2.2. Crystal Chemistry of the Type-II Clathrates
The unit cells of the three cubic type-II clathrates (space group
Fd3m, see
Table 1) decrease at 200 K from
a = 14.9354(5) Å for Cs
8Na
16Ga
22.7Si
113.3(1) (
1), to
a = 14.8822(10) Å for Rb
8.4Na
15.6(1)Ga
19.6Si
116.4(1) (
2), to
a = 14.8027(10) Å for Rb
8Na
16Zn
8.4Si
127.6(1) (
3). This reduction is in agreement with the observed decreased amount of Ga in
1 vs. 3; the relatively larger difference in the unit cell volumes of
2 vs. 3 can be attribute to the larger size of the atoms Ga compared to Zn (
rGa ≈ 1.25 Å,
rZn ≈ 1.21 Å,
rSi ≈ 1.17 Å, according to Pauling [
25]). The guest atoms must also have an effect, but it appears to be more subtle (the difference in the metallic radii for the alkali metal Cs and Rb is quite large,
rCs ≈ 2.35 Å,
rRb ≈ 2.16 Å according to Pauling [
25]). As both Zn and Ga are larger than Si, any substitutions to the framework result in enlargement of the unit cell, clearly seen by comparing our data with the data for the known ternary type-II clathrates Cs
8Na
16Si
136 [
15] and Rb
8Na
16Si
136 [
6] have with
a = 14.7560(4) Å and
a = 14.7400(4) Å, respectively.
The compositions of the previously reported type-II compounds Cs
8Na
16Ga
21Si
115 and Rb
7.3Na
16Ga
20Si
116 [
17] and the herein reported
1 and
2 are worth considering too. Note that from the structure refinements, the Ga-substitution rates appear rather similar, with the new Cs-containing phase
1 having slightly higher amount of Ga than in the earlier reported Cs
8Na
16Ga
21Si
115. This conjecture can be seen by comparing the corresponding unit cells as well—Cs
8Na
16Ga
21Si
115 has a cell parameter of
a = 14.918(2) Å, which expand slightly to
a = 14.9354(5) Å for Cs
8Na
16Ga
22.7Si
113.3(1), in a manner consistent with the increased Ga-content (the unit cell difference might actually be smaller due to the fact that the previous measurement was taken at 120 K, while the current is at 200 K). The comparison of the Rb-containing compounds shows nearly identical cell parameter with
a = 14.883(2) Å for Rb
7.3Na
16Ga
20Si
116 and
a = 14.8822(10) Å for Rb
8.4Na
15.6(1)Ga
19.6Si
116.4(1). Interpretation of this result is hindered again by the different temperatures at which the unit cell parameters have been determined. The slight discrepancy in the refined formulae with regard to the alkali metals is another issue to be considered (
vide infra).
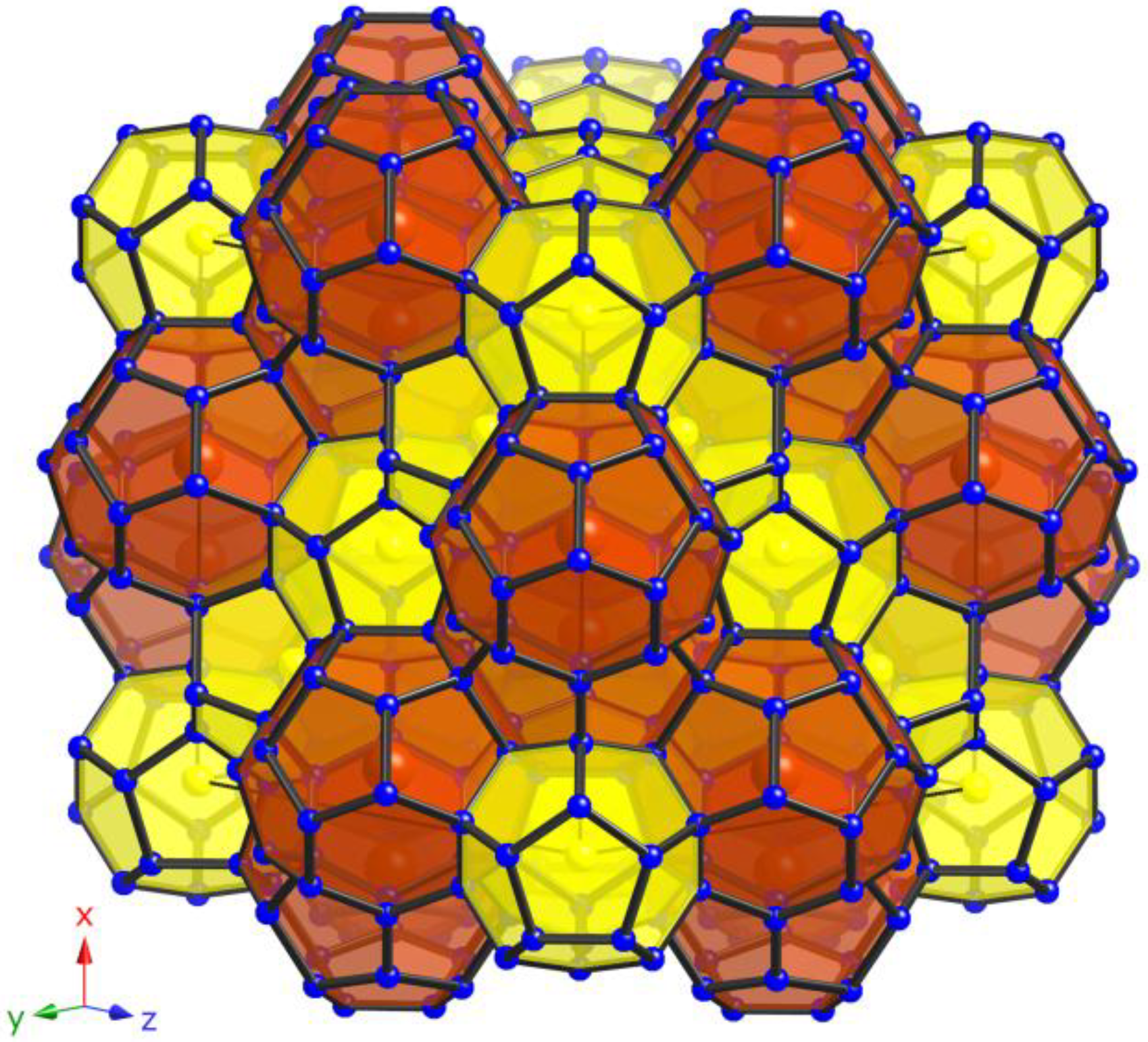
The open-framework of the type-II clathrates comprises of 136 tetrahedrally coordinated
M and Si atoms (
M = Ga, Zn) per unit cell located on the Wyckoff sites 96
g, 32
e, and
8a. The structure is best viewed as constituting a space-filling network of 16 (
M,Si)
20 pentagonal dodecahedra and 8 (
M,Si)
28 hexakaidecahedra (see
Figure 1). The larger 28-atom polyhedra are linked in a diamond-like fashion via their four hexagonal faces, while the smaller 20-atom polyhedra generate face-shared layers running along [111] in ABC-sequence, typical for the face-centered cubic symmetry. In the herein presented Si-based clathrates, the framework is substituted with Ga and Zn, respectively, and the framework site 96
g is favored by these larger atoms, while site 8
a is the least preferred site. This is visible in the atom contribution (see
