stress Caused by Three Polycationic Chitin Derivatives
Abstract
:1. Introduction
2. Results
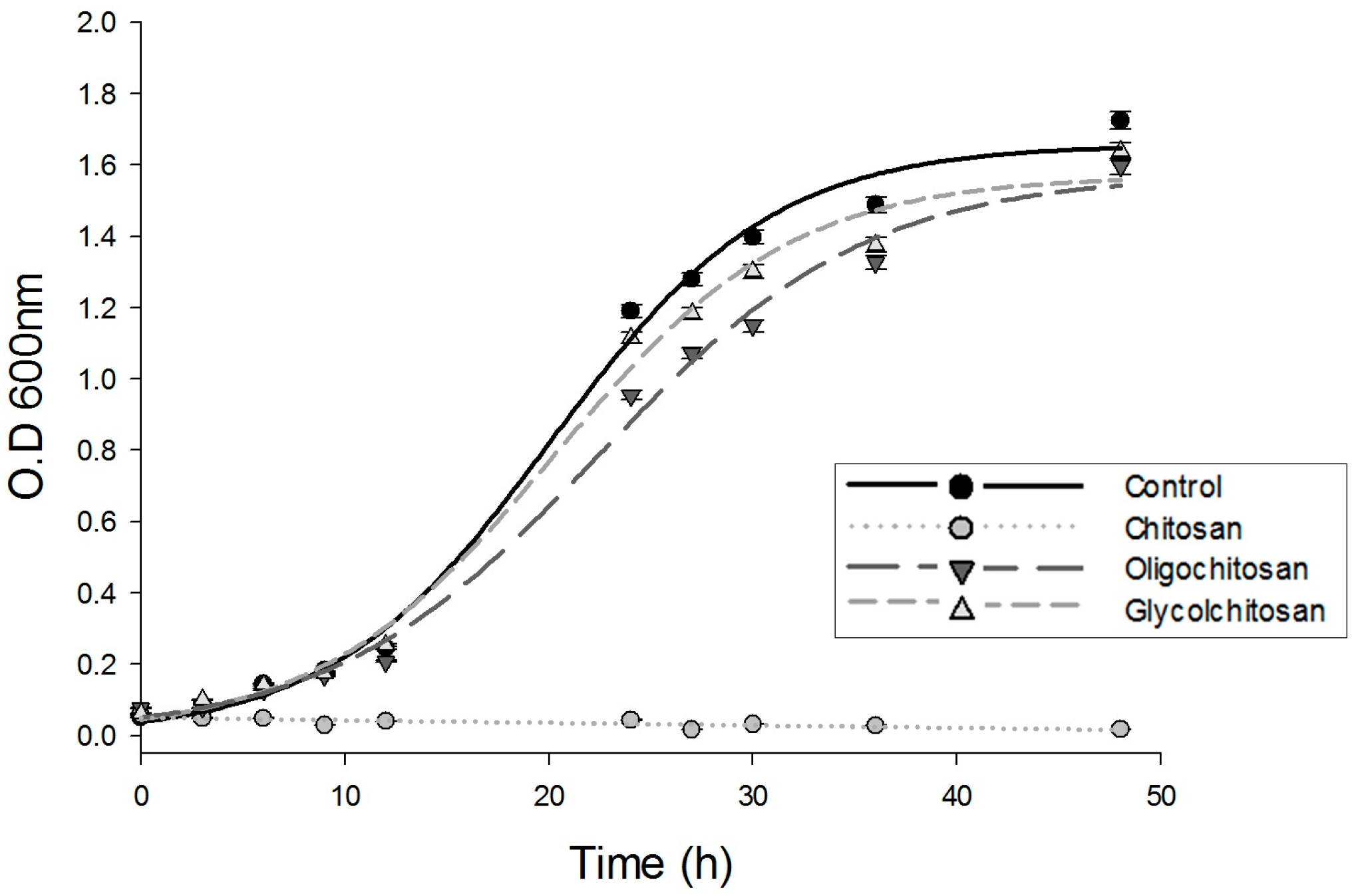
2.1. Growth of U. maydis at Different Osmolarities: Effects of CH, OCH, and GCH
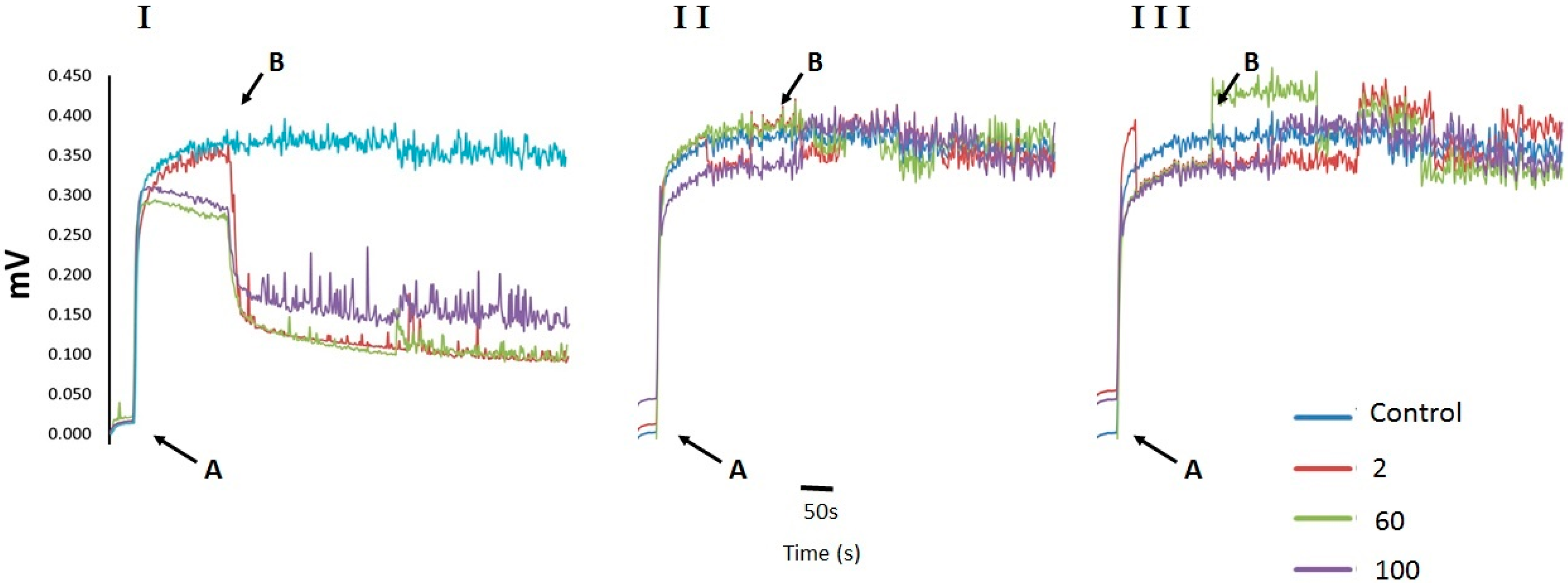
2.2. Cell Membrane Permeability Changes in U. maydis upon Addition of CH, OCH or GCH
2.3. Determination of ROS Released by U. maydis as a Response to CH, OCH or GCH
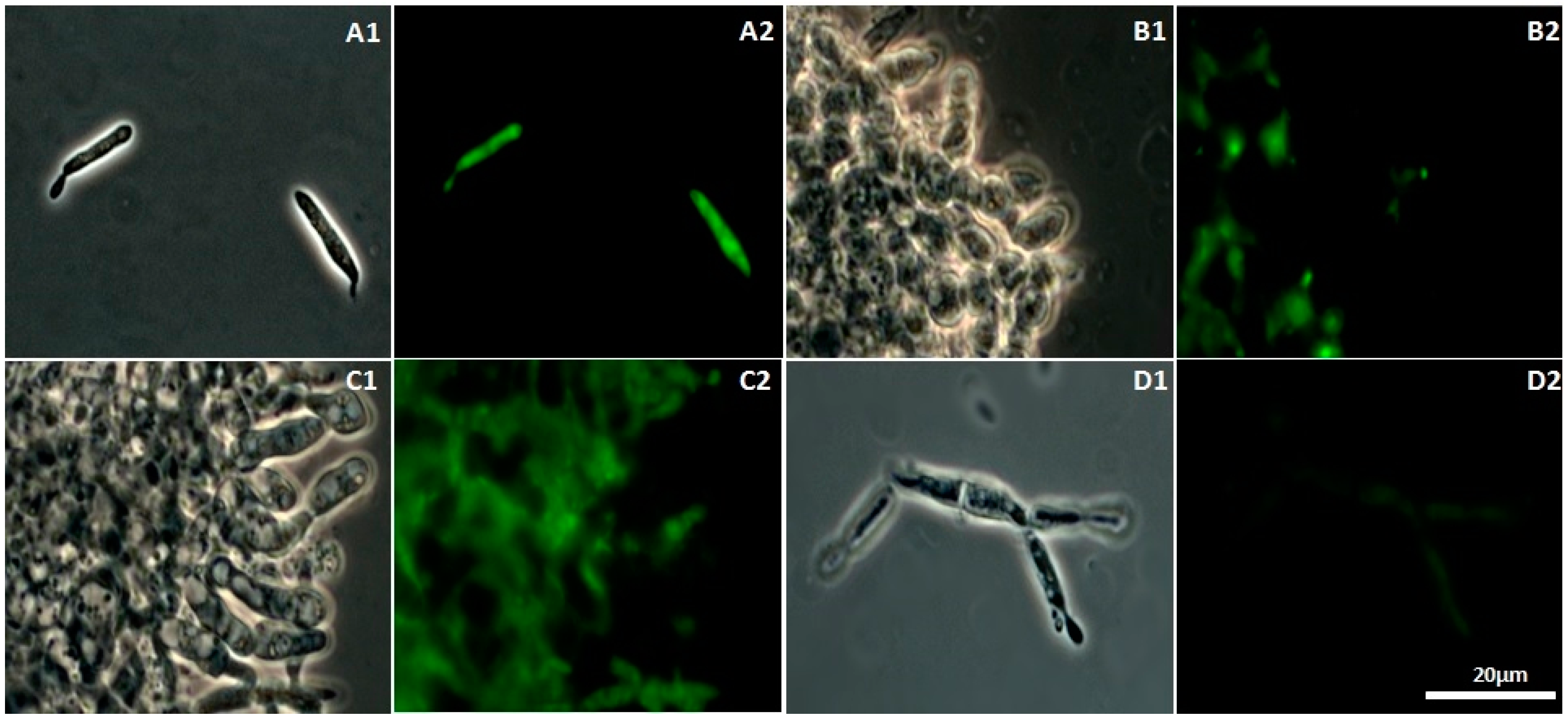
2.4. CH-, OCH-, or GCH-Mediated Damage of the Mitochondrial Structure in U. maydis
2.5. Chitin Derivatives Modify Total Phospholipid Contents
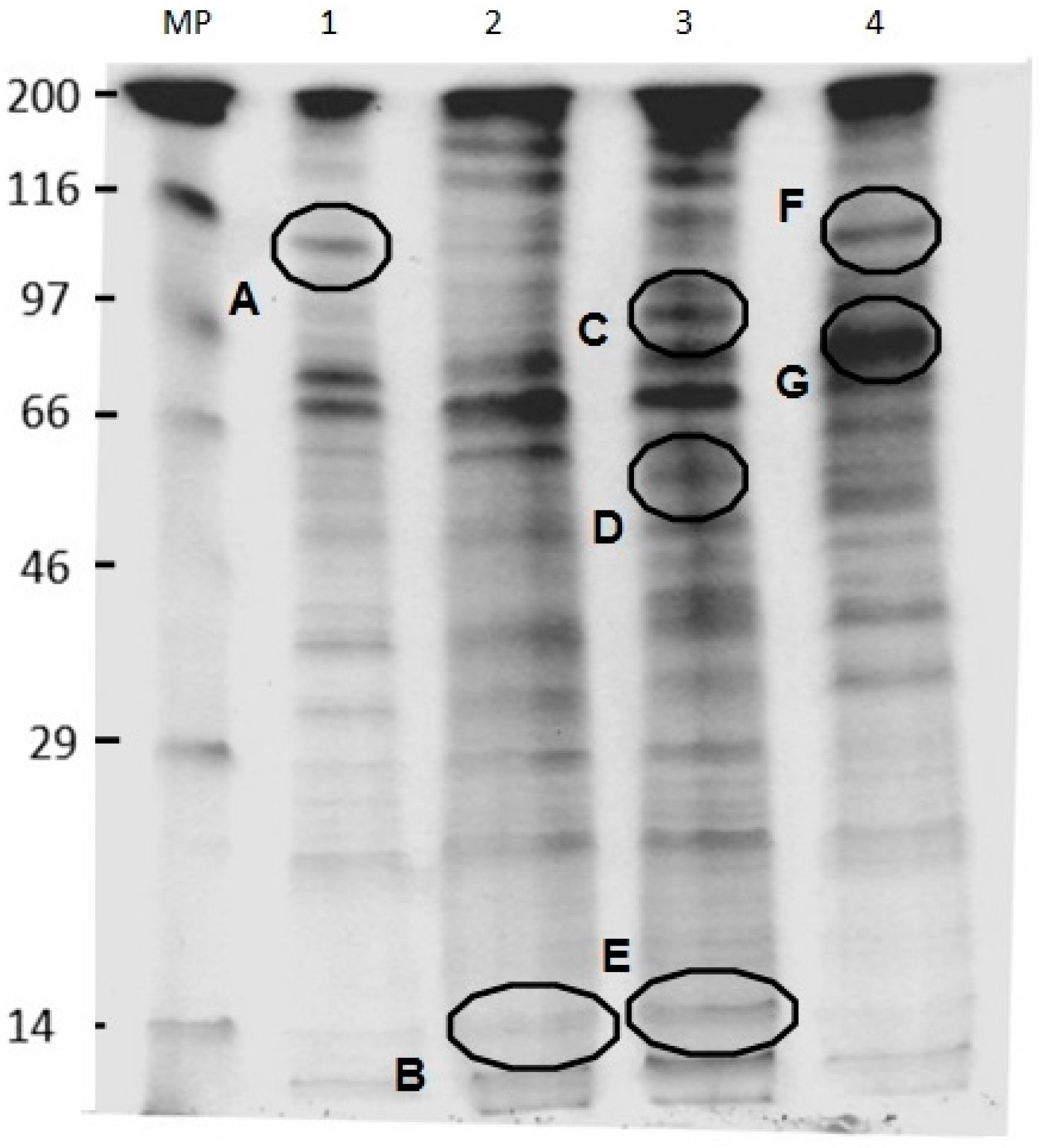
2.6. SDS-PAGE Analysis of U. maydis Membrane Proteins in the Absence and Presence of CH, OCH or GCH
2.7. Effects of CH, OCH or GCH on the Accumulation of Glycogen by U. maydis
3. Discussion
4. Materials and Methods
4.1. Reagents and Solutions
4.2. Growth in Isosmolar Medium
4.3. Transmembrane Potential
4.4. H2O2 Production Measured by the Amplex Red® Method
4.5. Catalase Activity
4.6. Mitochondrial Staining
4.7. Total Phospholipid Quantification
4.8. SDS-PAGE of the Membrane Fraction
4.9. Glycogen Accumulation and PAS Staining
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Martin, F.; Cullen, D.; Hibbett, D.; Pisabarro, A.; Spatafora, J.W.; Baker, S.E.; Grigoriev, I.V. Sequencing the fungal tree of life. New Phytol. 2011, 190, 818–821. [Google Scholar] [CrossRef] [PubMed]
- Lackner, D.H.; Schmidt, M.W.; Wu, S.; Wolf, D.A.; Bähler, J. Regulation of transcriptome, translation, and proteome in response to environmental stress in fission yeast. Genome Biol. 2012, 13, R25. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Urquiza, A.; Keyhani, N.O. Stress response signaling and virulence: Insights from entomopathogenic fungi. Curr. Genet. 2015, 61, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Gasch, A.P. Comparative genomics of the environmental stress response in ascomycete fungi. Yeast 2007, 24, 961–976. [Google Scholar] [CrossRef] [PubMed]
