Synthesis, Molecular Docking and Preliminary in-Vitro Cytotoxic Evaluation of Some Substituted Tetrahydro-naphthalene (2',3',4',6'-Tetra-O-Acetyl-β-D-Gluco/-Galactopyranosyl) Derivatives
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
| Comp No. | Ar | R1 | R2 | MP (°C) | Cryst.Solv. | Yield % | Molecular Formula (Mol. Wt.) |
|---|---|---|---|---|---|---|---|
| 2a | -C4H3O | --- | --- | 245–247 | Ethanol/H2O | 80 | C20H16N2OS (332) |
| 3a | -C4H3O | H | OAc | 194–196 | Ethanol/H2O | 76 | C34H34N2O10S (662) |
| 3b | 4-NO2-C6H5 | H | OAc | 172–174 | Ethanol/H2O | 75 | C36H35N3O11S (717) |
| 3c | 3-EtO,4-OH-C6H4 | H | OAc | 118–120 | Ethanol/H2O | 68 | C38H40N2O11S (732) |
| 3d | -C4H3O | OAc | H | 215–217 | Ethanol/H2O | 75 | C34H34N2O10S (662) |
| 3e | 4-NO2-C6H5 | OAc | H | 140–142 | Ethanol/H2O | 76 | C36H35N3O11S (717) |
| 3f | 3-EtO-4-OH-C6H4 | OAc | H | 175–177 | Ethanol/H2O | 79 | C38H40N2O11S (732) |
| 5a | 2,6-Cl2-C6H4 | H | OAc | 192–194 | Ethanol/H2O | 65 | C36H35Cl2N3O9 (724) |
| 5b | 2,6-Cl2-C6H4 | OAc | H | 205–207 | Ethanol/H2O | 70 | C36H35Cl2N3O9 (724) |
| 5c | 2,6-F2-C6H4 | H | OAc | 107–109 | Ethanol/H2O | 66 | C36H35F2N3O9 (691) |
| 5d | 2,6-F2-C6H4 | OAc | H | 132–134 | Ethanol/H2O | 72 | C36H35F2N3O9 (691) |
| 7a | 2,6-Cl2-C6H4 | --- | --- | 180–182 | Ethanol/H2O | 95 | C22H18Cl2N2O2 (413) |