Table 2 and
Table 3) with Si:(Ga,Zn) ratios of 78:22 for
1, 82:18 for
2 and 92:8 for
3 on 96
g, 96:4 for
1, 94:6 for
2 and 98:2 for
3 on 32
e, and 97:3 for
1 and 96:1 for
2 on 8
a. Only Si is occupying site 8
a in the Zn-containing compound
3. The same trend is occurring as well in both known Ga-substituted type-II clathrates Cs
8Na
16Ga
21Si
115 (ratio Si:Ga is 80:20 on 96
g, 95:5 on 32
e, and 98:2 on 8
a) and Rb
7.3Na
16Ga
20Si
116 (ratio Si:Ga is 80:20 on 96
g, 96:4 on 32
e, and 99:1 on 8
a) with comparable ratios. In all cases, the larger substituted anions increase the cage sizes and therefore an expansion of the unit cells (
vide supra) occur in accordance with the increasing amount of incorporated Zn and Ga, respectively. This can be also observed in the atomic distances (see
Table 4), which fall in the intervals of
dSi/Ga–Si/Ga = 2.385(1) to 2.430(1) Å for
1,
dSi/Ga–Si/Ga = 2.380(1) to 2.423(1) Å for
2, and
dSi/Zn–Si/(Zn) = 2.373(1) to 2.412(1) Å for
3, respectively. The distances in Cs
8Na
16Ga
21Si
115 (
dSi/Ga–Si/Ga = 2.386(1)–2.428(2) Å) and Rb
7.3Na
16Ga
20Si
116 (
dSi/Ga–Si/Ga = 2.378(1)–2.426(2) Å) [
17] also agree well. As expected, all distance have longer values than these for the ternary clathrates Cs
8Na
16Si
136 (
dSi–Si = 2.3584(4)–2.3924(5) Å at room temperature) [
15] and Rb
8Na
16Si
136 (
dSi–Si = 2.362(1)–2.395(1) Å at room temperature) [
6].
Figure 1.
View of the polyanionic framework of clathrates with type-II structure (yellow: pentagonal dodecahedra; red: hexakaidecahedra).
Figure 1.
View of the polyanionic framework of clathrates with type-II structure (yellow: pentagonal dodecahedra; red: hexakaidecahedra).
Table 2.
Atomic coordinates and equivalent isotropic displacement parameters (Ueq/Å2) for Cs8Na16Ga22.7Si113.3(1) (1); Rb8.4Na15.6(1)Ga19.6Si116.4(1) (2); and Rb8Na16Zn8.4Si127.6(1) (3). Coordinates are reported in the origin choice No. 2 for the cubic space group Fd3m.
Table 2.
Atomic coordinates and equivalent isotropic displacement parameters (Ueq/Å2) for Cs8Na16Ga22.7Si113.3(1) (1); Rb8.4Na15.6(1)Ga19.6Si116.4(1) (2); and Rb8Na16Zn8.4Si127.6(1) (3). Coordinates are reported in the origin choice No. 2 for the cubic space group Fd3m.
| Atom | site | x/a | y/b | z/c | occupancy/% | Ueq [a] |
|---|
| Cs8Na16Ga22.7Si113.3(1) | | | | |
| Na | 16c | 0 | 0 | 0 | 100 | 0.0212(6) |
| Cs | 8b | 3/8 | 3/8 | 3/8 | 100 | 0.0205(2) |
| Si1/Ga1 | 96g | 0.06746(2) | x | 0.37069(3) | 78/22 [b] | 0.0087(2) |
| Si2/Ga2 | 32e | 0.21719(4) | x | x | 95.6/4.4(4) | 0.0079(4) |
| Si3/Ga3 | 8a | 1/8 | 1/8 | 1/8 | 96.8/3.3(7) | 0.0084(7) |
| Rb8.4Na15.6(1)Ga19.6Si116.4(1) | | | | |
| Na1/Rb1 | 16c | 0 | 0 | 0 | 97.9/2.1(7) | 0.020(1) |
| Rb2 | 8b | 3/8 | 3/8 | 3/8 | 100 | 0.0283(4) |
| Si1/Ga1 | 96g | 0.06734(3) | x | 0.37088(5) | 82/18 [b] | 0.0084(2) |
| Si2/Ga2 | 32e | 0.21728(6) | x | x | 93.9/6.1(5) | 0.0083(5) |
| Si3/Ga3 | 8a | 1/8 | 1/8 | 1/8 | 96/4(1) | 0.0078(9) |
| Rb8Na16Zn8.4Si127.6(1) | | | | |
| Na | 16c | 0 | 0 | 0 | 100 | 0.0212(10) |
| Rb | 8b | 3/8 | 3/8 | 3/8 | 100 | 0.0296(5) |
| Si1/Zn1 | 96g | 0.06740(4) | x | 0.37091(6) | 92/8 [b] | 0.0086(3) |
| Si2/Zn2 | 32e | 0.21755(7) | x | x | 97.6/2.4(6) | 0.0087(4) |
| Si3 | 8a | 1/8 | 1/8 | 1/8 | 100 | 0.0074(7) |
Table 3.
