Chemical and Biological Research on Herbal Medicines Rich in Xanthones
Abstract
:1. Introdution
2. Plant Sources of Xanthones
3. Bioactivities of Xanthones
3.1. Effects on Cytotoxicity and Proliferation
3.2. Free Radical Scavenging Activity
3.3. Anti-Microbial Activity
3.4. α-Glucosidase Inhibitory Activity
3.5. Anti-Virus Activity
3.6. Anti-Inflammatory Activity
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vieira, L.M.; Kijjoa, A. Naturally-occurring xanthones: Recent developments. Curr. Med. Chem. 2005, 12, 2413–2446. [Google Scholar] [CrossRef] [PubMed]
- Peres, V.; Nagem, T.J.; de Oliveira, F.F. Tetraoxygenated naturally occurring xanthones. Phytochemistry 2000, 55, 683–710. [Google Scholar] [CrossRef]
- Han, Q.B.; Xu, H.X. Caged Garcinia xanthones: Development since 1937. Curr. Med. Chem. 2009, 16, 3775–3796. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Orozco, F.; Failla, M.L. Biological activities and bioavailability of mangosteen xanthones: A critical review of the current evidence. Nutrients 2013, 5, 3163–3183. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, W. Gambogic Acid is a Novel Anti-cancer Agent that Inhibits Cell Proliferation, Angiogenesis and Metastasis. Anticancer Agents. Med. Chem. 2012, 12, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, W.; Zhao, Q.; Qi, Q.; Lu, N.; Yang, Y.; Nei, F.F.; Rong, J.J.; You, Q.D.; Guo, Q.L. Synergistic effect of 5-fluorouracil with gambogic acid on BGC-823 human gastric carcinoma. Toxicology 2009, 256, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, L.; You, Q.D.; Nie, F.F.; Gu, H.Y.; Zhao, L.; Wang, X.T.; Guo, Q.L. Differential apoptotic induction of gambogic acid, a novel anticancer natural product, on hepatoma cells and normal hepatocytes. Cancer Lett. 2007, 256, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Wei, J.; Qian, X.; Ding, Y.; Yu, L.; Liu, B. Gambogic acid, a potent inhibitor of survivin, reverses docetaxel resistance in gastric cancer cells. Cancer Lett. 2008, 262, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.T.; Wang, J.W. Phase I human tolerability trial of gambogic acid. Chin. J. New Drugs 2007, 1, 79–83. [Google Scholar]
- Tang, Y.P.; Li, P.G.; Kondo, M.; Ji, H.P.; Kou, Y.; Ou, B. Effect of a mangosteen dietary supplement on human immune function: A randomized, double-blind, placebo-controlled trial. J. Med. Food 2009, 12, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Dua, V.K.; Verma, G.; Dash, A.P. In vitro antiprotozoal activity of some xanthones isolated from the roots of Andrographis paniculata. Phytother. Res. 2009, 23, 126–128. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.E.; Zambrano, R.; Sepúlveda, B.; Simirgiotis, M.J. Antioxidant properties and hyphenated HPLC-PDA-MS profiling of chilean pica mango fruits (Mangifera indica L. Cv. piqueño). Molecules 2013, 19, 438–458. [Google Scholar] [CrossRef] [PubMed]
- Singh, O.; Ali, M.; Akhtar, N. New antifungal xanthones from the seeds of Rhus coriaria L. Z. Naturforsch. C 2011, 66, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Sabphon, C.; Sermboonpaisarn, T.; Sawasdee, P. Cholinesterase inhibitory activities of xanthones from Anaxagorea luzonensis A. Gray. J. Med. Plant Res. 2012, 6, 3781–3785. [Google Scholar]
- Emmanoel, V.C.; Assis, M.F.; Lúcia, B.M.; Braga, R.M. Chemical constituents isolated from the bark of Guatteria blepharophylla (Annonaceae) and their antiproliferative and antimicrobial activities. J. Braz. Chem. Soc. 2011, 22, 1111–1117. [Google Scholar]
- Waller, C.P.; Thumser, A.E.; Langat, M.K.; Crouch, N.R.; Mulholland, D.A. COX-2 inhibitory activity of homoisoflavanones and xanthones from the bulbs of the Southern African Ledebouria socialis and Ledebouria ovatifolia (Hyacinthaceae: Hyacinthoideae). Phytochemistry 2013, 95, 284–290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, J.; Xu, C.H.; Xue, R.; Jiang, W.X.; Wu, B.; Huang, C.G. Cytotoxic activities of chemical constituents from rhizomes of Anemarrhena asphodeloides and their analogues. Arch. Pharm. Res. 2015, 38, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, D.A.; Koorbanally, C.; Crouch, N.R.; Sandor, P. Xanthones from Drimiopsis maculate. J. Nat. Prod. 2004, 67, 1726–1728. [Google Scholar] [CrossRef] [PubMed]
- Cottiglia, F.; Casu, L.; Bonsignore, L.; Casu, M.; Floris, C.; Sosa, S.; Altinier, G.; Loggia, R.D. Topical anti-inflammatory activity of flavonoids and a new xanthone from Santolina insularis. Z. Naturforsch. C 2005, 60, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Pauletti, P.M.; Castro-Gamboa, I.; Silva, D.; Helena, S. New antioxidant C-glucosylxanthones from the stems of Arrabidaea samydoides. J. Nat. Prod. 2003, 66, 1384–1387. [Google Scholar] [CrossRef] [PubMed]
- Sati, S.C.; Sati, M.D.; Sharma, A. Isolation and characterization of flavone di-glucoside and acetoxyxanthone from the flowers of Bombex ceiba. J. Appl. Nat. Sci. 2011, 3, 128–130. [Google Scholar]
- Nkengfack, A.E.; Azebaze, G.A.; Vardamides, J.C.; Fomum, Z.T.; Heerden, F.R. A prenylated xanthone from Allanblackia floribunda. Phytochemistry 2002, 60, 381–384. [Google Scholar] [CrossRef]
- Azebaze, A.G.B.; Ouahouo, B.M.W.; Vardamides, J.C.; Valentin, A.; Kuete, V.; Acebey, L.; Beng, V.P.; Nkengfack, A.E.; Meyer, M. Antimicrobial and antileishmanial xanthones from the stem bark of Allanblackia gabonensis. Chem. Nat. Compd. 2008, 44, 582–587. [Google Scholar] [CrossRef]
- Azebaze, A.G.; Menasria, F.; Noumi, L.G.; Nguemfo, E.L. Xanthones from the seeds of Allanblackia monticola and their apoptotic and antiproliferative activities. Planta Med. 2009, 75, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, M.P.; Lima, M.M.C.; Santos, M.G.; Rocha, L.M.; Kuster, R.M. Anthraquinones and xanthone from Bonnetia stricta and their chemosystematic significance. Biochem. Syst. Ecol. 2013, 48, 73–75. [Google Scholar] [CrossRef]
- Blanco-Ayala, T.; Lugo-Huitron, R.; Serrano-Lopez, E.M.; Reyes-Chilpa, R.; Rangel-Lopez, E.; Pineda, B.; Medina-Campos, O.N.; Sanchez-Chapul, L.; Pinzon, E.; Cristina, T.S. Antioxidant properties of xanthones from Calophyllum brasiliense: Prevention of oxidative damage induced by FeSO4. BMC Complement. Altern. Med. 2013, 13, 262. [Google Scholar] [CrossRef] [PubMed]
