Methyl Jasmonate: An Alternative for Improving the Quality and Health Properties of Fresh Fruits
Abstract
:1. Introduction
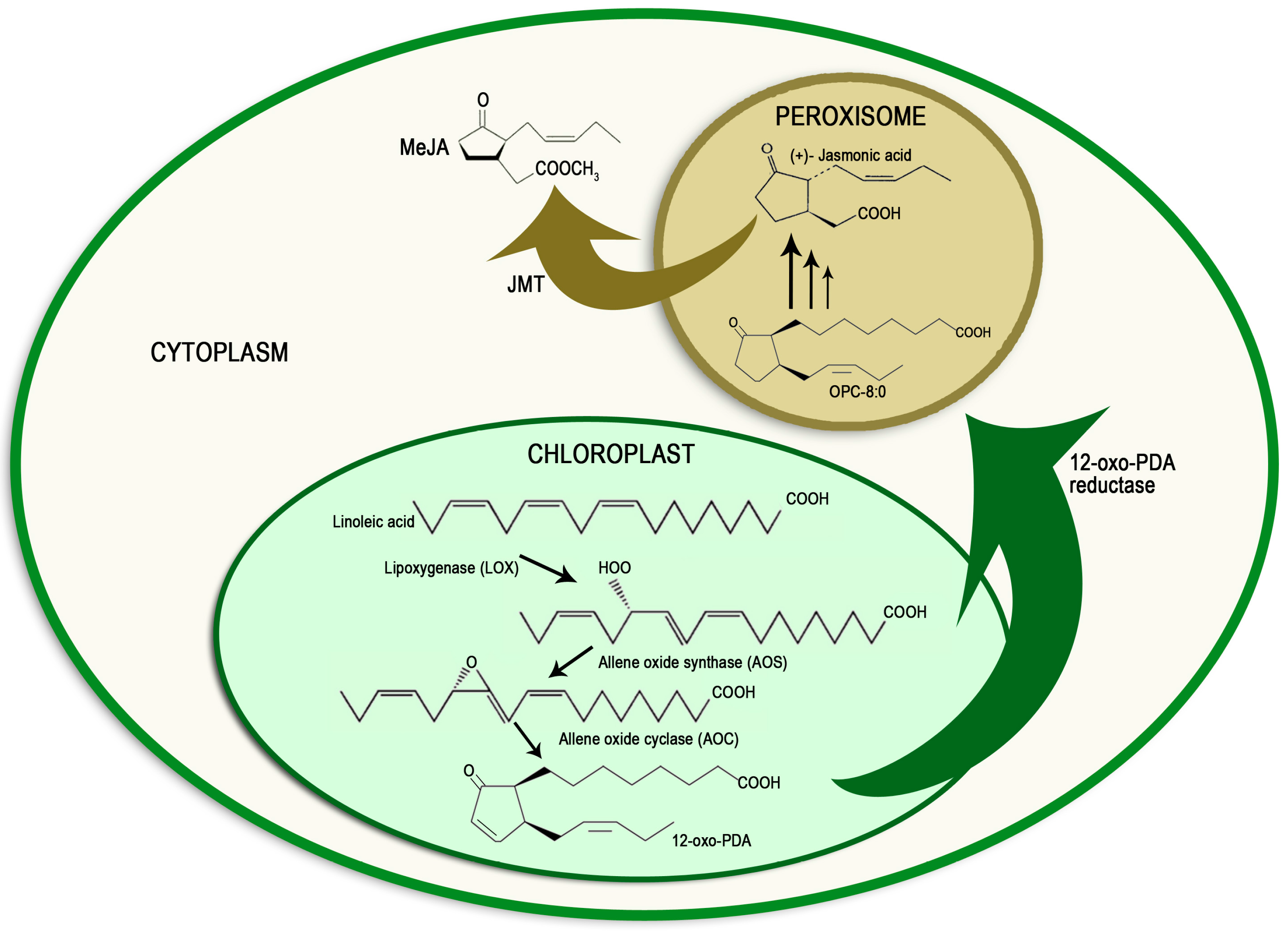
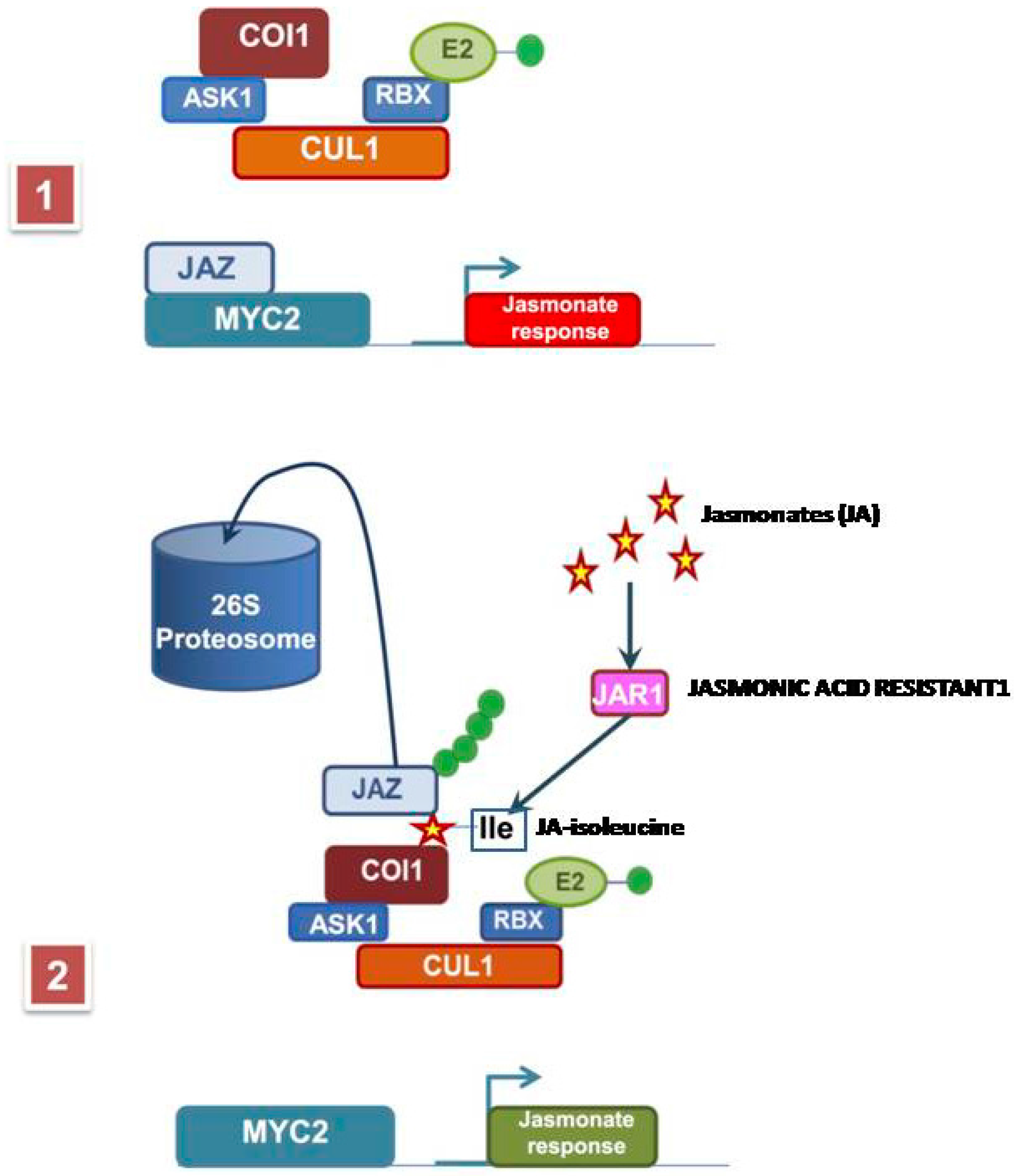
2. Methyl Jasmonate Biosynthesis and Signal Transduction Pathway
3. Pre-Harvest Responses to MeJA Applications
4. Post-Harvest Responses to MeJA Applications
5. MeJA and Its Association with Other Post-Harvest Technologies
6. JAs and MeJA as Health Molecules
7. Concluding Remarks and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chen, H.; Jones, A.; Howe, G. Constitutive activation of the jasmonate signaling pathway enhances the production of secondary metabolites in tomato. FEBS Lett. 2006, 580, 2540–2546. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S. Effects of leaf age on transpiration and energy exchange of Ficus glomerata, a multipurpose tree species of central Himalayas. Physiol. Mol. Biol. Plants 2003, 9, 255–260. [Google Scholar]
- Ueda, J.; Saniewski, J. Methyl jasmonate-induced stimulation of chlorophyll formation in the basal part of tulip bulbs kept under natural light conditions. J. Fruit Ornam. Plant Res. 2006, 14, 199–210. [Google Scholar]
- Wasternack, C. Jasmonates: An update on biosynthesis, signal transduction an action in plant stress response, growth and development. Ann. Bot. 2007, 100, 681–697. [Google Scholar] [CrossRef] [PubMed]
- Wasternack, C.; Hause, B. Jasmonates-Biosynthesis and Role in Stress Responses and Developmental Processes. Ann. Bot. 2013, 111, 1021–1058. [Google Scholar] [CrossRef] [PubMed]
- Creelman, R.A.; Mullet, J.E. Biosynthesis and action of jasmonates in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 355–381. [Google Scholar] [CrossRef] [PubMed]
- Rudell, D.; Mattheis, J.; Fan, X.; Fellman, J. Methyl jasmonate enhances anthocyanin accumulation and modifies production of phenolics and pigments in ‘Fuji’ Apples. J. Am. Soc. Hortic. Sci. 2002, 127, 435–441. [Google Scholar]
- Ghasemnezhad, M.; Javaherdashti, M. Effect of methyl jasmonate treatment on antioxidant capacity, internal quality and postharvest life of raspberry fruit. Casp. J. Environ. Sci. 2008, 6, 73–78. [Google Scholar]
- Blanch, G.; del Castillo, M. Changes in strawberry volatile constituents after pre-harvest treatment with natural hormonal compounds. Flavour Fragr. J. 2012, 27, 180–187. [Google Scholar] [CrossRef]
- Bari, R.; Jones, J. Role of plant hormones in plant defense responses. Plant Mol. Biol. 2008, 69, 473–488. [Google Scholar] [CrossRef] [PubMed]
- Stanley, D. Keeping freshness in fresh-cut produce. Agric. Res. 1998, 46, 12–14. [Google Scholar]
- Cao, S.; Zheng, Y.; Wang, K.; Jin, P.; Rui, H. Methyl jasmonate reduces chilling injury and enhances antioxidant enzyme activity in postharvest loquat fruit. Food Chem. 2009, 115, 1458–1463. [Google Scholar] [CrossRef]
- Sembdner, G.; Parthier, B. The biochemistry and the physiological and molecular actions of jasmonates. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1993, 44, 569–589. [Google Scholar] [CrossRef]
- Hamberg, M.; Gardner, H.W. Oxylipin pathway to jasmonates: Biochemistry and biological significance. Biochim. Biophys. Acta 1992, 1165, 1–18. [Google Scholar] [CrossRef]
- Avanci, N.; Luche, D.; Goldman, G.; Goldman, M. Jasmonates are phytohormones with multiple functions, including plant defense and reproduction. Genet. Mol. Res. 2010, 9, 484–505. [Google Scholar] [CrossRef] [PubMed]
- Cheong, J.J.; Choi, Y.D. Methyl jasmonate as a vital substance in plants. Trends Genet. 2003, 19, 409–413. [Google Scholar] [CrossRef]
- Dar, T.A.; Uddin, M.; Khan, M.M.A.; Hakeem, K.R.; Jaleel, H. Jasmonates counter plant stress: A review. Environ. Exp. Bot. 2015, 115, 49–57. [Google Scholar] [CrossRef]
- Vick, B.; Zimmerman, D. Biosynthesis of jasmonic acid by several plant species. Plant Physiol. 1984, 75, 458–461. [Google Scholar] [CrossRef] [PubMed]
