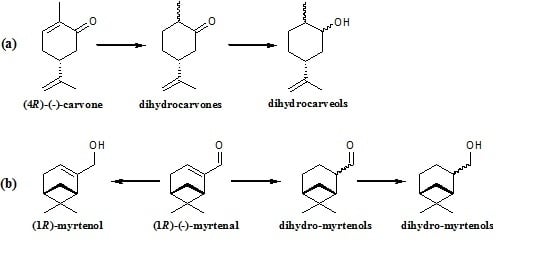
Production of Flavours and Fragrances via Bioreduction of (4R)-(-)-Carvone and (1R)-(-)-Myrtenal by Non-Conventional Yeast Whole-Cells
Abstract
:1. Introduction
2. Results and Discussion
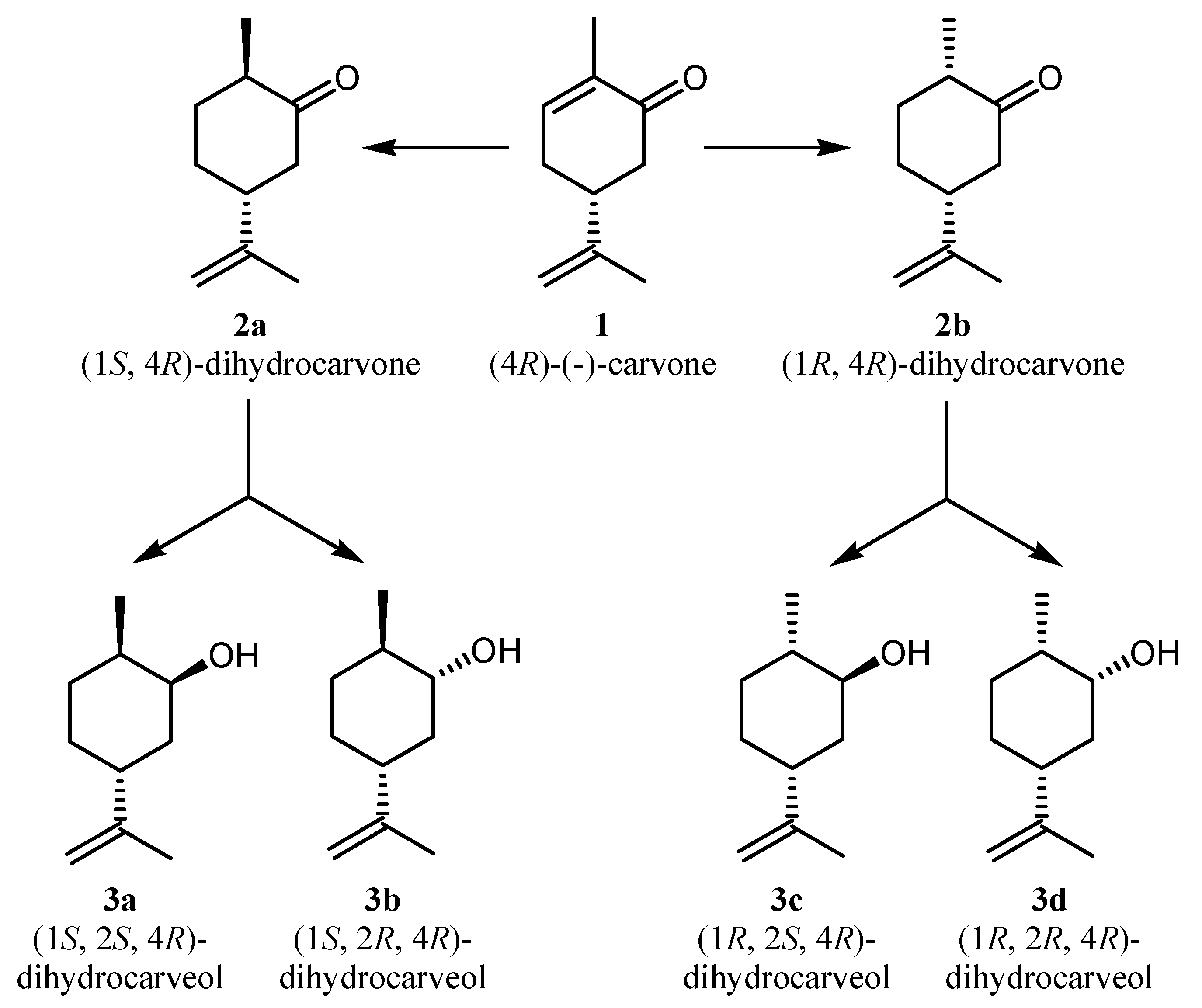
| Species and strain | Conversion (mol %) | Products (mol %) | |||||
|---|---|---|---|---|---|---|---|
| 2b | 2a | 3b | 3a | 3d | 3c | ||
| C. maltosa DBVPG 6021 | 12.93 ± 3.87 | 9.76 ± 4.49 | 0.55 ± 0.04 | 2.63 ± 0.93 | 0 | 0 | 0 |
| Cr. gastricus DBVPG 6057 | 4.71 ± 0.15 | 3.73 ± 0.25 | 0.33 ± 0.03 | 0.66 ± 0.09 | 0 | 0 | 0 |
| C. oregonensis DBVPG 6149 | 14.81 ± 4.46 | 6.69 ± 1.22 | 0.57 ± 0.08 | 7.40 ± 3.19 | 0 | 0.15 ± 0.13 | 0 |
| C. sake DBVPG 6162 | 0.05 ± 0.05 | 0.05 ± 0.04 | 0.01 ± 0.01 | 0 | 0 | 0 | 0 |
| C. freyschussii DBVPG 6208 | 1.09 ± 0.26 | 1.00 ± 0.23 | 0.01 ± 0.01 | 0.07 ± 0.02 | 0 | 0 | 0 |
| W. canadensis DBVPG 6211 | 2.05 ± 0.24 | 0.36 ± 0.12 | 0 | 1.32 ± 0.15 | 0.13 ± 0.22 | 0.24 ± 0.22 | 0 |
| Cr. albidus DBVPG 6237 | 0.74 ± 1.08 | 0.43 ± 0.56 | 0.30 ± 0.53 | 0 | 0 | 0 | 0 |
| Cr. terreus DBVPG 6242 | 7.38 ± 12.43 | 7.24 ± 12.21 | 0.14 ± 0.22 | 0 | 0 | 0 | 0 |
| V. polyspora DBVPG 6243 | 13.45 ± 17.77 | 13.35 ± 17.65 | 0.10 ± 0.13 | 0 | 0 | 0 | 0 |
| K. lodderae DBVPG 6308 | 0.17 ± 0.04 | 0.17 ± 0.04 | 0 | 0 | 0 | 0 | 0 |
| L. amylophila DBVPG 6346 | 7.79 ± 4.91 | 7.37 ± 4.64 | 0.42 ± 0.28 | 0 | 0 | 0 | 0 |
| K. exigua DBVPG 6469 | 4.29 ± 0.83 | 2.87 ± 0.29 | 0.22 ± 0.04 | 1.20 ± 0.53 | 0 | 0 | 0 |
| Cr.s terreus DBVPG 6685 | 0.94 ± 0.42 | 0.90 ± 0.40 | 0.05 ± 0.02 | 0 | 0 | 0 | 0 |
| Kaz. spencerorum DBVPG 6746 | 11.38 ± 16.72 | 10.85 ± 16.23 | 0.46 ± 0.54 | 0.06 ± 0.10 | 0 | 0 | 0 |