- Tesei, D.; Marzban, G.; Zakharova, K.; Isola, D.; Selbmann, L.; Sterflinger, K. Alteration of protein patterns in black rock inhabiting fungi as a response to different temperatures. Fungal Biol. 2012, 116, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Francois, J.M.; Formosa, C.; Schiavone, M.; Pillet, F.; Martin-Yken, H.; Dague, E. Use of atomic force microscopy (afm) to explore cell wall properties and response to stress in the yeast saccharomyces cerevisiae. Curr. Genet. 2013, 59, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Gunde-Cimerman, N.; Plemenitaš, A.; Buzzini, P. Changes in lipids composition and fluidity of yeast plasma membrane as response to cold. In Cold-Adapted Yeasts: Biodiversity, Adaptation Strategies and Biotechnological Significance; Buzzini, P., Margesin, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 225–242. [Google Scholar]
- Lushchak, V.I. Adaptive response to oxidative stress: Bacteria, fungi, plants and animals. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2011, 153, 175–190. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Oñate, M.A.; Herrera-Estrella, A. Damage response involves mechanisms conserved across plants, animals and fungi. Curr. Genet. 2015, 61, 359–372. [Google Scholar]
- Dash, M.; Chiellini, F.; Ottenbrite, R.M.; Chiellini, E. Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog. Polym. Sci. 2011, 36, 981–1014. [Google Scholar] [CrossRef]
- Bautista-Baños, S.; Hernández-Lauzardo, A.N.; Velázquez-del Valle, M.G.; Hernández-López, M.; Ait Barka, E.; Bosquez-Molina, E.; Wilson, C.L. Chitosan as a potential natural compound to control pre and postharvest diseases of horticultural commodities. Crop Prot. 2006, 25, 108–118. [Google Scholar]
- Vinsova, J.; Vavrikova, E. Chitosan derivatives with antimicrobial, antitumour and antioxidant activities—A review. Curr. Pharma. Des. 2011, 17, 3596–3607. [Google Scholar] [CrossRef]
