Ginsenoside Rb1 Protects Rat Neural Progenitor Cells against Oxidative Injury
Abstract
:1. Introduction
2. Results and Discussion
2.1. In Vitro Characterization of NPCs

2.2. Rb1 Pretreatment Reduced Oxidative Stress on Culture NPCs
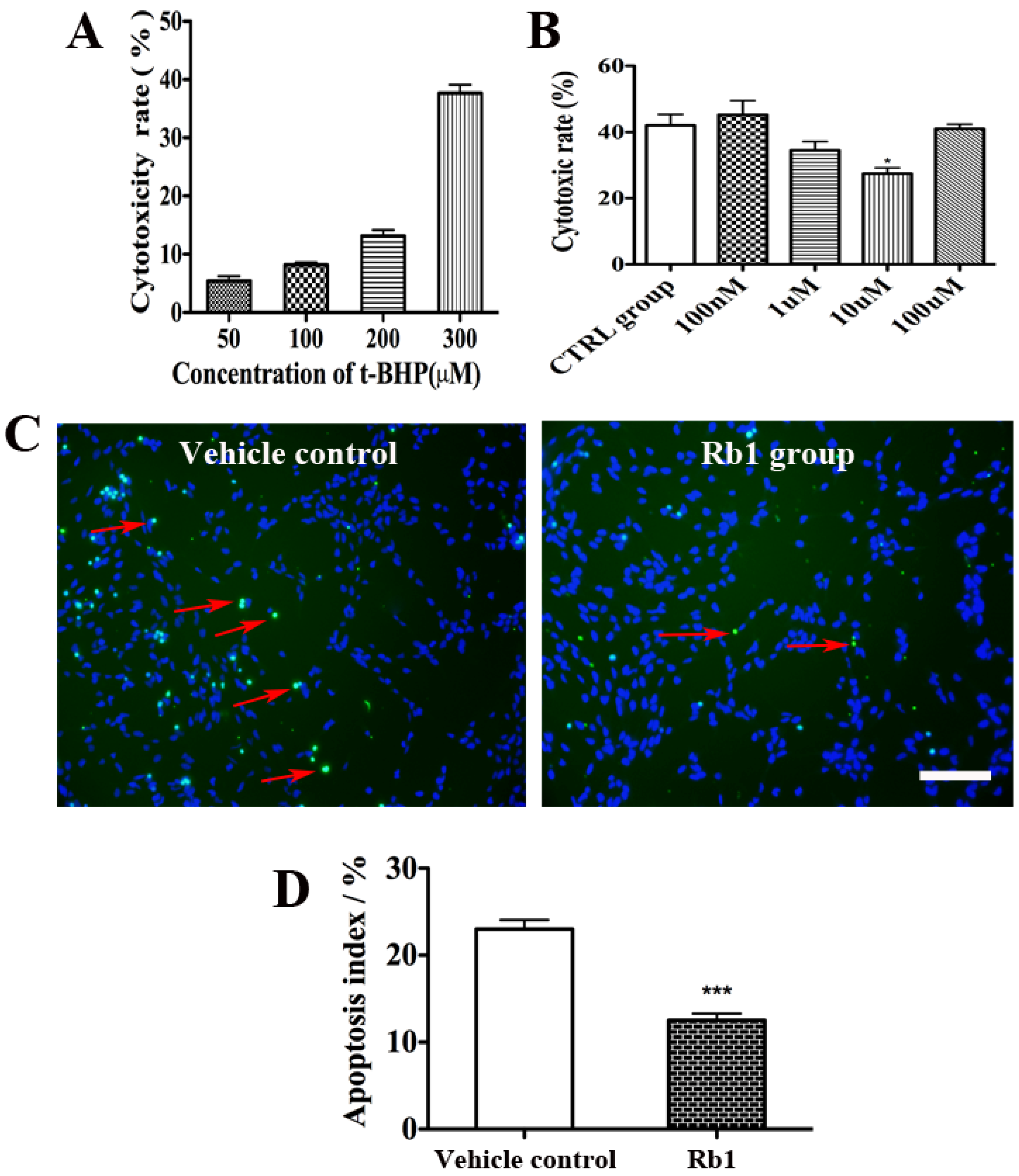
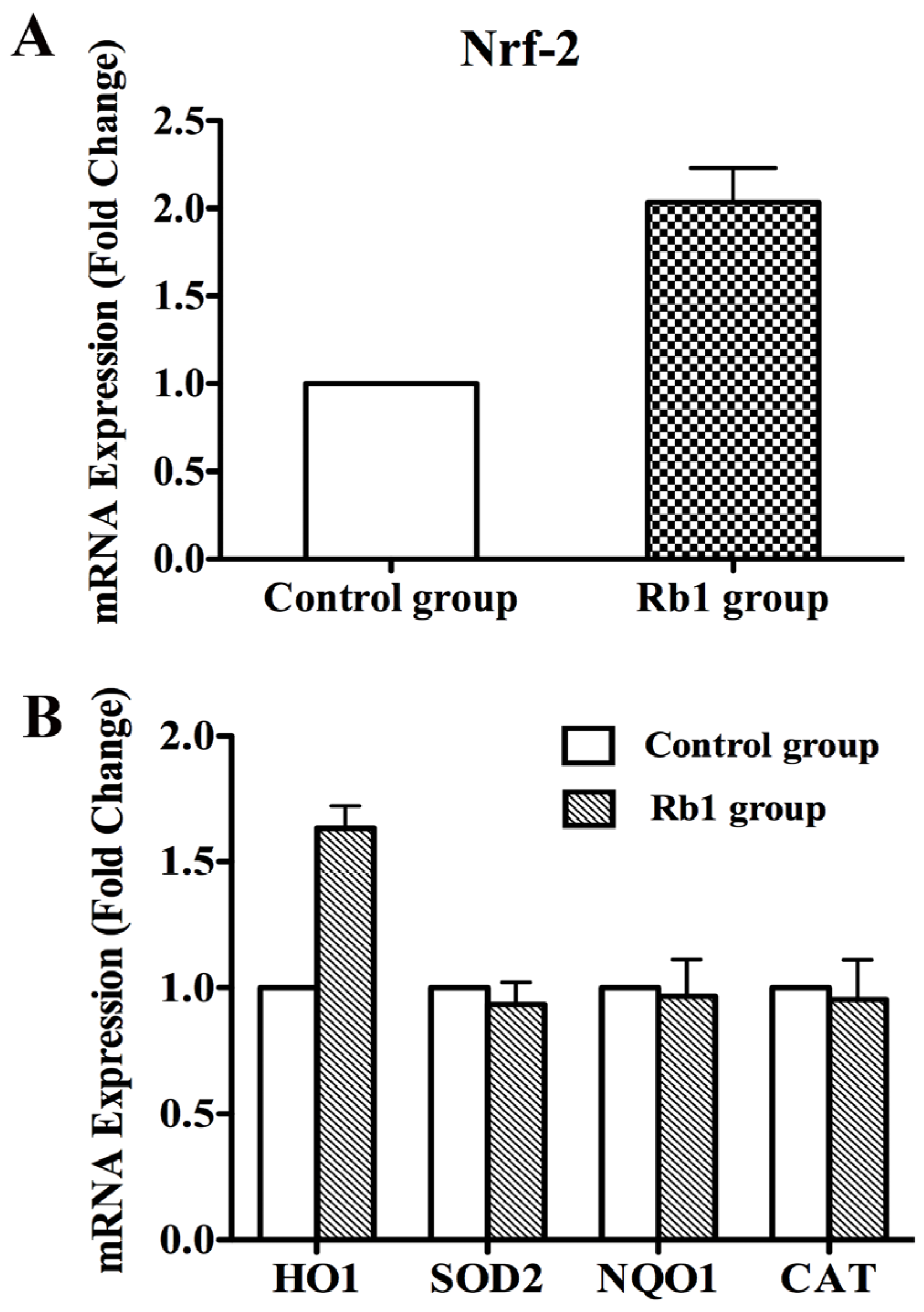
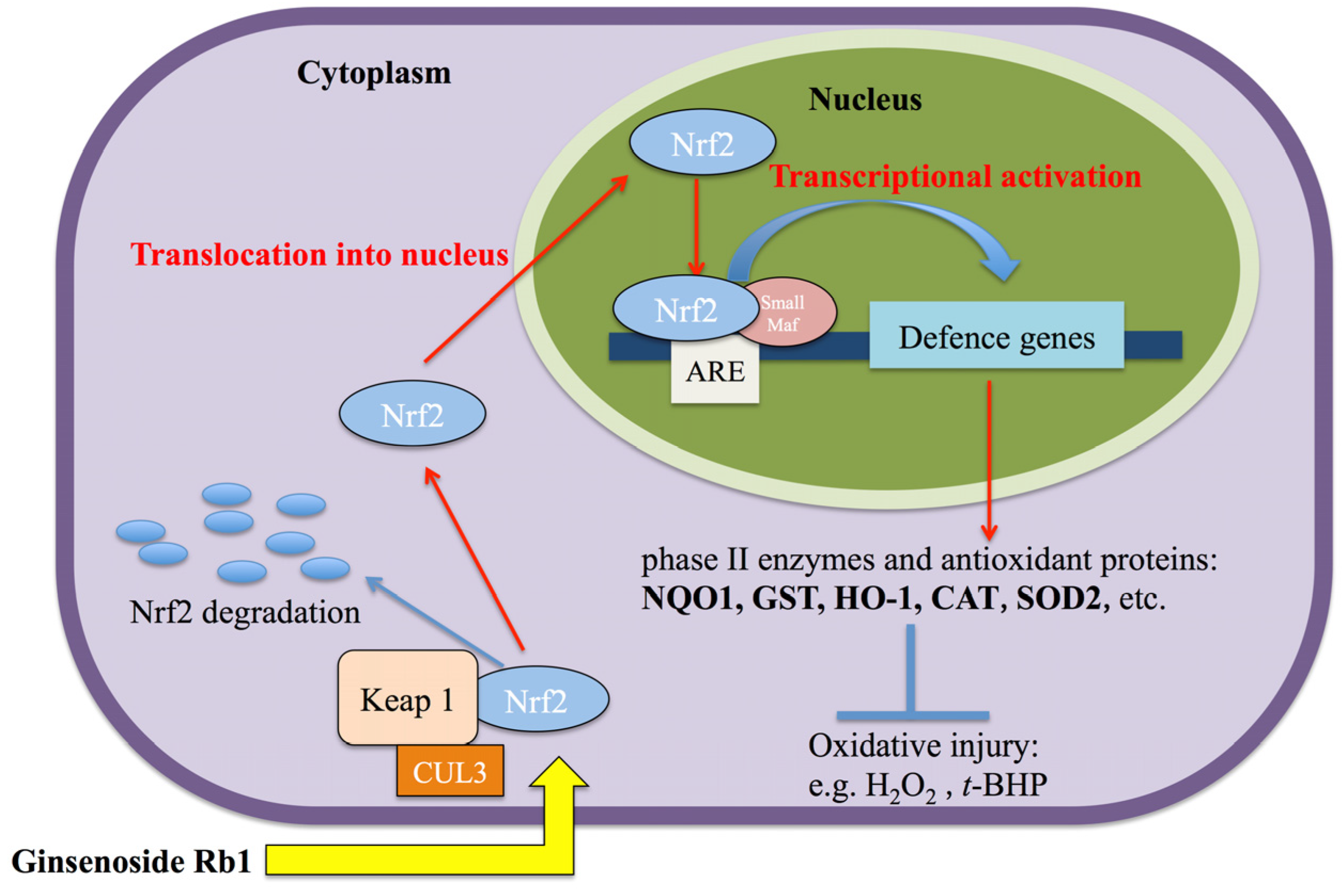
2.3. Rb1 Pretreatment Upregulated Antioxidant Genes in Cultured NPCs
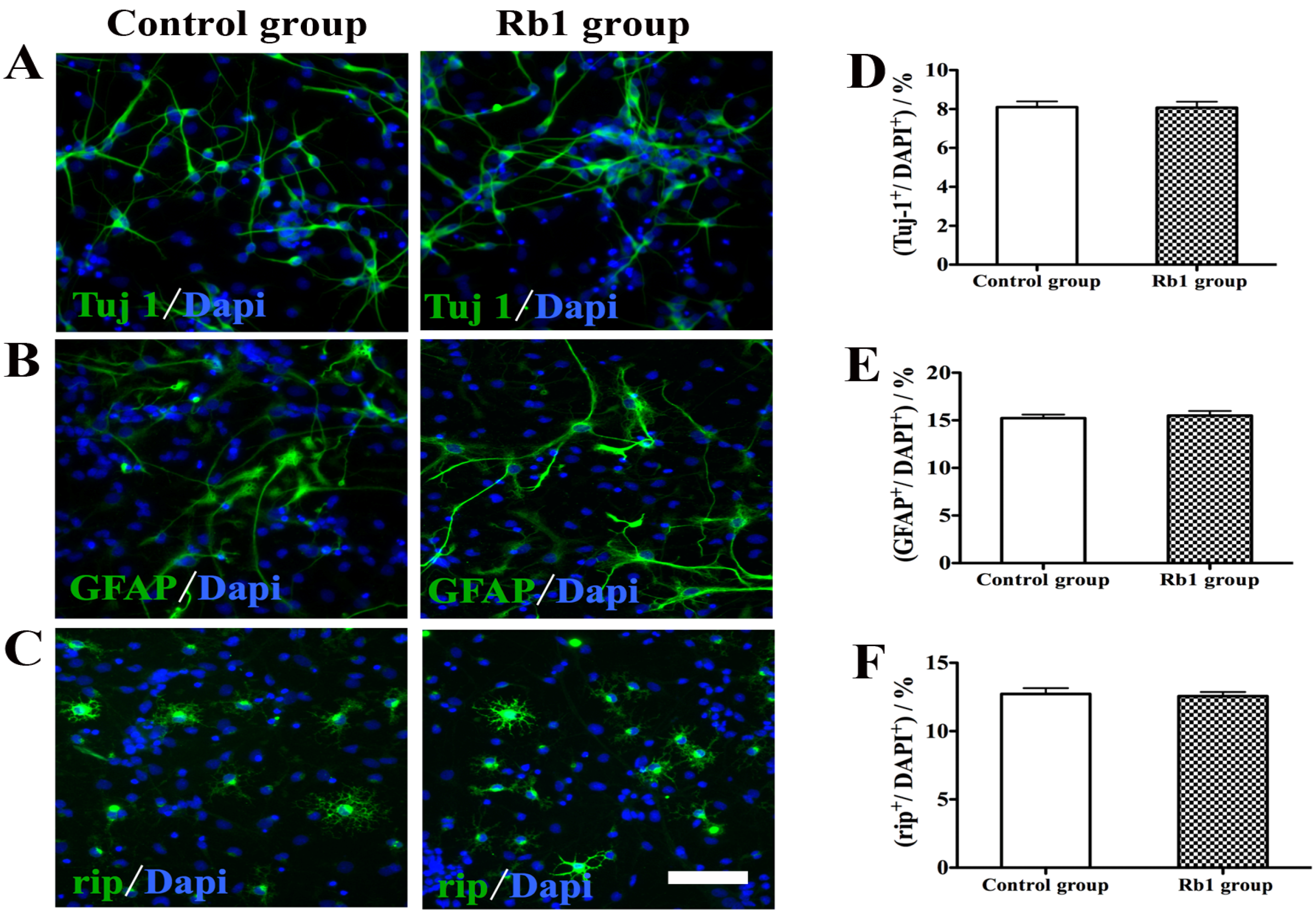
2.4. Pretreatment of Rb1 Showed No Effects on Promoting Proliferation and Neuronal Differentiation of NPCs

3. Exprerimental
3.1. Materials
3.2. Cell Isolation and Culture
3.3. Establishment of t-BHP Induced Oxidative Injury Model
3.4. Lactate Dehydrogenase (LDH) Assay
3.5. Treatment of NPCs with Rb1 in a t-BHP Oxidative Injury Model
3.6. TUNEL Staining
3.7. Immunocytochemistry
3.8. Quantitative Real-Time PCR
| Gene name | Forward (5') primer | Reverse (3') primer | Association No. |
|---|---|---|---|
| Beta actin | 5'-GTCGTACCACTGGCATTCTG-3' | 5'-CTCTCAGCTGTGGTGGTGAA-3' | NM_031144 |
| NRF-2 | 5'-gCAACTCCAgAAggAACAgg-3' | 5'-CAgTgAggggATCgATgAgT-3' | NM-031789.1 |
| HO-1 | 5'-TgCTCgCATgAACACTCTg-3' | 5'-TCCTCTgTCAgCAgTgCCT | NM_012580.2 |
| SOD2 | 5'-ggCCAAgggAgATgTTACAA-3' | 5'-gCTTgATAgCCTCCAgCAAC-3' | NM_001274771 |