Anisotropic displacement parameters (Uij/Å2) for Cs8Na16Ga22.7Si113.3(1) (1); Rb8.4Na15.6(1)Ga19.6Si116.4(1) (2); and Rb8Na16Zn8.4Si127.6(1) (3).
Table 3.
Anisotropic displacement parameters (Uij/Å2) for Cs8Na16Ga22.7Si113.3(1) (1); Rb8.4Na15.6(1)Ga19.6Si116.4(1) (2); and Rb8Na16Zn8.4Si127.6(1) (3).
| Atom | U11 | U22 | U33 | U23 | U13 | U12 |
|---|
| Cs8Na16Ga22.7Si113.3(1) |
| Na | 0.0212(6) | =U11 | =U11 | –0.0031(6) | =U23 | =U23 |
| Cs | 0.0205(2) | =U11 | =U11 | 0 | 0 | 0 |
| Si1/Ga1 | 0.0086(2) | =U11 | 0.0091(3) | –0.0001(1) | =U23 | 0.0003(2) |
| Si2/Ga2 | 0.0079(4) | =U11 | =U11 | 0.0001(2) | =U23 | =U23 |
| Si3/Ga3 | 0.0084(7) | =U11 | =U11 | 0 | 0 | 0 |
| Rb8.4Na15.6(1)Ga19.6Si116.4(1) |
| Na1/Rb1 | 0.0233(7) | =U11 | =U11 | –0.0013(8) | =U23 | =U23 |
| Rb2 | 0.0299(4) | =U11 | =U11 | 0 | 0 | 0 |
| Si1/Ga1 | 0.0082(3) | =U11 | 0.0089(4) | –0.0003(2) | =U23 | 0.0002(3) |
| Si2/Ga2 | 0.0080(3) | =U11 | =U11 | 0.0007(3) | =U23 | =U23 |
| Si3/Ga3 | 0.0075(4) | =U11 | =U11 | 0 | 0 | 0 |
| Rb8Na16Zn8.4Si127.6(1) |
| Na | 0.0212(10) | =U11 | =U11 | –0.0025(10) | =U23 | =U23 |
| Rb | 0.0296(5) | =U11 | =U11 | 0 | 0 | 0 |
| Si1/Zn1 | 0.0083(4) | =U11 | 0.0093(5) | 0.0001(2) | =U23 | 0.0000(3) |
| Si2/Zn2 | 0.0087(4) | =U11 | =U11 | 0.0005(4) | =U23 | =U23 |
| Si3 | 0.0074(7) | =U11 | =U11 | 0 | 0 | 0 |
Table 4.
Selected interatomic distances for Cs8Na16Ga22.7Si113.3(1) (1); Rb8.4Na15.6(1)Ga19.6Si116.4(1) (2); and Rb8Na16Zn8.4Si127.6(1) (3). Tt denotes the mixed occupied Si/Ga and Si/Zn, respectively.
Table 4.
Selected interatomic distances for Cs8Na16Ga22.7Si113.3(1) (1); Rb8.4Na15.6(1)Ga19.6Si116.4(1) (2); and Rb8Na16Zn8.4Si127.6(1) (3). Tt denotes the mixed occupied Si/Ga and Si/Zn, respectively.
| Compound 1 | d/Å | Compound 2 | d/Å | Compound 3 | d/Å |
|---|
| Tt1–Tt1 (2×) | 2.4016(5) | Tt1–Tt1 (2×) | 2.3894(8) | Tt1–Tt1 (2×) | 2.377(1) |
| Tt1–Tt2 | 2.4067(5) | Tt1–Tt2 | 2.3989(8) | Tt1–Tt2 | 2.385(1) |
| Tt1–Tt1 | 2.430(1) | Tt1–Tt1 | 2.437(1) | Tt1–Tt1 | 2.412(2) |
| Tt2–Tt3 | 2.3847(9) | Tt2–Tt3 | 2.380(1) | Tt2–Si3 | 2.373(2) |
| Tt2–Tt1 (3×) | 2.4066(5) | Tt2–Tt1 (3×) | 2.3988(8) | Tt2–Tt1 (3×) | 2.385(1) |
| Tt3–Tt2 (4×) | 2.3847(9) | Tt3–Tt2 (4×) | 2.379(1) | Si3–Tt2 (4×) | 2.373(2) |
| Na–Tt3 (2×) | 3.2336(1) | Na1/Rb1–Tt3 (2×) | 3.2221(2) | Na–Si3 (2×) | 3.2049(2) |
| Na–Tt2 (6×) | 3.3140(4) | Na1/Rb1–Tt2 (6×) | 3.3066(6) | Na–Tt2 (6×) | 3.2912(7) |
| Na–Tt1 (12×) | 3.4260(3) | Na1/Rb1–Tt1 (12×) | 3.4102(5) | Na–Tt1 (12×) | 3.3939(6) |
| Cs–Tt1 (12×) | 3.9879(4) | Rb2–Tt1 (12×) | 3.9719(7) | Rb–Tt1 (12×) | 3.950(1) |
| Cs–Tt1 (12×) | 4.0657(5) | Rb2–Tt1 (12×) | 4.049(1) | Rb–Tt1 (12×) | 4.028(1) |
| Cs–Tt2 (4×) | 4.0825(9) | Rb2–Tt2 (4×) | 4.064(1) | Rb–Tt2 (4×) | 4.037(2) |
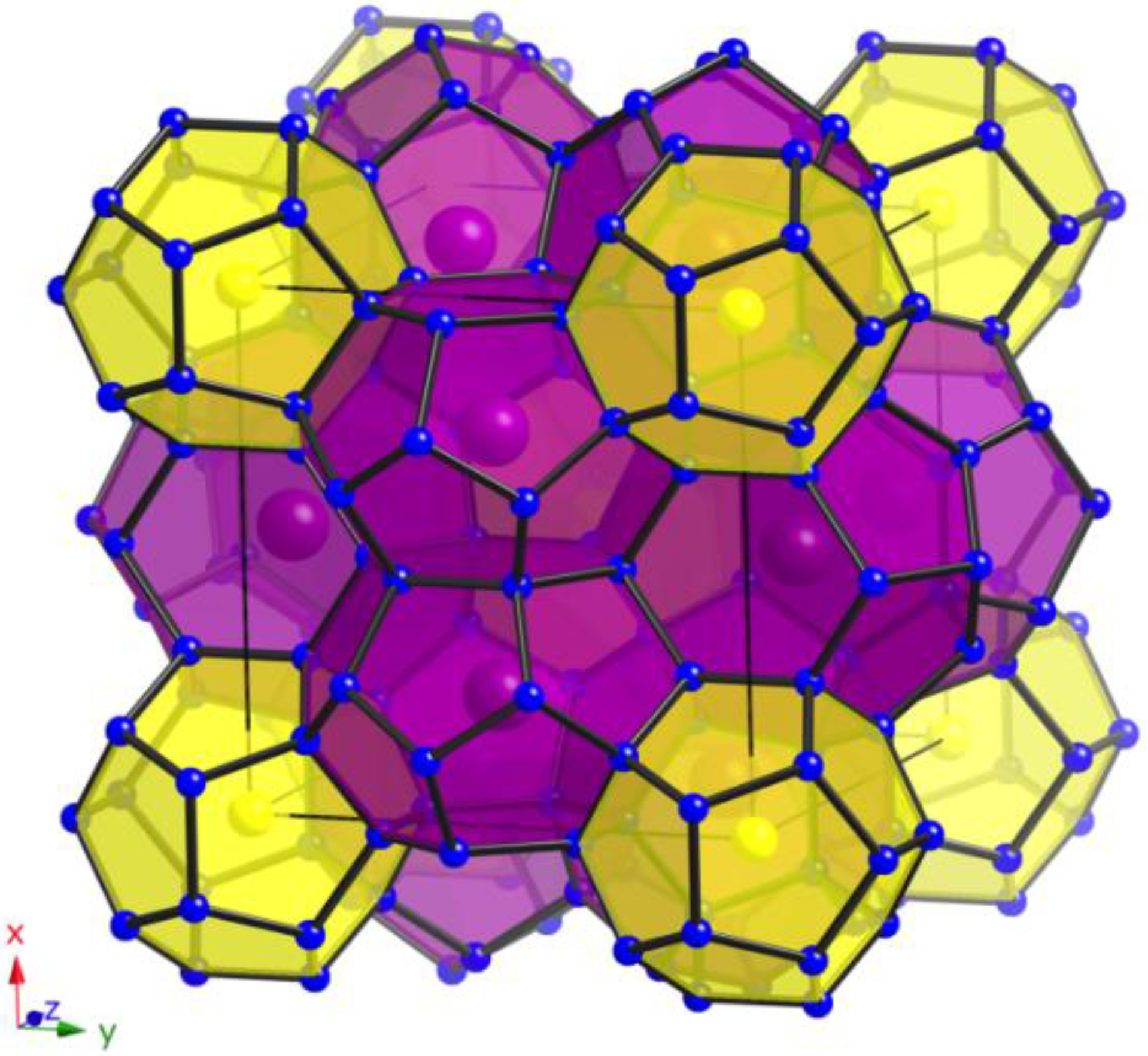