| Kaz. spencerorum DBVPG 6748 | 4.43 ± 0.28 | 3.98 ± 0.25 | 0.46 ± 0.03 | 0 | 0 | 0 | 0 |
| H. guilliermondii DBVPG 6790 | 63.59 ± 15.44 | 62.11 ± 14.99 | 1.49 ± 0.45 | 0 | 0 | 0 | 0 |
| H. occidentalis DBVPG 6798 | 6.26 ± 1.45 | 4.90 ± 1.05 | 0.41 ± 0.09 | 0.95 ± 0.33 | 0 | 0 | 0 |
| C. shehatae DBVPG 6850 | 16.93 ± 3.20 | 7.59 ± 0.24 | 0.63 ± 0.02 | 8.71 ± 2.99 | 0 | 0 | 0 |
| K. marxianus DBVPG 6854 | 0.24 ± 0.04 | 0.24 ± 0.04 | 0 | 0 | 0 | 0 | 0 |
| Kaz. africana DBVPG 6934 | 1.44 ± 0.12 | 1.28 ± 0.11 | 0.16 ± 0.01 | 0 | 0 | 0 | 0 |
| N. bacillisporus DBVPG 6945 | 0.33 ± 0.01 | 0.30 ± 0.02 | 0.03 ± 0.00 | 0 | 0 | 0 | 0 |
| N. bacillisporus DBVPG 6962 | 1.27 ± 0.80 | 1.16 ± 0.70 | 0.10 ± 0.11 | 0 | 0 | 0 | 0 |
| D. nepalensis DBVPG 7123 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0 | 0 | 0 | 0 | 0 |
| D. coudertii DBVPG 7124 | 0.020± 0.04 | 0.02 ± 0.04 | 0 | 0 | 0 | 0 | 0 |
| Kaz. naganishii DBVPG 7133 | 12.25 ± 10.56 | 11.68 ± 10.26 | 0.57 ± 0.30 | 0 | 0 | 0 | 0 |
| Strain | Conversion (mol %) | Main products (mol %) | ||
|---|---|---|---|---|
| 5 | 6 | 7 | ||
| C. maltosa DBVPG 6021 | 97.86 ± 0.47 | 95.56 ± 1.34 | 0 | 2.29 ± 0.98 |
| Cr. gastricus DBVPG 6057 | 78.22 ± 20.3 | 65.64 ± 4.77 | 0 | 2.41 ± 0.15 |
| C. oregonensis DBVPG 6149 | 98.82 ± 0.29 | 97.93 ± 0.33 | 0 | 0.89 |
| C. sake DBVPG 6162 | 91.36 ± 1.24 | 0 | 74.80 ± 5.25 | 16.56 ± 4.81 |
| C. freyschussii DBVPG 6208 | 100 ± 0.00 | 96.58 ± 1.87 | 0 | 4.15 ± 0.65 |
| W. canadensis DBVPG 6211 | 52.58 ± 38.44 | 27.62 ± 47. 84 | 1.89 ± 1.68 | 23.07 ± 8.65 |
| Cr. albidus DBVPG 6237 | 99.52 ± 0.82 | 2.26 ± 1.96 | 0 | 67.48 ± 38.82 |
| Cr. terreus DBVPG 6242 | 38.49 ± 53.49 | 36.27 ± 55.37 | 0.58 ± 0.51 | 1.63 ± 1.49 |
| V. polyspora DBVPG 6243 | 83.34 ± 4.43 | 79.06 ± 7.20 | 0 | 1.80 ± 0.78 |
