Synthesis and Cytotoxic Activity of Some Novel N-Pyridinyl-2-(6-phenylimidazo[2,1-b]thiazol-3-yl)acetamide Derivatives
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
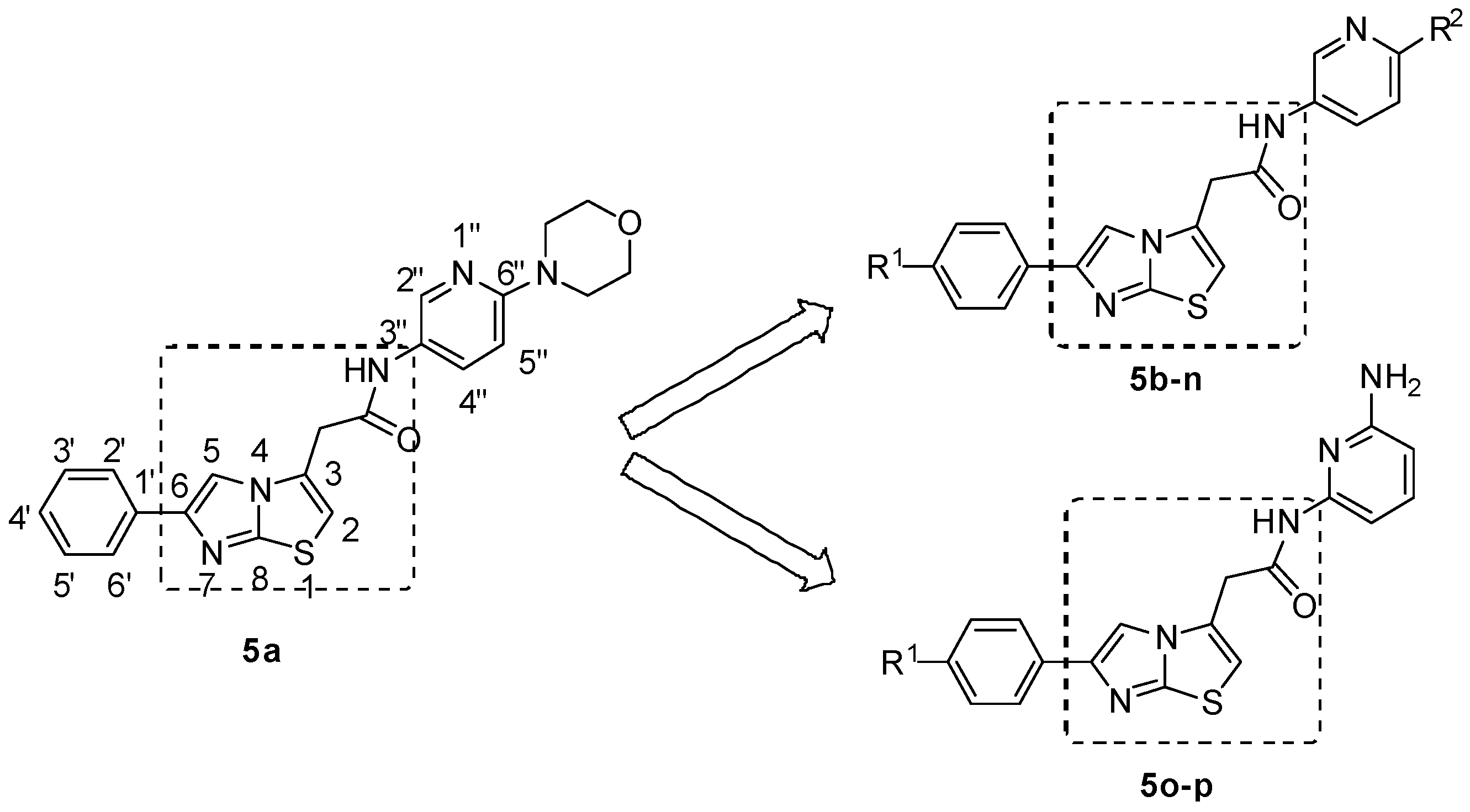
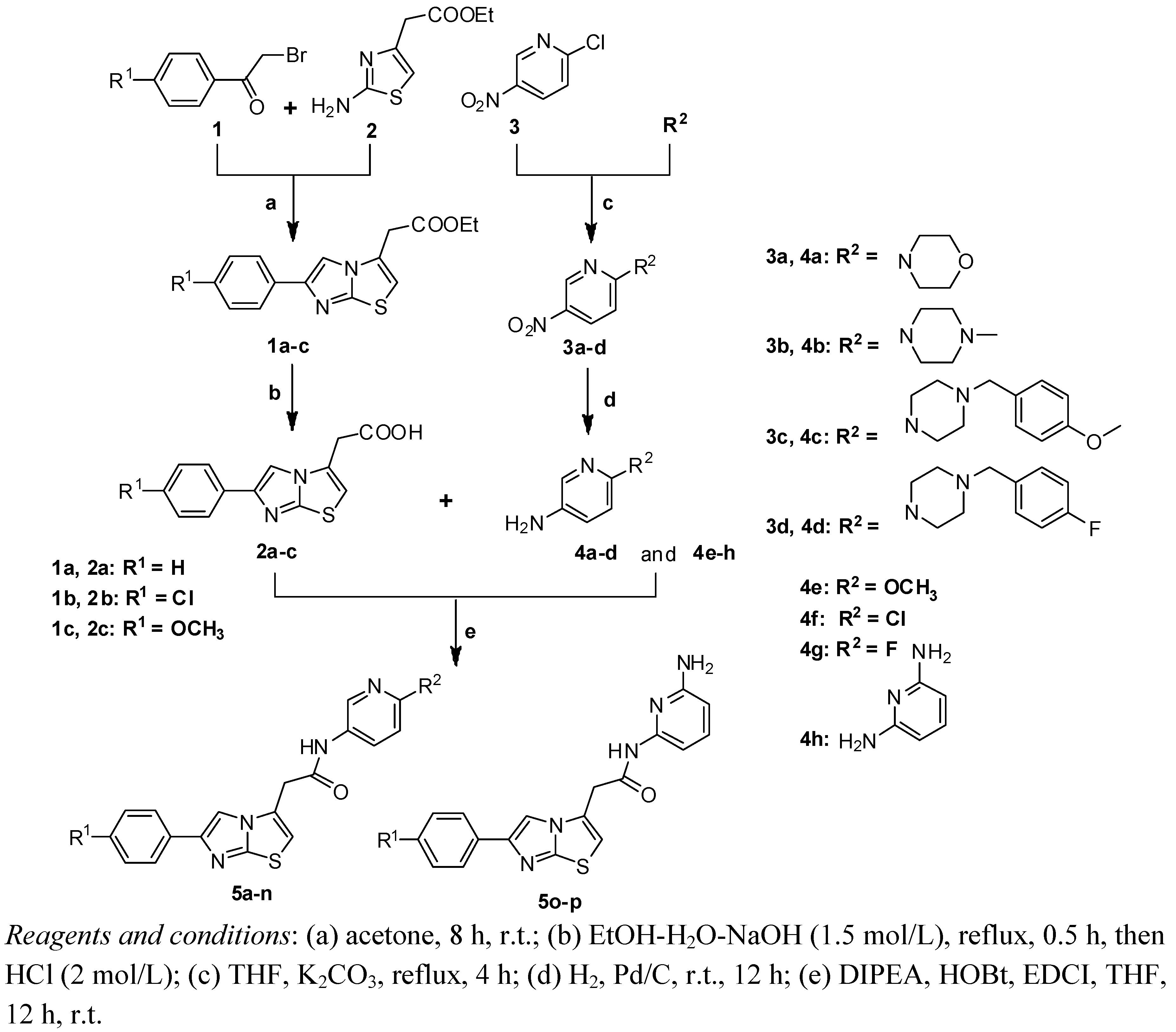
2.2. In Vitro Bioactivity

| Compound | R1 | R2 | IC50* (µM) | |
|---|---|---|---|---|
| HepG2 | MDA-MB-231 | |||
| 5a | H |  | 74.2 ± 2.5 | 27.1 ± 0.4 |
| 5b | H | OCH3 | >100 | >100 |
| 5c | Cl | OCH3 | 63.7 ± 0.4 | 40.1 ± 1.3 |
| 5d | H | Cl | 62.0 ± 3.7 | 22.8 ± 4.6 |
| 5e | H | F | >100 | 79.0 ± 3.8 |
| 5f | Cl | Cl | 50.0 ± 1.4 | 13.0 ± 0.2 |
| 5g | Cl | F | 53.4 ± 0.5 | 22.3 ± 1.3 |
| 5h | OCH3 | Cl | >100 | 51.8 ± 0.8 |
| 5i | H |  | 71.5 ± 1.7 | >100 |
| 5j | Cl |  | 28.2 ± 0.6 | >100 |
| 5k | H |  | 39.4 ± 1.9 | 6.0 ± 0.7 |
| 5l | Cl |  | 22.6 ± 1.5 | 1.4 ± 0.1 |
| 5m | H |  | 55.2 ± 1.5 | 19.8 ± 2.2 |
| 5n | Cl |  | 34.7 ± 0.4 | 12.9 ± 0.2 |
| 5o | H | >100 | 62.6 ± 3.7 | |
| 5p | Cl | 48.9 ± 1.4 | 35.1 ± 0.5 | |
| sorafenib | 33.7 ± 1.3 | 5.2 ± 0.2 | ||
3. Experimental
3.1. Materials and Reagents
3.2. Chemical Synthesis
3.2.1. General Procedure for Preparation of 2-(6-Phenylimidazo[2,1-b]thiazol-3-yl)acetic Acid Derivatives 2a–c
3.2.2. General Procedure for Preparation of Compounds 3a–d
3.2.3. General Procedure for Preparation of Compounds 4a–d
3.2.4. General Procedure for Preparation of Compounds 5a–p
3.3. Biological Methods
3.3.1. In Vitro Cytotoxic Assay
3.3.2. In Vitro VEGFR Inhibitory Activity Assay
4. Conclusions
Acknowledgments
References and Notes
- Dangi, R.R.; Hussain, N.; Talesara, G.L. Synthesis characterization and biological evaluation of some alkoxyphthalimide derivatives of 3-(4-substituted phenyl)-6,6-diphenyl-3,3a-dihydro-2H-imidazo[2,1-b]pyrazolo[3,4-d][1,3]thiazol-7(6H)-one. Med. Chem. Res. 2011, 20, 1490–1498. [Google Scholar] [CrossRef]