Due to the very different sizes of the two kinds of polyhedra, two very different guest atoms are required in order to achieve the complete and ordered occupation of the respective cages (see
Figure 2). This warranted our choice of the pairs of alkali metals, namely Na/Cs and Na/Rb, respectively (
rNa ≈ 1.57 Å,
rRb ≈ 2.16 Å,
rCs ≈ 2.35 Å [
25]). The optimal sizes for an ordering are realized with Na for the smaller pentagon dodecahedra and Cs for the larger hexakaidecahedra in
1 and in Cs
8Na
16Ga
21Si
115 [
17] and as well with Na again in the (Si,Zn)
20 cages and Rb in the (Si,Zn)
28 cages in compound
3. In contrast, the Ga-substituted framework of the Rb-containing clathrate
2 is intermediate in size (between
1 and
3), therefore, one can surmise that the larger cavities will still favor occupation by Rb atoms only, while the smaller ones are now large enough to host Na atoms, admixed with a small amount of Rb (refined as statistical ratio of Na:Rb = 97:3). For comparison, in Rb
7.3Na
16Ga
20Si
116 [
17], the pentagonal dodecahedra are fully occupied by Na, while the hexakaidecahedra are only 90% occupied by the Rb guest atoms, despite the similar amount of Ga substituting for Si in the framework and the correspondingly similar sizes of the (Ga,Si)
20 pentagonal dodecahedra and the (Ga,Si)
28 hexakaidecahedra (between Rb
7.3Na
16Ga
20Si
116 [
17] and structure
2). We were intrigued by this difference in the filling patterns for Rb
8.4Na
15.6(1)Ga
19.6Si
116.4(1) and the previously reported Rb
7.3Na
16Ga
20Si
116 [
17]. We speculated that differences in the reaction conditions might explain why this happens, and attempted to synthesize a new clathrate type-II from Rb, Na, Ga, and Si, using a modified synthetic procedure. Several random crystals selected from this reaction were subjected to crystallographic analysis and showed again the exact same phenomenology—one crystal was indexed with a unit cell
a = 14.8722(5) Å, and another one with
a = 14.8765(5) Å, both at 200 K. Despite these values being within a few standard deviations from each other, structure refinements showed the former to be an analog of
2,
i.e., the structure with the small Na-Rb disorder within the pentagonal dodecahedra, while the latter turned out to be analogous to the previously reported Rb–Na–Ga–Si phase with underoccupied hexakaidecahedral cages. The slight decrease in the unit cell parameters for these two structures compared to Rb
8.4Na
15.6(1)Ga
19.6Si
116.4(1) and Rb
7.3Na
16Ga
20Si
116 is apparently due to the slight difference in the Ga content (an artifact of the change in the synthesis scheme). Based on the above, it is evident that the Rb–Na–Ga–Si system requires more detailed studies to fully understand this behavior, which is not mirrored by the analogous Cs–Na–Ga–Si system.