- Oger, J.M.; Morel, C.; Helesbeux, J.J.; Litaudon, M.; Seraphin, D.; Dartiguelongue, C.; Larcher, G.; Richomme, P.; Duval, O. First 2-hydroxy-3-methylbut-3-enyl substituted xanthones isolated from plants: Structure elucidation, synthesis and antifungal activity. Nat. Prod. Res. 2003, 17, 195–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ajithabai, M.D.; Rameshkumar, B.; Jayakumar, G.; Varma, L.; Nair, M.S.; Ajaikumar Nair, G.P. Decipic acid and 12-acetyl apetalic acid from Calophyllum decipiens Wight. Indian J. Chem. B 2012, 51B, 393–397. [Google Scholar]
- Nasir, N.M.; Rahmani, M.; Shaari, K.; Kassim, N.K.; Go, R.; Stanslas, J.; Jeyaraj, E.J. Xanthones from Calophyllum gracilipes and their cytotoxic activity. Sains Malays. 2013, 42, 1261–1266. [Google Scholar]
- Iinuma, M.; Tosa, H.; Tanaka, T.; Yonemori, S. Two xanthones from roots of Calophyllum inophyllum. Phytochemistry 1995, 38, 725–728. [Google Scholar] [CrossRef]
- Ming, M.; Zhang, X.; Chen, H.F.; Zhu, L.J.; Zeng, D.Q.; Yang, J.; Wu, G.X.; Wu, Y.Z.; Yao, X.S. RXRα transcriptional inhibitors from the stems of Calophyllum membranaceum. Fitoterapia 2016, 108, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Ito, C.; Miyamoto, Y.; Rao, K.S.; Furukawa, H. A novel dibenzofuran and two new xanthones from Calophyllum panciflorum. Chem. Pharm. Bull. 1996, 44, 441–443. [Google Scholar] [CrossRef]
- Alarcon, A.B.; Cuesta-Rubio, O.; Cardenas Perez, J.; Piccinelli, A.L.; Rastrelli, L. Constituents of the Cuban endemic species Calophyllum pinetorum. J. Nat. Prod. 2008, 71, 1283–1286. [Google Scholar] [CrossRef] [PubMed]
- Mah, S.H.; Ee, G.C.; The, S.S.; Rahmani, M.; Lim, Y.M.; Go, R. Phylattrin, a new cytotoxic xanthone from Calophyllum soulattri. Molecules 2012, 17, 8303–8311. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, F.; Muhamud, A.; Hashim, R.; Sulaiman, O.; Ohara, S. Two antifungal xanthones from the heartwood of Calophyllum symingtonianum. Jpn. Agric. Res. 2012, 46, 181–185. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Nguyen, D.M.; Nguyen, L.H.D. A new xanthone from the bark of Calophyllum thorelii. Nat. Prod. Res. 2013, 27, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Dharmaratne, H.R.W.; Wanigasekera, W.M.A.P. Xanthones from root bark of Calophyllum thwaitesii. Phytochemistry 1996, 42, 249–250. [Google Scholar] [CrossRef]
- Molinar-Toribio, E.; Gonzalez, J.; Ortega-Barria, E.; Capson, T.L.; Coley, P.D.; Kursar, T.A.; McPhail, K.; Cubilla-Rios, L. Antiprotozoal activity against Plasmodium falciparum and Trypanosoma cruzi of xanthones isolated from Chrysochlamys tenuis. Pharm. Biol. 2006, 44, 550–553. [Google Scholar] [CrossRef]
- Silva, E.M.; Araujo Renata, M.; Freire-Filha, L.G.; Silveira, E.R.; Lopes, N.P.; Elias, P.J.; Braz-Filho, R.; Espindola, L.S. Clusiaxanthone and tocotrienol series from Clusia pernambucensis and their antileishmanial activity. J. Braz. Chem. Soc. 2013, 24, 1314–1321. [Google Scholar]
- Wilawan, M.; Rattanaburi, S.; Phongpaichit, S.; Kanjana-Opas, A. Antibacterial and cytotoxic xanthones from Cratoxylum cochinchinense. Phytochem. Lett. 2008, 1, 211–214. [Google Scholar]
- Boonnak, N.; Chantrapromma, S.; Tewtrakul, S.; Sudsai, T. Inhibition of nitric oxide production in lipopolysaccharide-activated RAW264.7 macrophages by isolated xanthones from the roots of Cratoxylum formosum ssp. pruniflorum. Arch. Pharm. Res. 2014, 37, 1329–1335. [Google Scholar] [CrossRef] [PubMed]
- Kamdem, W.; Mulholland, D.; Wansi, J.D.; Mbaze, L.M.; Powo, R. Afzeliixanthones A and B, 2 new prenylated xanthones from Garcinia afzelii ENGL. (Guttiferae). Chem. Pharm. Bull. 2006, 54, 448–451. [Google Scholar] [CrossRef]
- Niu, S.L.; Li, Z.L.; Ji, F.; Liu, G.Y.; Zhao, N.; Liu, X.Q.; Jing, Y.K.; Hua, H.M. Xanthones from the stem bark of Garcinia bracteata with growth inhibitory effects against HL-60 cells. Phytochemistry 2012, 77, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Milena, M.; Bassarello, C.; Bifulco, G.; Piacente, S. Polyisoprenylated benzophenone derivatives from the fruits of Garcinia cambogia and their absolute configuration by quantum chemical circular dichroism calculations. Tetrahedron 2010, 66, 139–145. [Google Scholar]
- Jantan, I.; Saputri, F.C. Benzophenones and xanthones from Garcinia cantleyana var. cantleyana and their inhibitory activities on human low-density lipoprotein oxidation and platelet aggregation. Phytochemistry 2012, 80, 58–63. [Google Scholar] [PubMed]
- Ritthiwigrom, T.; Laphookhieo, S.; Pyne, S.G. Chemical constituents and biological activities of Garcinia cowa Roxb. Maejo. Int. J. Sci. Technol. 2013, 7, 212–231. [Google Scholar]
- Iinuma, M.; Tosa, H.; Tanaka, T.; Riswan, S. Three new xanthones from the bark of Garcinia dioica. Chem. Pharm. Bull. 1996, 44, 232–234. [Google Scholar] [CrossRef]
- Deachathai, S.; Mahabusarakam, W.; Phongpaichit, S.; Taylor, W.C. Phenolic compounds from the fruit of Garcinia dulcis. Phytochemistry 2005, 66, 2368–2375. [Google Scholar] [CrossRef] [PubMed]
- Mian, J.V.Y.; Lian, E.G.C.; Aspollah, S.M.; Hin, T.Y.Y.; Yen, K.H.; Yok, C.M.K. Benzophenone constituents from the roots of Garcinia eugenifolia. Res. J. Chem. Environ. 2012, 16, 36–39. [Google Scholar]
- Ito, C.; Itoigawa, M.; Takakura, T.; Ruangrungsi, N.; Enjo, F.; Tokuda, H.; Nishino, H.; Furukawa, H. Chemical constituents of Garcinia fusca: Structure elucidation of eight new xanthones and their cancer chemopreventive activity. J. Nat. Prod. 2003, 66, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Mahamodo, S.; Riviere, C.; Neut, C.; Abedini, A.; Ranarivelo, H.; Duhal, N.; Roumy, V. Antimicrobial prenylated benzoylphloroglucinol derivatives and xanthones from the leaves of Garcinia goudotiana. Phytochemistry 2014, 102, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Alkadi, K.A.A.; Adam, A.; Taha, M.; Hasan, M.H.; Shah, S.A.A. Prenylated xanthone and rubraxanthone with antiplatelet aggregation activity in human whole blood isolated from Garcinia griffithii. Orient. J. Chem. 2013, 29, 1291–1295. [Google Scholar] [CrossRef]
- Deng, Y.X.; Pan, S.L.; Zhao, S.Y.; Wu, M.Q.; Sun, Z.Q.; Chen, X.H.; Shao, Z.Y. Cytotoxic alkoxylated xanthones from the resin of Garcinia hanburyi. Fitoterapia 2012, 83, 1548–1552. [Google Scholar] [CrossRef] [PubMed]