- Juspin, T.; Laget, M.; Terme, T.; Azas, N.; Vanelle, P. TDAE-assisted synthesis of new imidazo[2,1-b]thiazole derivatives as anti-infectious agents. Eur. J. Med. Chem. 2010, 45, 840–845. [Google Scholar]
- Atta, K.F.M.; Farahat, O.O.M.; Ahmed, A.Z.A.; Marei, M.G. Synthesis and antibacterial activities of novel imidazo[2,1-b]-1,3,4-thiadiazoles. Molecules 2011, 16, 5496–5506. [Google Scholar]
- Guzeldemirci, N.U.; Kucukbasmaci, O. Synthesis and antimicrobial activity evaluation of new 1,2,4-triazoles and 1,3,4-thiadiazoles bearing imidazo[2,1-b]thiazole moiety. Eur. J. Med. Chem. 2010, 45, 63–68. [Google Scholar]
- Shetty, N.S.; Koti, R.S.; Lamani, R.S.; Badiger, N.P.; Khazi, I.A.M. Synthesis and antimicrobial activities of some ethyl 2-arylthio-6-arylimidazo[2,1-b]thiazole-3-carboxylates and their sulfones. J. Sulfur. Chem. 2008, 29, 539–547. [Google Scholar]
- Lamani, R.S.; Shetty, N.S.; Kamble, R.R.; Khazi, I.A.M. Synthesis and antimicrobial studies of novel methylene bridged benzisoxazolyl imidazo[2,1-b][1,3,4]thiadiazole derivatives. Eur. J. Med. Chem. 2009, 44, 2828–2833. [Google Scholar]
- Jadhav, V.B.; Kulkarni, M.V.; Rasal, V.P.; Biradar, S.S.; Vinay, M.D. Synthesis and anti-inflammatory evaluation of methylene bridged benzofuranyl imidazo[2,1-b][1,3,4]thiadiazoles. Eur. J. Med. Chem. 2008, 43, 1721–1729. [Google Scholar]
- Budriesi, R.; Ioan, P.; Locatelli, A.; Cosconati, S.; Leoni, A.; Ugenti, M.P.; Andreani, A.; di Toro, R.; Bedini, A.; Spampinato, S.; et al. Imidazo[2,1-b]thiazole system: A scaffold endowing dihydropyridines with selective cardiodepressant activity. J. Med. Chem. 2008, 51, 1592–1600. [Google Scholar]
- Budriesi, R.; Ioan, P.; Leoni, A.; Pedemonte, N.; Locatelli, A.; Micucci, M.; Chiarini, A.; Galietta, L.J.V. Cystic fibrosis: A new target for 4-imidazo[2,1-b]thiazole-1,4-dihydropyridines. J. Med. Chem. 2011, 54, 3885–3894. [Google Scholar]