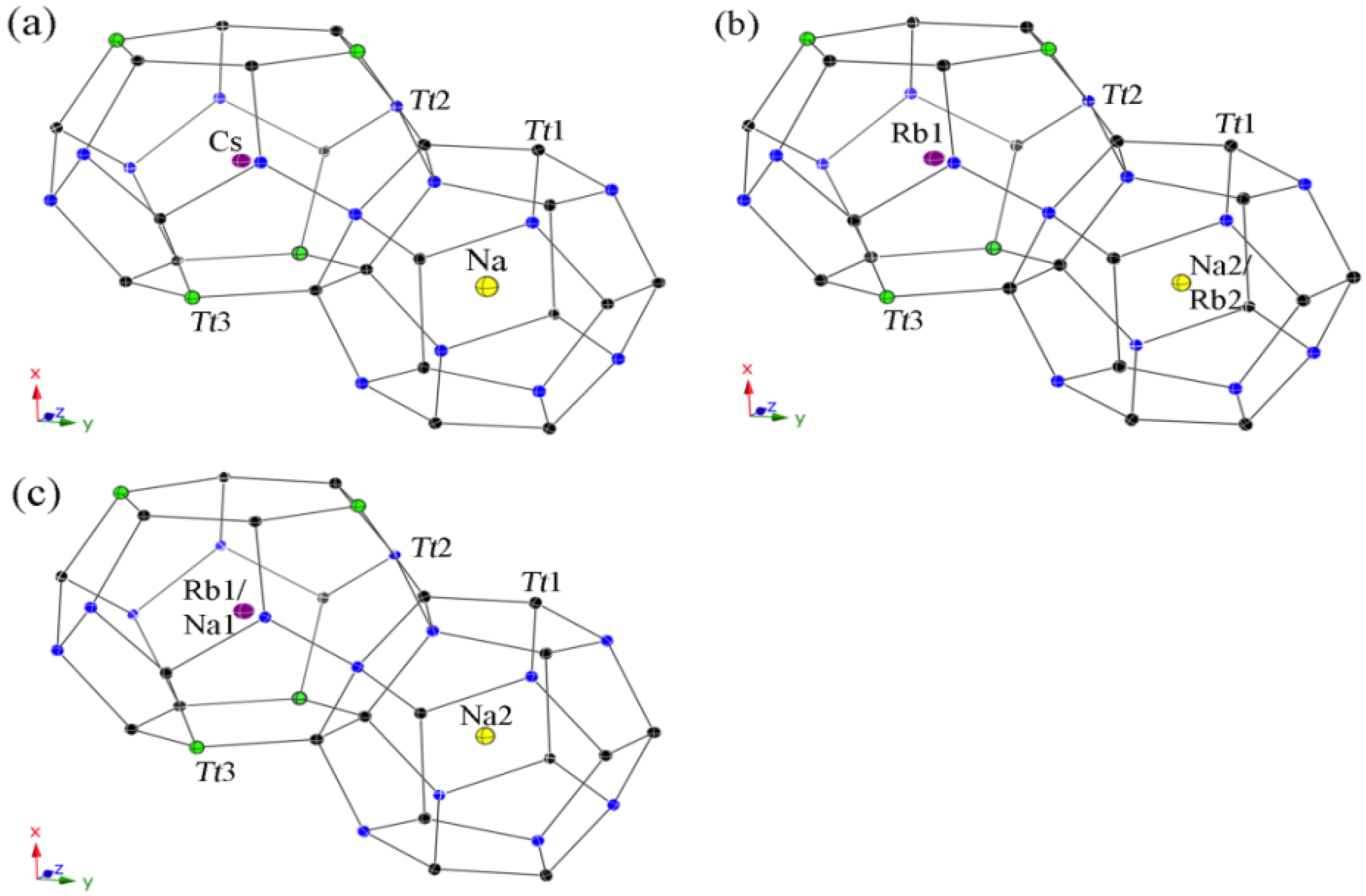
Figure 2.
Representation of the polyhedral cages in type-II structures with anisotropic displacement parameters, drawn at the 95% probability level. Cs8Na16Ga22.7Si113.3(1) (a); Rb8.4Na15.6(1)Ga19.6Si116.4(1) (b); and Rb8Na16Zn8.4Si127.6(1) (c).
Figure 2.
Representation of the polyhedral cages in type-II structures with anisotropic displacement parameters, drawn at the 95% probability level. Cs8Na16Ga22.7Si113.3(1) (a); Rb8.4Na15.6(1)Ga19.6Si116.4(1) (b); and Rb8Na16Zn8.4Si127.6(1) (c).