| NQO1 | 5'-gCCCggATATTgTAgCTgAA-3' | 5'-gTggTgATggAAAgCAAggT-3' | NM_017000.3 |
| CAT | 5'-TTATggCCTCCgAgATCTTTTC-3' | 5'-ACCTTggTCAggTCAAATggAT-3' | NM_012520 |
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- Aksenov, M.Y.; Aksenova, M.V.; Butterfield, D.A.; Geddes, J.W.; Markesbery, W.R. Protein oxidation in the brain in Alzheimer’s disease. Neuroscience 2001, 103, 373–383. [Google Scholar] [CrossRef]
- Lovell, M.A.; Gabbita, S.P.; Markesbery, W.R. Increased DNA oxidation and decreased levels of repair products in Alzheimer's disease ventricular CSF. J. Neurochem. 1999, 72, 771–776. [Google Scholar] [CrossRef]
- Halliwell, B. Oxidative stress and neurodegeneration: Where are we now? J. Neurochem. 2006, 97, 1634–1658. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Lu, T.T.; Yue, X.Y.; Wei, N.; Jiang, Y.J.; Chen, M.G.; Ni, G.Z.; Liu, X.F.; Xu, G.L. Neuroprotective effect of ginsenoside Rb1 on glutamate-induced neurotoxicity: With emphasis on autophagy. Neurosci. Lett. 2010, 482, 264–268. [Google Scholar] [CrossRef]
- Radad, K.; Gille, G.; Moldzio, R.; Saito, H.; Rausch, W.D. Ginsenosides Rb1 and Rg1 effects on mesencephalic dopaminergic cells stressed with glutamate. Brain Res. 2004, 1021, 41–53. [Google Scholar]