- Lorbeth, R.; Dammann, C.; Ebneth, M.; Amati, S.; Sanchez-Serrano, J. Promoter elements involved in environmental and developmental control of potato proteinase inhibitor II expression. Plant J. 1992, 2, 477–486. [Google Scholar]
- Furch, A.; Zimmermann, M.; Kogel, K.; Reichelt, M.; Mithöfer, A. Direct and individual analysis of stress-related phytohormone dispersion in the vascular system of Cucurbita maxima after flagellin 22 treatment. New Phytol. 2014, 201, 1176–1182. [Google Scholar] [CrossRef] [PubMed]
- Kazan, K.; Manners, J. Jasmonate signaling: Toward an integrated view. Plant Physiol. 2008, 146, 1459–1468. [Google Scholar] [CrossRef] [PubMed]
- Santner, A.; Estelle, M. Recent advances and emerging trends in plant hormone signaling. Nature 2009, 459, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Staswick, P.E.; Tiryaki, I.; Rowe, M.L. Jasmonate response locus JAR1 and several related Arabidopsis genes encode enzymes of the firefly luciferase superfamily that show activity on jasmonic, salicylic, and indole-3-acetic acids in an assay for adenylation. Plant Cell 2002, 14, 1405–1415. [Google Scholar] [CrossRef] [PubMed]
- Kazan, K.; Manners, J. The interplay between light and jasmonatesignalling during defense and development. J. Exp. Bot. 2011, 62, 4087–4100. [Google Scholar] [CrossRef] [PubMed]
- Robson, F.; Okamoto, H.; Patrick, E.; Harris, S.R.; Wasternack, C.; Brearley, C.; Turner, J.G. Jasmonate and phytochrome a signaling in Arabidopsis wound and shade responses are integrated through JAZ1 stability. Plant Cell 2010, 22, 1143–1160. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, S.; Chico, J.; Solano, R. The jasmonate pathway: The ligand, the receptor and the core-signaling module. Curr. Opin. Plant Biol. 2009, 12, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Maciejewska, B.; Kopcewicz, J. Inhibitory effect of methyl jasmonate on flowering and elongation growth in Pharbitis nil. J. Plant Growth Regul. 2002, 21, 216–223. [Google Scholar] [CrossRef]
- Anjum, S.; Xie, X.; Farooq, M.; Wang, L.; Xue, L.; Shahbaz, M.; Salhab, J. Effect of exogenous methyl jasmonate on growth, gas exchange and chlorophyll contents of soybean subjected to drought. Afr. J. Biotechnol. 2011, 10, 9640–9646. [Google Scholar]
- Jung, S. Effect of chlorophyll reduction in Arabidopsis thaliana by methyl jasmonate or norflurazon on antioxidant systems. Plant Physiol. Biochem. 2004, 42, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J. Effect of methyl jasmonate on phenolic compounds and carotenoids of romaine lettuce (Lactuca sativa L.). J. Agric. Food Chem. 2007, 55, 10366–10372. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Jin, P.; Cao, S.; Shang, H.; Yang, Z.; Zheng, Y. Methyl jasmonate reduces decay and enhances antioxidant capacity in chinese bayberries. J. Agric. Food Chem. 2009, 57, 5809–5815. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Chen, F.; Wang, X.; Rajapakse, N.C. Effect of Methyl Jasmonate on Secondary Metabolites of Sweet Basil (Ocimum basilicum L.). J. Agric. Food Chem. 2006, 54, 2327–2332. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhao, Y.; McCraig, B.; Wingerd, B.; Wang, J.; Whalon, M.; Pichersky, E.; Howe, G. The tomato homolog of CORONATINE-INSENSITIVE1 is required for the maternal control of seed maturation, jasmonate-signaled defense responses, and glandular trichome development. Plant Cell 2004, 16, 126–143. [Google Scholar] [CrossRef] [PubMed]
- Pourcel, L.; Irani, N.; Koo, A.; Bohorquez-Restrepo, A.; Howe, G.; Grotewold, E. A chemical complementation approach reveals genes and interactions of flavonoids with other pathways. Plant J. 2013, 74, 383–397. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Hao, Y.; Yang, Y.; Deng, W. Molecular cloning and expression analysis of a cytochrome P450 gene in tomato. Plant Growth Regul. 2010, 61, 297–304. [Google Scholar] [CrossRef]
