Influence of the Culinary Treatment on the Quality of Lactarius deliciosus
Abstract
:1. Introductions
2. Experimental Section
2.1. Material
- Product prepared with unblanched mushroom caps,
- Product prepared with blanched mushroom caps,
- Product prepared with unblanched mushroom caps with the following ingredients (quantity per 1 kg of mushrooms): 100 g of onion, 10 g of garlic, 20 g of ground black pepper, 1 g of allspice grains, and 1 g of bay leaf.
2.2. Analysis of Proximate Composition
2.3. Analysis of Antioxidant Properties
2.4. Color Analysis
2.5. Texture Analysis
2.6. Sensory Analysis
2.7. Microbiological Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Proximate Composition
| Component | Type of product | |||
|---|---|---|---|---|
| Fresh mushrooms | I Fried unblanched mushrooms | II Fried blanched mushrooms | III Fried unblanched mushrooms with onions and spices | |
| Moisture | 90.68 ± 0.73 a | 81.59 ± 1.24 b | 80.18 ± 0.95 b | 81.62 ± 1.01 b |
| Proteins | 2.35 ± 0.06 a | 2.38 ± 0.17 b | 2.40 ± 0.11 b | 2.38 ± 0.09 b |
| Fat | 1.04 ± 0.01 a | 9.25 ± 0.02 b | 9.21 ± 0.13 b | 9.28 ± 0.11 b |
| Total carbohydrates | 5.17 ± 0.09 a | 5.84 ± 0.11 b | 6.98 ± 0.14 b | 5.70 ± 0.07 b |
| Ash | 0.78 ± 0.99 a | 0.99 ± 0.04 b | 1.04 ± 0.01 c | 1.21 ± 0.05 d |
| Energy (kcal) | 38 ± 0 a | 114 ± 0 b | 119 ± 1 b | 114 ± 0 b |
3.2. Antioxidant Properties
| Antioxidant properties | Storage | Type of product | |||