The interatomic distances between the filler atoms and the framework are within the intervals of
dNa–Si/Ga = 3.234(1) to 3.420(1) Å and
dCs–Si/Ga = 3.988(1) to 4.083(1) Å for
1,
dNa1/Rb1–Si/Ga = 3.222(1) to 3.410(1) Å and
dRb2–Si/Ga = 3.972(1) to 4.064(1) Å for
2, and
dNa–Si/Ga = 3.205(1) to 3.392(1) Å and
dRb–Si/Ga = 3.950(1) to 4.037(1) Å for
3, respectively (see
Table 4). The distances for Cs
8Na
16Ga
21Si
115 (
dNa–Si/Ga = 3.230(1)–3.416(1) Å and
dCs–Si/Ga = 3.984(1)–4.077(2) Å) and Rb
7.3Na
16Ga
20Si
116 (
dNa–Si/Ga = 3.222(1)–3.411(1) Å and
dRb–Si/Ga = 3.971(1)–4.066(2) Å) [
17] are very close in values as well. According to the Zintl-Klemm rules [
26,
27], the formulae for charge balanced compounds are [(Cs or Rb)
+]
8[Na
+]
16[4b-Ga1−]
24[4b-Si0]
112 for
1 and
2 and [Rb
+]
8[Na
+]
16[4b-Zn2−]
12[4b-Si0]
124 for
3. Hence, the composition of the Cs-containing compound
1 is the closest to the ideal formula, and a semiconducting-like (heavily doped, or even poorly metallic) behavior can be expected, while these for both Rb-containing clathrates
2 and
3 are obviously not charge-balanced and a metallic behavior can be predicted for both phases. In support of this reasoning, we can cite the known germanium clathrates Cs
8Na
16Ag
6.7Ge
129.3 [
18] and Cs
8Na
16Cu
5Ge
131 [
19], whose formulae also deviate from the ideal, charge-balanced compositions.
2.3. Crystal Chemistry of the Type-I Clathrates
Like for the above reported type-II clathrates, the cell parameters for the individual type-I clathrates are also decreasing at 200 K with
a = 10.4710(2) Å for Cs
6Na
2Ga
8.25Si
37.75(3) (
4), to
a = 10.4466(4) for Rb
6.34Na
1.66(2)Ga
8.02Si
37.98(3) (
5), to
a = 10.3543(4) Å for Rb
5.20Na
2.80(4)Zn
3.85Si
42.15(2) (
6) corresponding to the amount of Ga or Zn in the Si-based framework and the used alkali metals as guest atoms (see
Table 5,
Table 6 and
Table 7). The insertion of Na in Rb
6.34Na
1.66(2)Ga
8.02Si
37.98(3) has very little influence on the unit cell, as evident by comparing it to that of the ternary compound Rb
8Ga
8Si
38 (
a = 10.469(2) Å at room temperature) [
11], while the cell parameter for the Zn-containing clathrate is even significantly smaller than that of K
8Ga
7.9Si
38.1 (
a = 10.427(1) Å at room temperature) [
10].
Table 5.
Selected crystal data and structure refinement parameters for Cs6Na2Ga8.25Si37.75(3) (4); Rb6.34Na1.66(2)Ga8.02Si37.98(3) (5); and Rb5.20Na2.80(4)Zn3.85Si42.15(2) (6).
Table 5.
Selected crystal data and structure refinement parameters for Cs6Na2Ga8.25Si37.75(3) (4); Rb6.34Na1.66(2)Ga8.02Si37.98(3) (5); and Rb5.20Na2.80(4)Zn3.85Si42.15(2) (6).
| Compound | 4 | 5 | 6 |
|---|
| Fw/g/mol | 2479.03 | 2205.85 | 1944.48 |
| Crystal system | Cubic |
| Space group | Pm3n (No. 223) |
| a/Å | 10.4710(2) | 10.4466(4) | 10.3543(4) |
| V/Å3 | 1148.06(4) | 1140.05(8) | 1110.10(7) |
| Z | 1 |
| T/K | 200(2) |
| Radiation | MoKα, λ= 0.71073 Å |
| ρ/g·cm−3 | 3.59 | 3.21 | 2.91 |
| μ/cm−1 | 10.48 | 12.42 | 8.91 |
| Rint | 0.053 | 0.089 | 0.075 |
| data/restraints/parameters | 241/0/18 | 241/0/20 | 241/0/19 |
| R1 [I > 2σ(I)] [a] | 0.0159 | 0.0167 | 0.0135 |
| wR2 [I > 2σ(I)] [a] | 0.0394 | 0.0362 | 0.0295 |
| GOF | 1.202 | 1.072 | 1.128 |
| largest peak & hole/e−·Å−3 | 0.59 & −0.42 | 0.37 & −0.30 | 0.40 & −0.23 |
Table 6.
Atomic coordinates and equivalent isotropic displacement parameters (Ueq/Å2) for Cs6Na2Ga8.25Si37.75(3) (4), Rb6.34Na1.66(2)Ga8.02Si37.98(3) (5), and Rb5.20Na2.80(4)Zn3.85Si42.15(2) (6).
Table 6.
Atomic coordinates and equivalent isotropic displacement parameters (Ueq/Å2) for Cs6Na2Ga8.25Si37.75(3) (4), Rb6.34Na1.66(2)Ga8.02Si37.98(3) (5), and Rb5.20Na2.80(4)Zn3.85Si42.15(2) (6).
| Atom | Site | x/a | y/b | z/c | occupancy/% | Ueq(a) |
|---|
| Cs6Na2Ga8.25Si37.75(3) |
| Cs | 6d | 1/4 | 1/2 | 0 | 100 | 0.0131(2) |
| Na | 2a | 0 | 0 | 0 | 100 | 0.0176(13) |
| Si1/Ga1 | 24k | 0 | 0.30307(8) | 0.11830(8) | 86(1)/14(1) | 0.0069(2) |
| Si2/Ga2 | 16i | 0.18385(7) | x | x | 96(1)/4(1) | 0.0076(3) |
| Si3/Ga3 | 6c | 1/4 | 0 | 1/2 | 30(1)/70(1) | 0.0079(4) |
| Rb6.34Na1.66(2)Ga8.02Si37.98(3) |
| Rb1 | 6d | 1/4 | 1/2 | 0 | 100 | 0.0159(2) |
| Na2/Rb2 | 2a | 0 | 0 | 0 | 83(1)/17(1) | 0.0120(6) |
| Si1/Ga1 | 24k | 0 | 0.30439(7) | 0.11798(7) | 85(1)/15(1) | 0.0079(2) |
| Si2/Ga2 | 16i | 0.18411(5) | x | x | 97(1)/3(1) | 0.0078(3) |
| Si3/Ga3 | 6c | 1/4 | 0 | 1/2 | 34(1)/66(1) | 0.0076(2) |
| Rb5.20Na2.80(4)Zn3.85Si42.15(2) |
| Rb1/Na1 | 6d | 1/4 | 1/2 | 0 | 86(1)/14(1) | 0.0161(2) |
| Na2 | 2a | 0 | 0 | 0 | 100 | 0.0124(7) |
| Si1/Zn1 | 24k | 0 | 0.30472(6) | 0.11742(6) | 97(1)/3(1) | 0.0071(2) |
| Si2/Zn2 | 16i | 0.18363(4) | x | x | 98(1)/2(1) | 0.0067(3) |
| Si3/Zn3 | 6c | 1/4 | 0 | 1/2 | 54(1)/46(1) | 0.0074(2) |
Table 7.