- Rudell, D.; Fellman, J.; Mattheis, J. Preharvest application of methyl jasmonate to Fuji apples enhances red coloration and affects fruit size, splitting and bitter pit incidence. HortScience 2005, 40, 1760–1762. [Google Scholar]
- Shafiq, M.; Singh, Z.; Kha, A. Time of methyl jasmonate application influences the development of ‘Cripps Pink’ apple fruit colour. J. Sci. Food Agric. 2013, 93, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Flores, G.; Ruiz del Castillo, M.L. Influence of preharvest and postharvest methyl jasmonatetreatmentson flavonoid content and metabolomic enzymes in red raspberry. Postharvest Biol. Technol. 2014, 97, 77–82. [Google Scholar] [CrossRef]
- Kucuker, E.; Ozturk, B.; Celik, S.M.; Aksitc, H. Pre-Harvest spray application of methyl jasmonate plays an important role in fruit ripening, fruit quality and bioactive compounds of Japanese plums. Sci. Hortic. 2014, 176, 162–169. [Google Scholar] [CrossRef]
- Martínez-Esplá, A.; Zapata, P.J.; Castillo, S.; Guilléna, F.; Martínez-Romero, D.; Valero, D.; Serrano, M. Preharvestapplication of methyljasmonate (MeJA) in twoplumcultivars. 1. Improvement of fruit growth and quality attributes at harvest. Postharvest Biol. Technol. 2014, 98, 98–105. [Google Scholar] [CrossRef]
- Zapata, P.J.; Martínez-Esplá, A.; Guillén, F.; Díaz-Mula, H.M.; Martínez-Romero, D.; Serrano, M.; Valero, D. Preharvest application of methyl jasmonate (MeJA) in two plum cultivars. 2. Improvement of fruit quality and antioxidant systems during postharvest storage. Postharvest Biol. Technol. 2014, 98, 115–122. [Google Scholar] [CrossRef]
- Janisiewicz, W.; Korsten, L. Biological control of postharvest diseases of fruits. Annu. Rev. Phytopathol. 2002, 40, 411–441. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.; Miersch, O.; Buttner, C.; Dathe, W.; Sembdner, G. Occurrence of the plant growth regulator jasmonic acid in plants. J. Plant Growth Regul. 1984, 3, 1–8. [Google Scholar] [CrossRef]
- González-Aguilar, G.; Buta, J.; Wang, C. Methyl jasmonate and modified atmosphere packaging (MAP) reduce decay and maintain postharvest quality of papaya “Sunrise”. Postharvest Biol. Technol. 2003, 28, 361–370. [Google Scholar] [CrossRef]
- Nilprapruck, P.; Pradisthakarn, N.; Authanithee, F.; Keebjan, P. Effect of exogenous methyl jasmonate on chilling injury and quality of pineapple (Ananas comosus L.) cv. Pattavia. Silpakorn Univ. Sci. Technol. J. 2008, 2, 33–42. [Google Scholar]
- Meng, X.; Han, J.; Wang, Q.; Tian, S. Changes in physiology and quality of peach fruits treated by methyl jasmonate under low temperature stress. Food Chem. 2009, 114, 1028–1035. [Google Scholar] [CrossRef]
- Ziosi, V.; Bregoli, A.; Fregola, F.; Costa, G.; Torrigiani, P. Jasmonate-Induced ripening delay is associated with up-regulation of polyamine levels in peach fruit. J. Plant Physiol. 2008, 166, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.H.; Suo, B.; Kan, J.; Wang, H.M.; Wang, Z.J. Changes in cell wall polysaccharide of harvested peach fruit during storage. J. Plant Physiol. Mol. Biol. 2006, 32, 657–664. [Google Scholar]
- Fan, X.; Mattheis, J.; Fellman, J. A role for jasmonates in climacteric fruit ripening. Planta 1998, 204, 444–449. [Google Scholar] [CrossRef]
- González-Aguilar, G.; Wang, C.; Buta, G.; Krizek, D. Use of UV-C irradiation to prevent decay and maintain postharvest quality of ripe ‘Tommy Atkins’ mangoes. Int. J. Food Sci. Technol. 2001, 36, 767–773. [Google Scholar] [CrossRef]
- Haffner, K.; Rosenfeld, H.; Skrede, G.; Wang, L. Quality of red raspberry Rubus idaeus L. cultivars after storage in controlled and normal atmospheres. Postharvest Biol. Technol. 2002, 24, 279–289. [Google Scholar] [CrossRef]
- Osorio, G.T.; Oliveira, B.S.; Di Piero, R.B. Effect of fumigants on blue and gray molds of apple fruit. Trop. Plant Pathol. 2013, 38, 63–67. [Google Scholar]