| K. lodderae DBVPG 6308 | 87.13 ± 1.79 | 84.20 ± 4.05 | 0 | 0.33 ± 0.19 |
| L. amylophila DBVPG 6346 | 99.19 ± 0.17 | 0 | 0 | 82.65 ± 5.30 |
| K. exigua DBVPG 6469 | 43.46 ± 37.32 | 17.65 ± 28.62 | 0.14 ± 0.24 | 25.67 ± 44.15 |
| Cr.s terreus DBVPG 6685 | 0 | 0 | 0 | 0 |
| Kaz. spencerorum DBVPG 6746 | 100 ± 0.00 | 98.68 ± 0.44 | 0 | 1.67 ± 0.39 |
| Kaz. spencerorum DBVPG 6748 | 70.81 ± 0.92 | 70.23 ± 1.62 | 0 | 0.70 ± 0.81 |
| H. guilliermondii DBVPG 6790 | 99.33 ± 1.16 | 64.20 ± 55.60 | 0 | 27.13 ± 28.72 |
| H. occidentalis DBVPG 6798 | 78.53 ± 13.46 | 71.59 ± 12.25 | 0 | 6.10 ± 1.36 |
| C. shehatae DBVPG 6850 | 58.50 ± 33.41 | 57.08 ± 32.76 | 0 | 1.46 ± 0.35 |
| K. marxianus DBVPG 6854 | 32.65 ± 56.55 | 0 | 0 | 4.61 ± 5.32 |
| Kaz. africana DBVPG 6934 | 40.87 ± 13.58 | 35.85 ± 53.03 | 0 | 5.96 ± 2.66 |
| N. bacillisporus DBVPG 6945 | 51.64 ± 2.84 | 50.75 ± 2.58 | 0 | 1.15 ± 0.18 |
| N. bacillisporus DBVPG 6962 | 97.02 ± 3.27 | 49.78 ± 57.48 | 0 | 11.16 ± 7.85 |
| D. nepalensis DBVPG 7123 | 53.97 ± 51.15 | 53.97 ± 51.15 | 0 | 0 |
| D. coudertii DBVPG 7124 | 54.41 ± 51.27 | 54.41 ± 51.27 | 0 | 0 |
| Kaz. naganishii DBVPG 7133 | 0 | 0 | 0 | 0 |
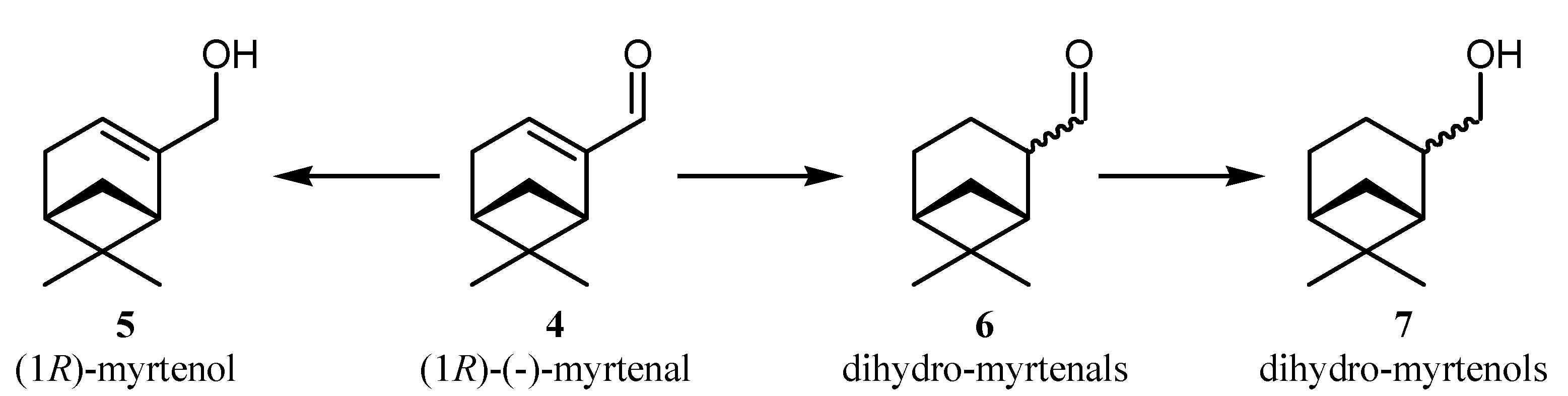
3. Experimental
3.1. Chemicals and Culture Media
3.2. Yeast Strains
| Pecies and Strain | Origin | Location |
|---|---|---|
| Candida maltosa DBVPG 6021 | Soil | Japan |
| Cryptococcus gastricus DBVPG 6057 | Soil | New Zealand |
| Candida oregonensis DBVPG 6149 | Frass of Tsuga heterophylla | USA |
| Candida sake DBVPG 6162 | Soil | Sweden |
| Candida freyschussii DBVPG 6208 | Wood pulp | Sweden |
| Wickerhamomyces canadensis DBVPG 6211 | Wood pulp | Sweden |
| Cryptococcus albidus DBVPG 6237 | Soil | Hungary |