- Klaiklay, S.; Sukpondma, Y.; Rukachaisirikul, V.; Phongpaichit, S. Friedolanostanes and xanthones from the twigs of Garcinia hombroniana. Phytochemistry 2013, 85, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Li, D.; Jia, C.C.; Xue, C.M.; Bai, J.; Li, Z.L.; Hua, H.M. Three new xanthones from the leaves of Garcinia lancilimba. J. Nat. Med. 2016, 70, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Lantvit, D.D.; Carcache, E.J.; Kardono, L.B.S.; Riswan, S. Proteasome-inhibitory and cytotoxic constituents of Garcinia lateriflora: Absolute configuration of caged xanthones. Tetrahedron 2010, 66, 5311–5320. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Chen, I.S.; Duh, C.Y. Cytotoxic xanthones and biphenyls from the root of Garcinia linii. Planta Med. 2004, 70, 1195–1200. [Google Scholar] [CrossRef] [PubMed]
- Chae, H.S.; Kim, E.Y.; Han, L.; Kim, N.R.; Lam, B.; Paik, J.H.; Yoon, K.D. Xanthones with pancreatic lipase inhibitory activity from the pericarps of Garcinia mangostana L. (Guttiferae). Eur. J. Lipid Sci. Technol. 2016, 118, 1416–1421. [Google Scholar] [CrossRef]
- Nguyen, L.H.D.; Vo, H.T.; Pham, H.D.; Connolly, J.D.; Harrison, L.J. Xanthones from the bark of Garcinia merguensis. Phytochemistry 2003, 63, 467–470. [Google Scholar] [CrossRef]
- Chiang, Y.M.; Kuo, Y.H.; Oota, S.; Fukuyama, Y. Xanthones and benzophenones from the stems of Garcinia multiflora. J. Nat. Prod. 2003, 66, 1070–1073. [Google Scholar] [CrossRef] [PubMed]
- Rukachaisirikul, V.; Ritthiwigrom, T.; Pinsa, A.; Sawangchote, P.; Taylor, W.C. Xanthones from the stem bark of Garcinia nigrolineata. Phytochemistry 2003, 64, 1149–1156. [Google Scholar] [CrossRef]
- Ee, G.C.L.; Foo, C.H.; Jong, V.Y.M.; Ismail, N.H.; Sukari, M.A.; Ya, Y.H.T.; Awang, K. A new xanthone from Garcinia nitida. Nat. Prod. Res. 2012, 26, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Fouotsa, H.; Tatsimo, S.J.N.; Neumann, B.; Michalek, C.; Mbazoa, C.D.; Nkengfack, A.E.; Sewald, N.; Lannang, A.M. A new xanthone derivative from twigs of Garcinia nobilis. Nat. Prod. Res. 2014, 28, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Xia, Z.X.; Qiao, S.P.; Jiang, C.; Shen, G.R.; Cai, M.X.; Tang, X.Y. Four new cytotoxic xanthones from Garcinia nujiangensis. Fitoterapia 2015, 102, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Shan, W.G.; Lin, T.D.; Yu, H.N.; Chen, Y.; Zhan, Z.J. Polyprenylated xanthones and benzophenones from the bark of Garcinia oblongifolia. Helv. Chim. Acta 2012, 95, 1442–1448. [Google Scholar] [CrossRef]
- Tang, Y.X.; Fu, W.W.; Wu, R.; Tan, H.S.; Shen, Z.; Xu, H.X. Bioassay-guided isolation of prenylated xanthone derivatives from the leaves of Garcinia oligantha. J. Nat. Prod. 2016, 79, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Ha, L.D.; Hansen, P.E.; Vang, O.; Duus, F.; Pham, H.D.; Nguyen, L.H.D. Cytotoxic geranylated xanthones and O-alkylated derivatives of α-mangostin. Chem. Pharm. Bull. 2009, 57, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Kardono, L.B.S.; Hanafi, M.; Sherley, G.; Kosela, S.; Harrison, L.J. Bioactive constituents of Garcinia porrecta and G. parvifolia grown in Indonesia. Pak. J. Biol. Sci. 2006, 9, 483–486. [Google Scholar]
- Wu, Y.P.; Zhao, W.; Xia, Z.Y.; Kong, G.H.; Lu, X.P.; Hu, Q.F.; Gao, X.M. Three novel xanthones from Garcinia paucinervis and their anti-TMV activity. Molecules 2013, 18, 9663–9669. [Google Scholar] [CrossRef] [PubMed]
- Vo, H.T.; Nguyen, N.T.T.; Maas, G.; Werz, U.R.; Pham, H.D.; Nguyen, L.H.D. Xanthones from the bark of Garcinia pedunculata. Phytochem. Lett. 2012, 5, 766–769. [Google Scholar] [CrossRef]
- Jabit, M.L.; Khalid, R.; Abas, F.; Shaari, K.; Hui, L.S.; Stanslas, J.; Lajis, N.H. Cytotoxic xanthones from Garcinia penangiana Pierre. Z. Naturforsch. C 2007, 62, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Lannang, A.M.; Komguem, J.; Ngninzeko, F.N.; Tangmouo, J.G.; Lontsi, D.; Ajaz, A.; Choudhary, M.I.; Ranjit, R.; Devkota, K.P.; Sondengam, B.L. Bangangxanthone A and B, two xanthones from the stem bark of Garcinia polyantha Oliv. Phytochemistry 2005, 66, 2351–2355. [Google Scholar] [CrossRef] [PubMed]
- Tantapakul, C.; Phakhodee, W.; Ritthiwigrom, T.; Cheenpracha, S.; Prawat, U.; Deachathai, S.; Laphookhieo, S. Rearranged benzophenones and prenylated xanthones from Garcinia propinqua Twigs. J. Nat. Prod. 2012, 75, 1660–1664. [Google Scholar] [CrossRef] [PubMed]
- Elya, B.; He, H.P.; Kosela, S.; Hanafi, M.; Hao, X.J. A new cytotoxic xanthone from Garcinia rigida. Fitoterapia 2008, 79, 182–184. [Google Scholar] [CrossRef] [PubMed]
- Sukandar, E.R.; Siripong, P.; Khumkratok, S.; Tip-pyang, S. New depsidones and xanthone from the roots of Garcinia schomburgkiana. Fitoterapia 2016, 111, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Sukpondma, Y.; Rukachaisirikul, V.; Phongpaichit, S. Xanthone and sesquiterpene derivatives from the fruits of Garcinia scortechinii. J. Nat. Prod. 2005, 68, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Fouotsa, H.; Lannang, A.M.; Dzoyem, J.P.; Tatsimo, S.J.N.; Neumann, B.; Mbazoa, C.D. Antibacterial and antioxidant xanthones and benzophenone from Garcinia smeathmannii. Planta Med. 2015, 81, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Ngoupayo, J.; Tabopda, T.K.; Ali, M.S. Antimicrobial and immunomodulatory properties of prenylated xanthones from twigs of Garcinia staudtii. Bioorg. Med. Chem. 2009, 17, 5688–5695. [Google Scholar] [CrossRef] [PubMed]
- Abe, F.; Nagafuji, S.; Okabe, H.; Higo, H.; Akahane, H. Trypanocidal constituents in plants 2. Xanthones from the stem bark of Garcinia subelliptica. Biol. Pharm. Bull. 2003, 26, 1730–1733. [Google Scholar] [CrossRef] [PubMed]
- Duangsrisai, S.; Choowongkomon, K.; Bessa, L.J.; Costa, P.M.; Amat, N.; Kijjoa, A. Antibacterial and EGFR-tyrosine kinase inhibitory activities of polyhydroxylated xanthones from Garcinia succifolia. Molecules 2014, 19, 19923–19934. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.E.; Wang, L.L.; Li, Z.L.; Niu, S.L.; Liu, X.Q.; Hua, H.M.; Chen, H.; Chu, J.; Zhang, T.C. Triterpenes and xanthones from the stem bark of Garcinia tetralata. J. Asian Nat. Prod. Res. 2011, 13, 440–443. [Google Scholar] [CrossRef] [PubMed]
- Hay, A.E.; Aumond, M.C.; Mallet, S.; Dumontet, V.; Litaudon, M.; Rondeau, D.; Richomme, P. Antioxidant Xanthones from Garcinia vieillardii. J. Nat. Prod. 2004, 67, 707–709. [Google Scholar] [CrossRef] [PubMed]
- Merza, J.; Aumond, M.C.; Rondeau, D.; Dumontet, V.; Le Ray, A.M. Prenylated xanthones and tocotrienols from Garcinia virgate. Phytochemistry 2004, 65, 2915–2920. [Google Scholar] [CrossRef] [PubMed]
- Zhong, F.F.; Chen, Y.; Wang, P.; Feng, H.J.; Yang, G.Z. Xanthones from the bark of Garcinia xanthochymus and their 1,1-diphenyl-2-picrylhydrazyl radical-scavenging activity. Chin. J. Chem. 2009, 27, 74–80. [Google Scholar] [CrossRef]