- Walters, D.; Wash, D.; Newton, A.; Lyon, G. Induced resistance for plant disease control: Maximizing the efficacy of resistance elicitors. Phytopathology 2005, 95, 1368–1373. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Fang, W.; Lu, H.; Zhu, R.; Lu, L.; Zheng, X.; Yu, T. Inhibition of green mold disease in mandarins by preventive applications of methyl jasmonate and antagonistic yeast Cryptococcus laurenti. Postharvest Biol. Technol. 2014, 88, 72–78. [Google Scholar] [CrossRef]
- Ayala-Zavala, J.; Wang, S.; Wang, C.; González-Aguilar, G. Methyl jasmonate in conjunction with ethanol treatment increases antioxidant capacity, volatile compounds and postharvest life of strawberry fruit. Eur. Food Res. Technol. 2005, 221, 731–738. [Google Scholar] [CrossRef]
- Belhadj, A.; Saigne, C.; Telef, N.; Cluzet, S. Methyl jasmonate induces defense responses in grapevine and triggers protection against Erysiphenecator. J. Agric. Food Chem. 2006, 54, 9119–9125. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Liao, Y.; Kan, J.; Han, L.; Zheng, Y. Response of direct or priming defense against Botrytis cinerea to methyl jasmonate treatment at different concentrations in grape berries. Int. J. Food Microbiol. 2015, 194, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Jin, P.; Zheng, Y.; Tang, S.; Rui, H.; Wang, C. Enhancing disease resistance in peach fruit with methyl jasmonate. J. Sci. Food Agric. 2009, 89, 802–808. [Google Scholar] [CrossRef]
- Tsao, R.; Zhou, T. Interaction of monoterpenoids, methyl jasmonate, and Ca2+ in controlling postharvest brown rot of sweet cherry. HortScience 2000, 35, 1304–1307. [Google Scholar]
- Yao, H.; Tiana, Sh. Effects of pre- and post-harvest application of salicylic acid or methyl jasmonate on inducing disease resistance of sweet cherry fruit in storage. Postharvest Biol. Technol. 2005, 35, 253–262. [Google Scholar] [CrossRef]
- Yao, H.J.; Tian, S.P. Effects of a biocontrol agent and methyl jasmonate on postharvest diseases of peach fruit and the possible mechanisms involved. J. Appl. Microbiol. 2005, 98, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Tian, S. Resistant responses of tomato fruit treated with exogenous methyl jasmonate to Botrytis cinerea infection. Sci. Hortic. 2012, 142, 38–43. [Google Scholar] [CrossRef]
- Zhang, H.; Ma, L.; Turner, M.; Dong, Y.; Jiang, S. Methyl jasmonate enhances biocontrol efficacy of Rhodotorulaglutinis to postharvest blue mold decay of pears. Food Chem. 2009, 117, 621–626. [Google Scholar] [CrossRef]
- Wang, K.; Jin, P.; Han, L.; Shang, H.; Tang, S.; Rui, H.; Duan, Y.; Kong, F.; Kai, X.; Zheng, Y. Methyl jasmonate induces resistance against Penicillium citrinumin Chinese bayberry by priming of defense responses. Postharvest Biol. Technol. 2014, 98, 90–97. [Google Scholar] [CrossRef]
- Cao, S.; Zheng, Y.; Yang, Z.; Tang, S.; Jin, P. Control of anthracnose rot and quality deterioration in loquat fruit with methyl jasmonate. J. Sci. Food Agric. 2008, 88, 1598–1602. [Google Scholar] [CrossRef]
- Walter, A.; Mazars, C.; Maitrejean, M.; Hopke, J.; Ranjeva, R.; Boland, W.; Mithöfer, A. Structural requirements of jasmonates and synthetic analogues as inducers of Ca2+ signals in the nucleus and the cytosol of plant cells. Angew. Chem. Int. Ed. 2007, 46, 4783–4785. [Google Scholar] [CrossRef] [PubMed]
- Stückrath, R.; Quevedo, R.; de la Fuente, L.; Hernández, A.; Sepúlveda, V. Effect of foliar application of calcium on the quality of blueberry fruits. J. Plant Nutr. 2008, 31, 1299–1312. [Google Scholar] [CrossRef]
- Hepler, P. Calcium: A central regulator of plant growth and development. Plant Cell 2005, 17, 2142–2155. [Google Scholar] [CrossRef] [PubMed]