- Benhabiles, M.S.; Salah, R.; Lounici, H.; Drouiche, N.; Goosen, M.F.A.; Mameri, N. Antibacterial activity of chitin, chitosan and its oligomers prepared from shrimp shell waste. Food Hydrocoll. 2012, 29, 48–56. [Google Scholar] [CrossRef]
- Raafat, D.; Sahl, H.-G. Chitosan and its antimicrobial potential—A critical literature survey. Microb. Biotechnol. 2009, 2, 186–201. [Google Scholar] [CrossRef] [PubMed]
- Olicón-Hernández, D.R.; Hernández-Lauzardo, A.N.; Pardo, J.P.; Peña, A.; Velázquez-del Valle, M.G.; Guerra-Sánchez, G. Influence of chitosan and its derivatives on cell development and physiology of Ustilago maydis. Int. J. Biol. Macromol. 2015, 79, 654–660. [Google Scholar]
- Ma, Z.; Yang, L.; Yan, H.; Kennedy, J.F.; Meng, X. Chitosan and oligochitosan enhance the resistance of peach fruit to brown rot. Carbohydr. Polym. 2013, 94, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Dey, A.; Gupta, B. Plant polyamines in abiotic stress responses. Acta Physiol. Plant. 2013, 35, 2015–2036. [Google Scholar] [CrossRef]
- Liu, L.; Zhou, Y.; Zhao, X.; Wang, H.; Wang, L.; Yuan, G.; Asim, M.; Wang, W.; Zeng, L.; Liu, X.; et al. Oligochitosan stimulated phagocytic activity of macrophages from blunt snout bream (megalobrama amblycephala) associated with respiratory burst coupled with nitric oxide production. Dev. Comp. Immunol. 2014, 47, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhao, X.; Wang, X.; Zhao, Z.; Du, Y. Oligochitosan inhibits phytophthora capsici by penetrating the cell membrane and putative binding to intracellular targets. Pestic. Biochem. Physiol. 2007, 88, 167–175. [Google Scholar] [CrossRef]