| Cryptococcus terreus DBVPG 6242 | Soil | Buthan |
| Vanderwaltozyma polyspora DBVPG 6243 | Soil | South Africa |
| Kazachstania lodderae DBVPG 6308 | Soil | South Africa |
| Lindnera amylophila DBVPG 6346 | Frass of Pinus taeda | USA |
| Kazachstania exigua DBVPG 6469 | Soil | South Africa |
| Cryptococcus terreus DBVPG 6685 | Soil | ex USSR |
| Kazachstania spencerorum DBVPG 6746 | Soil | South Africa |
| Kazachstania spencerorum DBVPG 6748 | Gut of Psychidae larva | South Africa |
| Hanseniaspora guilliermondii DBVPG 6790 | Trachea of bee | France |
| Hanseniaspora occidentalis DBVPG 6798 | Soil | West Indies |
| Candida shehatae DBVPG 6850 | Rain forest drosophilids | Brazil |
| Kluyveromyces marxianus DBVPG 6854 | Rain forest drosophilids | Brazil |
| Kazachstania africana DBVPG 6934 | Soil | Zimbabwe |
| Nakaseomyces bacillisporus DBVPG 6945 | Exudate of Quercus emoryi | USA |
| Nakaseomyces bacillisporus DBVPG 6962 | Exudate of Quercus emoryi | USA |
| Debaryomyces nepalensis DBVPG 7123 | Soil | Nepal |
| Debaryomyces coudertii DBVPG 7124 | Dropping of Aptenodytes patgonica | France |
| Kazachstania naganishii DBVPG 7133 | Decaying leaves | Japan |
3.3. Preparation of Lyophilized Whole-Cells Biocatalysts
3.4. Bioconversion Reactions
3.5. SPME and GC-MS Analyses
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Matsuda, T.; Yamanaka, R.; Nakamura, K. Recent progress in biocatalysis for asymmetric oxidation and reduction. Tetrahedron Asymmetry 2009, 20, 513–557. [Google Scholar] [CrossRef]
- Bastos Borges, K.; de Souza Borges, W.; Durán-Patrón, R.; Tallarico Pupo, M.; Sueli Bonato, P.; González Collado, I. Stereoselective biotransformations using fungi as biocatalysts. Tetrahedron Asymmetry 2009, 20, 385–397. [Google Scholar] [CrossRef]