- Gao, X.Q.; Yang, C.X.; Chen, G.J.; Wang, G.Y.; Chen, B.; Tan, S.K.; Liu, J.; Yuan, Q.L. Ginsenoside Rb1 regulates the expressions of brain-derived neurotrophic factor and caspase-3 and induces neurogenesis in rats with experimental cerebral ischemia. J. Ethnopharmacol. 2010, 132, 393–399. [Google Scholar] [CrossRef]
- Nishiyama, N.; Cho, S.I.; Kitagawa, I.; Saito, H. Malonylginsenoside Rb1 potentiates nerve growth factor (NGF)-induced neurite outgrowth of cultured chick embryonic dorsal root ganglia. Biol. Pharm. Bull. 1994, 17, 509–513. [Google Scholar] [CrossRef]
- Hashimoto, R.; Yu, J.; Koizumi, H.; Ouchi, Y.; Okabe, T. Ginsenoside Rb1 Prevents MPP(+)-induced apoptosis in pc12 cells by stimulating estrogen receptors with consequent activation of ERK1/2, Akt and inhibition of SAPK/JNK, p38 MAPK. Evid. Based Complement. Alternat. Med. 2012, 2012, 693717. [Google Scholar]
- Wang, Y.; Liu, J.; Zhang, Z.; Bi, P.; Qi, Z.; Zhang, C. Anti-neuroinflammation effect of ginsenoside Rbl in a rat model of Alzheimer disease. Neurosci. Lett. 2011, 487, 70–72. [Google Scholar] [CrossRef]
- Mook-Jung, I.; Hong, H.S.; Boo, J.H.; Lee, K.H.; Yun, S.H.; Cheong, M.Y.; Joo, I.; Huh, K.; Jung, M.W. Ginsenoside Rb1 and Rg1 improve spatial learning and increase hippocampal synaptophysin level in mice. J. Neurosci. Res. 2001, 63, 509–515. [Google Scholar] [CrossRef]
- Gage, F.H. Mammalian neural stem cells. Science 2000, 287, 1433–1438. [Google Scholar] [CrossRef]