- Han, Q.B.; Yang, N.Y.; Tian, H.L.; Qiao, C.F.; Song, J.Z.; Chang, D.C.; Chen, S.L.; Luo, K.Q.; Xu, H.X. Xanthones with growth inhibition against HeLa cells from Garcinia xipshuanbannaensis. Phytochemistry 2008, 69, 2187–2192. [Google Scholar] [CrossRef] [PubMed]
- Biesdorf, C.; Cortez, D.A.G.; Audi, E.A. Assessment of anxiolytic and panicolytic effects of dichloromethane fraction from stems of Kielmeyera coriacea. Phytomedicine 2012, 19, 374–377. [Google Scholar] [CrossRef] [PubMed]
- Coqueiro, A.; Choi, Y.H.; Verpoorte, R.; Gupta, K.B.S.S.; De Mieri, M.; Hamburger, M.; Young, M.C.M.; Stapleton, P.; Gibbons, S.; Bolzani, V.S. Antistaphylococcal prenylated acylphoroglucinol and xanthones from Kielmeyera variabilis. J. Nat. Prod. 2016, 79, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Laphookhieo, S.; Promnart, P.; Syers, J.K.; Kanjana-Opas, A.; Ponglimanont, C.; Karalai, C. Coumarins and xanthones from the seeds of Mammea siamensis. J. Braz. Chem. Soc. 2007, 18, 1077–1080. [Google Scholar] [CrossRef]
- Ee, G.C.L.; The, S.S.; Rahmani, M.; Taufiq-Yap, Y.H.; Go, R.; Mah, S.H. A new furanoxanthone from the root bark of Mesua ferrea. Lett. Org. Chem. 2012, 9, 457–459. [Google Scholar]
- Karunakaran, T.; Ee, G.C.L.; The, S.S.; Daud, S.; Mah, S.H.; Lim, C.K.; Jong, V.Y.M.; Awang, K. A new coumarin from stem bark of Mesua hexapetala. Nat. Prod. Res. 2016, 30, 1591–1597. [Google Scholar] [CrossRef] [PubMed]
- Maurice, T.; Marlyse, O.W.B.; Robert, N.J.; Pierre, M.; Victor, K.; Sterner, O.; Michelle, M.; Ephrem, N.A. Antimicrobial prenylated xanthones and anthraquinones from barks and fruits of Psorospermum adamauense (Engl.). Nat. Prod. J. 2013, 3, 60–65. [Google Scholar] [CrossRef]
- Abou-Shoer, M.; Suwanborirux, K.; Habib, A.A.M.; Chang, C.J.; Cassady, J.M. Xanthones and vismiones from Psorospermum febrifugum. Phytochemistry 1993, 34, 1413–1420. [Google Scholar] [CrossRef]
- Leet, J.E.; Liu, X.H.; Drexler, D.M.; Cantone, J.L.; Huang, S.; Mamber, S.W.; Fairchild, C.R.; Hussain, R.; Newman, D.J.; Kingston, D.G.I. Cytotoxic xanthones from Psorospermum molluscum from the madagascar rain forest. J. Nat. Prod. 2008, 71, 460–463. [Google Scholar] [CrossRef] [PubMed]
- Almanza, G.R.; Quispe, R.; Mollinedo, P.; Rodrigo, G.; Fukushima, O.; Villagomez, R.; Akesson, B.; Sterner, O. Antioxidant and antimutagenic polyisoprenylated benzophenones and xanthones from Rheedia acuminate. Nat. Prod. Commun. 2011, 6, 1269–1274. [Google Scholar] [PubMed]
- Cottet, K.; Neudorffer, A.; Kritsanida, M.; Michel, S.; Lallemand, M.C.; Largeron, M. Polycyclic polyprenylated xanthones from Symphonia globulifera: Isolation and biomimetic electrosynthesis. J. Nat. Prod. 2015, 78, 2136–2140. [Google Scholar] [CrossRef] [PubMed]
- Tala, M.F.; De Dien Tamokou, J.; Tchakam, P.D.; Tane, P.; Kuiate, J.R.; Wabo, H.K. Antioxidant xanthones, anthraquinones and semi-synthetic derivatives from Vismia rubescens and Vismia laurentii. Pharmacologyonline 2011, 3, 1410–1418. [Google Scholar]
- Zhao, H.; Wang, X.; Li, W.; Koike, K.; Bai, H. A new minor homoisoflavonoid from Caesalpinia sappan. Nat. Prod. Res. 2014, 28, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Sob, S.V.T.; Wabo, H.K.; Tane, P.; Ngadjui, B.T.; Ma, D. A xanthone and a polyketide derivative from the leaves of Cassia obtusifolia (Leguminosae). Tetrahedron 2008, 64, 7999–8002. [Google Scholar] [CrossRef]
- Kokotkiewicz, A.; Luczkiewicz, M.; Pawlowska, J.; Luczkiewicz, P.; Sowinski, P.; Witkowski, J.; Bryl, E.; Bucinski, A. Isolation of xanthone and benzophenone derivatives from Cyclopia genistoides (L.) Vent. (honeybush) and their pro-apoptotic activity on synoviocytes from patients with rheumatoid arthritis. Fitoterapia 2013, 90, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Sun, Y.N.; Yan, X.T.; Yang, S.Y.; Choi, C.W.; Kim, Y.H. Phenolic compounds from Desmodium caudatum. Nat. Prod. Sci. 2013, 19, 215–220. [Google Scholar]
- Rezanka, T.; Jachymova, J.; Dembitsky, V.M. Prenylated xanthone glucosides from Ural’s lichen Umbilicaria proboscidea. Phytochemistry 2003, 62, 607–612. [Google Scholar] [CrossRef]
- El-Shanawany, M.A.; Mohamed, G.A.; Nafady, A.M.; Ibrahim, S.R.M.; Radwan, M.M.; Ross, S.A. A new xanthone from the roots of Centaurium spicatum. Phytochem. Lett. 2011, 4, 126–128. [Google Scholar] [CrossRef]
- Zhou, M.; Zhou, K.; Zhao, Y.L.; Xiang, N.J.; Zhang, T.D.; Wang, Y.D.; Dong, W.; Ji, B.K.; Li, L.M.; Lou, J. Three new prenylated xanthones from Comastoma pedunculatum and their anti-tobacco mosaic virus activity. Phytochem. Lett. 2015, 11, 245–248. [Google Scholar] [CrossRef]
- Zhou, M.; Zhou, K.; Zhao, Y.L.; Xiang, N.J.; Zhang, T.D.; Wang, Y.D.; Zhang, C.M.; Wang, Y.D.; Dong, W.; Ji, B.K.; et al. New xanthones from Comastoma pulmonarium and their anti-tobacco mosaic virus activity. Heterocycles 2015, 91, 604–609. [Google Scholar]
- Krstic-Milosevic, D.; Jankovic, T.; Vinterhalter, B.; Menkovic, N.; Aljancic, I.; Vinterhalter, D. Influence of carbohydrate source on xanthone content in root cultures of Gentiana dinarica Beck. Plant Growth Regul. 2013, 71, 147–155. [Google Scholar] [CrossRef]
- Isakovic, A.; Jankovic, T.; Harhaji, L.; Kostic-Rajacic, S.; Nikolic, Z.; Vajs, V.; Trajkovic, V. Antiglioma action of xanthones from Gentiana kochiana: Mechanistic and structure-activity requirements. Bioorg. Med. Chem. 2008, 16, 5683–5694. [Google Scholar] [CrossRef] [PubMed]
- Aberham, A.; Schwaiger, S.; Stuppner, H.; Ganzera, M. Quantitative analysis of iridoids, secoiridoids, xanthones and xanthone glycosides in Gentiana lutea L. roots by RP-HPLC and LC-MS. J. Pharm. Biomed. Anal. 2007, 45, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Xu, X.Z.; Zhan, H.Y.; Wang, A.L. Two new O- and C-glycosylxanthones from Gentiana tizuensis Franch. Indian J. Chem. B 2003, 42B, 950–952. [Google Scholar]
- Jankovic, T.; Krstic-Milosevic, D.; Aljancic, I.; Savikin, K.; Menkovic, N.; Radanovic, D.; Milosavljevic, S. Phytochemical re-investigation of Gentiana utriculosa. Nat. Prod. Res. 2009, 23, 466–469. [Google Scholar] [PubMed]
- Petrovic, S.; Leskovac, A.; Joksic, G. Radioprotective properties of Gentiana dinarica polyphennols on human lymphocytes in vitro. Res. Artic. 2008, 95, 1035–1041. [Google Scholar]
- Lv, L.J.; Li, M.H. Terpenoilds, flavonoids and xanthones from Gentianella acuta (Gentianaceae). Biochem. Syst. Ecol. 2009, 37, 497–500. [Google Scholar]