- Hollmann, F.; Arends, I.W.C.E.; Holtmann, D. Enzymatic reductions for the chemist. Green Chem. 2011, 13, 2285–2313. [Google Scholar] [CrossRef]
- Hollmann, F.; Arends, I.W.C.E.; Buehler, K.; Schallmey, A.; Buehler, K. Enzyme-mediated oxidations for the chemist. Green Chem. 2011, 13, 226–265. [Google Scholar] [CrossRef]
- Winkler, C.K.; Tasnádi, G.; Clay, D.; Hall, M.; Faber, K. Asymmetric bioreduction of activated alkenes to industrially relevant optically active compounds. J. Biotechnol. 2012, 162, 381–389. [Google Scholar] [CrossRef]
- Patel, R.N. Biocatalysis: Synthesis of key intermediates for development of pharmaceuticals. ACS Catal. 2011, 1, 1056–1074. [Google Scholar] [CrossRef]
- Faber, K. Biotransformations in Organic Chemistry, 5th ed.; Springer-Verlag: Berlin/Heidelberg, Germany, 2004; pp. 3–11. [Google Scholar]
- Lomascolo, A.; Stentelaire, C.; Aster, M.; Lesage-Meessen, L. Basidiomycetes as new biotechnological tools to generate natural aromatic flavours for the food industry. TrendsBiotechnol. 1999, 17, 282–289. [Google Scholar]
- Krings, U.; Berger, R.G. Biotechnological production of flavours and fragrances. Appl. Microbiol. Biotechnol. 1998, 49, 1–8. [Google Scholar] [CrossRef]
- Serra, S.; Fuganti, C.; Brenna, E. Biocatalytic preparation of natural flavours and fragrances. Trends Biotechnol. 2005, 23, 193–198. [Google Scholar] [CrossRef]
- US Code of Federal Regulations; Food and Drug Administration: Washington, DC, USA, 1985; Volume 21, p. 101.22a.3.
- Council Directive 88/388/EEC on the approximation of the laws of the Member States relating to flavourings for use in foodstuffs and to source materials for their production. Official J. Eur. Union L 1988, 184, 61.