- Alvarez-Buylla, A.; Garcia-Verdugo, J.M. Neurogenesis in adult subventricular zone. J. Neurosci. 2002, 22, 629–634. [Google Scholar]
- Kim, H.J.; Kim, P.; Shin, C.Y. A comprehensive review of the therapeutic and pharmacological effects of ginseng and ginsenosides in central nervous system. J. Ginseng Res. 2013, 37, 8–29. [Google Scholar] [CrossRef]
- Liu, Z.C.; Chen, J.Y.; Huang, W.D.; Zeng, Z.; Yang, Y.F.; Zhu, B.H. Ginsenoside Rb1 protects rat retinal ganglion cells against hypoxia and oxidative stress. Mol. Med. Rep. 2013, 8, 1397–1403. [Google Scholar]
- Kim, S.; Na, J.Y.; Song, K.B.; Choi, D.S.; Kim, J.H.; Kwon, Y.B.; Kwon, J. Protective effect of ginsenoside Rb1 on hydrogen peroxide-induced oxidative stress in rat articular chondrocytes. J. Ginseng Res. 2012, 36, 161–168. [Google Scholar] [CrossRef]
- Tan, S.J.; Yu, Z.; Dong, Q.T. Effects of ginsenoside Rb1 on the oxidative stress in the skeletal muscles of rats with postoperative fatigue syndrome. Zhongguo Zhong Xi Yi Jie He Za Zhi 2012, 32, 1535–1538. [Google Scholar]
- Li, J.; O, W.; Li, W.; Jiang, Z.G.; Ghanbari, H.A. Oxidative stress and neurodegenerative disorders. Int. J. Mol. Sci. 2013, 14, 24438–24475. [Google Scholar] [CrossRef]
- Migliore, L.; Coppede, F. Environmental-induced oxidative stress in neurodegenerative disorders and aging. Mutat. Res. 2009, 674, 73–84. [Google Scholar] [CrossRef]