- Urbain, A.; Marston, A.; Batsuren, D.; Purev, O.; Hostettmann, K. Preparative isolation of closely-related xanthones from Gentianella amarella ssp. acuta by high-speed countercurrent chromatography. Phytochem. Anal. 2008, 19, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zou, D.Z.; Bai, S.; Li, Z.H.; Zhang, C.H.; Li, M.H. Chemical constituents from Gentianella turkestanorum (Gentianaceae). Biochem. Syst. Ecol. 2016, 65, 89–92. [Google Scholar] [CrossRef]
- Cui, Z.H.; Li, Y.; Wang, Z.W.; Zhang, J.; Zhang, C.H.; Zhang, N.; Li, M.H. Chemical constituents from Gentianopsis barbata var. sinensis Ma (Gentianaceae). Biochem. Syst. Ecol. 2013, 47, 101–103. [Google Scholar] [CrossRef]
- Yeung, M.F.; Lau, C.B.S.; Chan, R.C.Y.; Zong, Y.Y.; Che, C.T. Search for antimycobacterial constituents from a Tibetan medicinal plant, Gentianopsis paludosa. Phytother. Res. 2009, 23, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, S.; Wolfender, J.L.; Odontuya, G.; Purev, O.; Hostettmann, K. Xanthones, secoiridoids and flavonoids from Halenia corniculata. Phytochemistry 1995, 40, 1265–1272. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, J.G.; Wang, M.Z.; Che, C.T.; Yeung, J.H.K. Vasodilatory actions of xanthones isolated from a Tibetan herb, Halenia elliptica. Phytomedicine 2009, 16, 1144–1150. [Google Scholar] [PubMed]
- Li, L.; Li, M.H.; Zhang, N.; Huang, L.Q. Chemical constituents from Lomatogonium carinthiacum (Gentianaceae). Biochem. Syst. Ecol. 2011, 39, 766–768. [Google Scholar] [CrossRef]
- Terreaux, C.; Maillard, M.; Gupta, M.P.; Hostettmann, K. Xanthones from Schultesia lisianthoides. Phytochemistry 1995, 40, 1791–1795. [Google Scholar] [CrossRef]
- Singh, P.P.; Ambika Chauhan, S.M.S. Activity-guided isolation of antioxidant xanthones from Swertia chirayita (Roxb.) H. Karsten (Gentianaceae). Nat. Prod. Res. 2012, 26, 1682–1686. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.U.; Pervin, A.; Feroz, M.; Choudhary, M.I.; Qureshi, M.M.; Perveen, S.; Mir, I.; Khan, M.I. Phytochemical studies on Swertia cordata. J. Nat. Prod. 1994, 57, 134–137. [Google Scholar] [CrossRef]
- Uvarani, C.; Arumugasamy, K.; Chandraprakash, K.; Sankaran, M.; Ata, A.; Mohan, P.S. A new DNA-intercalative cytotoxic allylic xanthone from Swertia corymbosa. Chem. Biodivers. 2015, 12, 358–370. [Google Scholar] [CrossRef] [PubMed]
- Khetwal, K.S.; Rajput, P.S.; Sajwan, K.; Pathak, S.; Adhikari, A. 1,5-Dihydroxy-3,8-dimethoxyxanthone from Swertia cuneata. Indian J. Chem. B 2003, 42B, 953–955. [Google Scholar] [CrossRef]
- Jiang, W.; Zhu, D.L.; Wang, M.F.; Yang, Q.S.; Zuo, M.Y.; Zeng, L.; Li, G.P. Xanthones from the herb of Swertia elata and their anti-TMV activity. Nat. Prod. Res. 2015, 30, 1810–1815. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.G.; Zhang, X.; Xue, X.Y.; Zhang, Y.; Xiao, H.B.; Liang, X.M. Rapid identification of polyphenol C-glycosides from Swertia franchetiana by HPLC-ESI-MS-MS. J. Chromatogr. Sci. 2009, 47, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.S.; Min, Q.X.; Wang, Y.L.; Yue, Y.D.; Chen, J.C. Xanthone glycoside constituents of Swertia kouitchensis with α-glucosidase inhibitory activity. J. Nat. Prod. 2013, 76, 1248–1253. [Google Scholar] [CrossRef] [PubMed]
- Hajimehdipoor, H.; Dijoux-Franca, M.G.; Mariotte, A.M.; Amanzadeh, Y.; Sadat-Ebrahimi, S.E.; Ghazi-Khansari, M. Two new xanthone diglycosides from Swertia longifolia Boiss. Nat. Prod. Res. 2006, 20, 1251–1255. [Google Scholar] [CrossRef] [PubMed]
- Uvarani, C.; Chandraprakash, K.; Sankaran, M.; Ata, A.; Mohan, P.S. Antioxidant and structure-activity relationships of five tetraoxygenated xanthones from Swertia minor (Griscb.) Knobl. Nat. Prod. Res. 2012, 26, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.T.; Mao, S.S.; Liu, F.L.; Yang, M.X.; Chen, H.R.; Kurihara, H.; Li, Y.L. Antioxidant xanthones from Swertia mussotii, a high altitude plant. Fitoterapia 2013, 91, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Pant, N.; Misra, H.; Jain, D.C. A xanthone glycoside from aerial parts of Swertia paniculata. J. Saudi Chem. Soc. 2014, 18, 551–554. [Google Scholar] [CrossRef]
- Li, J.C.; Feng, L.; Sun, B.H.; Ikeda, T.; Nohara, T. Hepatoprotective activity of the constituents in Swertia pseudochinensis. Biol. Pharm. Bull. 2005, 28, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.Y.; Yang, Y.F.; Li, W.; Zhao, X.; Sun, Y.; Sun, H.; Wang, Y.H.; Pu, X.P. Two xanthones from Swertia punicea with hepatoprotective activities in vitro and in vivo. J. Ethnopharmacol. 2014, 153, 854–863. [Google Scholar] [CrossRef] [PubMed]
- Rana, V.S.; Rawat, M.S. A new xanthone glycoside and antioxidant constituents from the rhizomes of Swertia speciosa. Chem. Biodivers. 2005, 2, 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.F.; Luiz, F.; Lima, E.S.; Vasconcello, M.C. In vitro and in vivo antimalarial activity and cytotoxicity of extracts, fractions and a substance isolated from the Amazonian plant Tachia grandiflora (Gentianaceae). Memórias Instituto Oswaldo Cruz 2013, 108, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Periyar, S.S.; Magesh, V.; Raja, V.; Mahendran, T.S.; Muthumary, J.; Kalaichelvan, P.T.; Murugesan, K. Antimutagenicity of mangiferin purified from Salacia chinensis Linn. Int. Multidiscip. Res. J. 2011, 1, 1–5. [Google Scholar]
- Duarte, L.P.; Figueiredo, R.C.; Faria De Sousa, G.; Soares, D.B.S.; Rodrigues, S.B.V.; Silva, F.C.; Silva, G.D.F.; Filho, S.A.V. Chemical constituents of Salacia elliptica (Celastraceae). Quim. Nova 2010, 33, 900–903. [Google Scholar] [CrossRef]
- Nishida, Y.; Eto, M.; Miyashita, H.; Ikeda, T.; Yamaguchi, K.; Yoshimitsu, H.; Nohara, T.; Ono, M. A new homostilbene and two new homoisoflavones from the bulbs of Scilla scilloides. Chem. Pharm. Bull. 2008, 56, 1022–1025. [Google Scholar] [CrossRef] [PubMed]
- Hashida, W.; Tanaka, N.; Takaishi, Y. Prenylated xanthones from Hypericum ascyron. J. Nat. Med. 2007, 61, 371–374. [Google Scholar] [CrossRef]
- Zhou, Z.B.; Zhang, Y.M.; Luo, J.G.; Kong, L.Y. Cytotoxic polycyclic polyprenylated acylphloroglucinol derivatives and xanthones from Hypericum attenuatum. Phytochem. Lett. 2016, 15, 215–219. [Google Scholar] [CrossRef]
- Lou, J.; Wang, H.; Liu, G.Y.; Yang, J.X.; Li, L.M.; Hu, Q.F.; Gao, X.M. Isolation of new xanthone from Hypericum chinense and its cytotoxicity. Asian J. Chem. 2015, 27, 2102–2104. [Google Scholar] [CrossRef]