|---|---|---|---|---|---|
| Fresh mushrooms | I Fried unblanched mushrooms | II Fried blanched mushrooms | III Fried unblanched mushrooms with onions and spices | ||
| Total polyphenol content (mg of (+)-catechnin) | 0 | 0.49 ± 0.01 a | 0.50 ± 0.03 a | 0.39 ± 0.03 b | 0.50 ± 0.00 a |
| 48 h/20 °C | 0.44 ± 0.02 c | 0.36 ± 0.01 e | 0.53 ± 0.03 f | ||
| 48 h/4 °C | 0.47 ± 0.01 d | 0.36 ± 0.01 e | 0.50 ± 0.03 a | ||
| 96 h/4 °C | 0.46 ± 0.03 c | 0.36 ± 0.01 e | 0.52 ± 0.02 a,f | ||
| Total flavonoid content (mg of (+)-catechin) | 0 | 0.34 ± 0.02 a | 0.36 ± 0.02 b | 0.29 ± 0.02 c | 0.37 ± 0.03 b,d |
| 48 h/20 °C | 0.29 ± 0.02 c | 0.34 ± 0.01 a | 0.31 ± 0.01 e | ||
| 48 h/4 °C | 0.38 ± 0.02 d | 0.31 ± 0.02 e | 0.34 ± 0.02 a | ||
| 96 h/4 °C | 0.34 ± 0.02 a | 0.36 ± 0.02 b | 0.33 ± 0.01 a | ||
| Antioxidant activity against ABTS (μmol TE) | 0 | 36.3 ± 0.9 a | 28.9 ± 0.3 b | 15.1 ± 0.4 e | 30.4 ± 0.4 d |
| 48 h/20 °C | 26.9 ± 0.7 c | 9.2 ± 0.5 f | 32.3 ± 0.7 i | ||
| 48 h/4 °C | 28.3 ± 0.9 b | 11.5 ± 0.4 g | 27.5 ± 0.5 c | ||
| 96 h/4 °C | 30.1 ± 0.1 d | 14.1 ± 0.3 h | 32.8 ± 0.9 i | ||
| Antioxidant activity against DPPH (μmol TE) | 0 | 20.1 ± 0.9 a | 12.6 ± 0.9 b | 12.1 ± 0.5 b | 14.5 ± 0.6 c |
| 48 h/20 °C | 14.0 ± 0.7 c | 11.2 ± 0.9 d | 15.4 ± 0.8 f | ||
| 48 h/4 °C | 14.0 ± 0.8 c | 11.5 ± 0.6 d | 13.7 ± 0.6 c | ||
| 96 h/4 °C | 13.2 ± 0.9 b | 9.8 ± 0.6 e | 14.2 ± 0.9 c | ||
