1. Introduction
Due to concerns about environmental, long-term economic and national security, there has been increasing interest in renewable and domestic sources of fuels to replace fossil fuels in recent decades. [
1]. Bioethanol, produced from renewable materials, is regarded as an alternative to gasoline. There are multiple raw materials to produce bioethanol; one of the most widely adopted is sugar- or starch- based material, such as corn. Bioethanol produced from corn is called first generation biofuel. It has been commercialized in several places and is considered quite efficient. However, a problem arose because of land use and competition with food crops, the so-called food versus fuel debate [
2]. Bioethanol can also be produced from lignocellulosic biomass, which is known as second generation biofuel [
3]. In general, four major processes are involved in converting lignocellulosic biomass to bioethanol: pretreatment, hydrolysis, fermentation, and ethanol recovery [
4]. Among the four steps, pretreatment is critical because of the difficulties in removing the lignin-carbohydrate complex (LCC) structure in lignocellulosic biomass. With the assistance of pretreatment, the LCC structure could be removed, and the exposed cellulose could be broken down into monosaccharides, then the resulting glucose can be fermented into ethanol [
1].
Numerous efforts have been invested in exploring various pretreatment methods on various biomass to enhance enzymatic digestibility. Additionally, various pretreatment reagents have been studied, such as carbon dioxide, dilute acid, hot water, ammonia and alkaline. Based on the results of extensive research, each different reagent exhibited its unique characteristics. Several reagents are compared as following.
Carbon dioxide (CO
2) has many advantages, as it is environmentally friendly, inexpensive, and easy to recover after use. The pretreatment method based on CO
2 is supercritical carbon dioxide (SC-CO
2). It has been applied to a few lignocellulosic biomass, such as aspen and south yellow pine [
5], wheat straw [
6], guayule [
7], and corn stover [
8]. As for corn stover, the maximum glucose yield obtained under 3500 psi and 150 °C was 30 g/100 g dry corn stover [
8]. However, the need for high-pressure equipment by using the SC-CO
2 pretreatment method may result in high capital cost; besides, the low efficiency of this treatment may be a barrier as well to large-scale production [
5].
Hot water has also been used as a reagent in pretreatment studies. Hot water has been studied in materials like aspen [
9], soybean straw [
10], corn stover [
9,
10], alfalfa [
11], and cattails [
12]. As a convenient pretreatment method, liquid hot water is effective for soybean straw with the combination of fungal degradation pretreatment, but the combination of these two pretreatment methods is not efficient for corn stover, when compared with fungal degradation pretreatment alone [
10].
Another reagent, ammonia, is also broadly explored in this field. Pretreatment methods of ammonia have attracted much attention due to its effectiveness in delignification. For example, ammonia fiber explosion [
13,
14,
15,
16], ammonia fiber expansion [
17,
18,
19,
20], and aqueous ammonia soaking [
21,
22,
23] have been developed. In addition, the improvement in glucose yield is clearly observed. However, water consumption, environmental concerns, and high cost are problematic for ammonia-based pretreatment methods.
Yoo et al. [
24] developed the low moisture anhydrous ammonia (LMAA) pretreatment method to eliminate the washing step and reduce capital costs in the ammonia-based pretreatment method. In their study, corn stover pretreated with 3% glucan loading at 80 °C for 84 h resulted in the highest ethanol yield, that is: 89% of theoretical ethanol yield. However, the reactor used in the research conducted by Yoo et al. [
24] was a 2.9-inch (8.1 cm) internal diameter with a 6.5-inch (18.5 cm) length (690 mL internal volume). The small sealed reactor may not be capable of providing optimal conditions for bioethanol production at industrial scales. Yang and Rosentrater [
25] and Cayetano and Kim [
26] have expanded on this initial study. Yang and Rosentrater [
25] investigated the effectiveness of LMAA as a method to both pretreat and preserve corn stover prior to fermentation, and found that LMAA is beneficial to preserving sugar yields during storage, with sealed containers being more effective at ammonia treatment.
The main objective of this study was to investigate the LMAA pretreatment process with a larger-scale reactor; four pretreatment conditions (moisture content, particle size, pretreatment temperature, and pretreatment time) were considered in this study. Furthermore, optimal conditions for higher ethanol yield were explored.
2. Materials and Methods
2.1. Biomass
In this study, freshly-harvested, air-dried corn stover was collected from central Iowa in 2012 and stored at ambient temperature. Prior to pretreatment, the corn stover was ground and sieved into three size fractions (<0.09, 0.09–2.0, and >2.0 mm). Then, the sieved corn stover was stored at room temperature (~21 °C) until use.