- Noolvi, M.N.; Patel, H.M.; Singh, N.; Gadad, A.K.; Cameotra, S.S.; Badiger, A. Synthesis and anticancer evaluation of novel 2-cyclopropylimidazo[2,1-b][1,3,4]-thiadiazole derivatives. Eur. J. Med. Chem. 2011, 46, 4411–4418. [Google Scholar]
- Park, J.H.; Oh, C.H. Synthesis of new 6-(4-fluorophenyl)-5-(2-substituted pyrimidin-4-yl)imidazo[2,1-b] thiazole derivatives and their antiproliferative activity against melanoma cell line. Bull. Korean Chem. Soc. 2010, 31, 2854–2860. [Google Scholar]
- Kamal, A.; Dastagiri, D.; Ramaiah, M.J.; Reddy, J.S.; Bharathi, E.V.; Srinivas, C.; Pushpavalli, S.N.C.V.L.; Pal, D.; Pal-Bhadra, M. Synthesis of imidazothiazole-chalcone derivatives as anticancer and apoptosis inducing agents. ChemMedChem 2010, 5, 1937–1947. [Google Scholar]
- Meric, A.; Incesu, Z.; Hatipoglu, I. Synthesis of some 3,4-disubstituted-6,7-dihydro-imidazo[2,1-b][1,3]thiazole and 3,4-disubstituted-7,8-dihydro-6H-imidazo[2,1-b][1,3]thiazine derivatives and evaluation of their cytotoxicities against F2408 and 5RP7 cells. Med. Chem. Res. 2008, 17, 30–41. [Google Scholar] [CrossRef]
- Andreani, A.; Burnelli, S.; Granaiola, M.; Leoni, A.; Locatelli, A.; Morigi, R.; Rambaldi, M.; Varoli, L.; Calonghi, N.; Cappadone, C.; et al. Antitumor activity of new substituted 3-(5-Imidazo[2,1-b]thiazolylmethylene)-2-indolinones and 3-(5-Imidazo[2,1-b]thiadiazolylmethylene)-2-indolinones: Selectivity against colon tumor cells and effect on cell cycle-related events. J. Med. Chem. 2008, 51, 7508–7513. [Google Scholar]
- Andreani, A.; Burnelli, S.; Granaiola, M.; Leoni, A.; Locatelli, A.; Morigi, R.; Rambaldi, M.; Varoli, L.; Calonghi, N.; Cappadone, C.; et al. Potential Antitumor Agents. 42. New antitumor imidazo[2,1-b]thiazole guanylhydrazones and analogues. J. Med. Chem. 2008, 51, 809–816. [Google Scholar]
- Gursoy, E.; Guzeldemirci, N.U. Synthesis and primary cytotoxicity evaluation of new imidazo[2,1-b]thiazole derivatives. Eur. J. Med. Chem. 2007, 42, 320–326. [Google Scholar]