- Lopez-Moya, F.; Colom-Valiente, M.F.; Martinez-Peinado, P.; Martinez-Lopez, J.E.; Puelles, E.; Sempere-Ortells, J.M.; Lopez-Llorca, L.V. Carbon and nitrogen limitation increase chitosan antifungal activity in neurospora crassa and fungal human pathogens. Fungal Biol. 2015, 119, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Martinez, L.R.; Mihu, M.R.; Tar, M.; Cordero, R.J.B.; Han, G.; Friedman, A.J.; Friedman, J.M.; Nosanchuk, J.D. Demonstration of antibiofilm and antifungal efficacy of chitosan against candidal biofilms, using an in vivo central venous catheter model. J. Infect. Dis. 2010, 201, 1436–1440. [Google Scholar] [CrossRef] [PubMed]
- Cota-Arriola, O.; Cortez-Rocha, M.O.; Rosas-Burgos, E.C.; Burgos-Hernández, A.; López-Franco, Y.L.; Plascencia-Jatomea, M. Antifungal effect of chitosan on the growth of aspergillus parasiticus and production of aflatoxin b1. Polym. Int. 2011, 60, 937–944. [Google Scholar] [CrossRef]
- Xing, K.; Zhu, X.; Peng, X.; Qin, S. Chitosan antimicrobial and eliciting properties for pest control in agriculture: A review. Agron. Sustain. Dev. 2015, 35, 569–588. [Google Scholar] [CrossRef]
- Dean, R.; Van Kan, J.A.L.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef] [PubMed]
- Peña, A.; Uribe, S.; Pardo, J.P.; Borbolla, M. The use of a cyanine dye in measuring membrane potential in yeast. Arch. Biochem. Biophys. 1984, 231, 217–225. [Google Scholar]
- Hernández, A.; Cooke, D.T.; Clarkson, D.T. In vivo activation of plasma membrane h+-atpase hydrolytic activity by complex lipid-bound unsaturated fatty acids in Ustilago maydis. Eur. J. Biochem. 2002, 269, 1006–1011. [Google Scholar]
- Shor, E.; Perlin, D.S. Coping with stress and the emergence of multidrug resistance in fungi. PLoS Pathog. 2015, 11, e1004668. [Google Scholar] [CrossRef] [PubMed]
- Zakrzewska, A.; Boorsma, A.; Delneri, D.; Brul, S.; Oliver, S.G.; Klis, F.M. Cellular processes and pathways that protect saccharomyces cerevisiae cells against the plasma membrane-perturbing compound chitosan. Eukaryot. Cell. 2007, 6, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Krantz, M.; Becit, E.; Hohmann, S. Comparative genomics of the hog-signalling system in fungi. Curr. Genet. 2006, 49, 137–151. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Lauzardo, A.N.; Vega-Pérez, J.; Velázquez-del Valle, M.G.; Sánchez, N.S.; Peña, A.; Guerra-Sánchez, G. Changes in the functionality of plasma membrane of rhizopus stolonifer by addition of chitosan. J. Phytopathol. 2011, 159, 563–568. [Google Scholar]
- Pena, A.; Sanchez, N.S.; Calahorra, M. Effects of chitosan on candida albicans: Conditions for its antifungal activity. BioMed Res. Int. 2013, 2013, 15. [Google Scholar] [CrossRef] [PubMed]
- Palma-Guerrero, J.; Lopez-Jimenez, J.A.; Pérez-Berná, A.J.; Huang, I.C.; Jansson, H.B.; Salinas, J.; Villalaín, J.; Read, N.D.; Lopez-Llorca, L.V. Membrane fluidity determines sensitivity of filamentous fungi to chitosan. Mol. Microbiol. 2010, 75, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Alfaro-Gutiérrez, I.C.; Guerra-Sánchez, M.G.; Hernández-Lauzardo, A.N.; Velázquez-del Valle, M.G. Morphological and physiological changes on rhizopus stolonifer by effect of chitosan, oligochitosan or essential oils. J. Phytopathol. 2014, 162, 723–730. [Google Scholar]
- Lopez-Moya, F.; Kowbel, D.; Nueda, M.J.; Palma-Guerrero, J.; Glass, N.L.; Lopez-Llorca, L.V. Neurospora crassa transcriptomics reveals oxidative stress and plasma membrane homeostasis biology genes as key targets in response to chitosan. Mol. BioSyst. 2016, 12, 391–403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Rincón, J.; Vega-Pérez, J.; Guerra-Sánchez, M.G.; Hernández-Lauzardo, A.N.; Peña-Díaz, A.; Velázquez-Del Valle, M.G. Effect of chitosan on growth and plasma membrane properties of rhizopus stolonifer (ehrenb.:Fr.) vuill. Pestic. Biochem. Physiol. 2010, 97, 275–278. [Google Scholar]
- Cannon, R.D.; Lamping, E.; Holmes, A.R.; Niimi, K.; Tanabe, K.; Niimi, M.; Monk, B.C. Candida albicans drug resistance—Another way to cope with stress. Microbiology 2007, 153, 3211–3217. [Google Scholar] [CrossRef] [PubMed]