- Kaspar, J.W.; Niture, S.K.; Jaiswal, A.K. Nrf2:INrf2 (Keap1) signaling in oxidative stress. Free Radic. Biol. Med. 2009, 47, 1304–1309. [Google Scholar] [CrossRef]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-Antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef]
- Johnson, J.A.; Johnson, D.A.; Kraft, A.D.; Calkins, M.J.; Jakel, R.J.; Vargas, M.R.; Chen, P.C. The Nrf2-ARE pathway an indicator and modulator of oxidative stress in neurodegeneration. Mitochon. Oxidative Stress Neurodegener. Disord. 2008, 1147, 61–69. [Google Scholar]
- Van Muiswinkel, F.L.; Kuiperij, H.B. The Nrf2-ARE Signalling pathway: Promising drug target to combat oxidative stress in neurodegenerative disorders. Curr. Drug Targets CNS Neurol. Disord. 2005, 4, 267–281. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Nurulhuda, M.H.; Ismail, A.; Abas, F.; Hamid, M.; Roowi, S. Antioxidative and cardioprotective properties of anthocyanins from defatted dabai extracts. Evid. Based Complement. Alternat. Med. 2013, 2013, 434057. [Google Scholar]
- Li, L.; Dong, H.; Song, E.; Xu, X.; Liu, L.; Song, Y. Nrf2/ARE pathway activation, HO-1 and NQO1 induction by polychlorinated biphenyl quinone is associated with reactive oxygen species and PI3K/AKT signaling. Chem. Biol. Interact. 2014, 209, 56–67. [Google Scholar] [CrossRef]