- Matsuoka, E.; Machida, K.; Kikuchi, M. Chemical constituents of Hypericum erectum Thunb. J. Nat. Med. 2008, 62, 467–469. [Google Scholar] [CrossRef] [PubMed]
- Wabo, H.; Kowa, T.K.; Lonfouo, A.H.N.; Tchinda, A.T.; Tane, P.; Kikuchi, H.; Frederich, M.; Oshima, Y. Phenolic compounds and terpenoids from Hypericum lanceolatum. Rec. Nat. Prod. 2012, 6, 94–100. [Google Scholar]
- Ali, M.; Latif, A.; Zaman, K.; Arfan, M.; Maitland, D.; Ahmad, H.; Ahmad, M. Anti-ulcer xanthones from the roots of Hypericum oblongifolium Wall. Fitoterapia 2014, 95, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Ishiguro, K.; Nakajima, M.; Fukumoto, H.; Isoi, K. A xanthone substituted with an irregular monoterpene in cell suspension cultures of Hypericum patulum. Phytochemistry 1995, 39, 903–905. [Google Scholar] [CrossRef]
- Sun, L.W.; Sun, Y.N.; Yan, X.T.; Yang, S.Y.; Choi, C.W.; Hyun, J.W.; Kang, H.K.; Paek, K.Y.; Kim, Y.H. Isolation of xanthones from adventitious roots of St. John’s Wort (Hypericum perforatum L.) and their antioxidant and cytotoxic activities. Food Sci. Biotechnol. 2013, 22, 945–949. [Google Scholar]
- Xiao, Z.Y.; Shiu, W.K.P.; Zeng, Y.H.; Mu, Q.; Gibbons, S. A naturally occurring inhibitory agent from Hypericum sampsonii with activity against multidrug-resistant staphylococcus aureus. Pharm. Biol. 2008, 46, 250–253. [Google Scholar] [CrossRef]
- Tanaka, N.; Takaishi, Y.; Shikishima, Y.; Nakanishi, Y.; Bastow, K.; Lee, K.H.; Takeda, Y.; Kodzhimatov, O.K. Prenylated benzophenones and xanthones from Hypericum scabrum. J. Nat. Prod. 2004, 67, 1870–1875. [Google Scholar] [CrossRef] [PubMed]
- Gamiotea-Turro, D.; Cuesta-Rubio, O.; Prieto-Gonzalez, S.; De Simone, F.; Passi, S.; Rastrelli, L. Antioxidative constituents from the leaves of Hypericum styphelioides. J. Nat. Prod. 2004, 67, 869–871. [Google Scholar] [CrossRef] [PubMed]
- Al-Khalil, S.; Tosa, H.; Iinuma, M. A xanthone C-glycoside from Iris nigricans. Phytochemistry 1995, 38, 729–731. [Google Scholar] [CrossRef]
- Mbouangouere, R.N.; Tane, P.; Ngamga, D.; Khan, S.N.; Choudhary, M.I.; Ngadjui, B.T. A new steroid and α-glucosidase inhibitors from Anthocleista schweinfurthii. Res. J. Med. Plant 2007, 1, 106–111. [Google Scholar]
- Ateufack, G.; Nguelefack, T.B.; Mbiantcha, M.; Tane, P.; Kamanyi, A. Spasmogenic activity of 1-hydroxy-3,7,8-trimethoxyxanthone isolated from the methanol extract of the stem bark of Anthocleista vogelii planch. (Loganiaceae) in rats. Pharmacologyonline 2007, 3, 374–384. [Google Scholar]
- Ee, G.C.; Teo, S.H.; Rahmani, M.; Lim, C.K.; Lim, Y.M.; Go, R. Artomandin, a new xanthone from Artocarpus kemando (Moraceae). Nat. Prod. Res. 2011, 25, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Jayasinghe, U.L.; Samarakoon, T.B.; Kumarihamy, B.M.M.; Hara, N.; Fujimoto, Y. Four new prenylated flavonoids and xanthones from the root bark of Artocarpus nobilis. Fitoterapia 2008, 79, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Hashim, N.M.; Rahmani, M.; Sukari, M.A.; Ali, A.M.; Alitheen, N.B.; Go, R.; Ismail, H.B.M. Two new xanthones from Artocarpus obtusus. J. Asian Nat. Prod. Res. 2010, 12, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhou, Q.; Li, B.; Liu, S.J.; Dong, J.X. A new flavonoid from Cudrania cochinchinensis. Nat. Prod. Res. 2015, 29, 1217–1221. [Google Scholar] [CrossRef] [PubMed]
- Liang, B.; Li, H.R.; Xu, L.Z.; Yang, S.L. Xanthones from the roots of Cudrania fruticosa Wight. J. Asian Nat. Prod. Res. 2007, 9, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Quang, T.H.; Ngan, N.T.T.; Yoon, C.S.; Cho, K.H.; Kang, D.G.; Lee, H.S.; Kim, Y.G.; Oh, H. Protein tyrosine phosphatase 1B inhibitors from the roots of Cudrania tricuspidata. Molecules 2015, 20, 11173–11183. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.; Tanaka, T.; Murata, H.; Kaburagi, K.; Inoue, M. Xanthones from the roots of Maclura cochinchinensis var. gerontogea and their retinoic acid receptor-α agonistic activity. Bioorg. Med. Chem. Lett. 2015, 25, 1998–2001. [Google Scholar] [PubMed]
- Ahmad, A.; Singh, D.K.; Fatima, K.; Tandon, S.; Luqman, S. New constituents from the roots of Oenothera biennis and their free radical scavenging and ferric reducing activity. Ind. Crops Prod. 2014, 58, 125–132. [Google Scholar] [CrossRef]
- Rezanka, T.; Sigler, K. Hirtusneanoside, an unsymmetrical dimeric tetrahydroxanthone from the Lichen Usnea hirta. J. Nat. Prod. 2007, 70, 1487–1491. [Google Scholar] [CrossRef] [PubMed]
- Silveira, E.R.; Falcao, M.J.C.; Kingston, D.G.I.; Glass, T.E. Pentaoxygenated xanthones from Bredemeyera floribunda. Phytochemistry 1995, 39, 1433–1436. [Google Scholar] [CrossRef]
- Filho, H.D.S.; Pacheco, L.C.; Andrade, E.S.; Correa, M.J.C.; Araujo, R.N.M.; Guilhon, G.M.S.P.; Da Silva, J.K.R.; Santos, L.S. Xanthones from the roots of Moutabea guianensis Aubl. Molecules 2015, 20, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.L.; Huang, F.; Chen, S.B.; Yang, D.J.; Chen, S.L.; Yang, J.S.; Xiao, P.G. Xanthones from the roots of Polygala caudata and their antioxidation and vasodilatation activities in vitro. Planta Med. 2005, 71, 372–375. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.M.; Yang, X.H.; Zhao, Y.X.; Zhou, J. New xanthones from Polygala crotalarioides. Chem. Res. Chin. Univ. 2006, 22, 400–402. [Google Scholar] [CrossRef]
- Klein, L.C.J.; Gandolfi, R.B.; Santin, J.R.; Lemos, M.; Cechinel Filho, V.; Andrade, S.F. Antiulcerogenic activity of extract, fract ions, and some compounds obtained from Polygala cyparissias St. Hillaire & Moquin (Polygalaceae). Naunyn Schmiedebergs Arch. Pharmacol. 2010, 381, 121–126. [Google Scholar] [PubMed]
- Wu, J.F.; Tu, P.F.; Zhan, H.T.; Gao, J.C. Dioxyxanthones from Polygala hongkongensis and their cytotoxicity. Chem. Res. Chin. Univ. 2011, 27, 777–779. [Google Scholar]
- Qing, C.X.; Li, C.J.; Zuo, L.; Yang, J.Z.; Zhang, D.M. Three new xanthones from the roots of Polygala japonica Houtt. J. Asian Nat. Prod. Res. 2009, 11, 465–469. [Google Scholar]
- Dao, T.T.; Dang, T.T.; Nguyen, P.H.; Kim, E.; Thuong, P.T.; Won, K. Xanthones from Polygala karensium inhibit neuraminidases from influenza a viruses. Bioorg. Med. Chem. Lett. 2012, 22, 3688–3692. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Zhang, W.; Tu, P.F.; Xu, X.J. Xanthone glycosides from Polygala tenuifolia and their conformational analyses. J. Nat. Prod. 2005, 68, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.H.; Guo, Q.; Jiang, Y.; Tu, P.F. Chemical constituents from the roots of Polygala wattersii Hance. J. Chin. Pharm. Sci. 2014, 23, 723–730. [Google Scholar] [CrossRef]