2.2. Equipment
The reactor (
Figure 1), which was purchased from Pall Corporation, Ann Arbor, MI, USA, was used in the ammoniation process. Compared to Yoo’s [
24] study, this sealed reactor was about 4.35 times larger (the internal capacity is 3 L). It is anticipated that the potential errors caused by different ammonia loadings and reaction times could be eliminated by the use of a larger reactor. High Performance Liquid Chromatography (HPLC) with a Bio-Rad Aminex HPX-87P column (Aminex HPX-87P, Bio-Rad Laboratories, Hercules, CA, USA) and a refractive index detector (Varian 356-LC, Varian, Inc., Palo Alto, CA, USA) were used to measure sugar contents. Acid soluble lignin (ASL) content was determined by UV-Visible spectrophotometer (UV-2100 Spectrophotometer, Unico, United Products & Instruments, Inc., Dayton, NY, USA).
2.3. Enzymes
In this study, GC 220 cellulase, purchased from Genencor International, Inc. (Rochester, NY, USA), was a mixture of endogluconases and cellobiohydrolases. The cellulase activity was expressed in filter paper units (FPU); the average activity of GC 220 was determined to be 45 FPU/mL. The β-glucosidase enzyme (Novozyme 188), provided by Sigma-Aldrich, Inc. (St. Louis, MO, USA), was used to convert cellobiose to glucose. The activity of Novozyme 188 was 750 cellobiase units (CBU)/mL.
2.4. LMAA Pretreatment Process
The original moisture content was measured before ammoniation, then certain amounts of water were added to the corn stover in order to achieve the target moisture content (20, 50, and 80 w.b.%). Moisturized corn stover was equilibrated for 24 h afterwards.
The moisturized corn stover was placed in the sealed reactor, and ammonia gas was introduced. On top of the reactor, a pressure gauge and a temperature gauge were equipped to monitor the pressure and temperature change during the whole process. However, temperature change was not controlled during this study. The pressure of the anhydrous ammonia was maintained at 0.1 g NH3/g DM biomass for 30 min in order to achieve a complete reaction. After the ammoniation process, the reactor was cooled down for 5 min and the lid was removed in the fume hood. Then the ammoniated corn stover was transferred into several glass bottles (250 mL) with screw caps. A pipe was connected between the top of the reactor and the fume hood to ventilate surplus ammonia.