- Kroll, K.; Pähtz, V.; Kniemeyer, O. Elucidating the fungal stress response by proteomics. J. Proteom. 2014, 97, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.A.; Morgan, B.A.; Quinn, J. Stress signalling to fungal stress-activated protein kinase pathways. FEMS Microbiol. Lett. 2010, 306, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hayes, B.M.E.; Anderson, M.A.; Traven, A.; van der Weerden, N.L.; Bleackley, M.R. Activation of stress signalling pathways enhances tolerance of fungi to chemical fungicides and antifungal proteins. Cell. Mol. Life Sci. 2014, 71, 2651–2666. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Campbell, B.C.; Yu, J.; Mahoney, N.; Chan, K.L.; Molyneux, R.J.; Bhatnagar, D.; Cleveland, T.E. Examination of fungal stress response genes using saccharomyces cerevisiae as a model system: Targeting genes affecting aflatoxin biosynthesis by aspergillus flavus link. Appl. Microbiol. Biotechnol. 2005, 67, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Freitas, F.Z.; Virgilio, S.; Cupertino, F.B.; Kowbel, D.J.; Fioramonte, M.; Gozzo, F.C.; Glass, N.L.; Bertolini, M.C. The SEB-1 transcription factor binds to the STRE motif in Neurospora crassa and regulates a variety of cellular processes including the stress response and reserve carbohydrate metabolism. G3 Genes Genomes Genet. 2016, 6, 1327–1343. [Google Scholar] [CrossRef] [PubMed]
- Zakrzewska, A.; Boorsma, A.; Brul, S.; Hellingwerf, K.J.; Klis, F.M. Transcriptional response of saccharomyces cerevisiae to the plasma membrane-perturbing compound chitosan. Eukaryot. Cell 2005, 4, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, C.; Petrezsélyová, S.; Barreto, L.; Martínez, J.L.; Zahrádka, J.; Ariño, J.; Sychrová, H.; Ramos, J. Lack of main k+ uptake systems in saccharomyces cerevisiae cells affects yeast performance in both potassium-sufficient and potassium-limiting conditions. FEMS Yeast Res. 2010, 10, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Castillo, S.; Cabrera-Orefice, A.; Vázquez-Acevedo, M.; González-Halphen, D.; Uribe-Carvajal, S. During