- Lemos, B.J.; Dionsio, A.P.; Pastore, G.M. Bio-oxidation of terpenes: An approach for the flavor industry. Chem. Rev. 2009, 109, 4518–4531. [Google Scholar] [CrossRef]
- Cramarossa, M.R.; Nadini, A.; Bondi, M.; Messi, P.; Pagnoni, U.M.; Forti, L. Biocatalytic reduction of (+)- and (–)-carvone by bacteria. C. R. Chim. 2005, 8, 849–852. [Google Scholar] [CrossRef]
- Ponzoni, C.; Gasparetti, C.; Goretti, M.; Turchetti, B.; Pagnoni, U.M.; Cramarossa, M.R.; Forti, L.; Buzzini, P. Biotransformation of acyclic monoterpenoids by Debaryomyces sp., Kluyveromyces. sp., and Pichia. sp. strains of environmental origin. Chem. Biodivers. 2005, 5, 471–483. [Google Scholar]
- King, A.J.; Dickinson, J.R. Biotransformation of monoterpene alcohols by Saccharomyces cerevisiae, Torulaspora. delbrueckii and Kluyveromyces. lactis. Yeast 2000, 16, 499–506. [Google Scholar] [CrossRef]
- Demyttenaere, J.C.R.; del Carmen Herrera, M.; de Kimpe, N. Biotransformation of geraniol, nerol and citral by sporulated surface cultures of Aspergillus. niger and Penicillium. sp. Phytochemistry. 2000, 55, 363–373. [Google Scholar]
- Demyttenaere, J.C.R.; de Pooter, H.E. Biotransformation of citral and nerol by spores of Penicillium. digitatum. Flavour Fragrance J. 1998, 13, 173–176. [Google Scholar] [CrossRef]
- Demyttenaere, J.C.R.; de Pooter, H.E. Biotransformation of geraniol and nerol by spores of Penicillium. italicum. Phytochemistry 1996, 41, 1079–1082. [Google Scholar] [CrossRef]
- Van Dyk, M.S.; van Rensburg, E.; Rensburg, I.P.B.; Moleleki, N. Biotransformation of monoterpenoid ketones by yeasts and yeast-like fungi. J. Mol. Catal. B Enzymatic 1998, 5, 149–154. [Google Scholar] [CrossRef]
- Pinheiro, L.; Marsaioli, A.J. Microbial monooxygenases applied to fragrance compounds. J. Mol. Catal. B Enzym. 2007, 44, 78–86. [Google Scholar] [CrossRef]
- Brenna, E.; Fuganti, C.; Gatti, F.G.; Serra, S. Biocatalytic methods for the synthesis of enantioenriched odor active compounds. Chem. Rev. 2011, 111, 4036–4072. [Google Scholar] [CrossRef]
- Goretti, M.; Ponzoni, C.; Caselli, E.; Marchigiani, E.; Cramarossa, M.R.; Turchetti, B.; Buzzini, P.; Forti, L. Biotransformation of electron-poor alkenes by yeasts: asymmetric reduction of (4S)-(+)-carvone by yeast enoate reductases. Enzyme Microb. Technol. 2009, 45, 463–468. [Google Scholar] [CrossRef]
- Fryszkowska, A.; Toogood, H.; Sakuma, M.; Gardiner, J.M.; Stephens, G.M.; Scrutton, N.S. Asymmetric reduction of activated alkenes by pentaerythritol tetranitrate reductase: specificity and control of stereochemical outcome by reaction optimisation. Adv. Synth. Catal. 2009, 351, 2976–2990. [Google Scholar] [CrossRef]
- Goretti, M.; Branda, E.; Turchetti, B.; Cramarossa, M.R.; Onofri, A.; Forti, L.; Buzzini, P. Response surface methodology as optimization strategy for asymmetric bioreduction of (4S)-(+)-carvone by Cryptococcus gastricus. Biores. Technol. 2012, 121, 290–297. [Google Scholar] [CrossRef]
- Padhi, S.K.; Bougioukou, D.J.; Stewart, J.D. Site-saturation mutagenesis of tryptophan 116 of Saccharomyces pastorianus Old Yellow Enzyme uncovers stereocomplementary variants. J. Am. Chem. Soc. 2009, 131, 3271–3280. [Google Scholar] [CrossRef]
- Adalbjornsson, B.V.; Toogood, H.S.; Fryszkowska, A.; Pudney, C.R.; Jowitt, T.A.; Leys, D.; Scrutton, N.S. Biocatalysis with thermostable enzymes: structure and properties of a thermophilic ‘ene’-reductase related to Old Yellow Enzyme. Chem. Bio. Chem. 2010, 11, 197–207. [Google Scholar] [CrossRef]
- Hook, I.L.; Ryan, S.; Sheridan, H. Biotransformation of aliphatic and aromatic ketones, including several monoterpenoid ketones and their derivatives by five species of marine microalgae. Phytochemistry. 2003, 63, 31–36. [Google Scholar] [CrossRef]