| Ferric reducing/antioxidant potential—FRAP (μmol Fe2+) | 0 | 32.4 ± 1.7 a | 22.0 ± 0.5 b | 22.1 ± 0.7 b | 23.5 ± 1.0 b |
| 48 h/20 °C | 22.2 ± 0.5 b | 23.8 ± 2.1 b,c | 25.1 ± 1.3 c | ||
| 48 h/4 °C | 23.8 ± 1.2 b,c | 22.0 ± 1.9 b | 23.4 ± 0.8 b,c | ||
| 96 h/4 °C | 25.1 ± 1.4 c | 21.2 ± 1.1 b,d | 24.0 ± 1.0 b,c | ||
3.3. Color Analysis
| Parameter | Storage | Type of product | |||
|---|---|---|---|---|---|
| Fresh mushrooms | I Fried unblanched mushrooms | II Fried blanched mushrooms | III Fried unblanched mushrooms with onions and spices | ||
| L* | 0 | 40.65 ± 0.47 a | 45.84 ± 0.68 b | 46.83 ± 0.79 d | 43.40 ± 0.23 c |
| 48 h/20 °C | 43.40 ± 0.37 c | 46.25 ± 0.44 b,d | 42.93 ± 0.39 c | ||
| 96 h/4 °C | 42.86 ± 1.24 c | 46.21 ± 0.79 b,d | 43.08 ± 0.25 c | ||
| a* | 0 | 13.62 ± 0.44 a | 11.72 ± 0.52 b | 13.06 ± 0.64 d | 11.25 ± 1.14 b |
| 48 h/20 °C | 9.90 ± 0.23 c | 10.82 ± 0.32 e | 10.96 ± 0.18 e | ||
| 96 h/4 °C | 10.18 ± 0.39 c | 9.16 ± 0.11 f | 10.04 ± 0.29 c | ||
| b* | 0 | 28.68 ± 0.85 a | 28.35 ± 1.26 a | 29.73 ± 0.20 a,c | 27.29 ± 2.21 a,d |
| 48 h/20 °C | 25.88 ± 0.48 b | 27.86 ± 0.34 a,d | 27.64 ± 0.53 a,d | ||
| 96 h/4 °C | 24.28 ± 1.28 b | 24.85 ± 0.32 b | 25.41 ± 0.60 b | ||
| C* | 0 | 33.76 ± 0.85 a | 30.68 ± 1.36 b | 32.48 ± 0.41 a | 29.52 ± 2.46 d |
| 48 h/20 °C | 27.71 ± 0.53 c | 29.89 ± 0.42 d | 29.73 ± 0.53 d | ||
| 96 h/4 °C | 26.33 ± 1.32 c | 26.48 ± 0.30 c | 27.32 ± 0.65 c | ||
| h* | 0 | 64.59 ± 0.82 a | 67.53 ± 0.11 b | 66.29 ± 0.95 d | 67.61 ± 0.72 b |