| 7b | 2,6-F2-C6H4 | --- | --- | 170–172 | Ethanol/H2O | 92 | C22H18F2N2O2 (380) |
| 8a | 2,6-Cl2-C6H4 | OAc | H | 230–232 | Ethanol/H2O | 86 | C36H36Cl2N2O11 (743) |
| 8b | 2,6-F2-C6H4 | OAc | H | 155–157 | Ethanol/H2O | 85 | C36H36F2N2O11 (710) |
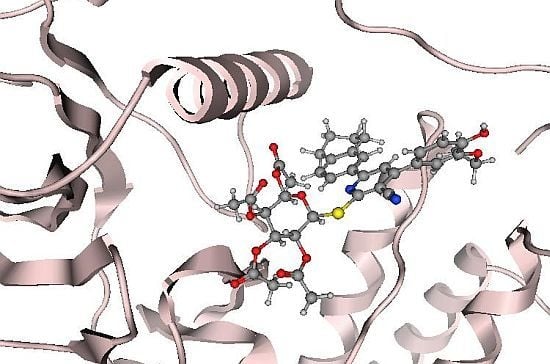
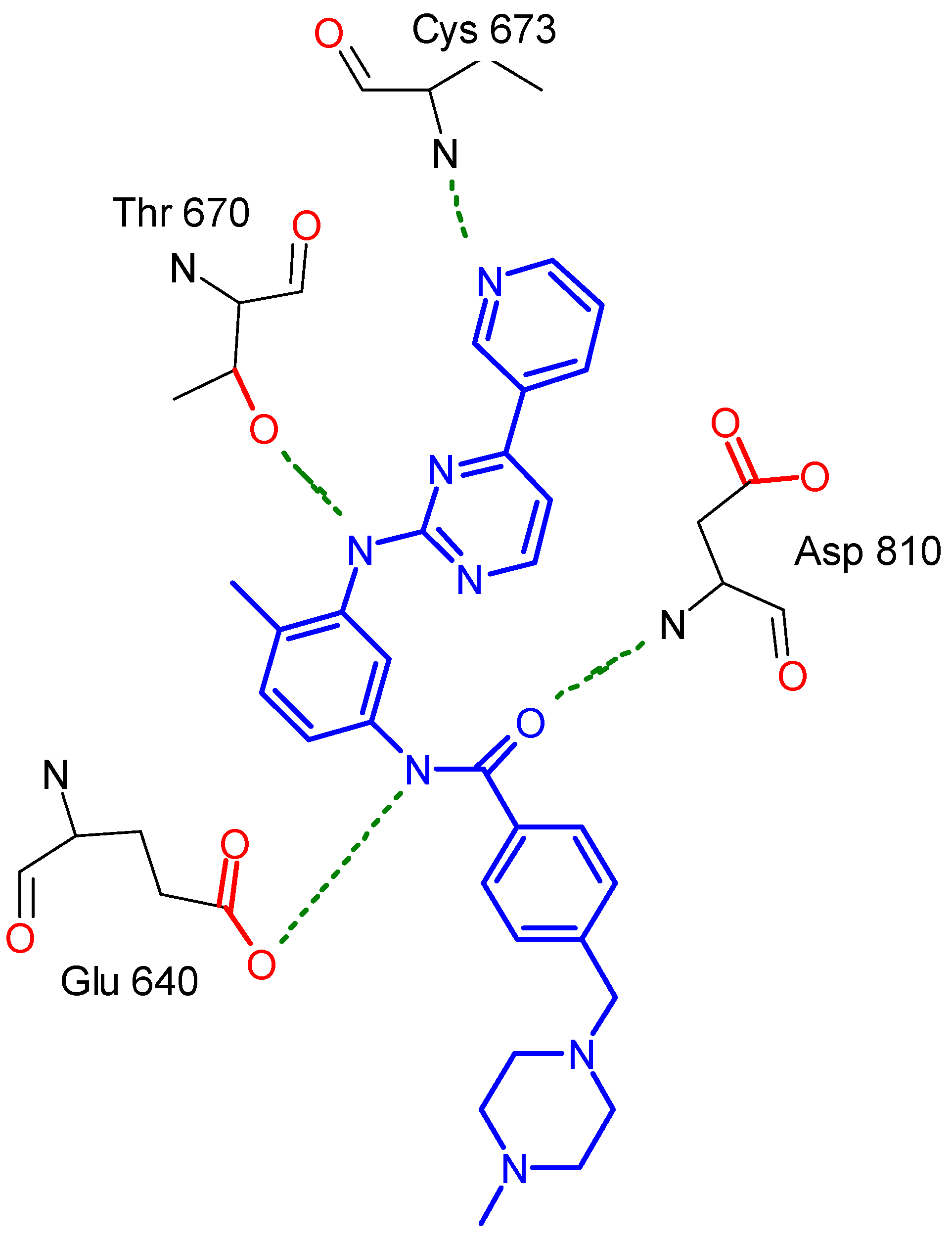
2.2. Molecular Modeling Study
Docking Using Autodock Vina


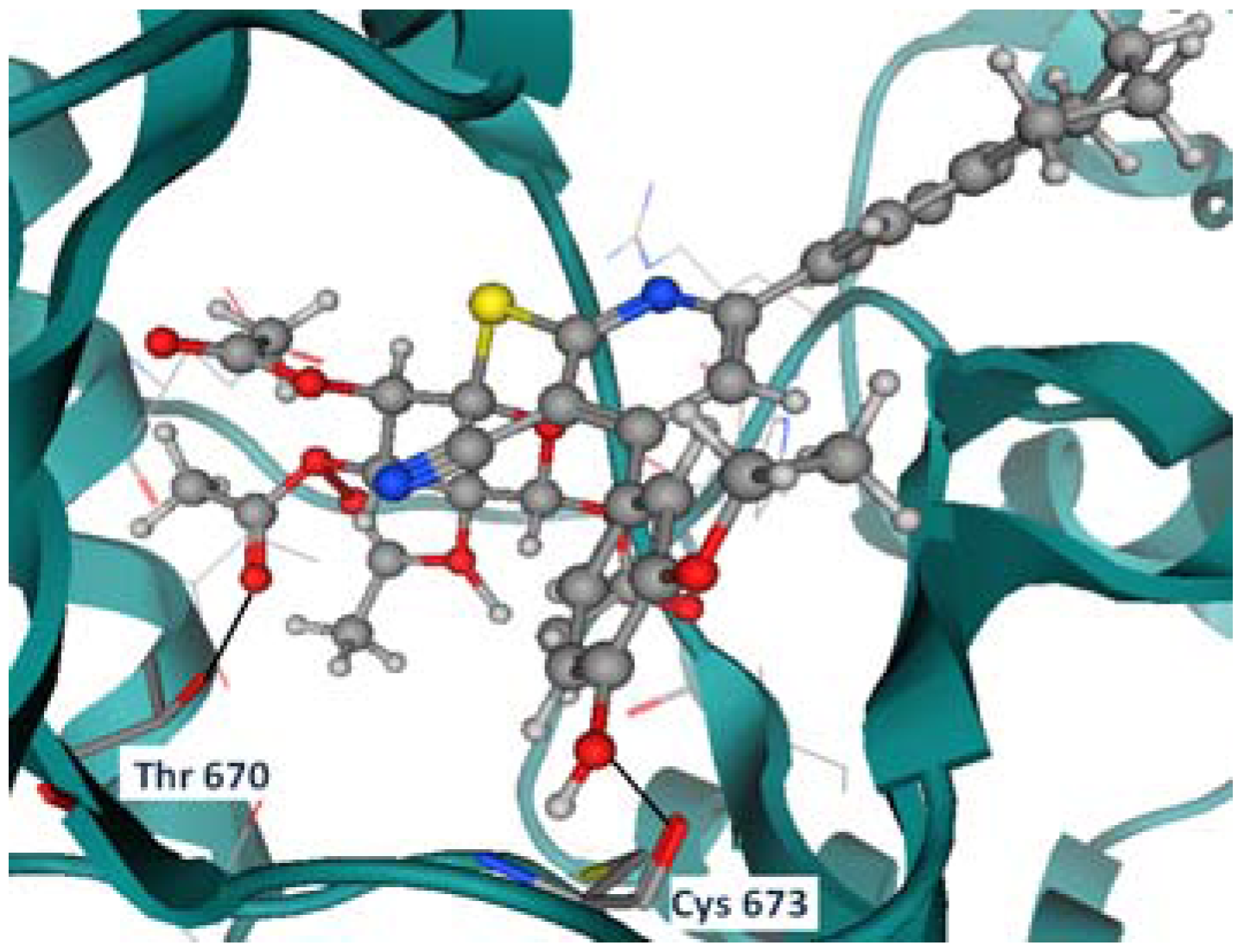


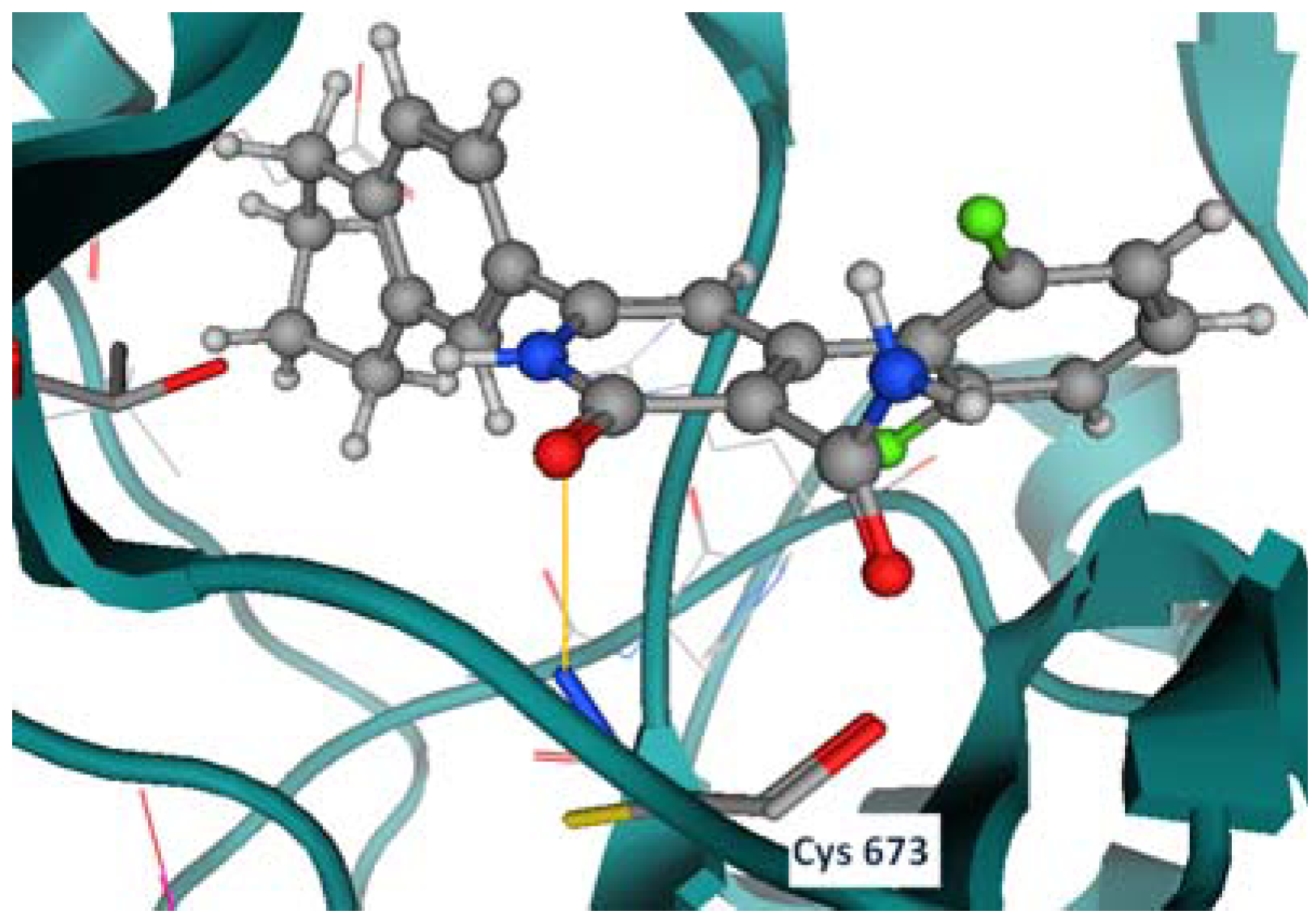
2.3. In Vitro Cytotoxic Screening
| Compound | Anticancer effect % inhibition of cell viability | Affinity Kcal/mol | RMSD Deviation from best mode | |
|---|---|---|---|---|
| 100 µg/mL | 50 µg/mL | |||
| 2b | 0 | 0 | −9.5 | 7.07 |
| 3a | 0 | 0 | −8.3 | 9.5 |
| 3b | 0 | 0 | −7.7 | 11.6 |
| 3c | 70% | 50% | −10.7 | 4.02 |
| 3d | 0 | 0 | −6.6 | 10.3 |
| 3e | 0 | 0 | −7.4 | 9.2 |
| 3f | 10% | 0 | −11.4 | 9.0 |
| 5b | 0 | 0 | −11.0 | 9.3 |
| 5c | 40% | 7% | −11.2 | 9.5 |
| 7a | 0 | 0 | −10.3 | 7.2 |
| 7b | 20% | 15% | −11.6 | 4.6 |
3. Experimental
3.1. Chemistry
3.1.1. General
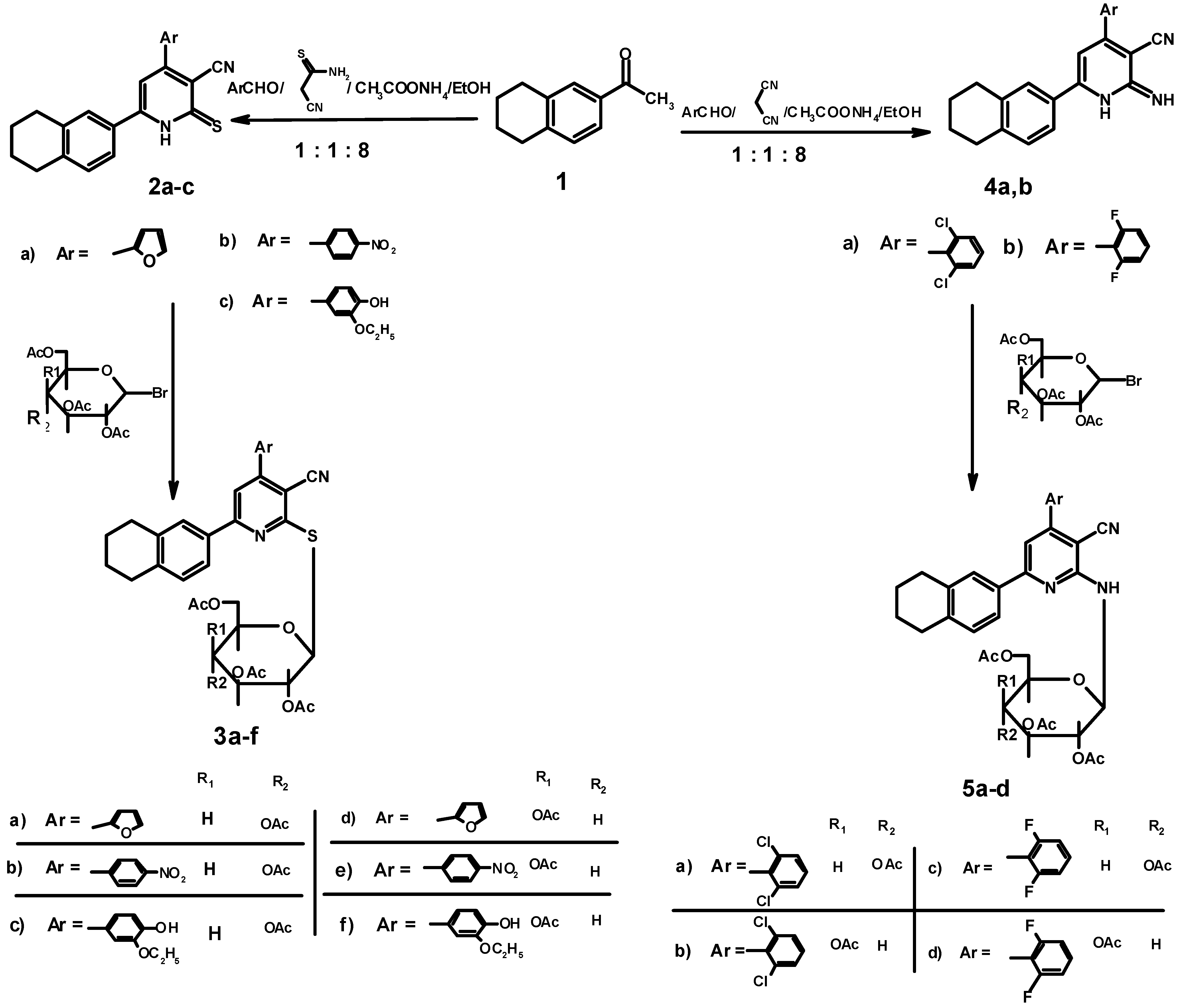
3.1.2. Synthesis of 4-Aryl-6-(1,2,3,4-tetrahydronaphthalen-6-yl)-2-thioxo-1,2-dihydropyridine-3-carbonitriles 2a–c
3.1.3. Synthesis of 4-Aryl-6-(1,2,3,4-tetrahydronaphthalen-6-yl)-2-(2',3',4',6'-tetra-O-acetyl-β-D-gluco and galactopyranosyl thio)pyridine-3-carbonitriles 3a–f