the stationary growth phase, yarrowia lipolytica prevents the overproduction of reactive oxygen species by activating an uncoupled mitochondrial respiratory pathway. Biochim. Biophys. Acta 2012, 1817, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Li, Y.; Schellhorn, H.E. Rapid kinetic microassay for catalase activity. J. Biomol. Tech. JBT 2007, 18, 185–187. [Google Scholar] [PubMed]
- Pham, C.D.; Yu, Z.; Sandrock, B.; Bölker, M.; Gold, S.E.; Perlin, M.H. Ustilago maydis rho1 and 14-3-3 homologues participate in pathways controlling cell separation and cell polarity. Eukaryot. Cell 2009, 8, 977–989. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds chitosan, oligochitosan and glycol-chitosan are not available from the authors. |

| Antifungal Tested (1mg·mL−1) | Total Phospholipid Concentration (mM Phospholipids g of Cells−1 Wet Weight) |
|---|---|
| Control | 4.54 ± 0.035 a |
| Chitosan | ND |
| Oligochitosan | 2.51 ± 0.12 b |
| Glycol-chitosan | 3.76 ± 0.043 c |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olicón-Hernández, D.R.; Uribe-Alvarez, C.; Uribe-Carvajal, S.; Pardo, J.P.; Guerra-Sánchez, G. Response of Ustilago maydis against the Stress Caused by Three Polycationic Chitin Derivatives. Molecules 2017, 22, 1745. https://doi.org/10.3390/molecules22121745
Olicón-Hernández DR, Uribe-Alvarez C, Uribe-Carvajal S, Pardo JP, Guerra-Sánchez G. Response of Ustilago maydis against the Stress Caused by Three Polycationic Chitin Derivatives. Molecules. 2017; 22(12):1745. https://doi.org/10.3390/molecules22121745
Chicago/Turabian StyleOlicón-Hernández, Dario Rafael, Cristina Uribe-Alvarez, Salvador Uribe-Carvajal, Juan Pablo Pardo, and Guadalupe Guerra-Sánchez. 2017. "Response of Ustilago maydis against the Stress Caused by Three Polycationic Chitin Derivatives" Molecules 22, no. 12: 1745. https://doi.org/10.3390/molecules22121745