| 48 h/20 °C | 69.06 ± 0.14 c | 68.77 ± 0.40 c | 68.38 ± 0.37 c | ||
| 96 h/4 °C | 67.25 ± 0.53 b | 69.75 ± 0.31 c,e | 68.45 ± 0.25 c | ||
3.4. Texture Analysis
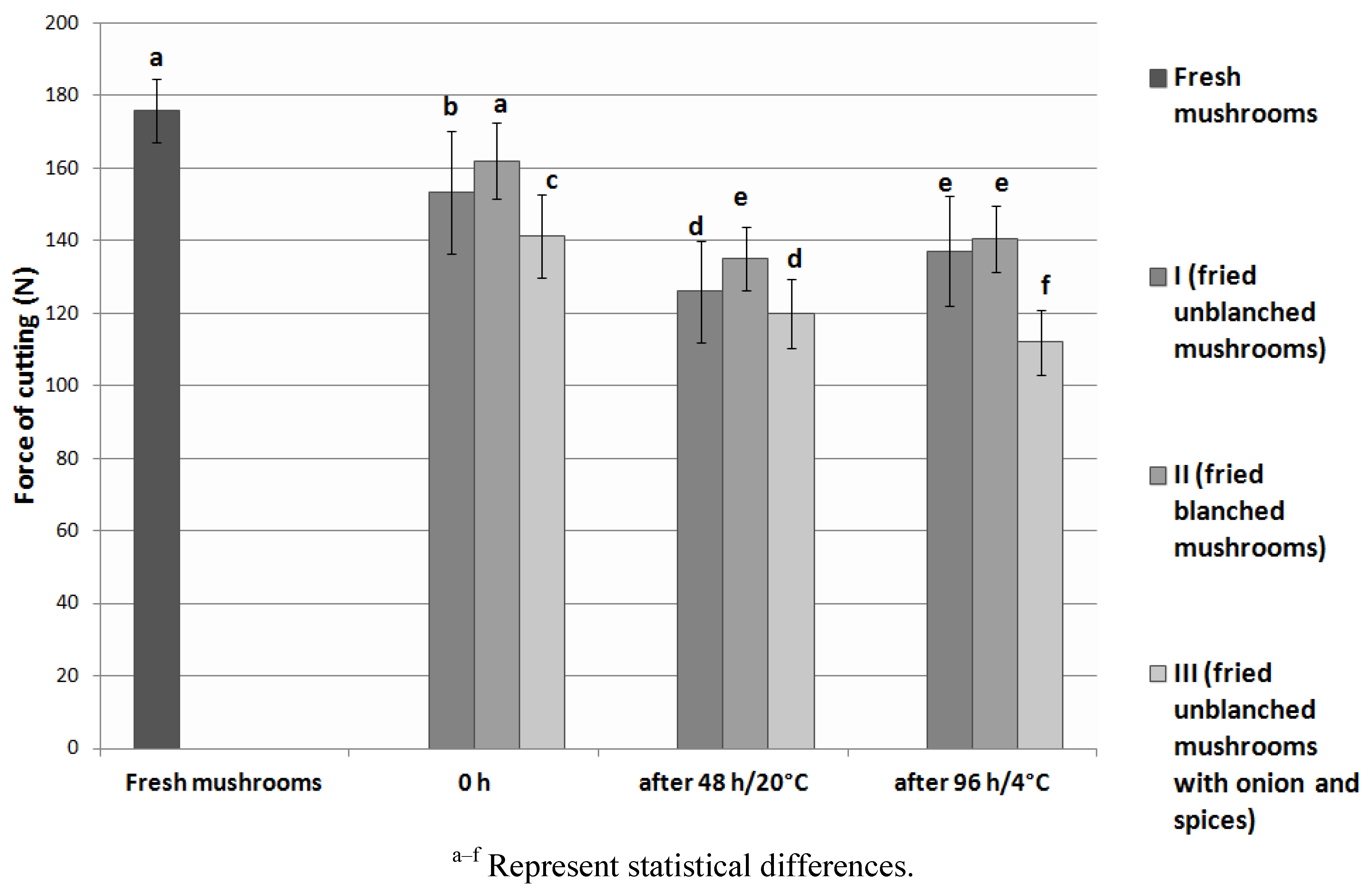
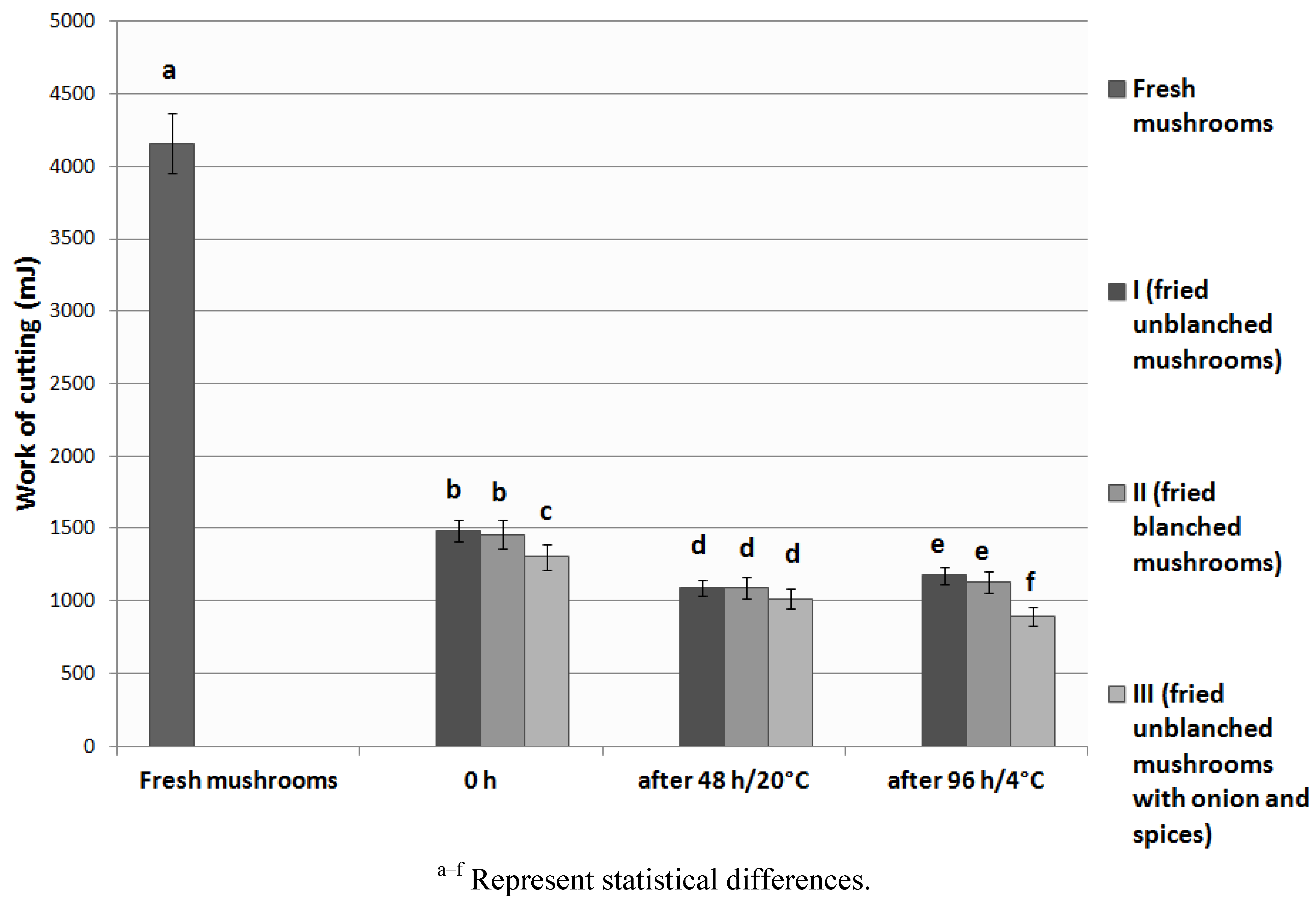
3.5. Sensory Analysis
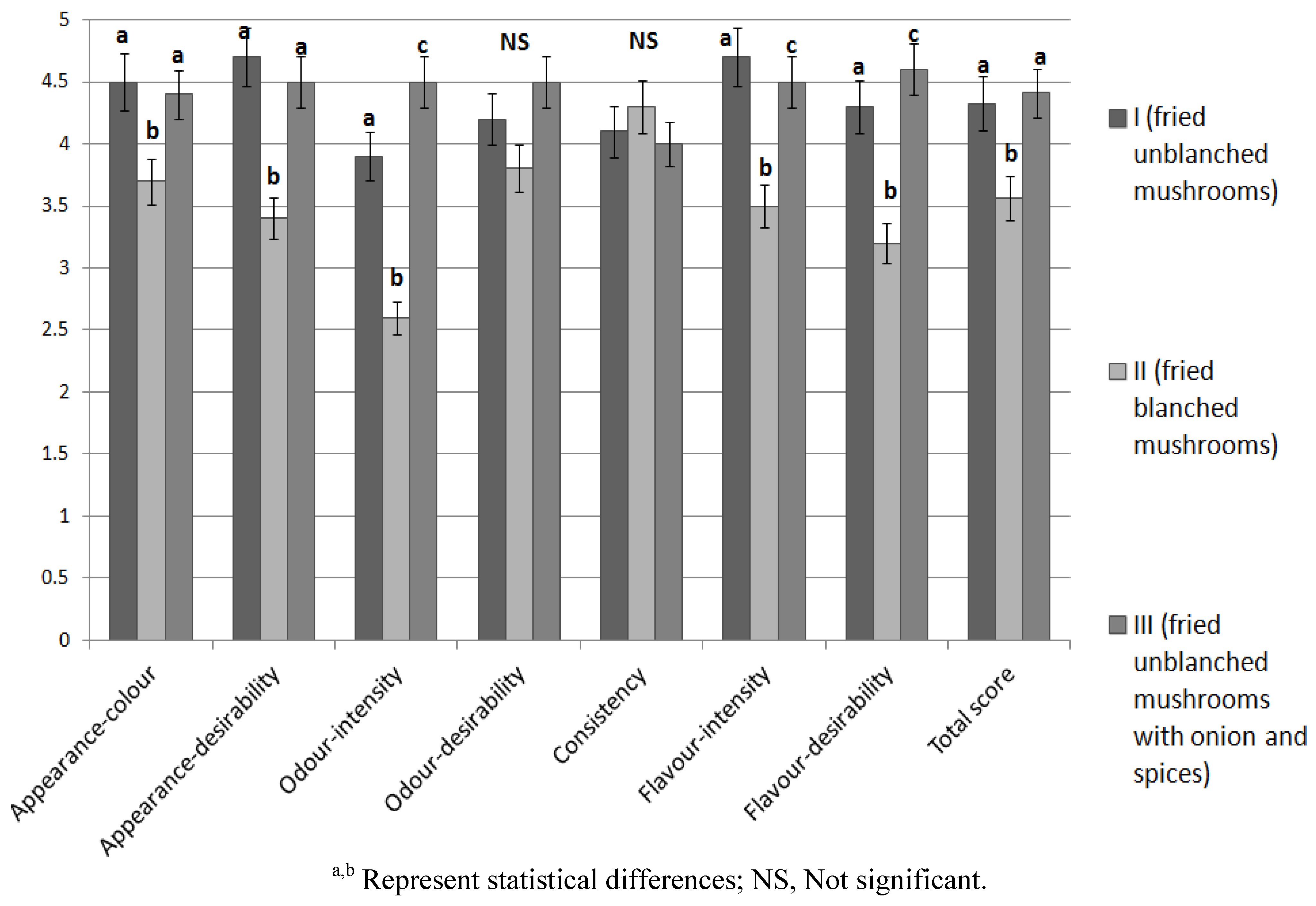
3.6. Microbiological Analysis
| Type of product | TVC | Yeast and molds | Lactic acid bacteria | E.coli | Bacillus |
|---|---|---|---|---|---|
| After 48 h in 20 °C | |||||
| I | +++ | Ø | ++ | Ø | Ø |
| II | +++ | Ø | ++ | Ø | Ø |
| III | +++ | Ø | Ø | Ø | Ø |
| After 96 h in 4 °C | |||||
| I | Ø | Ø | Ø | Ø | Ø |
| II | + | Ø | + | Ø | Ø |
| III | Ø | Ø | Ø | Ø | Ø |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Barros, L.; Baptista, P.; Correia, D.M.; Casal, S.; Oliveira, B.; Ferreira, I.C.F.R. Fatty acid and sugar compositions, and nutritional value of five wild edible mushrooms from Northeast Portugal. Food Chem. 2007, 105, 140–145. [Google Scholar]
- Kalač, P. Chemial composition and nutritional value of European species of wild growing mushrooms: A review. Food Chem. 2009, 113, 9–16. [Google Scholar] [CrossRef]
- Heleno, S.A.; Barros, L.; Sousa, M.J.; Martins, A.; Ferreira, I.C.F.R. Tocopherols composition of Portuguese wild mushrooms with antioxidant capacity. Food Chem. 2010, 119, 1443–1450. [Google Scholar] [CrossRef]