3.1.4. Synthesis of 4-Aryl-6-(1,2,3,4-tetrahydronaphthalen-6-yl)-2-(2',3',4',6'-tetra-O-acetyl-β-D-gluco and galactopyranosyl imino)pyridine-3-carbonitriles 5a–d
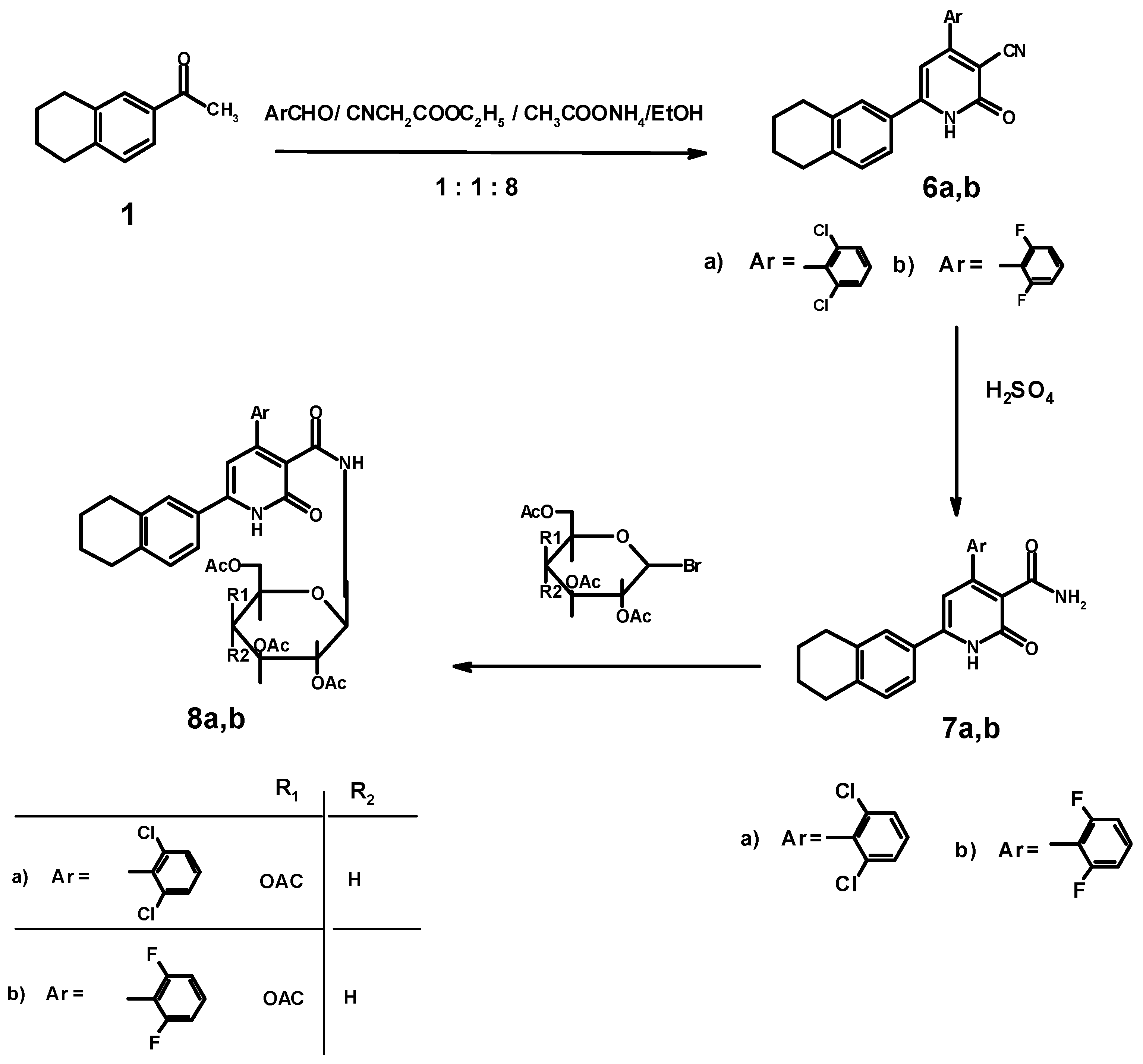
3.1.5. Synthesis of 4-Aryl-6-(1,2,3,4-tetrahydronaphthalen-6-yl)-2-oxo-1,2-dihydropyridine-3-carboxamides 7a,b
3.1.6. Synthesis of 4-Aryl-N-(2',3',4',6'-tetra-O-acetyl-β-D-galactopyranosyl)-6-(1,2,3,4-tetrahydro-naphthalen-6-yl)-2-oxo-1,2-dihydro pyridine-3-carboxamides 8a,b
3.2. Molecular Modelling
3.3. In Vitro Cytotoxic Screening
3.3.1. Tumor
3.3.2. Animals
3.3.3. In Vitro Test for Cytotoxic Effect
4. Conclusions
Acknowledgments
References and Notes
- Barker, M.; Clackers, M.; Copley, R.; Demaine, D.A.; Inglis, G.G.A.; Johnston, M.J.; Jones, H.T.; Haas, M.V.; House, D.; Loiseau, R.; et al. Dissociated nonsteroidal glucocorticoid receptor modulators; discovery of the agonist trigger in a tetrahydronaphthalene-benzoxazine series. J. Med. Chem. 2006, 49, 4216–4231. [Google Scholar]
- Amin, K.M.; El-Zahar, M.I.; Anwar, M.; Kamel, M.; Mohamad, M. Synthesis and anticancer activity of novel tetralin-6-yl pyridine and tetralin-6-yl pyrimidine derivatives. Acta Pol. Pharm. 2009, 66, 279–291. [Google Scholar]