- Szczegielniak, J.; Borkiewicz, L.; Szurmak, B.; Lewandowska-Gnatowska, E.; Statkiewicz, M.; Klimecka, M.; Ciesla, J.; Muszynska, G. Maize calcium-dependent protein kinase (ZmCPK11): Local and systemic response to wounding, regulation by touch and components of jasmonate signaling. Physiol. Plant. 2012, 146, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, J.; Saltveit, M. Inhibition or promotion of tomato fruit ripening by acetaldehyde and ethanol is concentration dependent and varies with fruit maturity. J. Am. Soc. Hortic. Sci. 1997, 122, 392–398. [Google Scholar]
- Lichter, A.; Zutkhy, Y.; Sonego, L.; Dvir, O.; Kaplunov, T.; Sarig, P.; Ben-Arie, R. Ethanol controls postharvest decay of table grapes. Postharvest Biol. Technol. 2002, 24, 301–308. [Google Scholar] [CrossRef]
- González-Aguilar, G.; Wang, C.; Buta, G. UV-C irradiation reduces breakdown and chilling injury of peaches during cold storage. J. Sci. Food Agric. 2004, 84, 415–422. [Google Scholar] [CrossRef]
- Fingrut, O.; Flescher, E. Plant stress hormones suppress the proliferation and induce apoptosis in human cancer cells. Leukemia 2002, 16, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Chan, A.T. Fruit, vegetables, and folate: Cultivating the evidence for cancer prevention. Gastroenterology 2011, 141, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.J.; Yen, G.C. Chemopreventive effects of dietary phytochemicals against cancer invasion and metastasis: Phenolic acids, monophenol, polyphenol, and their derivatives. Cancer Treat. Rev. 2012, 38, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.; Namdeo, A.G.; Chaugule, B.B.; Kavale, M.; Nanda, S. Broad-Spectrum sun-protective action of Porphyra-334 derived from Porphyravietnamensis. Pharmacogn. Res. 2010, 2, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, R.P.; Singh, S.P.; Häder, D.-P.; Sinha, R.P. Detection of reactive oxygen species (ROS) by the oxidant-sensing probe 2′,7′-dichlorodihydrofluorescein diacetate in the cyanobacterium Anabaena variabilis PCC 7937. Biochem. Biophys. Res. Commun. 2010, 397, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, M. Treatment of fibrocystic disease of the breast with myrtillus anthocyanins. Minerva Ginecol. 1993, 45, 617–621. [Google Scholar] [PubMed]
- Scharrer, A.; Ober, M. Anthocyanosides in the treatment of retinopathies. Klin Monatsbl Augenheikd 1981, 178, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Cao, G.; Prior, R.L. Oxygen radical absorbing capacity of anthocyanins. J. Agri. Food Chem. 1997, 45, 304–309. [Google Scholar] [CrossRef]
- Morazzoni, P.; Bombardelli, E. Vaccinium myrtillus L. Fitoterapia 1996, 67, 3–29. [Google Scholar]
- Sayyari, M.; Babalar, M.; Kalantari, S.; Martinez-Romero, D.; Guillén, F.; Serrano, M.; Valero, D. Vapour treatments with methyl salicylate or methyl jasmonate alleviated chilling injury and enhanced antioxidant potential during postharvest storage of pomegranates. Food Chem. 2011, 124, 964–970. [Google Scholar] [CrossRef]
- Wang, S.; Bowman, L.; Ding, M. Methyl jasmonate enhances antioxidant activity and flavonoid content in blackberries (Rubus sp.) and promotes antiproliferation of human cancer cells. Food Chem. 2008, 107, 1261–1269. [Google Scholar] [CrossRef]
- Asghari, M.; Hasanlooe, A.R. Methyl jasmonate effectively enhanced some defense enzymes activity and Total Antioxidant content in harvested “Sabrosa” strawberry fruit. Food Sci. Nutr. 2015. [Google Scholar] [CrossRef]
- Khan, A.S.; Singh, Z. Methyl jasmonate promotes fruit ripening and improves fruit quality in Japanese plum. J. Hortic. Sci. Biotechnol. 2007, 82. [Google Scholar] [CrossRef]
- Yu, L.; Liu, H.; Shao, X.; Yu, F.; Wei, Y.; Ni, Z.; Xu, F.; Wang, H. Effects of hot air and methyl jasmonate treatment on the metabolism of soluble sugars in peach fruit during cold storage. Postharvest Biol. Technol. 2016, 113, 8–16. [Google Scholar] [CrossRef]