- Palacios, I.; Lozano, M.; Moro, C.; D’arrigo, M.; Rostragno, M.A.; Martinez, J.A.; Garcia-Lafuente, A.; Guillamon, E.; Villares, A. Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chem. 2011, 128, 674–678. [Google Scholar] [CrossRef]
- Zhou, Z.Y.; Tan, J.W.; Liu, J.K. Two new polyols and a new phenylpropanoid glycoside from the basidiomycete Lactarius deliciosus. Fitoterapia 2011, 82, 1309–1312. [Google Scholar] [CrossRef]
- Guillamon, E.; Garcia-Lafunete, A.; Lozano, M.; D’arrigo, M.; Rostagno, M.; Villares, A.; Martinez, J.A. Edible mushrooms: Role in the prevention of cardiovascular diseases. Fitoterapia 2010, 81, 715–723. [Google Scholar] [CrossRef]
- Bognar, A. Comparative study on frying to other cooking techniques influence on the nutritive value. Grasas Aceit. 1998, 49, 250–260. [Google Scholar] [CrossRef]
- Faller, A.L.K.; Fiahlo, E. The antioxidant capacity and polyphenol content of organic and conventional retail vegetables after domestic cooking. Food Res. Int. 2009, 42, 210–215. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Mylona, A.; Chiou, A.; Ioannou, M.S.; Anrikopoulos, N.K. Retention and distribution of natural antioxidants (α-tocopherol, polyphenols and terpenic acids) after shallow frying of vegetables in virgin olive oil. LWT 2007, 40, 1008–1017. [Google Scholar] [CrossRef]
- AOAC (Association of Official Analytical Chemists), Official Methods of Analysis of the Association of Official Analytical Chemists, 16th ed.; AOAC: Arlington, VA, USA, 1995.