- Dong, Y.Z.; Shi, Q.; Nakagawa-Goto, K.; Wu, P.C.; Bastow, K.F.; Morris-Natschke, S.L.; Lee, K.H. Antitumor agents 269. Non-aromatic ring-A neotanshinlactone analog, TNO, as a new class of potent antitumor agents. Bioorg. Med. Chem. Lett. 2009, 19, 6289–6292. [Google Scholar]
- Zaghary, W.A. Synthesis of some tetrahydronaphthalene derivatives of expected analgesic and anti-inflammatory activities. Bull. Fac. Pharm. Cairo Univ. 2007, 45, 391–400. [Google Scholar]
- Ates-Alagoz, Z.; Yildiz, S.; Buyukbingol, E. Antimicrobial activities of some tetrahydronaphthalene-benzimidazole derivatives. Chemotherapy 2007, 53, 110–113. [Google Scholar]
- Takami, M.; Tsukada, W. Effect of DP-1904, a thromboxane synthetase inhibitor, on antigen- and spasmogen-induced bronchoconstriction in rodents. Eur. J. Pharmacol. 1999, 366, 253–259. [Google Scholar] [CrossRef]
- Hussain, R.A.; Dickey, J.K.; Rosser, M.P.; Matson, J.A.; Kozlowski, M.R.; Brittain, R.J.; Webb, M.L.; Rose, P.M.; Fernandes, P.A. Novel class of non-peptidic endothelin antagonists isolated from the medicinal herb phyllanthus niruri. J. Nat. Prod. 1995, 58, 1515–1520. [Google Scholar]
- Chalina, E.G.; Chakarova, L. Synthesis, hypotensive and antiarrhythmic activities of 3-alkyl-1-(2-hydroxy-5,8-dimethoxy-1,2,3,4-tetrahydro-3-naphthalenyl)ureas or thioureas and their guanidine analogues. Eur. J. Med. Chem. 1998, 33, 975–983. [Google Scholar]