- Lapierre, J.; Liu, Y.; Tandon, M. Inhibiteurs de raf et leurs utilisations. WO Patent WO/2010/065893, 10 June 2010. [Google Scholar]
- Fidanze, S.D.; Erickson, S.A.; Wang, G.T.; Mantei, R.; Clark, R.F.; Sorensen, B.K.; Bamaung, N.Y.; Kovar, P.; Johnson, E.F.; Swinger, K.K.; et al. Imidazo[2,1-b]thiazoles: Multitargeted inhibitors of both the insulin-like growth factor receptor and members of the epidermal growth factor family of receptor tyrosine kinases. Bioorg. Med. Chem. Lett. 2010, 20, 2452–2455. [Google Scholar]
- Borhani, D.W.; Calderwood, D.J.; Frank, K.E.; Davis, H.M.; Josephsohn, N.S.; Skinner, B.S. Novel imidazothiazoles and imidazoxazoles. WO Patent WO/2008/063287, 29 May 2008. [Google Scholar]
- Ivy, S.P.; Wick, J.Y.; Kaufman, B.M. An overview of small-molecule inhibitors of VEGFR signaling. Nat. Rev. Clin. Oncol. 2009, 6, 569–579. [Google Scholar]
- Sharma, P.S.; Sharma, R.; Tyagi, T. VEGF/VEGFR Pathway inhibitors as anti-angiogenic agents: Present and future. Curr. Cancer Drug Targets 2011, 11, 624–653. [Google Scholar]
- Han, L.; Ma, X.; Lin, H.; Jia, J.; Zhu, F.; Xue, Y.; Li, Z.; Cao, Z.; Ji, Z.; Chen, Y. A support vector machines approach for virtual screening of active compounds of single and multiple mechanisms from large libraries at an improved hit-rate and enrichment factor. J. Mol. Graph. Model. 2008, 26, 1276–1286. [Google Scholar]
- Glick, M.; Jenkins, J.L.; Nettles, J.H.; Hitchings, H.; Davies, J.W. Enrichment of high-throughput screening data with increasing levels of noise using support vector machines, recursive partitioning, and laplacian-modified naive bayesian classifiers. J. Chem. Inf. Model. 2006, 46, 193–200. [Google Scholar] [CrossRef]