- Manzi, P.; Aguzzi, A.; Pizzoferrato, L. Nutritional value of mushrooms widely consumed in Italy. Food Chem. 2001, 73, 321–325. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, C. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods in enzymology. Oxid. Aantioxid. 1999, 299, 152–178. [Google Scholar]
- Ardestani, A.; Yzadanparast, R. Antioxidant and free radical scavenging potential of Achillea santolina extacts. Food Chem. 2007, 104, 1041–1047. [Google Scholar]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Pekkarinen, S.S.; Heinonen, I.M.; Hopia, A.I. Flavonoids quercetin, myricetin, kaemferol and (+)-catechin and antioxidants in methyl linoleate. J. Sci. Food Agric. 1999, 79, 499–506. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- PN-90/A-75052.08. Processed Fruits, Vegetables and Meats. Methods of Microbiological Analysis. Enumeration of Yeasts And Molds (in polish); Polish Normalization Committee: Warsaw, Poland, 2008.
- PN-90/A-75052.07. Processed Fruits, Vegetables and Meats. Methods of Microbiological Analysis. Enumeration of Lactic Acid Bacteria (in polish); Polish Normalization Committee: Warsaw, Poland, 1990.
- PN-EN ISO 4833:2004/Ap1:2005. Microbiology of Foods And Fodders. Horizontal Method of Enumeration of Microorganisms. Plate Count at 30 °C (in Polish); Polish Normalization Committee: Warsaw, Poland, 2005.
- Manzi, P.; Marconi, S.; Aguzzi, A.; Pizzoferrato, L. Commercial mushrooms: Nutritional quality and effect of cooking. Food Chem. 2004, 84, 201–206. [Google Scholar] [CrossRef]
- Vaz, J.A.; Barros, L.; Martins, A.; Morais, J.S.; Vasconcelos, M.H.; Ferreira, I.C.F.R. Phenolic profile of seventeen Portuguese wild mushrooms. LWT 2011, 44, 343–346. [Google Scholar] [CrossRef]
- Barros, L.; Duenas, M.; Ferreira, I.C.F.R.; Baptista, P.; Santos-Buelga, C. Phenolic acids determination by HPLC-DAD-ESI/MS in sixteen different Portuguese wild mushrooms species. Food Chem. Toxicol. 2009, 47, 1076–1079. [Google Scholar] [CrossRef]
- Ferreira, I.C.F.R.; Baptista, P.; Vilas-Boas, M.; Barros, L. Free-radical scavenging capacity and reducing power of wild edible mushrooms from northeast Portugal: Individual cap and stipe activity. Food Chem. 2007, 100, 1511–1516. [Google Scholar] [CrossRef]
- Orhan, I.; Ustun, O. Determination of total phenol content, antioxidant activity and acetylcholinesterase inhibition in selected mushrooms from Turkey. J. Food Comp. Anal. 2011, 24, 386–390. [Google Scholar]
- Mazzeo, T.; N’dri, D.; Chiavaro, E.; Visconti, A.; Fogliano, V.; Pellegrini, N. Effect of two cooking procedures on phytochemical compounds, total antioxidant capacity and colour of selected frozen vegetables. Food Chem. 2011, 128, 627–633. [Google Scholar] [CrossRef]
- Turkmen, N.; Sari, F.; Velioglu, Y.S. The effect of cooking methods on total phenolics and antioxidant activity of selected green vegetables. Food Chem. 2005, 93, 713–718. [Google Scholar] [CrossRef]
- Chuah, A.M.; Lee, Y.C.; Yamaguci, T.; Takamura, H.; Yin, L.J.; Matoba, T. Effect of cooking on the antioxidant properties of coloured peppers. Food Chem. 2008, 111, 20–28. [Google Scholar] [CrossRef]