- Li, X.-N.; Lei, C.; Yang, L.-M.; Li, H.-M.; Huang, S.-X.; Du, X.; Pu, J.-X.; Xiao, W.-L.; Zheng, Y.-T.; Sun, H.-D. Three new arylnaphthalene lignans from Schisandra propinqua var sinensis. Fitoterapia 2012, 83, 249–252. [Google Scholar]
- Kamel, M.M.; Fathalla, O.A.; Rizk, M.Z.; Kassam, E.M.M.; Zaghary, W.A. Synthesis and the effect of some new 2(1H)pyridinethione on Biomphalaria Alexandrina snails enzymes. Egypt J. Pharm. Sci. 1994, 35, 355–364. [Google Scholar]
- Al-Abdullah, E.S. Synthesis and anticancer activity of some novel tetralin-6-yl-pyrazoline, 2-thioxopyrimidine, 2-oxopyridine, 2-thioxo-pyridine and 2-iminopyridine derivatives. Molecules 2011, 16, 3410–3419. [Google Scholar] [CrossRef]
- Reddy, T.R.; Reddy, G.R.; Reddy, L.S.; Jammula, S.; Lingappa, Y.; Kapavarapu, R.; Meda, C.L.T.; Parsa, K.V.L.; Pal, M. MontmorilloniteK-10 mediated green synthesis of cyano pyridines: Their evaluation as potential inhibitors of PDE4. Eur. J. Med. Chem. 2012, 48, 265–274. [Google Scholar]
- ztürk, G.; Erol, D.D.; Uzbay, T.; Aytemir, M.D. Synthesis of 4(1H)-pyridinone derivatives and investigation of analgesic and antiinflammatory activities. Farmaco 2001, 56, 251–256. [Google Scholar]