- Bartoskova, M.; Dobsikova, R.; Stancova, V.; Zivna, D.; Blahova, J.; Marsalek, P.; Zelnickova, L.; Bartos, M.; di Tocco, F.C.; Faggio, C. Evaluation of ibuprofen toxicity for zebrafish (Danio rerio) targeting on selected biomarkers of oxidative stress. Neuro Endocrinol. Lett. 2013, 34, 102–108. [Google Scholar]
- Otterbein, L.E.; Soares, M.P.; Yamashita, K.; Bach, F.H. Heme oxygenase-1: Unleashing the protective properties of heme. Trends Immunol. 2003, 24, 449–455. [Google Scholar] [CrossRef]
- Schulz, S.; Wong, R.J.; Jang, K.Y.; Kalish, F.; Chisholm, K.M.; Zhao, H.; Vreman, H.J.; Sylvester, K.G.; Stevenson, D.K. Heme oxygenase-1 deficiency promotes the development of necrotizing enterocolitis-like intestinal injury in a newborn mouse model. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, G991–G1001. [Google Scholar] [CrossRef]
- Reynolds, B.A.; Weiss, S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 1992, 255, 1707–1710. [Google Scholar]
- Su, H.X.; Zhang, W.M.; Guo, J.S.; Guo, A.C.; Yuan, Q.J.; Wu, W.T. Neural progenitor cells enhance the survival and axonal regeneration of injured motoneurons after transplantation into the avulsed ventral horn of adult rats. J. Neurotrauma 2009, 26, 67–80. [Google Scholar] [CrossRef]