- Yoshida, Y.; Niki, E.; Noguchi, N. Comparative study on the action of tocopherols and tocotrienols as antioxidant: chemical and physical effects. Chem. Phys. Lipids 2003, 123, 63–75. [Google Scholar] [CrossRef]
- Murcia, M.A.; Jimine, A.M.; Martinez-Tome, M. Vegetables antioxidant losses during industrial processing and refrigerated storage. Food Res. Int. 2009, 42, 1046–1052. [Google Scholar] [CrossRef]
- Zivanovic, S.; Buescher, R.; Kim, S.K. Mushroom texture, cell wall composition, color, and ultrastructure as affected by pH and temperature. J. Food Sci. 2003, 68, 1860–1865. [Google Scholar] [CrossRef]
- Hoffman, T. Characterization of the most intense coloured compounds from Maillard reactions of pentoses by application of colour dilution analysis. Carbohydr. Res. 1998, 313, 203–213. [Google Scholar] [CrossRef]
- Serpen, A.; Gokmen, V. Evaluation of the Maillard reaction in potato crisps by acrylamide, antioxidant capacity and color. J. Food Comp. Anal. 2009, 22, 589–595. [Google Scholar] [CrossRef]
- Jaworska, G.; Bernaś, E.; Biernacka, A.; Maciejaszek, I. Comparison of the texture of fresh and preserved Agaricus bisporus and Boletus edulis mushrooms. Int. J. Food Sci. Technol. 2010, 45, 1659–1665. [Google Scholar]
- Ko, W.C.; Liu, W.C.; Tsang, Y.T.; Hsieh, C.W. Kinetics of winter mushrooms (Flammulina velutipes) microstructure and quality changes during thermal processing. J. Food Eng. 2007, 81, 587–598. [Google Scholar] [CrossRef]
- Zivanovic, S.; Buescher, R. Changes in Mushroom texture and cell wall composition affected by thermal processing. J. Food Sci. 2004, 69, 44–49. [Google Scholar]
- Biekman, E.S.A.; Kroese-Hoedeman, H.I.; Schijvens, E.P.H.M. Loss of solutes during blanching of mushrooms (Agaricus bisporus) as a result of shrinkage and extraction. J. Food Eng. 1996, 28, 139–152. [Google Scholar] [CrossRef]
- Venturini, M.E.; Reyes, J.E.; Rivera, C.S.; Oria, R.; Blanco, D. Microbiological quality and safety of fresh cultivated and wild mushrooms commercialized in Spain. Food Microbiol. 2011, 28, 1492–1498. [Google Scholar] [CrossRef]
- Shan, B.; Cai, Y.Z.; Brooks, J.D.; Corke, H. The in vitro antibacterial activity of dietary spice and medicinal herb extracts. Int. J. Food Microbiol. 2007, 117, 112–119. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pogoń, K.; Jaworska, G.; Duda-Chodak, A.; Maciejaszek, I. Influence of the Culinary Treatment on the Quality of Lactarius deliciosus. Foods 2013, 2, 238-253. https://doi.org/10.3390/foods2020238
Pogoń K, Jaworska G, Duda-Chodak A, Maciejaszek I. Influence of the Culinary Treatment on the Quality of Lactarius deliciosus. Foods. 2013; 2(2):238-253. https://doi.org/10.3390/foods2020238
Chicago/Turabian StylePogoń, Krystyna, Grażyna Jaworska, Aleksandra Duda-Chodak, and Ireneusz Maciejaszek. 2013. "Influence of the Culinary Treatment on the Quality of Lactarius deliciosus" Foods 2, no. 2: 238-253. https://doi.org/10.3390/foods2020238
APA StylePogoń, K., Jaworska, G., Duda-Chodak, A., & Maciejaszek, I. (2013). Influence of the Culinary Treatment on the Quality of Lactarius deliciosus. Foods, 2(2), 238-253. https://doi.org/10.3390/foods2020238