Anisotropic displacement parameters (Uij/Å2) for Cs6Na2Ga8.25Si37.75(3) (4); Rb6.34Na1.66(2)Ga8.02Si37.98(3) (5); and Rb5.20Na2.80(4)Zn3.85Si42.15(2) (6).
Table 7.
Anisotropic displacement parameters (Uij/Å2) for Cs6Na2Ga8.25Si37.75(3) (4); Rb6.34Na1.66(2)Ga8.02Si37.98(3) (5); and Rb5.20Na2.80(4)Zn3.85Si42.15(2) (6).
| Atom | U11 | U22 | U33 | U23 | U13 | U12 |
|---|
| Cs6Na2Ga8.25Si37.75(3) |
| Cs | 0.0098(3) | 0.0147(2) | =U22 | 0 | 0 | 0 |
| Na | 0.0176(13) | =U11 | =U11 | 0 | 0 | 0 |
| Si1/Ga1 | 0.0060(4) | 0.0071(4) | 0.0077(4) | 0.0004(3) | 0 | 0 |
| Si2/Ga2 | 0.0076(3) | =U11 | =U11 | –0.0002(3) | =U23 | =U23 |
| Si3/Ga3 | 0.0080(5) | 0.0079(4) | =U22 | 0 | 0 | 0 |
| Rb6.34Na1.66(2)Ga8.02Si37.98(3) |
| Rb | 0.0116(4) | 0.0181(3) | =U22 | 0 | 0 | 0 |
| Na/Rb1 | 0.0120(6) | =U11 | =U11 | 0 | 0 | 0 |
| Si1/Ga1 | 0.0077(4) | 0.0082(4) | 0.0077(4) | 0.0006(3) | 0 | 0 |
| Si2/Ga2 | 0.0078(3) | =U11 | =U11 | 0.0000(2) | =U23 | =U23 |
| Si3/Ga3 | 0.0080(5) | 0.0074(3) | =U22 | 0 | 0 | 0 |
| Rb5.20Na2.80(4)Zn3.85Si42.15(2) |
| Rb1/Na1 | 0.0124(3) | 0.0179(3) | =U22 | 0 | 0 | 0 |
| Na2 | 0.0124(7) | =U11 | =U11 | 0 | 0 | 0 |
| Si1/Zn1 | 0.0065(4) | 0.0077(4) | 0.0069(3) | 0.0004(3) | 0 | 0 |
| Si2/Zn2 | 0.0067(3) | =U11 | =U11 | –0.0001(2) | =U23 | =U23 |
| Si3/Zn3 | 0.0081(4) | 0.0070(3) | =U22 | 0 | 0 | 0 |
In the cubic type-I structure (
Pm3n) of these clathrates, statistically disordered Si and
M atoms (
M = Ga, Zn) form the open-framework with 46 tetrahedrally coordinated atoms per unit cell located on the Wyckoff sites 24
k, 16
i, and 6
c. Overall, one unit cell encompasses two (Si,
M)
20 pentagonal dodecahedra and six (Si,
M)
24 tetrakaidecahedra (see
Figure 3) creating a space-filling network. The 24-atom polyhedra share their two hexagonal faces to create three perpendicular, but not interpenetrating columns running along [100], [010], and [001] of the cube. These columns encapsulate the isolated 20-atom polyhedra in the created voids. The
M atoms substitute all three framework sites in every clathrate (
Table 6) with an apparent preference to occupy site 6
c. The ratios are in agreement with the literature, whereby the preference for the 6
c site is confirmed. Our data show the following ratios—Si:Ga = 86:14 on 24
k, 96:4 on 16
i and 30:70 on 6
c for
4 and Si:Ga = 85:15 on 24
k, 97:3 on 16
i and 34:66 on 6
c for
5 similar and comparable to these of the ternary clathrates K
8Ga
7.9Si
38.1 (Si amounts in percents of the site occupation factors: 83%, 98%, and 43%, respectively) [
10] and Rb
8Ga
8Si
38 (Si amounts in percents of the site occupation factors: 78%, 99%, and 59%, respectively) [
11], while the ratio of Si:Zn = 97:3 on 24
k, 98:2 on 16
i, and 54:46 on 6
c for
6 follow the reduced amount of Zn.
Figure 3.
View of the polyanionic framework of clathrates with type-I structure (yellow: pentagonal dodecahedra; purple: tetrakaidecahedra).
Figure 3.
View of the polyanionic framework of clathrates with type-I structure (yellow: pentagonal dodecahedra; purple: tetrakaidecahedra).
All guest atoms are located in the center of the polyhedra with usual anisotropic displacement parameters (see
Figure 4). Both kinds of cavities are closer in size than for the type-II clathrates, yet, a strict ordering occurs again in the Cs-containing clathrate
4 with only Na atoms in the pentagon dodecahedra on site 2
a and Cs atoms in the tetrakaidecahedra on site 6
d. In contrast, the polyhedra sizes are not so unambiguous for both Rb-containing clathrates. Hence, the (Si,Ga)
20 cavities in
5 are large enough for the Rb cations and a statistical mixed occupancies occurs with a ratio of Na2:Rb2 = 83:17, while only Rb cations are in the (Si,Ga)
24 cavities. In the Zn-containing clathrate
6, the tetrakaidecahedra are already too small to encapsulate only Rb cations, resulting in a mixed occupancy of Rb1:Na1 with the ratio 86:14, and the pentagon dodecahedra are filled completely with Na atoms.