- Maquart, M.; Temmam, S.; Heraud, J.M.; Leparc-Goffart, I.; Cetre-Sossah, C.; Dellagi, K.; Cardinale, E.; Pascalis, H. Development of real-time RT-PCR for the detection of low concentrations of Rift Valley fever virus. J. Virol. Methods 2014, 195, 92–99. [Google Scholar] [CrossRef]
- Saw, C.L.; Yang, A.Y.; Guo, Y.; Kong, A.N. Astaxanthin and omega-3 fatty acids individually and in combination protect against oxidative stress via the NRF2-ARE pathway. Food Chem. Toxicol. 2013, 62, 869–875. [Google Scholar] [CrossRef]
- Sample Availability: Sample of ginsenoside Rb1 is available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ni, N.; Liu, Q.; Ren, H.; Wu, D.; Luo, C.; Li, P.; Wan, J.-B.; Su, H. Ginsenoside Rb1 Protects Rat Neural Progenitor Cells against Oxidative Injury. Molecules 2014, 19, 3012-3024. https://doi.org/10.3390/molecules19033012
Ni N, Liu Q, Ren H, Wu D, Luo C, Li P, Wan J-B, Su H. Ginsenoside Rb1 Protects Rat Neural Progenitor Cells against Oxidative Injury. Molecules. 2014; 19(3):3012-3024. https://doi.org/10.3390/molecules19033012
Chicago/Turabian StyleNi, Na, Qiang Liu, Huixia Ren, Di Wu, Chuanming Luo, Peng Li, Jian-Bo Wan, and Huanxing Su. 2014. "Ginsenoside Rb1 Protects Rat Neural Progenitor Cells against Oxidative Injury" Molecules 19, no. 3: 3012-3024. https://doi.org/10.3390/molecules19033012