- Huang, X.; Li, J.; Shang, H.; Meng, X. Effect of methyl jasmonate on the anthocyanin content and antioxidant activity of blueberries during cold storage. J. Sci. Food Agric. 2015, 95, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Flores, G.; Blanch, G.P.; Ruiz del Castillo, M.L. Postharvest treatment with (−) and (+)-methyl jasmonate stimulates anthocyanin accumulation in grapes. LWT-Food Sci. Technol. 2015, 62, 807–812. [Google Scholar] [CrossRef]
- Franceschi, V.R.; Grimes, H.D. Induction of soybean vegetative storage proteins and anthocyanins by low-level atmospheric methyl jasmonate. Proc. Natl. Acad. Sci. USA 1991, 88, 6745–6749. [Google Scholar] [CrossRef] [PubMed]
- Saniewski, M.; Miyamoto, K.; Ueda, J. Methyl jasmonate induces gums and stimulates anthocyanin accumulation in peach shoots. J. Plant Growth Regul. 1998, 17, 121–124. [Google Scholar] [CrossRef]
- Kondo, S.; Tsukada, N.; Niimi, Y.; Seto, H. Interactions between jasmonates and abscisic acid in apple fruit, and stimulative effect of jasmonates on anthocyanin accumulation. J. Soc. Hortic. Sci. 2001, 70, 546–552. [Google Scholar] [CrossRef]
- Pérez, A.G.; Sanz, C.; Olías, R.; Ríos, J.J.; Olías, J.M. Effect of modified atmosphere packaging on strawberry quality during shelf-life. In CA’97, Proceedings of the Fruits Other than Apples and Pears, Davis, CA, USA, 13–18 July 1997; Kader, A.A., Ed.; University of California Davis: Davis, CA, USA, 1997; Volume 3, pp. 153–158. [Google Scholar]
- Larrondo, F.; Gaudillère, J.P.; Krisa, S.; Decendi, A.; Deffieux, G.; Mérillon, J.M. Airborne methyl jasmonate induces stilbene accumulation in leaves and berries of grapevine plants. Am. J. Enol. Vitic. 2003, 54, 63–66. [Google Scholar]
- Wu, Q.J.; Xie, L.; Zheng, W.; Vogtmann, E.; Li, H.L.; Yang, G.; Ji, B.T.; Gao, Y.T.; Shu, X.O.; Xiang, Y.B. Cruciferous vegetables consumption and the risk of female lung cancer: A prospective study and a meta-analysis. Ann. Oncol. 2013, 24, 1918–1924. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.J.; Yang, Y.; Wang, J.; Han, L.H.; Xiang, Y.B. Cruciferous vegetable consumption and gastric cancer risk: A meta-analysis of epidemiological studies. Cancer Sci. 2013, 104, 1067–1073. [Google Scholar] [CrossRef] [PubMed]
- Kassie, F.; Parzefall, W.; Musk, S.; Johnson, I.; Lamprecht, G.; Sontag, G.; Knasmüller, S. Genotoxic effects of crude juices from Brassica vegetables and juices and extracts from phytopharmaceutical preparations and spices of cruciferous plants origin in bacterial and mammalian cells. Chem. Biol. Interact. 1996, 102, 1–16. [Google Scholar] [CrossRef]
- Martínez, A.; Ikken, Y.; Cambero, M.I.; Marin, M.L.; Haza, A.I.; Casas, C.; Morales, P. Mutagenicity and cytotoxicity of fruits and vegetables evaluated by the Ames test and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Food Sci. Technol. Int. 1999, 5, 431–437. [Google Scholar] [CrossRef]
- Baasanjav-Gerber, C.; Hollnagel, H.M.; Brauchmann, J.; Iori, R.; Glatt, H.R. Detection of genotoxicants in Brassicales using endogenous DNA as a surrogate target and adducts determined by 32P-postlabelling as an experimental end point. Mutagenesis 2011, 26, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.J.; Yang, Y.; Vogtmann, E.; Wang, J.; Han, L.H.; Li, H.L.; Xiang, Y.B. Cruciferous vegetables intake and the risk of colorectal cancer: A meta-analysis of observational studies. Ann. Oncol. 2013, 24, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Baasanjav-Gerber, C.; Monien, B.H.; Mewis, I.; Schreiner, M.; Barillari, J.; Iori, R.; Glatt, H.R. Identification of glucosinolate congeners able to form DNA adducts and to induce mutations upon activation by myrosinase. Mol. Nutr. Food Res. 2011, 55, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, C.M.J.; Dicke, M. Plant interactions with microbes and insects: From molecular mechanisms to ecology. Trends Plant Sci. 2007, 12, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Wiesner, M.; Schreiner, M.; Glatt, H. High mutagenic activity of juice from pakchoi (Brassica rapa ssp. chinensis) sprouts due to its content of 1-methoxy-3-indolylmethyl glucosinolate, and its enhancement by elicitation with methyl jasmonate. Food Chem. Toxicol. 2014, 67, 10–16. [Google Scholar] [PubMed]