- Yang, X.D.; An, N.; Xu, L.Z.; Yang, S.L. New xanthone glycosides from Securidaca inappendiculata. J. Asian Nat. Prod. Res. 2002, 4, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Dibwe, D.F.; Awale, S.; Kadota, S.; Tezuka, Y. Muchimangins A-D: Novel diphenylmethyl-substituted xanthones from Securidaca longepedunculata. Tetrahedron Lett. 2012, 53, 6186–6190. [Google Scholar] [CrossRef]
- Pascale, T.; Mondolot, L.; Gargadennec, A.; Kochko, A.; Hamon, S.; Fruchier, A.; Campa, C. First report on mangiferin (C-glucosyl-xanthone) isolated from leaves of a wild coffee plant, Coffea pseudozanguebariae (Rubiaceae). Acta Bot. Gallica 2008, 155, 513–519. [Google Scholar]
- Siddiqui, B.S.; Sattar, F.A.; Begum, S.; Gulzar, T.; Ahmad, F. Chemical constituents from the stems of Morinda citrifolia linn. Arch. Pharm. Res. 2007, 30, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Tala, M.F.; Wabo, H.K.; Zeng, G.Z.; Ji, C.J.; Tane, P.; Tan, N.H. A prenylated xanthone and antiproliferative compounds from leaves of Pentadesma butyracea. Phytochem. Lett. 2013, 6, 326–330. [Google Scholar] [CrossRef]
- Abdissa, N.; Heydenreich, M.; Midiwo, J.O.; Ndakala, A.; Majer, Z.; Neumann, B.; Stammler, H.G.; Sewald, N.; Yenesew, A. A xanthone and a phenylanthraquinone from the roots of Bulbine frutescens, and the revision of six seco-anthraquinones into xanthones. Phytochem. Lett. 2014, 9, 67–73. [Google Scholar] [CrossRef]
- Carvalho, M.J.; Carvalho, L.M.; Ferreira, A.M.; Silva, A.M.S. A new xanthone from Hedychium gardnerianum. Nat. Prod. Res. 2003, 17, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Li, D.H.; Li, C.X.; Jia, C.C.; Sun, Y.T.; Xue, C.M.; Bai, J.; Hua, H.M.; Liu, X.Q.; Li, Z.L. Xanthones from Garcinia paucinervis with in vitro anti-proliferative activity against HL-60 cells. Arch. Pharm. Res. 2016, 39, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Kaennakam, S.; Siripong, P.; Tip-pyang, S. Kaennacowanols A–C, three new xanthones and their cytotoxicity from the roots of Garcinia cowa. Fitoterapia 2015, 102, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Sukandar, E.R.; Ersam, T.; Fatmawati, S.; Siripong, P.; Aree, T.; Tip-pyang, S. Cylindroxanthones A–C, three new xanthones and their cytotoxicity from the stem bark of Garcinia cylindrocarpa. Fitoterapia 2016, 108, 62–65. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.; Hiep, N.T.; Kim, D.W.; Hwang, B.Y.; Lee, H.J.; Mar, W.; Lee, D. Neuroprotective xanthones from the root bark of Cudrania tricuspidata. J. Nat. Prod. 2014, 77, 1893–1901. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Fan, H.; Yang, G.Z.; Jiang, Y.; Zhong, F.F.; He, H.W. Prenylated xanthones from the bark of Garcinia xanthochymus and their 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging activities. Molecules 2010, 15, 7438–7449. [Google Scholar] [CrossRef] [PubMed]
- Trisuwan, K.; Ritthiwigrom, T. Benzophenone and xanthone derivatives from the inflorescences of Garcinia cowa. Arch. Pharm. Res. 2012, 35, 1733–1738. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; He, S.W.; Tang, C.; Li, J.; Yang, G.Z. Caged polyprenylated xanthones from the resin of Garcinia hanburyi. Fitoterapia 2016, 109, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Ee, G.C.; Mah, S.H.; Rahmani, M.; Taufiq-Yap, Y.H.; The, S.S.; Yang, M. A new furanoxanthone from the stem bark of Calophyllum inophyllum. J. Asian Nat. Prod. Res. 2011, 13, 956–960. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.S.; Brandão, P.; Fernandes, C.S.G.; da Silva, M.R.P.C.; de Sousa, M.E.D.S.P.; Pinto, M.M.M. Drug-like properties and ADME of xanthone derivatives: The antechamber of clinical trials. Curr. Med. Chem. 2016, 23, 3654–3686. [Google Scholar] [CrossRef] [PubMed]
- Han, S.Y.; You, B.H.; Kim, Y.C.; Chin, Y.W.; Choi, Y.H. Dose-independent ADME properties and tentative identification of metabolites of α-mangostin from Garcinia mangostana in mice by automated microsampling and UPLC-MS/MS methods. PLoS ONE 2015, 10, e0131587. [Google Scholar] [CrossRef] [PubMed]

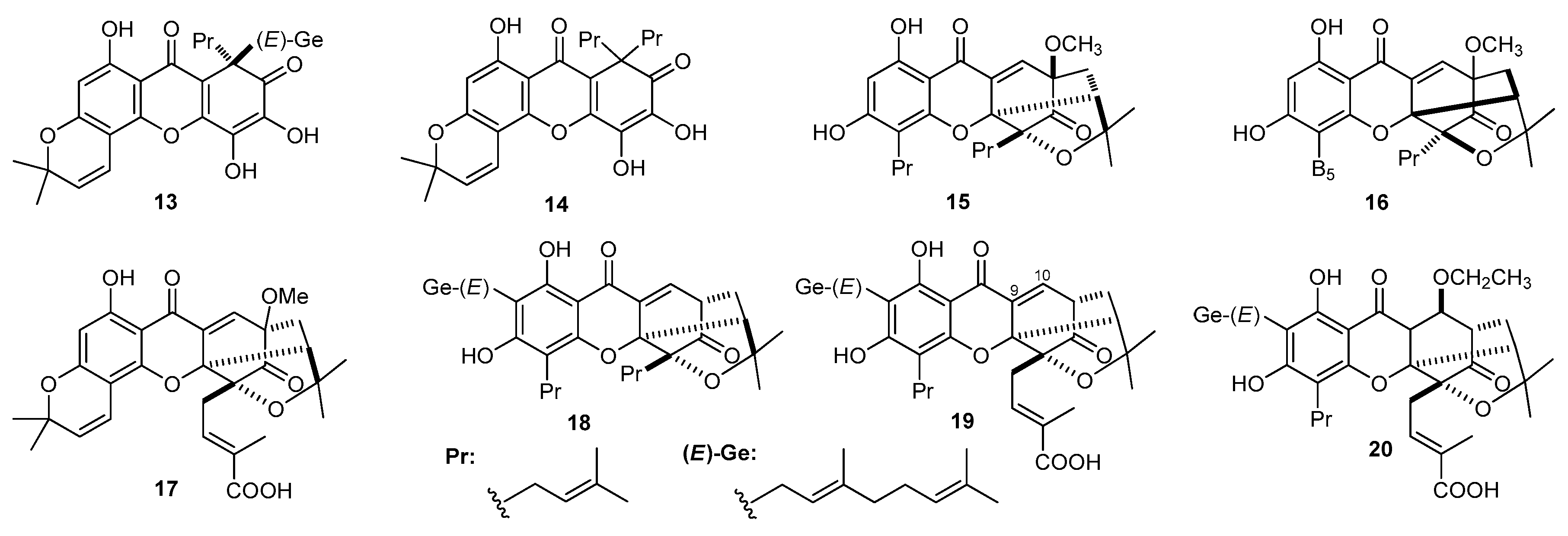
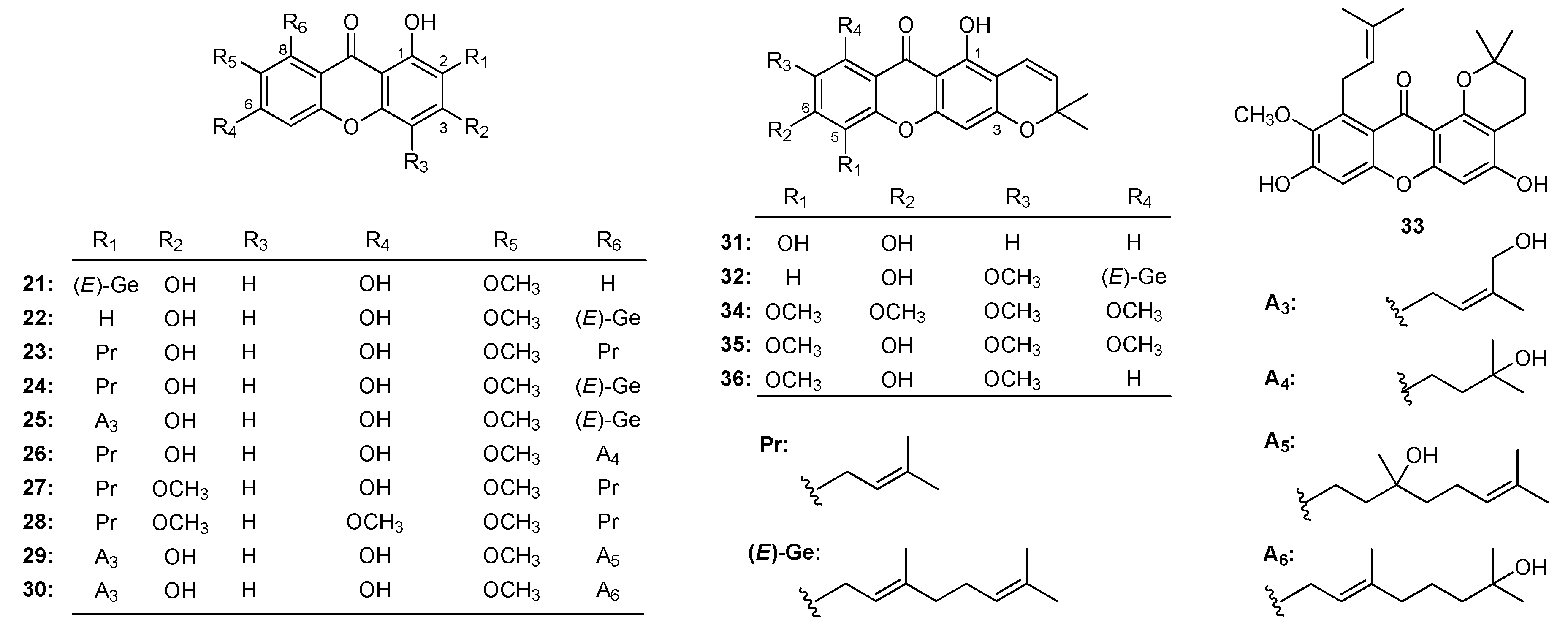
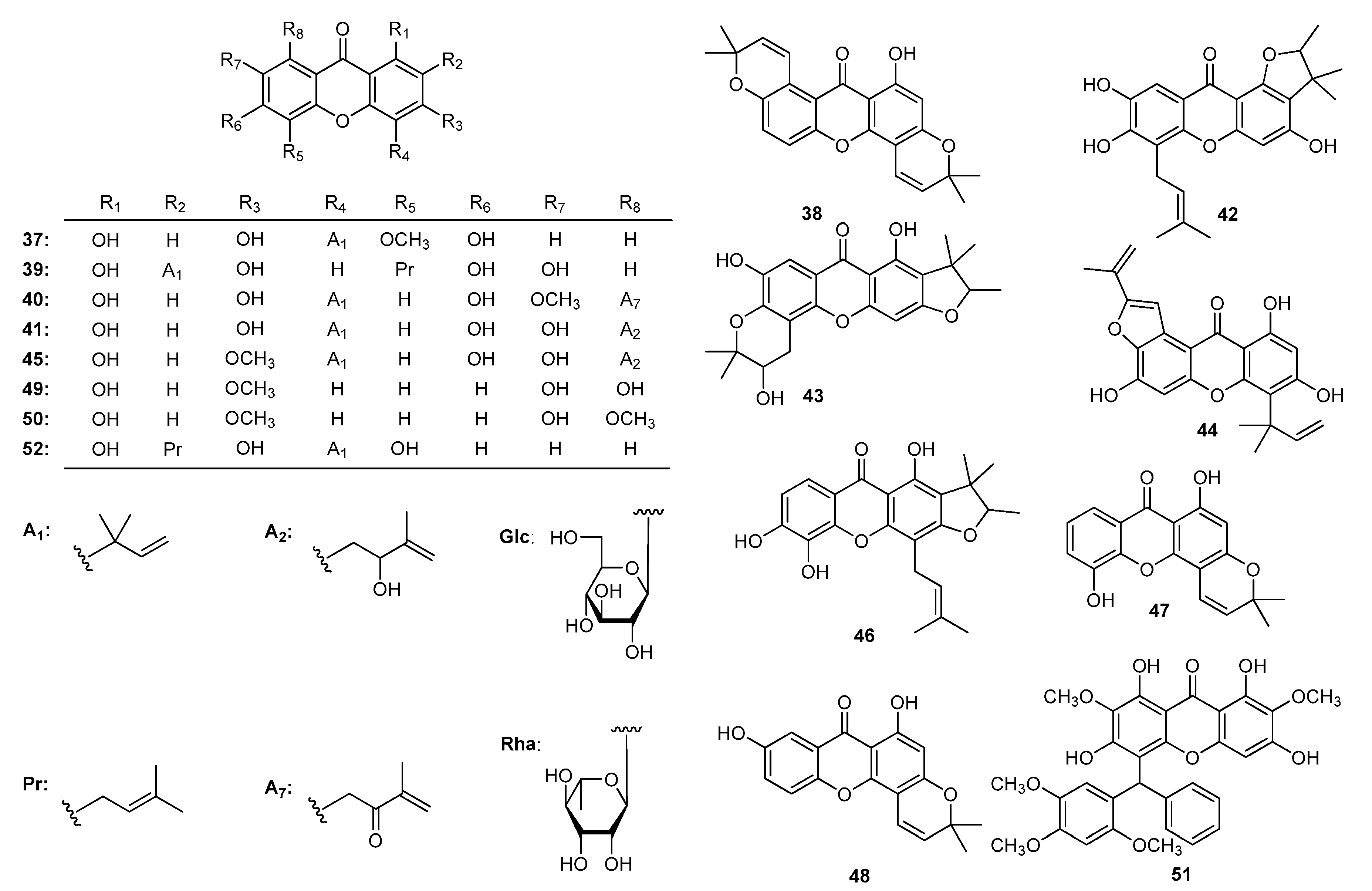
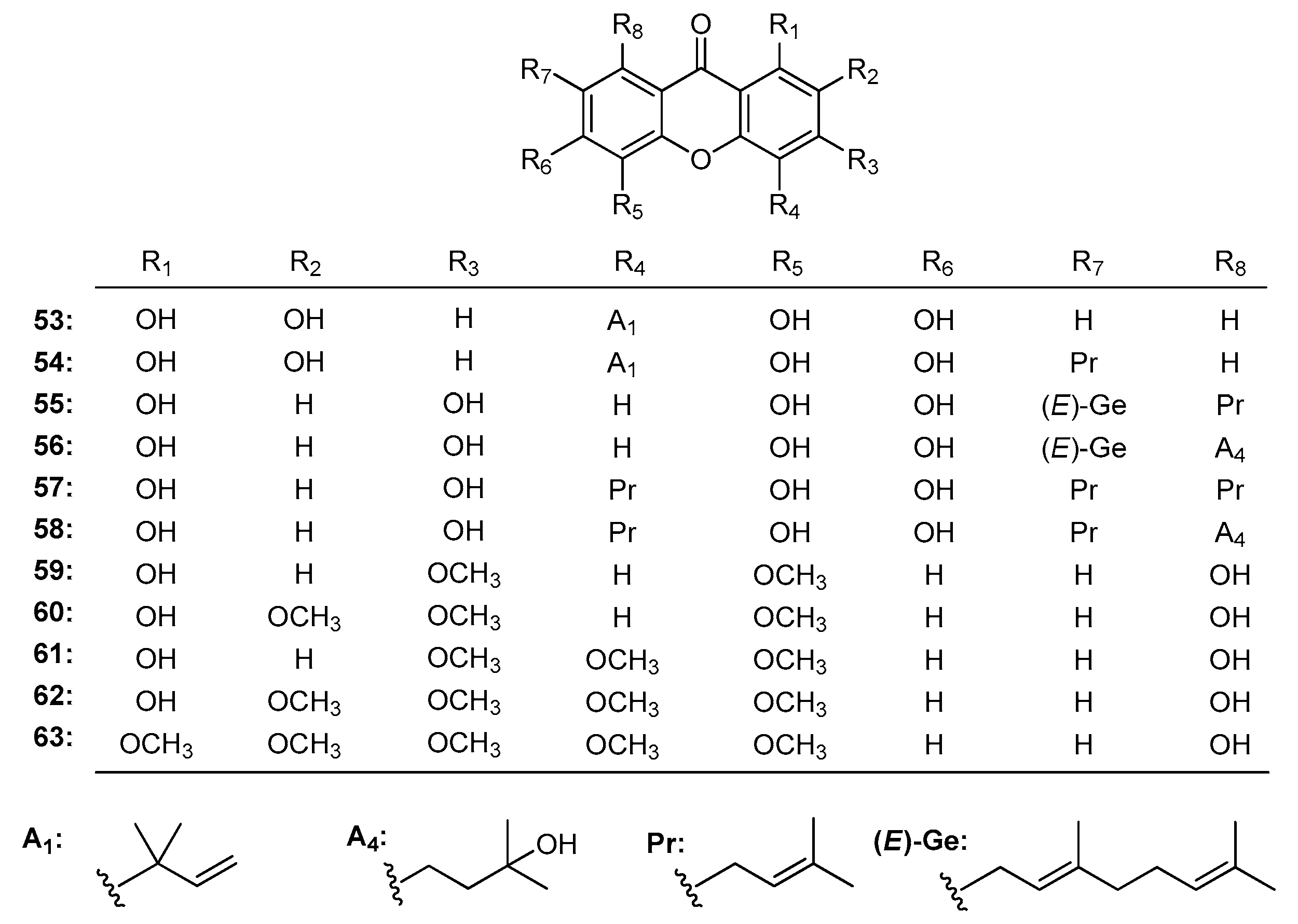
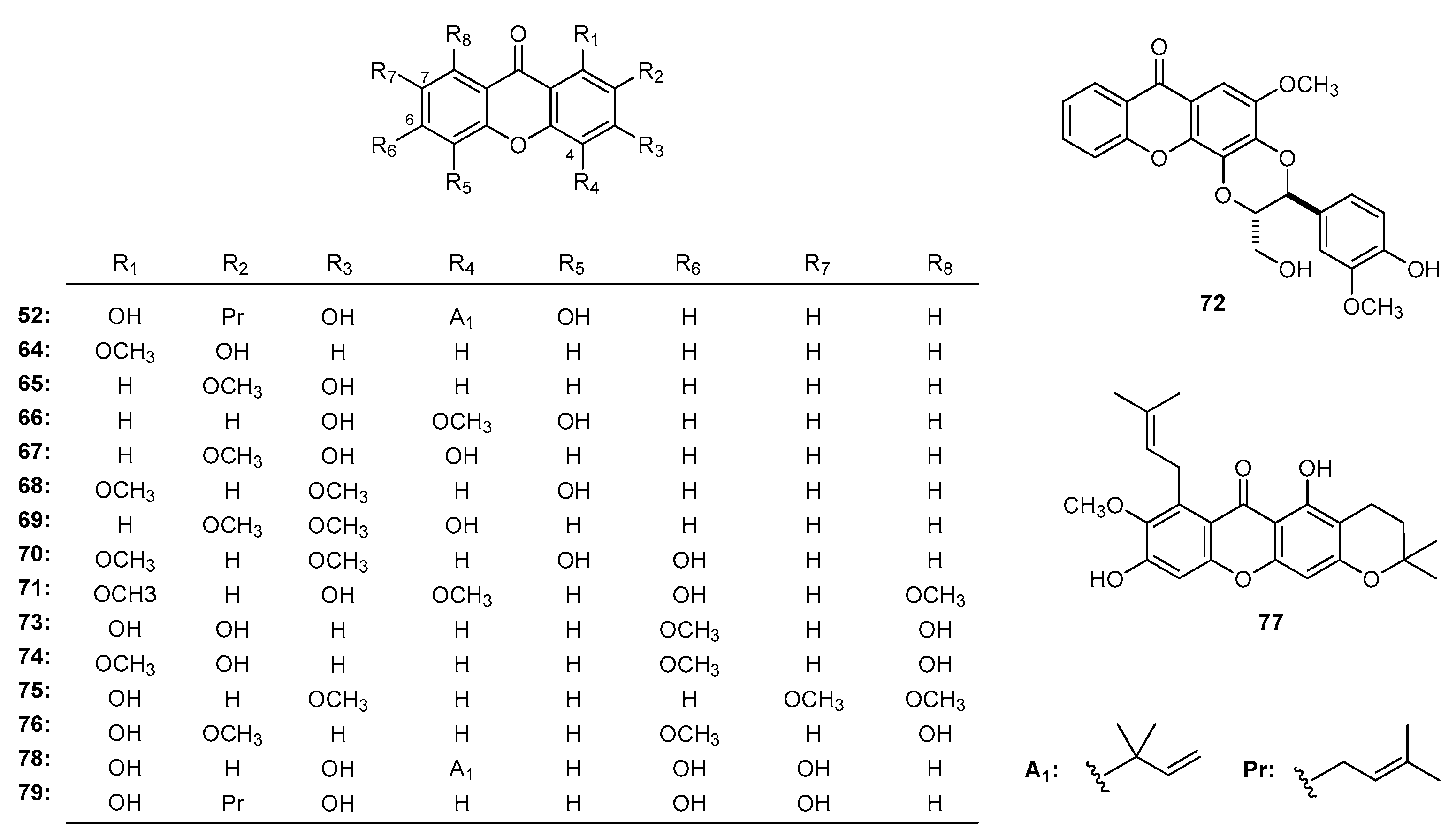
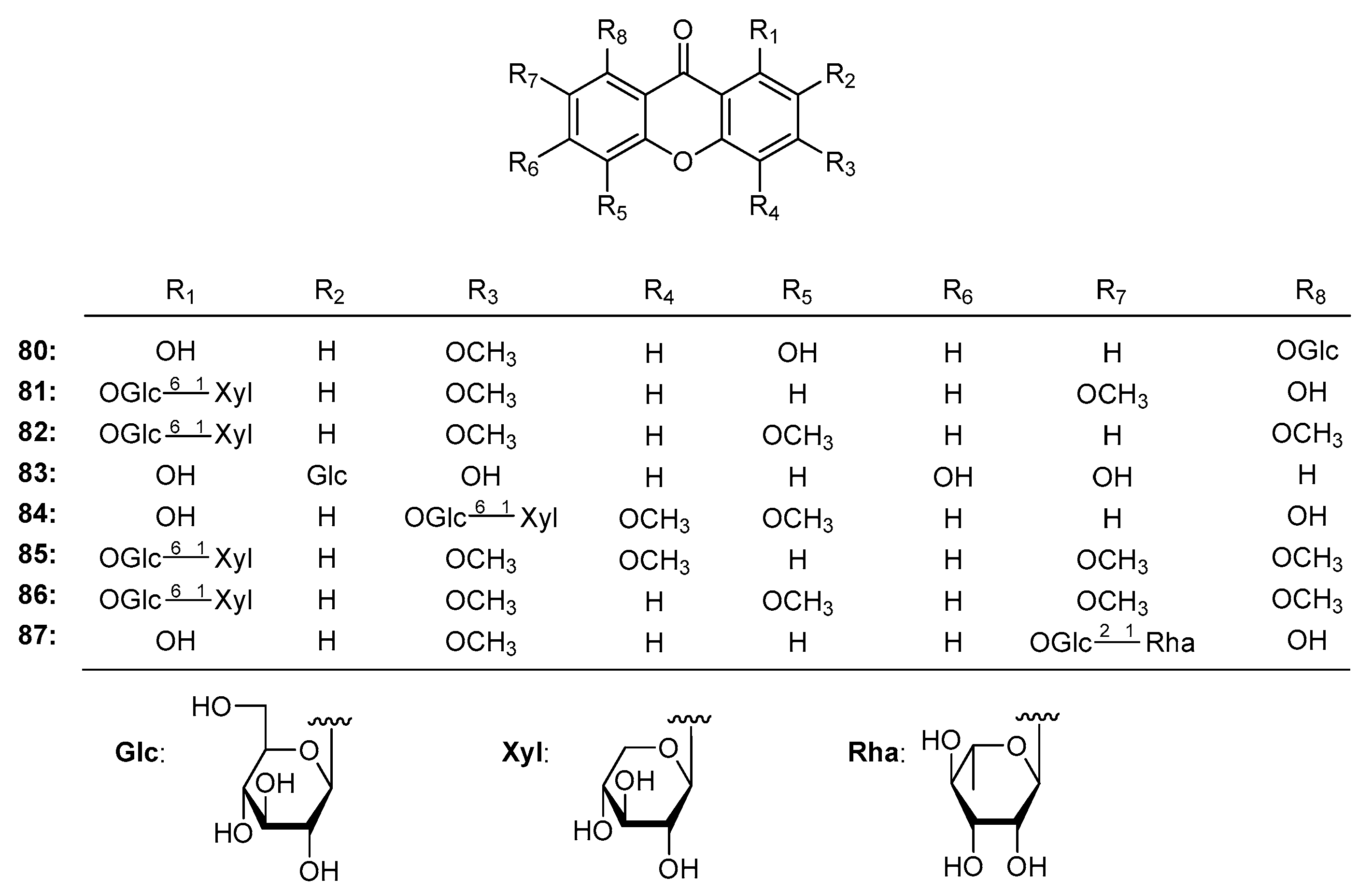
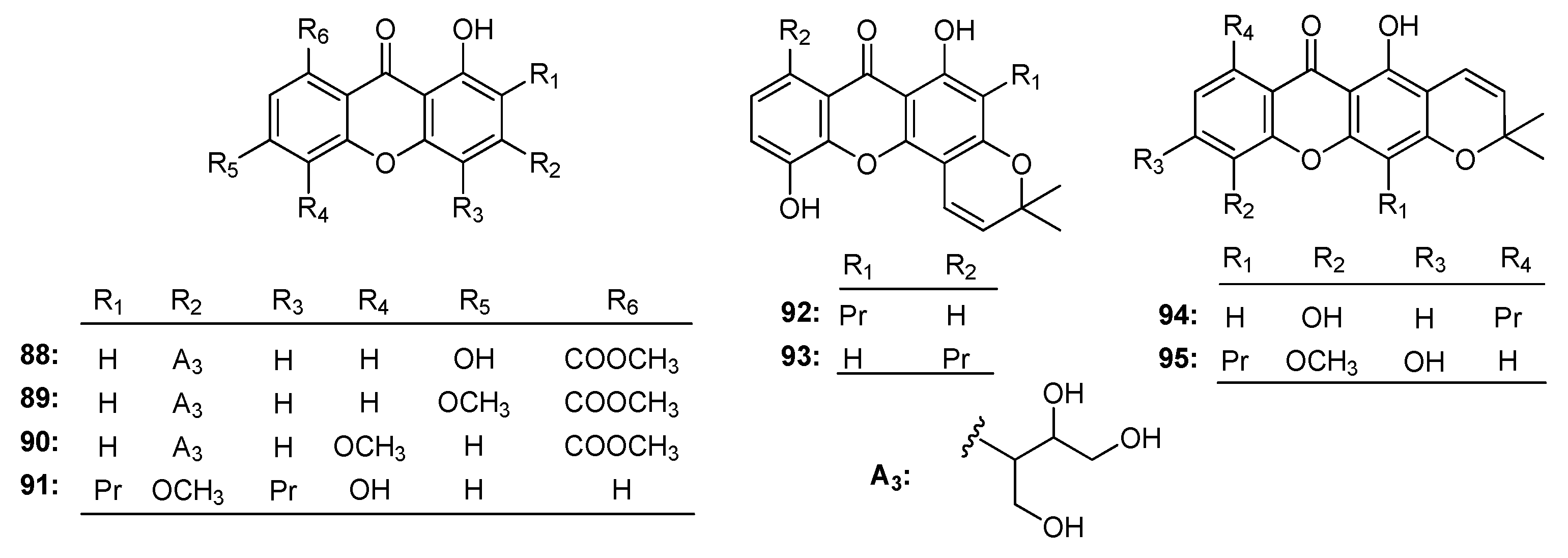
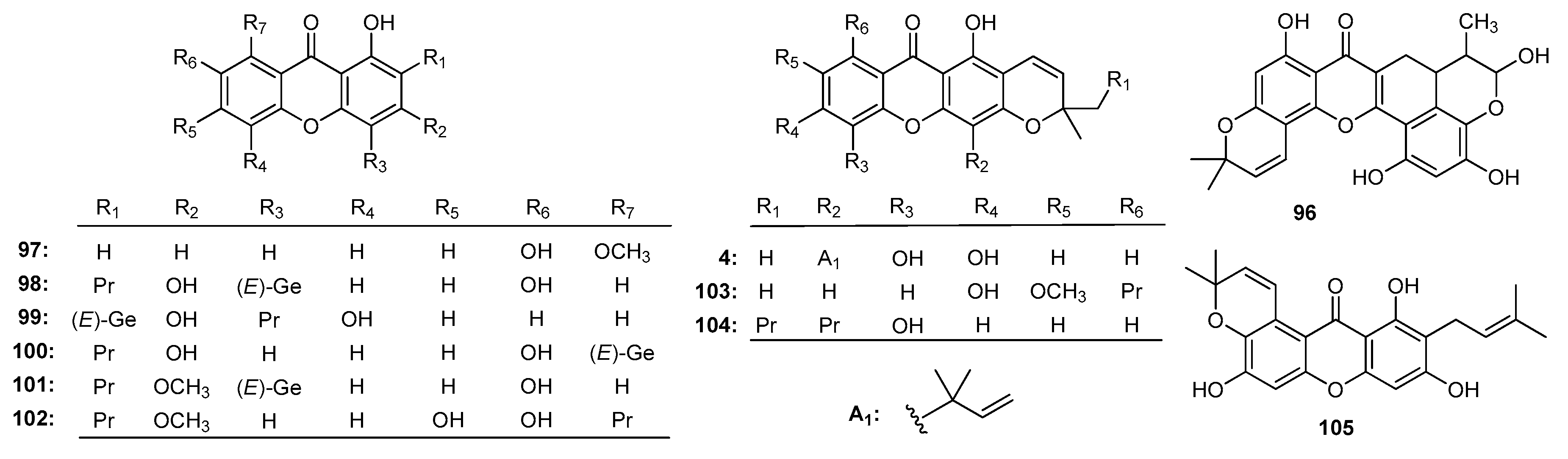
| Family | Genus | Species | Reference |
|---|---|---|---|
| Acanthaceae | Andrographis | A. paniculata (Burm. f.) Nees | [11] |
| Anacardiaceae | Mangifera | M. indica L. | [12] |
| Rhus | R. coriaria L. | [13] | |
| Annonaceae | Anaxagorea | A. luzonensis A. Gray | [14] |
| Guatteria | G. blepharophylla Mart. | [15] | |
| Asparagus | Ledebouria | L. ovatifolia (Schrad.) Jessop | [16] |
| Asparagaceae | Anemarrhena | A. asphodeloides Bunge | [17] |
| Drimiopsis | D. maculate Lindl. & Paxton | [18] | |
| Asteraceae | Santolina | S. insularis (Gennari ex Fiori) Arrigoni | [19] |
| Bignoniaceae | Arrabidaea | A. samydoides (Cham.) Sandwith | [20] |
| Bombacaceae | Bombax | B. ceiba L. | [21] |
| Clusiaceae (or Guttiferae) | Allanblackia | A. floribunda Oliv. | [22] |
| A. gabonensis (Pellegr.) Bamps | [23] | ||
| A. monticola Staner L. C. | [24] | ||
| Bonnetia | B. stricta Mart. | [25] | |
| Calophyllum | C. brasiliense Cambess. | [26] | |
| C. caledonicum Vieill. ex Planch. & Triana | [27] | ||
| C. decipiens Wight | [28] | ||
| C. gracilipes Merr. | [29] | ||
| C. inophyllum L. | [30] | ||
| C. membranaceum Gardner & Champ. | [31] | ||
| C. panciflorum A. C. Smith | [32] | ||
| C. pinetorum Bisse | [33] | ||
| C. soulattri Burm. f. | [34] | ||
| C. symingtonianum M.R. Hend. & Wyatt-Sm. | [35] | ||
| C. thorelii Pierre | [36] | ||
| C. thwaitesii Planch. & Triana | [37] | ||
| Chrysochlamys | C. tenuis Hammel | [38] | |
| Clusia | C. pernambucensis G. Mariz | [39] | |
| Cratoxylum | C. cochinchinensis (Lour.) Blume | [40] | |
| C. formosum sp. Pruniflorum (Kurz) Gogelein | [41] | ||
| Garcinia | G. afzelii Engl. | [42] | |
| G. bracteata C.Y. Wu ex Y.H. Li | [43] | ||
| G. cambogia (Gaertn.) Desr. | [44] | ||
| G. cantleyana Whitmore | [45] | ||
| G. cowa Roxb. ex Choisy | [46] | ||
| G. dioica Blume | [47] | ||
| G. dulcis (Roxb.) Kurz | [48] | ||
| G. eugenifolia Wall. ex T. Anderson | [49] | ||
| G. fusca Pierre | [50] | ||