- Hamada, H.; Yasumune, H.; Fuchikami, Y.; Hirata, T.; Sattler, I.; Williams, H.J.; Ian Scott, A. Biotransformation of geraniol, nerol and (+)- and (−)-carvone by suspension cultured cells of Catharanthus. roseus. Phytochemistry. 1997, 44, 615–621. [Google Scholar] [CrossRef]
- Shimoda, K.; Hirata, T. Biotransformation of enones with biocatalysts—Two enone reductases from Astasia. longa. J. Mol. Catal. B Enzym. 2000, 8, 255–264. [Google Scholar] [CrossRef]
- Dutra Silva, V.; Ugarte Stambuk, B.; da Graça Nascimento, M. Asymmetric reduction of (4R)-(−)-carvone catalyzed by Baker’s yeast in aqueous mono- and biphasic systems. J. Mol. Catal. B Enzym. 2012, 77, 98–104. [Google Scholar] [CrossRef]
- Chen, X.; Gao, X.; Wu, Q.; Zhu, D. Synthesis of optically active dihydrocarveol via a stepwise or one-pot enzymatic reduction of (R)- and (S)-carvone. Tetrahedron Asymmetry 2012, 23, 734–738. [Google Scholar] [CrossRef]
- de Carvalho, C.C.C.R.; da Fonseca, M.M.R. Carvone: why and how should one bother to produce this terpene. Food Chem. 2006, 95, 413–422. [Google Scholar] [CrossRef]
- Porto, C.; Stuker, C.Z.; Mallmann, A.S.; Simionatto, E.; Flach, A.; do Canto-Dorow, T.; da Silva, U.F.; Dalcol, I.I.; Morel, A.F. (R)-(−)-carvone and (1R,4R)-trans-(+)-dihydrocarvone from Poiretia. latifolia Vogel. J. Brazil. Chem. Soc. 2010, 21, 782–786. [Google Scholar] [CrossRef]
- Aubin, Y.; Audran, G.; Monti, H. Improved enantioselective synthesis of natural striatenic acid and its methyl ester. Tetrahedron Lett. 2006, 47, 3669–3671. [Google Scholar] [CrossRef]
- Blay, G.; Garcia, B.; Molina, E.; Pedro, J.R. Synthesis of (+)-pechueloic acid and (+)-aciphyllene. Revision of the structure of (+)-aciphyllene. Tetrahedron 2007, 63, 9621–9626. [Google Scholar] [CrossRef]
- Harrowven, D.C.; Pascoe, D.D.; Demurtas, D.; Bourne, H.O. Total synthesis of (−)-colombiasin A and (−)-elisapterosin B. Angew. Chem. Int. Ed. 2005, 44, 1221–1222. [Google Scholar]
- Dong, Y.; McCullough, K.J.; Wittlin, S.; Chollet, J.; Vennerstrom, J.L. The structure and antimalarial activity of dispiro-1,2,4,5-tetraoxanes derived from (+)-dihydrocarvone. Bioorg. Med. Chem. Lett. 2010, 20, 6359–6361. [Google Scholar] [CrossRef]
- de Rouville, H.P.J.; Vives, G.; Tur, E.; Rapenne, G.; Crassous, J. Synthesis and analytical resolution of chiral pyrazoles derived from (5R)-dihydrocarvone. New J. Chem. 2009, 33, 293–299. [Google Scholar] [CrossRef]
- Krawczyk, H.; Sliwinski, M.; Kedzia, J.; Wojciechowski, J.; Wolf, W.M. Asymmetric synthesis of enantiomerically pure 7-isopropenyl-4a-methyl-3-methyleneoctahydrochromen-2-ones. Tetrahedron Asymmetry 2007, 18, 2712–2718. [Google Scholar] [CrossRef]
- Bhatia, S.P.; Letizia, C.S.; Api, A.M. Fragrance material review on dihydrocarveol (R,R,R). Food Chem. Toxicol. 2008, 46, S121–S122. [Google Scholar] [CrossRef]
- Demain, A.L.; Phaff, H.J.; Kurtzman, C.P. The Idustrial and Agricultural Significance of Yeasts. In The Yeasts. A Taxonomic Study, 4th ed.; Kurtzman, C.P., Fell, J.W., Eds.; Elsevier Science: Amsterdam, The Netherlands, 1998; pp. 13–19. [Google Scholar]
- Johnson, E.A.; Echavarri-Erasun, C. Yeast biotechnology. In The Yeasts. A Taxonomy Study; Kurtzman, C.P., Fell, J.W., Boekhout, T., Eds.; Elsevier: New York, NY, USA, 2011; Volume 1, pp. 21–44. [Google Scholar]
- Buzzini, P.; Vaughan-Martini, A. Yeast Biodiversity and Biotechnology. In The Yeast Handbook. Biodiversity and Ecophysiology of Yeasts; Rosa, C.A., Gabor, P., Eds.; Springer-Verlag: Berlin, Germany, 2006; pp. 533–559. [Google Scholar]
- Industrial Biotransformations, 2nd ed.; Liese, A.; Seelbach, K.; Wandrey, C. (Eds.) Wiley-VCH: Weinheim, Germany, 2006.