- Storck, P.; Aubertin, A.; Grierson, D.S. Tosylation/mesylation of 4-hydroxy-3-nitro-2-pyridinones as an activation step in the construction of dihydropyrido[3,4-b] benzo[f][1,4]thiazepin-1-one based anti-HIV agents. Tetrahedron Lett. 2005, 46, 2919–2922. [Google Scholar]
- Ebeid, M.Y.; Kamel, M.M.; Abdalla, N.; Kassem, E.M.M.; Abdou, W.A. New tetrahydronaphthyl pyridones of possible analgesic activity. Bull. Fac. Pharm. Cairo Univ. 1991, 29, 15–19. [Google Scholar]
- Cocco, M.T.; Congiu, C.; Onnis, V. New bis(pyridyl)methane derivatives from 4-hydroxy-2-pyridones: Synthesis and antitumoral activity. Eur. J. Med. Chem. 2003, 38, 37–47. [Google Scholar]
- Patel, N.B.; Patel, H.R. Characterization and pharmacological evaluation of new pyridine analogs. Arab. J. Chem. 2012, 5, 81–91. [Google Scholar]
- Roskoski, R. Scr kinase regulation by phosphorylation and dephosphorylation. Biochem. Biophys. Res. Commun. 2005, 331, 1–14. [Google Scholar]
- Weisheit, S.; Liebmann, C. Allosteric modulation by protein kinase Cε leads to modified responses of EGF receptor towards tyrosine inhibitors. Cell. Signal. 2012, 24, 422–434. [Google Scholar]
- Dickson, R.B.; Salomon, D.S.; Lippman, M.E. Tyrosine kinase receptor-nuclear protooncogene interactions in breast cancer. Cancer Treat Res. 1992, 61, 249–273. [Google Scholar]