| G. goudotiana (Planch. & Triana) P. Sweeney & Z.S. Rogers | [51] | ||
| G. griffthii T. Anderson | [52] | ||
| G. hanburyi Hook. f. | [53] | ||
| G. hombroniana Pierre | [54] | ||
| G. lancilimba C.Y. Wu ex Y.H. Li | [55] | ||
| G. lateriflora Blume | [56] | ||
| G. linii C.E. Chang | [57] | ||
| G. mangostana L. | [58] | ||
| G. merguensis Wight | [59] | ||
| G. multiflora Champ. ex Benth. | [60] | ||
| G. nigrolineata Planch. ex T. Anderson | [61] | ||
| G. nitida Pierre | [62] | ||
| G. nobilis Engl. | [63] | ||
| G. nujiangensis C.Y. Wu & Y.H. Li | [64] | ||
| G. oblongifolia Champ. ex Benth. | [65] | ||
| G. oligantha Merr. | [66] | ||
| G. oliveri Pierre | [67] | ||
| G. parvifolia (Miq.) Miq. | [68] | ||
| G. paucinervis Chun & F.C. How | [69] | ||
| G. pedunculata Roxb. ex Buch.-Ham. | [70] | ||
| G. penangiana Pierre | [71] | ||
| G. polyantha Oliv. | [72] | ||
| G. porrecta Laness. | [68] | ||
| G. propinqua Craib | [73] | ||
| G. rigida Miq. | [74] | ||
| G. schomburgkiana Pierre | [75] | ||
| G. scortechinii King | [76] | ||
| G. smeathmannii (Planch. & Triana) Oliv. | [77] | ||
| G. staudtii Engl. | [78] | ||
| G. subelliptica Merr. | [79] | ||
| G. succifolia Kurz | [80] | ||
| G. tetralata C.Y. Wu ex Y.H. Li | [81] | ||
| G. vieillardii Pierre | [82] | ||
| G. virgate Vieill. | [83] | ||
| G. xanthochymus Hook. f. ex T. Anderson | [84] | ||
| G. xipshuanbannaensis Y.H. Li | [85] | ||
| Kielmeyera | K. coriacea Mart. | [86] | |
| K. variabilis Mart. & Zucc. | [87] | ||
| Mammea | M. siamensis T. Anderson | [88] | |
| Mesua | M. ferrea L. | [89] | |
| M. hexapetala (Hook. f.) P.S. Ashton | [90] | ||
| Psorospermum | P. adamauense Engl. | [91] | |
| P. febrifugum Spach | [92] | ||
| P. molluscum (Pers.) Hochr. | [93] | ||
| Rheedia | R. acuminata (Ruiz & Pav.) Planch. & Triana | [94] | |
| Symphonia | S. globulifera L.f. | [95] | |
| Vismia | V. laurentii De Wild. | [96] | |
| V. rubescens Oliv. | [96] | ||
| Fabaceae | Caesalpinia | C. sappan L. | [97] |
| Cassia | C. obtusifolia L. | [98] | |
| Cyclopia | C. genistoides (L.) Vent. | [99] | |
| Desmodium | D. caudatum (Thunb.) DC. | [100] | |
| Ganodermataceae | Gyrophora | G. proboscidea (L.) Ach. | [101] |
| Gentianaceae | Centaurium | C. spicatum (L.) Fritsch | [102] |
| Comastoma | C. pedunculatum (Royle ex G. Don) Holub | [103] | |
| C. pulmonarium (Turcz.) Toyok. | [104] | ||
| Gentiana | G. dinarica Beck | [105] | |
| G. kochiana Perr. & Songeon | [106] | ||
| G. lutea L. | [107] | ||
| G. tizuensis Franch. | [108] | ||
| G. utriculosa L. | [109] | ||
| Gentiana dinarica Beck. | [110] | ||
| Gentianella | G. acuta (Michx.) Hiitonen | [111] | |
| G. amarella (L.) Harry Sm. | [112] | ||
| G. turkestanorum (Gand.) Holub | [113] | ||
| Gentianopsis | G. barbata (Froel.) Ma | [114] | |
| G. paludosa (Hook. f.) Ma | [115] | ||
| Halenia | H. corniculata (L.) Cornaz | [116] | |
| H. elliptica D. Don | [117] | ||
| Lomatogonium | L. carinthiacum (Wulfen) A. Braun | [118] | |
| Schultesia | S. lisianthoides (Griseb.) Benth. & Hook. f. ex Hemsl. | [119] | |
| Swertia | S. chirayita (Roxb.) H. Karsten | [120] | |
| S. cordata (Wall. ex G. Don) C.B. Clarke | [121] | ||
| S. corymbosa Wight ex Griseb. | [122] | ||
| S. cuneata Wall. ex D. Don | [123] | ||
| S. elata Harry Sm. | [124] | ||
| S. franchetiana Harry Sm. | [125] | ||
| S. kouitchensis Franch. | [126] | ||
| S. longifolia Boiss. | [127] | ||
| S. minor (Griscb.) Knobl. | [128] | ||
| S. mussotii Franch. | [129] | ||
| S. paniculata | [130] | ||
| S. pseudochinensis H. Hara | [131] | ||
| S. punicea Hemsl. | [132] | ||
| S. speciosa Wall. | [133] | ||
| Tachia | T. grandiflora Maguire & Weaver | [134] | |
| Hippocrateaceae | Salacia | S. chinensis L. | [135] |
| S. elliptica (Mart.) G. Don | [136] | ||
| Hyacinthaceae | Scilla | S. scilloides (Lindl.) Druce | [137] |
| Hypericaceae | Hypericum | H. ascyron L. | [138] |
| H. attenuatum Fisch. ex Choisy | [139] | ||
| H. chinense L. | [140] | ||
| H. erectum Thunb. | [141] | ||
| H. lanceolatum Lam. | [142] | ||
| H. oblongifolium Choisy | [143] | ||
| H. patulum Thunb. | [144] | ||
| H. perforatum L. | [145] | ||
| H. sampsonii Hance | [146] | ||
| H. scabrum L. | [147] | ||
| H. styphelioides A. Rich. | [148] | ||
| Iridaceae | Iris | I. nigricans Dinsm. | [149] |
| Loganiaceae | Anthocleista | A. schweinfurthii Gilg | [150] |
| A. vogelii Planch. | [151] | ||
| Moraceae | Artocarpus | A. kemando Miq. | [152] |
| A. nobilis Thwaites | [153] | ||
| A. obtusus F.M. Jarrett | [154] | ||
| Cudrania | C. cochinchinensis (Lour.) Yakuro Kudo & Masam. | [155] | |
| C. fruticosa (Roxb.) Wight ex Kurz | [156] | ||
| C. tricuspidata (Carrière) Bureau ex Lavallée | [157] | ||
| Maclura | M. cochinchinensis (Lour.) Corner | [158] | |
| Onagraceae | Oenothera | O. biennis L. | [159] |
| Parmeliaceae | Usnea | U. hirta (L.) Weber ex F.H. Wigg | [160] |
| Polygalaceae | Bredemeyera | B. floribunda Willd. | [161] |
| Moutabea | M. guianensis Aubl. | [162] | |
| Polygala | P. caudata Rehder & E.H. Wilson | [163] | |
| P. crotalarioides Buch.-Ham. ex DC. | [164] | ||
| P. cyparissias A. St.-Hil. & Moq. | [165] | ||
| Securidaca | P. hongkongensis Hemsl. | [166] | |
| P. japonica Houtt. | [167] | ||
| P. karensium Kurz | [168] | ||
| P. tenuifolia Willd. | [169] | ||
| P. wattersii Hance | [170] | ||
| S. inappendiculata Hassk. | [171] | ||
| S. longepedunculata Fresen. | [172] | ||
| Rubiaceae | Coffea | C. pseudozanguebariae Bridson | [173] |
| Morinda | M. citrifolia L. | [174] | |
| Theaceae | Pentadesma | P. butyrace Sabine | [175] |
| Xanthorrhoeaceae | Bulbine | B. frutescens (L.) Willd. | [176] |
| Zingiberaceae | Hedychium | H. gardnerianum Sheppard ex Ker Gawl. | [177] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruan, J.; Zheng, C.; Liu, Y.; Qu, L.; Yu, H.; Han, L.; Zhang, Y.; Wang, T. Chemical and Biological Research on Herbal Medicines Rich in Xanthones. Molecules 2017, 22, 1698. https://doi.org/10.3390/molecules22101698
Ruan J, Zheng C, Liu Y, Qu L, Yu H, Han L, Zhang Y, Wang T. Chemical and Biological Research on Herbal Medicines Rich in Xanthones. Molecules. 2017; 22(10):1698. https://doi.org/10.3390/molecules22101698
Chicago/Turabian StyleRuan, Jingya, Chang Zheng, Yanxia Liu, Lu Qu, Haiyang Yu, Lifeng Han, Yi Zhang, and Tao Wang. 2017. "Chemical and Biological Research on Herbal Medicines Rich in Xanthones" Molecules 22, no. 10: 1698. https://doi.org/10.3390/molecules22101698