The bottles packed with ammoniated corn stover were placed in various heating ovens at varying pretreatment temperatures (20, 75, and 130 °C) for 24, 96, and 168 h. As soon as the pretreatment process was complete, the lid of the glass bottles was removed in the fume hood and surplus ammonia was evaporated for 12 h before compositional analysis.
2.5. Experimental Design
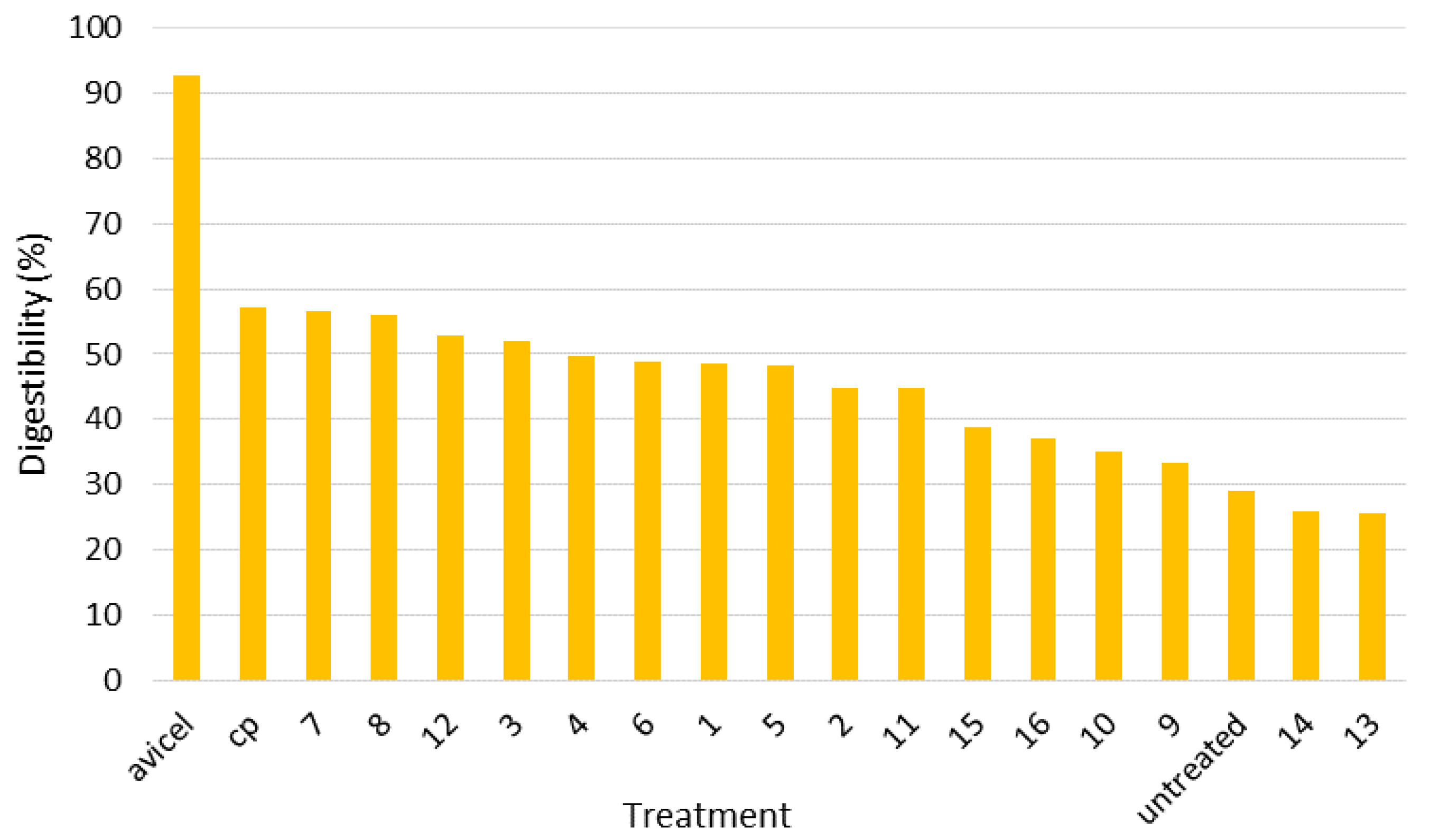
In this study, four independent variables that may have influence on the reaction severity were investigated. Biomass moisture contents were 20, 50 and 80 wet basis (w.b.) %; the pretreatment times were 24, 96, and 168 h; the pretreatment temperatures were 20, 75, and 130 °C; and the particle sizes were <0.9, 0.9–2.0 and >2.0 mm, respectively. By combining different levels of these four independent variables, 17 treatments were designed in this study, i.e., 2 × 2 × 2 × 2 + 1 center point. As dependent variables, moisture content, lignin, glucan, xylan, galactan, arabinan, mannan and ash content were measured and compared in the experiment. The experimental design for this study is shown in
Table 1.
2.6. Compositional Analysis
Carbohydrates and lignin (both acid-soluble lignin and acid-insoluble lignin) contents were determined by NREL LAP [
27]. Each sample was analyzed in duplicate. The glucan and xylan content in the corn stover were analyzed by HPLC, following the NREL standards. Acid soluble lignin was measured by UV-Visible Spectrophotometer. Moisture content was determined by an oven drying method [
27].
2.7. Enzymatic Digestibility Test
The enzymatic digestibility test was done in duplicate under conditions of pH 4.8 (0.1 M sodium citrate buffer) with 40 mg/L tetracycline and 30 mg/L cyclohexamide in 250 mL Erlenmeyer flasks according to NREL LAP [
28]. The initial glucan concentration was 1% (
w/
v). Cellulase enzyme (GC 220) loading was 15 FPU/g of glucan, and β-glucosidase enzyme (Novozyme 188) loading was equal to 30 CBU/g of glucan. Flasks were incubated at 50 ± 1 °C and 150 rpm in an incubator shaker (Excella E24 Incubator Shaker Series, New Brunswick Scientific, Edison, NJ, USA). Time for enzymatic digestibility test ranged from 0 to 168 h for sugar analysis.
Total glucose detected from HPLC was used to calculate the glucan digestibility following Equation (1) below. The conversion factor for glucose to equivalent glucan was 0.9 based on the calculation. The quantification of glucose in HPLC is based on the separation of the solvent into its constituent parts due to the different affinities of different molecules for the mobile phase and stationary phase. All the statistical results were anaylzed by SAS 9.4 (SAS Institute Inc., Cary, NC, USA).