- Arighi, E.; Borrello, M.G.; Sariola, H. RET tyrosine kinase signaling in development and cancer. Cytokine Growth Factor Rev. 2005, 16, 441–467. [Google Scholar]
- Kim, H.G.; Hwang, S.Y.; Aaronson, S.A.; Mandinova, A.; Lee, S.W. DDR1 receptor tyrosine kinase promotes prosurvival pathway through Notch1 activation. J. Biol. Chem. 2011, 286, 17672–17681. [Google Scholar]
- Elgemeie, G.H.; Zaghary, W.A.; Amin, K.M.; Nasr, T.M. A direct route to a new class of acrylamide thioglycosides. J. Carbohydr. Chem. 2008, 27, 373–378. [Google Scholar]
- Elgemeie, G.H.; Zaghary, W.A.; Amin, K.M.; Nasr, T.M. First synthesis of thiophene thioglycosides. J. Carbohydr. Chem. 2009, 28, 161–178. [Google Scholar]
- Sanner, M.F. Python: A programming language for software integration and development. J. Mol. Graph. Model. 1999, 17, 57–61. [Google Scholar]
- Trott, O.; Olson, A.J. Autodock vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar]
- Mclimans, W.F.; Davis, E.V.; Glover, F.L.; Rake, G.W. The submerged culture of mammalian cells: The spinner culture. J. Immunol. 1957, 79, 428–433. [Google Scholar]
- Protein Data Bank. Available online: http://www.pdb.org/pdb/ (accessed on 9 April 2012).
- PDBsum. Available online: http://www.ebi.ac.uk/pdbsum/ (accessed on 9 April 2012).