- De Carvalho, C.C.R.; da Fonseca, M.M.R. Biotransformation of terpenes. Biotechnol. Adv. 2006, 24, 134–142. [Google Scholar] [CrossRef]
- Wolf, K.; Breunig, K.; Barth, G. Non Conventional Yeasts in Genetics, Biochemistry and Biotechnology; Springer: Berlin, Germany, 2003. [Google Scholar]
- Van der Donk, W.A.; Zhao, H. Recent developments in pyridine nucleotide regeneration. Curr. Opin. Biotechnol. 2003, 14, 421–426. [Google Scholar] [CrossRef]
- Velonia, K.; Smonou, I. Dismutation of aldehydes catalyzed by alcohol dehydrogenases. J. Chem. Soc. Perkin Trans. 1 2000, 2283–2287. [Google Scholar] [CrossRef]
- Morgan, G.K., III; Liu, Z.; Wene, M.J.; King, D.F. Dryer-added fabric care articles. 2 August 2007. [Google Scholar] [PubMed]
- Noma, Y.; Takahashi, H.; Asakawa, Y. Biotransformation of terpene aldehydes by Euglena gracilis Z. Phytochemistry. 1991, 30, 1147–1151. [Google Scholar] [CrossRef]
- Bhatia, S.P.; McGinty, D.; Letizia, C.S.; Api, A.M. Fragrance material review on myrtenol. Food Chem. Toxicol. 2008, 46, S237–S240. [Google Scholar] [CrossRef]
- The University of Perugia, Available online:. Available online: http://www.agr.unipg.it/dbvpg (accessed on 16 May 2013).
- Carballeira, J.D.; Alvarez, E.; Sinisterra, J.V. Biotransformation of cyclohexanone using immobilized Geotrichum candidum NCYC49 - Factors affecting the selectivity of the process. J. Mol. Catal. B. Enzym. 2004, 28, 25–32. [Google Scholar] [CrossRef]
- Goretti, M.; Ponzoni, C.; Caselli, E.; Marchegiani, E.; Cramarossa, M.R.; Turchetti, B.; Forti, L.; Buzzini, P. Bioreduction of a,b-unsaturated ketones and aldehydes by non-conventional yeast (NCY) whole-cells. Biores. Technol. 2011, 102, 3993–3998. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the (4R)-(−)-carvone and (1R)-(−)-myrtenal and all strains used in the present study are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goretti, M.; Turchetti, B.; Cramarossa, M.R.; Forti, L.; Buzzini, P. Production of Flavours and Fragrances via Bioreduction of (4R)-(-)-Carvone and (1R)-(-)-Myrtenal by Non-Conventional Yeast Whole-Cells. Molecules 2013, 18, 5736-5748. https://doi.org/10.3390/molecules18055736
Goretti M, Turchetti B, Cramarossa MR, Forti L, Buzzini P. Production of Flavours and Fragrances via Bioreduction of (4R)-(-)-Carvone and (1R)-(-)-Myrtenal by Non-Conventional Yeast Whole-Cells. Molecules. 2013; 18(5):5736-5748. https://doi.org/10.3390/molecules18055736
Chicago/Turabian StyleGoretti, Marta, Benedetta Turchetti, Maria Rita Cramarossa, Luca Forti, and Pietro Buzzini. 2013. "Production of Flavours and Fragrances via Bioreduction of (4R)-(-)-Carvone and (1R)-(-)-Myrtenal by Non-Conventional Yeast Whole-Cells" Molecules 18, no. 5: 5736-5748. https://doi.org/10.3390/molecules18055736