- Palagiano, F.; Arenare, L.; Luraschi, E.; Decaprariis, P.; Abignente, E.; Damico, M.; Filippelli, W.; Rossi, F. Research on heterocyclic-compounds. 34. Synthesis and sar study of some imidazo[2,1,B]thiazole carboxylic and acetic-acids with antiinflammatory and analgesic activities. Eur. J. Med. Chem. 1995, 30, 901–909. [Google Scholar] [CrossRef]
- Bolin, D.R.; Michoud, C. Inhibitors of daicylglycerol acyltransferase (DGAT). EP Patent 1,848,687,31, October 2007. [Google Scholar]
- Swanson, D.M.; Shah, C.R.; Lord, B.; Morton, K.; Dvorak, L.K.; Mazur, C.; Apodaca, R.; Xiao, W.; Boggs, J.D.; Feinstein, M.; et al. Heterocyclic replacement of the central phenyl core of diamine-based histamine H(3) receptor antagonists. Eur. J. Med. Chem. 2009, 44, 4413–4425. [Google Scholar]
- Zegzouti, H.; Zdanovskaia, M.; Hsiao, K.; Goueli, S.A. ADP-Glo: A bioluminescent and homogeneous ADP monitoring assay for kinases. Assay Drug Dev. Technol. 2009, 7, 560–572. [Google Scholar]
- Sample Availability: Samples of the compounds 5a–p are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ding, H.; Chen, Z.; Zhang, C.; Xin, T.; Wang, Y.; Song, H.; Jiang, Y.; Chen, Y.; Xu, Y.; Tan, C. Synthesis and Cytotoxic Activity of Some Novel N-Pyridinyl-2-(6-phenylimidazo[2,1-b]thiazol-3-yl)acetamide Derivatives. Molecules 2012, 17, 4703-4716. https://doi.org/10.3390/molecules17044703
Ding H, Chen Z, Zhang C, Xin T, Wang Y, Song H, Jiang Y, Chen Y, Xu Y, Tan C. Synthesis and Cytotoxic Activity of Some Novel N-Pyridinyl-2-(6-phenylimidazo[2,1-b]thiazol-3-yl)acetamide Derivatives. Molecules. 2012; 17(4):4703-4716. https://doi.org/10.3390/molecules17044703
Chicago/Turabian StyleDing, Huaiwei, Zhe Chen, Cunlong Zhang, Tian Xin, Yini Wang, Hongrui Song, Yuyang Jiang, Yuzong Chen, Yongnan Xu, and Chunyan Tan. 2012. "Synthesis and Cytotoxic Activity of Some Novel N-Pyridinyl-2-(6-phenylimidazo[2,1-b]thiazol-3-yl)acetamide Derivatives" Molecules 17, no. 4: 4703-4716. https://doi.org/10.3390/molecules17044703