- Fahim, F.A.; Esmat, A.Y.; Mady, E.A.; Amin, M.A. Serum LDH and ALP isozyme activities in mice bearing solid Ehrlich carcinoma and/or treated with maximum tolerated dose (MTD) of aloin. Dis. Markers 1997, 13, 183–193. [Google Scholar]
- Sample Availability: Samples of the compounds 1, 2 and 4 are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Al-Mutairi, M.S.; Al-Abdullah, E.S.; Haiba, M.E.; Khedr, M.A.; Zaghary, W.A. Synthesis, Molecular Docking and Preliminary in-Vitro Cytotoxic Evaluation of Some Substituted Tetrahydro-naphthalene (2',3',4',6'-Tetra-O-Acetyl-β-D-Gluco/-Galactopyranosyl) Derivatives. Molecules 2012, 17, 4717-4732. https://doi.org/10.3390/molecules17044717
Al-Mutairi MS, Al-Abdullah ES, Haiba ME, Khedr MA, Zaghary WA. Synthesis, Molecular Docking and Preliminary in-Vitro Cytotoxic Evaluation of Some Substituted Tetrahydro-naphthalene (2',3',4',6'-Tetra-O-Acetyl-β-D-Gluco/-Galactopyranosyl) Derivatives. Molecules. 2012; 17(4):4717-4732. https://doi.org/10.3390/molecules17044717
Chicago/Turabian StyleAl-Mutairi, Maha S., Ebtehal S. Al-Abdullah, Mogedda E. Haiba, Mohammed A. Khedr, and Wafaa A. Zaghary. 2012. "Synthesis, Molecular Docking and Preliminary in-Vitro Cytotoxic Evaluation of Some Substituted Tetrahydro-naphthalene (2',3',4',6'-Tetra-O-Acetyl-β-D-Gluco/-Galactopyranosyl) Derivatives" Molecules 17, no. 4: 4717-4732. https://doi.org/10.3390/molecules17044717