- Kwon, K.H.; Barve, A.; Yu, S.; Huang, M.T.; Kong, A. Cancer chemoprevention by phytochemicals: Potential molecular targets, biomarkers and animal models. Acta Pharmacol. Sin. 2007, 28, 1409–1421. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.J.; Shin, S.H.; Lee, H.J.; Lee, K.W. Polyphenols as small molecular inhibitors of signaling cascades in carcinogenesis. Pharmacol. Ther. 2011, 130, 310–324. [Google Scholar] [CrossRef] [PubMed]
- Fuhrman, B.; Lavv, A.; Aviram, M. Consumption of red wine with meals reduces the susceptibility of human plasma and low-density lipoprotein to lipid peroxidation. Am. J. Clin. Nutr. 1995, 61, 549–554. [Google Scholar] [PubMed]
- Visioli, F.; Galli, C. Olive oils phenols and their potential effects on human health. J. Agric. Food Chem. 1998, 46, 4292–4296. [Google Scholar] [CrossRef]
- Kniazhanski, T.; Jackman, A.; Heyfets, A.; Gonen, P.; Flescher, E.; Sherman, L. Methyl jasmonate induces cell death with mixed characteristics of apoptosis and necrosis in cervical cancer cells. Cancer Lett. 2008, 271, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Yeruva, L.; Pierre, K.J.; Bathina, M.; Elegbede, A.; Carper, S.W. Delayed cytotoxic effects of methyl jasmonate and cis-jasmone induced apoptosis in prostate cancer cells. Cancer Investig. 2008, 26, 890–899. [Google Scholar] [CrossRef] [PubMed]
- Reischer, D.; Heyfets, A.; Shimony, S.; Nordenberg, J.; Kashman, Y.; Flescher, E. Effects of natural and novel synthetic jasmonates in experimental metastatic melanoma. Br. J. Pharmacol. 2007, 150, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Fingrut, O.; Reischer, D.; Rotem, R.; Goldin, N.; Altboum, I.; Zan-Bar, I.; Flescher, E. Jasmonates induce nonapoptotic death in high-resistance mutant p53-expressing B-lymphoma cells. Br. J. Pharmacol. 2005, 146, 800–808. [Google Scholar] [CrossRef] [PubMed]
- Flescher, E. Jasmonates—A new family of anti-cancer agents. Anti-Cancer Drugs 2005, 16, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Willis, C.; Chen, X. The promise and obstacle of p53 as a cancer therapeutic agent. Curr. Mol. Med. 2002, 2, 329–345. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.J. p53, the cellular gatekeeper for growth and division. Cell 1997, 88, 323–331. [Google Scholar] [CrossRef]
- Seeman, S.; Maurici, D.; Olivier, M.; De Formentel, C.C.; Hainaut, P. The tumor suppressor gene TP53: Implications for cancer management and therapy. Crit. Rev. Clin. Lab. Sci. 2004, 41, 551–583. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Flescher, E. Methyl jasmonate: A plant stress hormone as an anti-cancer drug. Phytochemistry 2009, 70, 1600–1609. [Google Scholar] [CrossRef] [PubMed]
- Umukoro, S.; Akinyinka, A.O.; Aladeokin, A.C. Antidepressant activity of methyl jasmonate, a plant stress hormone in mice. Pharmacol. Biochem. Behav. 2011, 98, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Zomkowski, A.D.; Santos, A.R.; Rodrigues, A.L. Putrescine produces antidepressant-like effects in the forced swimming test and in the tail suspension test in mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2006, 30, 1419–1425. [Google Scholar] [CrossRef] [PubMed]
- Umukoro, S.; Eduviere, A.T.; Aladeokin, A.C. Anti-Aggressive activity of methyl jasmonate and the probable mechanism of its action in mice. Pharmacol. Biochem. Behav. 2012, 101, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Wellman, M.L.; Krakowka, S.; Jabobs, R.M.; Kociba, G.J. A macrophage-monocyte cell line from a dog with malignant histiocytosis. In vitro. In Vitro Cell. Dev. Biol. 1988, 24, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Hernandes, C.; Cardozo, G.P.; França, S.C.; Fachin, A.L.; Marins, M.; Lourenço, M.V. Cytotoxic effect of jasmonate and methyl jasmonate on a canine macrophage tumor cell line. Rev. Bras. Plantas Med. 2012, 14, 122–124. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