Figure 4.
Representation of the polyhedral cages in the type-I structures with anisotropic displacement parameters, drawn at the 95% probability level. (a) Cs6Na2Ga8.25Si37.75(3); (b) Rb6.34Na1.66(2)Ga8.02Si37.98(3); and (c) Rb5.20Na2.80(4)Zn3.85Si42.15(2).
Figure 4.
Representation of the polyhedral cages in the type-I structures with anisotropic displacement parameters, drawn at the 95% probability level. (a) Cs6Na2Ga8.25Si37.75(3); (b) Rb6.34Na1.66(2)Ga8.02Si37.98(3); and (c) Rb5.20Na2.80(4)Zn3.85Si42.15(2).
The dependence of content and size of the framework building atoms is once more visible in the decreasing intervals for the distances (see
Table 8) with
dSi/Ga–Si/Ga = 2.395(1)–2.481(1) Å,
dNa–Si/Ga = 3.334(1)–3.407(1) Å,
dCs–Si/Ga = 3.555(1)–4.035(1) Å for
4, dSi/Ga–Si/Ga = 2.384(2)–2.465(1) Å,
dNa2/Rb2–Si/Ga = 3.331(1)–3.410(1) Å,
dRb1–Si/Ga = 3.538(1)–4.031(1) Å for
5, and
dSi/Ga–Si/Ga = 2.378(1)–2.444(1) Å,
dNa2–Si/Ga = 3.293(1)–3.381(1) Å,
dRb1/Na1–Si/Ga = 3.503(1)–4.002(1) Å for
6 as well as for the ternary clathrates Rb
8Ga
8Si
38 (
dSi/Ga–Si/Ga = 2.365(1)–2.494(1) Å
dRb2–Si/Ga = 3.351(1)–3.437(1) Å
dRb1–Si/Ga = 3.540(1)–4.030(1) Å) [
11] and K
8Ga
7.9Si
38.1 (
dSi/Ga–Si/Ga = 2.366(1)–2.463(1) Å
dK2–Si/Ga = 3.332(1)–3.418(1) Å
dK1–Si/Ga = 3.523(1)–4.025(1) Å) [
10] and in good agreement with the bond lengths of the corresponding type-II clathrates (
vide supra).
Table 8.
Selected interatomic distances for Cs6Na2Ga8.25Si37.75(3) (4); Rb6.34Na1.66(2)Ga8.02Si37.98(3) (5); and Rb5.20Na2.80(4)Zn3.85Si42.15(2) (6). Tt denotes the mixed occupied Si/Ga and Si/Zn, respectively.
Table 8.
Selected interatomic distances for Cs6Na2Ga8.25Si37.75(3) (4); Rb6.34Na1.66(2)Ga8.02Si37.98(3) (5); and Rb5.20Na2.80(4)Zn3.85Si42.15(2) (6). Tt denotes the mixed occupied Si/Ga and Si/Zn, respectively.
| Compound 4 | d/Å | Compound 5 | d/Å | Compound 6 | d/Å |
|---|
| Tt1–Tt2 (2×) | 2.3949(6) | Tt1–Tt2 (2×) | 2.3991(5) | Tt1–Tt2 (2×) | 2.3784(4) |
| Tt1–Tt1 | 2.478(2) | Tt1–Tt1 | 2.465(1) | Tt1–Tt1 | 2.432(1) |
| Tt1–Tt3 | 2.4807(8) | Tt1–Tt3 | 2.4653(7) | Tt1–Tt3 | 2.4440(6) |
| Tt2–Tt2 | 2.3949(6) | Tt2–Tt2 | 2.384(2) | Tt2–Tt2 | 2.3784(4) |
| Tt2–Tt1 (3×) | 2.399(2) | Tt2–Tt1 (3×) | 2.3991(5) | Tt2–Tt1 (3×) | 2.381(2) |
| Tt3–Tt1 (4×) | 2.4807(8) | Tt3–Tt1 (4×) | 2.4654(7) | Tt3–Tt1 (4×) | 2.4440(6) |
| Cs–Tt1 (8×) | 3.5552(5) | Rb1–Tt1 (8×) | 3.5377(5) | Rb1/Na1–Tt1 (8×) | 3.5025(4) |
| Cs–Tt2 (8×) | 3.8916(4) | Rb1–Tt2 (8×) | 3.8811(3) | Rb1/Na1–Tt2 (8×) | 3.8465(3) |
| Cs–Tt3 (4×) | 3.702(1) | Rb1–Tt3 (4×) | 3.6934(1) | Rb1/Na1–Tt3 (4×) | 3.6608(1) |
| Cs–Tt1 (4×) | 4.0352(8) | Rb1–Tt1 (4×) | 4.031(1) | Rb1/Na1–Tt1 (4×) | 4.002(1) |
| Na–Tt2 (8×) | 3.334(1) | Na2/Rb2–Tt2 (8×) | 3.331(1) | Na–Tt2 (8×) | 3.2932(7) |
| Na–Tt1 (12×) | 3.4066(8) | Na2/Rb2–Tt1 (12×) | 3.4103(7) | Na–Tt1 (12×) | 3.3813(6) |
The refined formulae for the type-I clathrates are satisfying the Zintl-Klemm ideas for valence electrons counting and charge balance with [(Na,Cs or Rb)+]8[4b-Ga1−]8[4b-Si0]38 for Cs6Na2Ga8.25Si37.75(3) and Rb6.34Na1.66(2)Ga8.02Si37.98(3), and [(Na,Rb)+]8[4b-Zn2−]4[4b-Si0]42 for Rb5.20Na2.80(4)Zn3.85Si42.15(2), hence, semiconducting behavior can be expected for all three compounds. Given this and the robust open framework with many possibilities for substitutions, such compounds could be candidate-materials for thermoelectric applications. However, these speculations could not be confirmed as part of this study, as the synthesis requirements for phase-pure sample have not been yet worked out.