Pharmaceutics 2023, 15(7), 1864; https://doi.org/10.3390/pharmaceutics15071864 - 1 Jul 2023
Cited by 3 | Viewed by 1814
Abstract
Amphibian secretions have been extensively investigated for the production of bioactive molecules. Salamandrin-I is an antioxidant peptide, isolated from the skin secretion of the fire salamander, that has induced no toxicity in microglia or erythrocytes. Importantly, the administration of antioxidants may constitute an
[...] Read more.
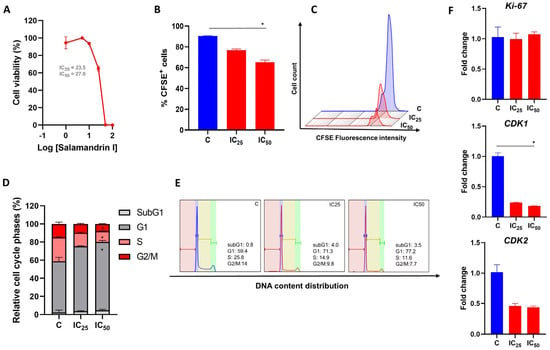
Amphibian secretions have been extensively investigated for the production of bioactive molecules. Salamandrin-I is an antioxidant peptide, isolated from the skin secretion of the fire salamander, that has induced no toxicity in microglia or erythrocytes. Importantly, the administration of antioxidants may constitute an adequate therapeutic approach to cancer treatment. Here, with the purpose of better characterizing the therapeutic potential of salamandrin-I, we investigated whether this antioxidant peptide also exerts anticancer activity, using the human leukemia cell line HL-60 as a cancer model. Salamandrin-I treatment induced a significant reduction in HL-60 proliferation, which was accompanied by cell cycle arrest. Furthermore, the peptide-induced cell death showed a significant increase in the LDH release in HL-60 cells. The cellular toxicity exerted by salamandrin-I is possibly related to pyroptosis, since the HL-60 cells showed loss of mitochondrial membrane potential and hyperexpression of inflammasome components following the peptide treatment. This is the first demonstration of the anticancer potential of the salamandrin-I peptide. Such results are important, as they offer relevant insights into the field of cancer therapy and allow the design of future bioactive molecules using salamandrin-I as a template.
Full article
(This article belongs to the Special Issue Peptides and Peptide Mimetics: Potential Tools for Therapy)
►
Show Figures