| Crop | Doses | Application Method | Fungal Species | Fungal Effect | Reference |
|---|---|---|---|---|---|
| Strawberry | 0.1 mM | Vapor | Epiphyas postvittana | Inhibition | Ayala-Zavala et al. [55] |
| Grapevine | 5 or 15 mM | Spray | Erysiphe necator | Inhibition | Belhadj et al. [56] |
| 0.01 mM | Vapor | Botrytis cinerea | Inhibition | Wang et al. [57] | |
| Loquat | 0.01 mM | Vapor | Colletotrichumacutatum | Inhibition | Cao et al. [12] |
| Papaya | 0.01 mM | Vapor | Colletotrichumgloeosporioides | Inhibition | González-Aguilar et al. [44] |
| Peach | 0.001 mM | Vapor | Botrytis cinerea | Inhibition | Jin et al. [58] |
| Sweet cherry | 10 mM | Vapor | Monilinia fructicola | No effect | Tsao and Zhou [59] |
| Sweet cherry | 0.2 mM | Spray | Monilinia fructicola | Inhibition | Yao and Tian [60] |
| Peach | 0.2 mM | Vapor | Monilinia fructicola and Penicillium expansum | Inhibition | Yao and Tian [61] |
| Dipping | |||||
| Tomato | 0.1 or 10 mM | Dipping | Botrytis cinerea | Inhibition | Zhu and Tian [62] |
| Pear | 0.2 mM | Vapor | Penicillium expansum | No effect | Zhang et al. [63] |
| Chinese bayberry | 0.01 mM | Vapor | Penicillium citrinum | Inhibition | Wang et al. [64] |
| Mandarins | 0.1 mM | Penicillium digitatum | Inhibition | Guo et al. [54] |
| Crop | MeJA Doses | Application Method | Enzymatic and Non-Enzymatic Antioxidants | Reference |
|---|---|---|---|---|
| Strawberry | 0.1 mM | Vapor | Anthocyanins, phenolic acid | Ayala-Zavala et al. [55] |
| Strawberry | 8 and 16 µM | Vapor | CAT, POD and polyphenol oxidase (PPO) | Asghari and Hasanlooe [84] |
| Raspberry | 0.01 and 0.1 mM | Vapor | Flavonoids, PAL, flavanone 3β-hydroxylase (FHT) and flavonol synthase (FLS) | Flores et al. [38] |
| Raspberry | 0.024 mM | Vapor | Anthocyanins | Ghasemnezhad and Javaherdashti [8] |
| Blackberry | 0.1 mM | Spray | Anthocyanins, phenolic acid | Wang et al. [83] |
| Blueberry | 0.01–0.1 mM | Vapor | Anthocyanins | Huang et al. [87] |
| Grapes | 1.78 mM | Vapor | Anthocyanins, total phenols | Flores et al. [88] |
| Loquat | 0.01 mM | Vapor | Superoxide dismutase (SOD), chloramphenicol acetyltransferase, ascorbate peroxidase (APX) | Cao et al. [12] |
| Pomegranates | 0.01–0.1 mM | Vapor | Total phenolic and anthocyanins | Sayyari et al. [82] |
| Apple | 1 mM | Dipping | Anthocyanins | Rudellet al. [36] |
| Plum | 0–1 mM | Vapor | 1-Aminocyclopropane-1-carboxylic acid synthase (ACS) and 1-amino-cyclopropane-1-carboxylic acid oxidase (ACO) | Khana and Singha [85] |
| Peach | 10 µM | Vapor | Sucrose phosphate synthase (SPS) | Yu et al. [86] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes-Díaz, M.; Lobos, T.; Cardemil, L.; Nunes-Nesi, A.; Retamales, J.; Jaakola, L.; Alberdi, M.; Ribera-Fonseca, A. Methyl Jasmonate: An Alternative for Improving the Quality and Health Properties of Fresh Fruits. Molecules 2016, 21, 567. https://doi.org/10.3390/molecules21060567
Reyes-Díaz M, Lobos T, Cardemil L, Nunes-Nesi A, Retamales J, Jaakola L, Alberdi M, Ribera-Fonseca A. Methyl Jasmonate: An Alternative for Improving the Quality and Health Properties of Fresh Fruits. Molecules. 2016; 21(6):567. https://doi.org/10.3390/molecules21060567
Chicago/Turabian StyleReyes-Díaz, Marjorie, Tomas Lobos, Liliana Cardemil, Adriano Nunes-Nesi, Jorge Retamales, Laura Jaakola, Miren Alberdi, and Alejandra Ribera-Fonseca. 2016. "Methyl Jasmonate: An Alternative for Improving the Quality and Health Properties of Fresh Fruits" Molecules 21, no. 6: 567. https://doi.org/10.3390/molecules21060567






